1. Introduction
The photoplethysmography (PPG) signal reflects the volumetric changes in microcirculation. PPG signals can be measured by detecting light absorption at different body sites with dense microvasculature, such as the fingertips or the ear lobe [
1,
2]. Hemodynamic parameters can be extracted from the AC components of PPG signals for daily cardiovascular healthcare applications. The development of PPG-based remote healthcare infrastructure is creating a promising pathway toward next-generation healthcare by enabling a low-cost real-time healthcare framework, especially for low-resource communities in underdeveloped countries [
3].
The PPG signal and its derivatives including velocity photoplethysmography (VPG) and acceleration photoplethysmography (APG) signals have been applied in investigating the physiological properties of the cardiovascular and respiratory systems including heart rate (HR), heart rate variability (HRV), and pulse rate variability (PRV) [
4,
5]. HRV indicators are often extracted from electrocardiogram (ECG) signals while the PRV indicators are extracted from the PPG signal. PRV indicators can be classified in accordance with the measurement time as long-term (≥24 h), short-term (~5 min), and ultra-short-term (≤5 min) ones. Ultra-short-term PRV monitoring can be achieved using the PPG signal. Peak-to-peak interval, defined as the distance between two systolic peaks in the PPG signal or its derivatives and is one of the commonest evaluations of PRV [
6,
7]. Other common PRV indicators include inter-beat interval (IBI), SD of normal-to-normal P-P intervals (SDNN), and root mean square of successive differences between normal heartbeats (RMSSD). However, the PPG signal and its derivatives can be affected by device properties, motion artifacts, and physiological factors such as anatomic variations, arterial stiffness, measurement site, posture, breathing pattern, and sentiment [
8,
9].
Several studies have examined the accuracy of PPG and APG signals in various scenarios, such as standing at rest, sitting with a deep breathing rhythm, and exercising [
10]. Jan et al. investigated the coherence between ECG and PPG-derived PRV indicators during controlled and uncontrolled breathing patterns and found that PRV indicators were considerably less accurate during controlled breathing exercises [
11]. Królak et al. studied the reliability of PRV extraction from prerecorded PPG data at rest and achieved a high approximation to ECG-derived HRV indicators [
12]. PRV indicators from PPG and APG signals during rest and exercise have been extensively explored in controlled scenarios, with time-domain PRV indicators being accurately detectable at rest, while their accuracy during exercise varies across different studies [
13,
14,
15,
16,
17]. Although comparison algorithms have been proposed to enhance the accuracy of time-domain PRV indicators, they have not been tested in real-time PPG signal extraction [
10]. Moreover, there is currently a lack of comprehensive evaluation of different physiological factors and their effects on the accuracy of PPG-derived ultra-short-term PRV indicators, which are crucial in remote healthcare monitoring.
The integration of commercial pulse oximeters, or PPG sensors, with intelligent systems provides an affordable means for remote healthcare monitoring in low-resource settings, though their accuracy in many cases still needs validation [
18]. Bluetooth low energy (BLE) is becoming increasingly popular in techniques for remote healthcare monitoring due to its low energy consumption and ability to transfer small data sets, especially in Internet of Things (IoT)-based applications. The use of PPG-based remote healthcare monitoring has the potential to revolutionize healthcare by reducing costs, enabling the agile implementation of healthcare infrastructure, providing universal healthcare access to entire populations, and decentralizing healthcare services [
19,
20].
Introducing new e-health technologies in Latin America holds great promise, as they have the potential to bring about numerous benefits. For example, IoT devices can enable healthcare providers to monitor the health status of patients in real time, facilitating early detection and treatment of potential health issues and ultimately improving overall patient outcomes. Additionally, remote monitoring can reduce the need for hospital readmissions, resulting in cost savings for both patients and healthcare providers. Furthermore, remote monitoring can empower patients to take an active role in managing their health, enhancing patient engagement [
21].
When it comes to monitoring PRV remotely, previous studies have only examined PRV data in two or three different body postures. Additionally, although researchers have explored extracting PRV from pre-recorded remote PPG data, there has not been a thorough investigation into real-time PRV monitoring using affordable PPG systems [
22,
23].
To address the abovementioned gaps, the purpose of this paper is to investigate the standardized integration of commercial PPG and BLE devices for the extraction of HR, IBI, SDNN, and RMSSD at different body postures (sitting at 90°, sitting at 90° while deep breathing, standing, and supine).
2. Materials and Methods
2.1. Subjects
Thirty healthy male and female subjects (mean ± SD of age: 24 ± 5 years) from local communities without any known cardiovascular or respiratory disease participated in this study with written informed consent. The protocol was approved under resolution no. PE-OB-02-2021 of the Santa Paula University Ethical Committee. Before the experiment, the procedure was explained in detail to each participant.
2.2. Device Integration Architecture
The integration proposed in this study was proposed for low-resource centers that comply with the following premises:
Very low budget for medical equipment
Total absence of WiFi equipment
Presence of a basic computer
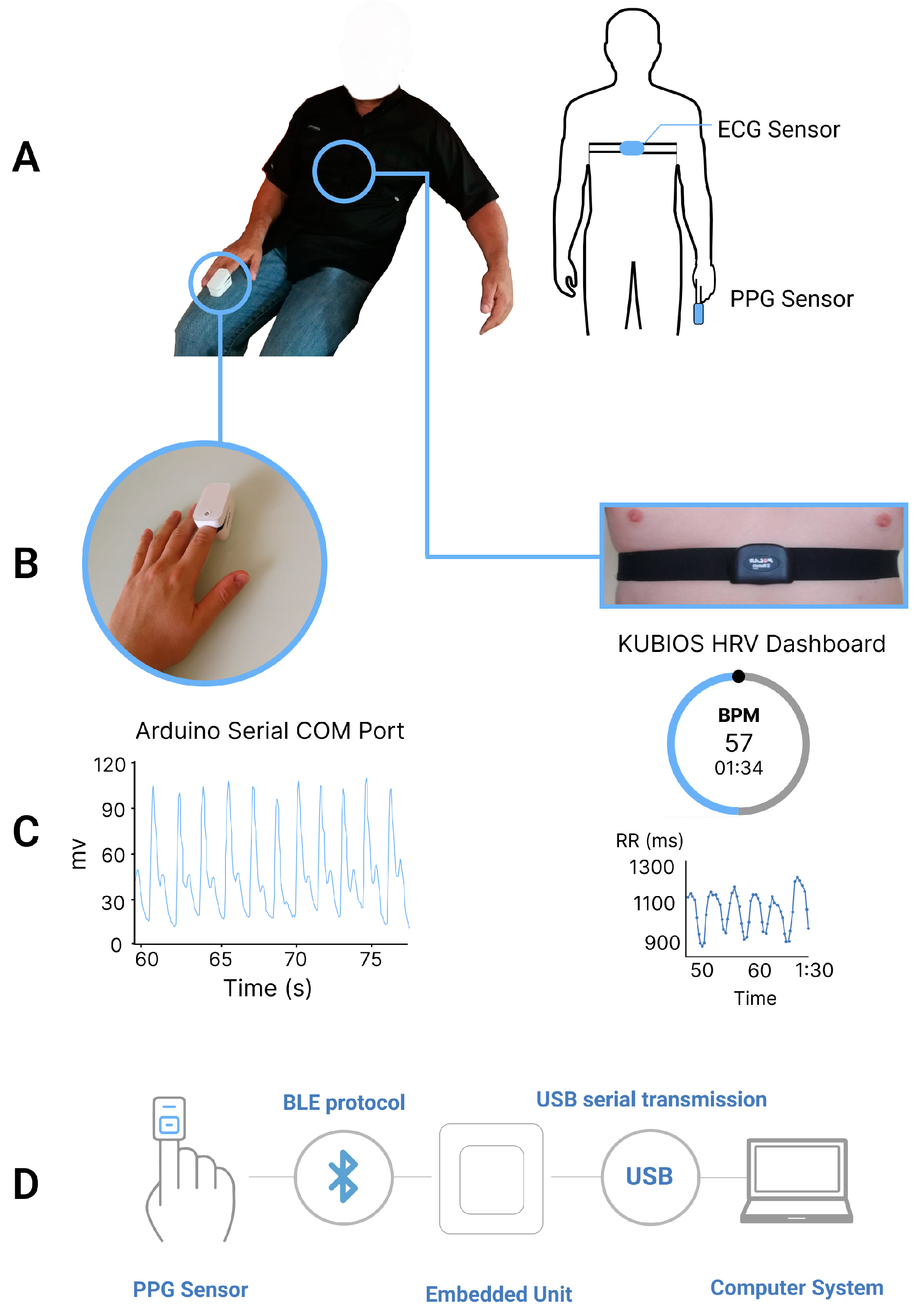
Therefore, the integration (shown in
Figure 1D) only includes the commercial PPG sensor, the BLE receiver unit connected to a basic computer that can locally store the data.
2.3. Equipment
The commercial Bluetooth Low Energy Pulse Oximeter (manufactured by Jumper Medical Equipment Co., Shenzhen, China, under the device model JPD-500G with a cost of USD 35) was used in this study as a non-invasive device that measures the oxygen saturation level in the blood through the PPG signal. It was equipped with an nRF52832 multiprotocol SoC chipset. The PPG sensor had a frequency range of 2.402–2.480 GHz and a power output of 0.4 mW, which adhered to the 15C radio frequency legislation. The sampling frequency of the PPG sensor was set to 40 Hz.
In contrast, the reference ECG sensor used in this study was the Polar H7 BLE Strap (manufactured by Polar Electro Oy, Helsinski, Finland), which has been validated in previous literature [
24,
25,
26]. This sensor was placed on the participant’s chest to record the electrical activity of the heart. It had a sampling frequency of 1000 Hz, which provided a high temporal resolution and allowed for the capture of subtle changes in the heart’s electrical activity. The ECG sensor was wirelessly connected to the Kubios HRV software (ver. 1.1.10) through Bluetooth Low Energy technology, which ensured reliable and fast data transmission.
The receiving unit is composed of a 32-bit Extensa LX6 (manufactured by the M5Stack Company, Shenzhen, China, with a cost of 15 USD) dual nucleus chipset along with an in-built link transmitter and receiver of 2.4 GHz band compliant to Bluetooth v4.2/EDR and BLE specification. In addition, a laptop computer with 32 Gb of RAM and 1.5 TB of memory was used for data processing.
2.4. Measurement Procedure
Initially, the Polar H7 ECG strap was adjusted and positioned at the xiphoid process by the participant. Next, the heart rate BLE data transmission between the ECG sensor and the Kubios HRV app was confirmed for each participant. Following that, the commercial PPG sensor was placed on the participant’s right-hand index finger (as shown in
Figure 1A), and the transmission of the PPG signal was validated through the serial port of the researcher’s personal computer. The HRV parameters were extracted using the commercial PPG sensor in four randomized scenarios: sitting at 90°, standing, and supine positions, all while breathing normally (as depicted in
Figure 2), as well as sitting at 90° with deep breathing. Each test had a duration of three minutes [
27] and was repeated twice with a 120 s break between the two trials. For the three postures under normal breathing, the participants were instructed to maintain stillness, avoid speaking, and breathe normally during each test. In the deep breathing scenario, the respiratory rate was controlled at approximately 10 breaths per minute. Throughout the experiment, a clinician was present to ensure that all the steps were correctly followed. The study protocol was designed to establish a foundation for physiological measurement in real-life situations. Therefore, we selected the three postures that are common in daily activities and have been widely implemented in prior studies, while the supine and deep breathing scenarios can simulate resting and sleeping states [
28,
29,
30].
2.5. Pre-Processing of PPG Signals
As shown in
Figure 3, to reduce the baseline wandering and signal saturation, a simple moving average filter was implemented on the real-time received raw data. Then, an adaptive iteratively reweighted penalized test [
30] was implemented to stabilize the baseline of the PPG data. Consequently, a digital IIR sixth-order low-pass filter with a cutoff frequency of 18 Hz was applied. Filtering performance can be visualized in
Figure 3, where noise is reduced significantly after the filter is applied (superimposed image). Once the filtering is implemented, the second derivative is applied to the PPG signal to utilize the APG component.
2.6. Extraction of Time-Domain HRV/PRV Indicators
2.6.1. Extraction of HRV Indicators from ECG Signals
The ECG data were processed using Kubios HRV (ver. 1.1.10) software to extract HR, IBI, SDNN, and RMSSD [
31].
2.6.2. Extraction of PRV Indicators from PPG Signals
The precise detection of peaks was achieved using a Python-based algorithm developed from the Scipy Package (
Figure 4) [
32], which utilized a flexible decision-making rule that had been previously developed and compared in [
33] to extract inter-beat intervals. In Formulas (1) and (2) the counter “k” for the cardiac events has been used. This counter runs from 1 to n, the total number of cardiac events to begin with. Cardiac event detection was initiated by identifying global maxima of the APG cardiac cycle, utilizing the outlined formula below to accentuate cardiac pulses, and assigning a value of zero to the remainder of the APG wave.
The logical array zι is created with i and n representing the first and nth elements, respectively, while ɵ denotes the acceptable threshold limit for peaks. N refers to the overall number of samples contained within the APG signal record, and Θ is the device’s sample frequency. The value of i is determined by the relative position within the data window ω, where . In the context of this study, the data window was established as 20 s.
is the local peak threshold.
Then, the extraction of peak-to-peak intervals was performed using the following formula:
where
is the discrete distance from detected peaks (
).
Then, heart rate was extracted as:
RMSSD was derived as:
where RR is the average peak-to-peak interval and N the number of cardiac events.
2.7. Statistical Methods
2.7.1. Initial Analysis of HRV and PRV Indicators
All the statistical analyses were performed in the R programming language (Version 4.1.2) due to its extensive and powerful packages for data analysis and visualization such as ggplot2 and dplyr [
34]. For descriptive statistics (mean ± standard deviation [SD]), Pearson correlation coefficient, percentage bias (P bias), and difference factors (DF%) [
16] were calculated for all HRV and PRV indicators.
2.7.2. Comparison between HRV and PRV Indicators in Four Physiological Status
In order to determine if PRV indicators can serve as reliable indicators for HRV, an assessment of normality was first conducted using the Shapiro–Wilk test. Normality was defined as having a p-value greater than 0.05 for all indicators in each of the four physiological statuses (i.e., three postures and deep breathing). Subsequently, a paired t-test was performed for each pair of HRV and PRV indicators that demonstrated normal distribution, while the Wilcoxon signed-rank test was utilized if normality was not met, in order to identify any significant differences between the two sets of indicators.
2.7.3. Effect of Posture and Breathing Pattern on the Difference between HRV and PRV Indicators
To further explore how different postures may impact the difference between corresponding HRV and PRV indicators, an analysis of variance (ANOVA) or its non-parametric equivalent, the Scheirer–Ray–Hare test, was conducted. Prior to this, the homogeneity of variance was evaluated using Levene’s test, with homogeneity defined as having a p-value greater than 0.05. If homogeneity of variance was satisfied, the ANOVA was performed, whereas if it was violated, the Scheirer–Ray–Hare test was used instead. The independent variables for both tests included posture (sitting at 90°, standing, and supine), indicator type (HR, IBI, SDNN, and RMSSD), and their interaction. Post hoc analysis was then conducted using Tukey’s test for ANOVA, and Dunn’s test or Kruskal–Wallis test for each scenario in the Scheirer–Ray–Hare test, in order to identify the most reliable posture and HRV indicator. In a similar vein, the impact of breathing patterns on the relative difference between HRV and PRV indicators was examined through a comparison of results using either the paired t-test or the Wilcoxon signed-rank test for each type of indicator.
2.7.4. Bland–Altman Analysis
To evaluate the accuracy of PRV indicators, the Bland–Altman analysis was performed between PRV and corresponding HRV indicators in all the postures and breathing patterns.
2.7.5. Linear Regression Analysis
Correlation of PRV data between the PPG results and the ECG results was constructed to inspect if the data follow a linear pattern. Datasets from all the indicators were fitted to a linear regression model and the correlation was evaluated based on the regression coefficient (R2).
2.8. Data Loss Analysis
Average data loss was defined as the percentage of missing sampling points during the measurement. The mean and SD of data loss were calculated in each physiological status. In addition, the effect of posture on data loss was studied similarly to that of the difference between HRV and PRV indicators.
5. Conclusions
Physiological abnormalities and body posture, particularly sitting, can affect the accuracy of PPG signals. However, there is a lack of research on the impact of the supine position. This study demonstrated that using commercial PPG and BLE devices with real-time extraction of time-domain HRV indicators is achievable with acceptable accuracy, scalability, stability, and minimal data loss. The study introduced an evaluation method that considers data normality, correlation analysis, Bland–Altman plots, and parametric or non-parametric statistical tests. The standardized approach provides an accurate and dependable reference for time-domain PRV indicators at different respiratory rates, utilizing cost-effective equipment compared to high-performing commercial systems. Although all postures showed acceptable accuracy, it is not recommended to use motion-induced scenarios for the real-time extraction of HRV and PPG-related indicators. The solution is scalable, low-cost, and integrable.
The proposed approach is not only accurate and reliable but also has the advantage of being less expensive compared to compared systems. This makes it an ideal solution for low-income countries where healthcare resources may be limited. Additionally, the proposed evaluation method provides a standardized and objective way to assess the quality of the data, which can be particularly useful in resource-limited settings where expert interpretation of data may not be readily available. Overall, this approach has the potential to facilitate the widespread use of PRV analysis as a non-invasive and low-cost tool for cardiovascular health monitoring in low-income countries.