2.1. Mutant Selection and Resistance Test
According to our previous experience in the treatment of
P. purpurogenum G59 spores by gentamicin in aqueous DMSO at 4 °C to introduce drug resistance [
27], and further, in view of the documented effect of ultrasound on the membrane permeability [
42,
43,
44,
45,
46,
47,
48,
49], we tested the introduction of drug resistance in
A. versicolor ZBY-3 by neomycin treatment of ZBY-3 spores at 4 °C for different times after ultrasound irradiation of the spores in the presence of neomycin at different concentrations. In preliminary tests searching for suitable conditions, the treatment of ZBY-3 spores with 200–800 mg/mL neomycin at 4 °C did not inhibit strain growth on potato dextrose agar (PDA) plates at 28 °C. The strain grew as well as the ZBY-3 strain without the spores having been treated by neomycin, as described below in the resistance test, indicating the insensitivity of the ZBY-3 strain to neomycin. Similarly, treatment of the ZBY-3 spores only with ultrasound irradiation by 2 s of irradiation every 4 s with an 8-min duration cycle at 200 or 400 W of output power also did not inhibit the strain growth on PDA plates at 28 °C. In contrast, ultrasound irradiation of the spores by 2 s of irradiation every 4 s with an 8-min duration cycle at 800 W of output power resulted in the complete inhibition of the strain’s growth, in spite of the presence or absence of neomycin. Fortunately, ultrasound irradiation for 2 s every 4 s with an 8-min duration cycle at 200 or 400 W of output power in the presence of 200 or 400 mg/mL neomycin properly inhibited the strain’s growth, which made its use suitable for selecting drug-resistant mutants from PDA plates by single colony isolation.
Thus, in the present study, ultrasound irradiation of the ZBY-3 spores was run by the 2-s irradiation every 4 s with an 8-min duration cycle at 200 or 400 W of output power. Fresh spore suspensions with the same spore density and 200 or 400 mg/mL neomycin in water were subjected to ultrasound irradiation. After ultrasound irradiation, the irradiated spore suspension was kept at 4 °C for up to 72 h for further treatment of the spores with the neomycin contained therein. During the treatment period, each 100 µL portion of the treated spore suspensions was spread on PDA plates at 3, 12, 24 and 48 h of the treatment and incubated at 28 °C for 3–5 days. Mutant colonies developed on the PDA plates were selected by single colony isolation during the incubation period with different appearances. A total of 30 mutants were selected, including 12 from the 200-W ultrasound irradiation and 18 from the 400-W ultrasound irradiation groups. Among them, 15 of the 30 mutants were from the 200 and 15 from the 400 mg/mL neomycin treatment groups, respectively (
Table 1).
All of the above-mentioned 30 mutants and their parent ZBY-3 strain were serially passaged four times by transfers under nonselective conditions onto PDA plates, followed by a further four times of passage over two years, to obtain the first to eighth passages of the mutants and the parent strain. Of them, the fourth, sixth and eighth passages were used in the first, second and third rounds of fermentations for the MTT assay to evaluate the effects of their culture extracts on K562 cells, respectively, and the extracts from the third round of fermentation of seven bioactive mutants and the parent ZBY-3 strain were also used in the HPLC-PDAD-UV and HPLC-ESI-MS analyses. The eighth passage of a bioactive mutant, u2n2h3-3, and the ZBY-3 strain was used in the resistance test and also in the large-scale fermentation for the investigation of newly produced metabolites by the mutant, u2n2h3-3. All of the 30 mutants and parent ZBY-3 strain maintained their genetic stability well over two years during the eight passages, as indicated by the effects of their culture extracts on the K562 cells, as shown in
Section 2.2.
Table 1.
Mutant numbers selected by ultrasound-mediated neomycin treatment of ZBY-3 spores a.
Table 1.
Mutant numbers selected by ultrasound-mediated neomycin treatment of ZBY-3 spores a.
| Ultrasound (W) | Neomycin (mg/mL) | Treatment Time at 4 °C | Total |
|---|
| 3 h | 12 h | 24 h | 48 h |
|---|
| 200 | 200 | 4 | 1 | 1 | 2 | 8 |
| 400 | 1 | 2 | 1 | 0 | 4 |
| Sum | 5 | 3 | 2 | 2 | 12 |
| 400 | 200 | 1 | 0 | 2 | 4 | 7 |
| 400 | 2 | 1 | 3 | 5 | 11 |
| Sum | 3 | 1 | 5 | 9 | 18 |
| Total | 8 | 4 | 7 | 11 | 30 |
The strain growth and spore formation of the 30 mutants and the parent ZBY-3 strain were not so obviously different, but their major difference mainly appeared in the morphology, the color of mycelia or pigment formation, when grown on PDA plates at 28 °C. The phenotypic differences of the parent ZBY-3 strain and the mutants are shown by photographs of some selected mutants and the ZBY-3 strain growing on PDA plates as typical examples in
Figure 2. The phenotypes of these mutants and parent ZBY-3 strain were not fully identical with the appearances of their corresponding colonies observed during mutant selection on the PDA plates on which the spore suspensions were spread soon after neomycin treatment. The stress from the ultrasound-mediated neomycin treatment likely affected the phenotypic development of these colonies. However, once the colonies were picked up, streaked out on PDA plates and then incubated at 28 °C, their own phenotypes appeared, as shown by typical examples in
Figure 2. Corresponding to the phenotypic differences on the PDA plates, as shown in
Figure 2, similar differences in the colors of mycelia and culture broths were also observed by fermentation in liquid medium.
The acquired resistance of the mutants to neomycin was testified by a resistance test using a mutant, u2n2h3-3, and the parent strain, ZBY-3. After ultrasound irradiation of the fresh u2n2h3-3 and ZBY-3 spore suspensions by 2-s irradiation every 4 s with an 8-min duration cycle at 200 W of ultrasound power in the presence of 200 mg/mL of neomycin, the spore suspensions were kept at 4 °C for 3 h for further treatment of the spores with neomycin, as done for selecting the mutant, u2n2h3-3. Each 100 μL of the spore suspensions was then spread on PDA plates and incubated at 28 °C for five days to examine their day-by-day growth. Many colonies of the mutant, u2n2h3-3, appeared on the second day of incubation and quickly grew up to become confluent, indicating the acquired resistance of the mutant to neomycin treatment in comparison to the mutants’ growth with the parent ZBY-3 strain (
Figure 3). The mutant grew far better than the control ZBY-3 strain in the resistance test, as seen in
Figure 3. In contrast to the mutant growth, although two single colonies of the ZBY-3 strain appeared early on the first day of incubation and grew well with incubations, a few other colonies appeared as late as on the fourth day of incubation and, further, more colonies later on the fifth day of incubation (
Figure 3). These colonies, especially those developed at earlier, before the fourth day of incubation, should be mutant colonies resistant to the neomycin treatment, which just reproduced a similar phenomenon allowing single colony isolation in the mutant selection. The many colonies coming into appearance later on the fifth day of incubation, both for the mutant and parent strains, in
Figure 3, seem likely to be formed from fresh spores of the early developing colonies on the plates.
Figure 2.
Phenotypes of the ZBY-3 strain and selected mutants growing on PDA plates by incubation at 28 °C for three days.
Figure 2.
Phenotypes of the ZBY-3 strain and selected mutants growing on PDA plates by incubation at 28 °C for three days.
Figure 3.
The growth of A. versicolor ZBY-3 and its mutant, u2n2h3-3, on PDA plates by incubation at 28 °C for different times (day) after treatment of their spores with neomycin. The fresh ZBY-3 and u2n2h3-3 spore suspensions were treated with 200 mg/mL neomycin, as done for selecting the mutant, and each 100 μL portion of the treated spore suspensions was spread on PDA plates, incubated at 28 °C and photographed at the given incubation times (day).
Figure 3.
The growth of A. versicolor ZBY-3 and its mutant, u2n2h3-3, on PDA plates by incubation at 28 °C for different times (day) after treatment of their spores with neomycin. The fresh ZBY-3 and u2n2h3-3 spore suspensions were treated with 200 mg/mL neomycin, as done for selecting the mutant, and each 100 μL portion of the treated spore suspensions was spread on PDA plates, incubated at 28 °C and photographed at the given incubation times (day).
Incidentally, when 100 μL of the fresh ZBY-3 spore suspension were spread on the PDA plate without treatment, as in the resistance test, and incubated at 28 °C, the stain grew very well. A lot of fine colony species appeared throughout the surface of the PDA plate on the second day of incubation, quickly growing into a confluent lawn on the third day of incubation, and the color of colonies or the confluent lawn differed from those shown in
Figure 3 for the mutant and the control parent strain in the resistance test.
Thus, the resistance test results demonstrated that the treatment of ZBY-3 spores with neomycin by ultrasound radiation has successfully introduced the drug resistance into the mutant, u2n2h3-3.
2.5. Discussions
Neomycin has been identified for weak antioomycete activities on crop oomycete pathogens within several
Phytophthora species, known as pseudofungi, but not antifungal activities on tested 10 eufungi species, including some of the
Penicillium and
Aspergillus species [
62]. Indeed, neomycin was also inactive for
Aspergillus versicolor ZBY-3, as shown by our preliminary tests on treatment of the ZBY-3 spores with a high concentration of 400 or 800 mg/mL neomycin in the present study as described. In fact, we also tested, at first, selecting neomycin-resistant mutants from the ZBY-3 strain using drug-containing PDA plates by the routine method in bacterial ribosome engineering to obtain drug-resistant mutants [
22]. However, when fresh ZBY-3 spores were spread on PDA plates containing a high concentration of 400 or 800 mg/mL neomycin and incubated at 28 °C for 3–5 days, the ZBY-3 strain grew very well: a lot of fine colony species appeared throughout the PDA plate surface on the second day of incubation and quickly grew into a confluent lawn on the third day of incubation, the same as the growth of the strain on the PDA plates that did not contain neomycin, and no colonies resistant to the neomycin could develop on the PDA plates. These results also indicated the insensitiveness of the ZBY-3 strain to the neomycin. It was thus impossible to obtain drug-resistant mutants from the ZBY-3 strain by neomycin-containing plates, as in bacterial ribosome engineering [
22].
The low intracellular concentration of aminoglycosides restricted by fungal membrane permeability, coupled with their lower binding affinity to the eukaryotic rRNA than to the prokaryotic rRNA, was considered to be a major cause resulting in the insensitivity of fungi to aminoglycosides. This has been overcome by the combined use of DMSO on the basis of its effect on membrane permeability to enhance the sensitivity of fungi to aminoglycoside antibiotics, resulting in the successful introduction of gentamicin-resistance into a
Penicillium species [
27]. Instead of the use of DMSO [
27], in the present study, we tested the effect of ultrasound on regulating the penetration of the neomycin into fungal cells and found that the treatment of ZBY-3 spores by a high concentration of neomycin in combination with suitable ultrasound irradiation could inhibit strain growth, allowing the development of resistant colonies on PDA plates. This enabled us to carry out single colony isolation to select a total of 30 neomycin-resistant mutants in present study. The acquired resistance of the mutants to neomycin was testified by the resistance test with a mutant, u2n2h3-3, and the parent strain, ZBY-3. After treatment of their spores with the same 200 mg/mL of neomycin as that for selecting the mutant, the mutant grew well on a PDA plate at 28 °C and quickly grew to form confluent lawns early on the fourth day of incubation (
Figure 3), whereas the growth of the ZBY-3 strain was significantly inhibited, so as to develop limited colonies on the fourth day of incubation (
Figure 3), as also observed in the mutant selection. Thus, the resistance test demonstrated that the neomycin-resistance has been successfully introduced into the mutant, u2n2h3-3.
The effect of the introduction of neomycin resistance on the secondary metabolite production in the ZBY-3 strain was demonstrated both by bioassays and chemical analyses. The EtOAc extract of the ZBY-3 strain did not inhibit K562 cells, whereas those from 22 mutants significantly inhibited K562 cells compared to the ZBY-3 extract (
Table 2), indicating that the 22 mutants have acquired the metabolic capability to produce antitumor metabolites. HPLC-PDAD-UV and HPLC-ESI-MS analyses of the EtOAc extracts of seven bioactive mutants and the ZBY-3 stain also supported the altered secondary metabolite production in these mutants (
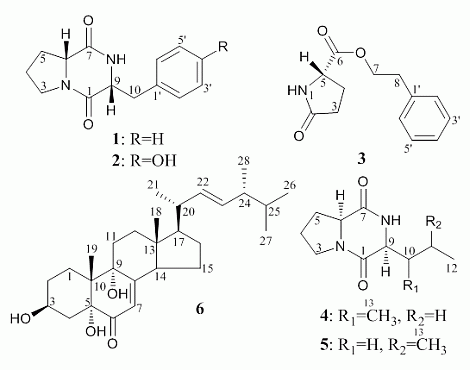
Figures S1 and S2 in the Supplementary Information). This was further evidenced by the following elucidation of six antitumor metabolites
1–
6 (
Figure 1) from one of the seven bioactive mutants, u2n2h3-3, confirming that all of them were newly produced by the mutant compared to the parent ZBY-3 strain (
Figures S3 and S4 in the Supplementary Information), which also resulted in the discovery of the new compound
3. All of
1–
6 showed inhibitory effects on the K562 cells and also on some of the other three human cancer cell lines tested, to varying extents (
Table 4 and
Table 5). These results indicated that the production of
1–
6 in mutant u2n2h3-3 was caused by the introduction of neomycin resistance, activating some metabolic pathways originally silent in parent ZBY-3 strain. It is of biogenetic interest that the production of both dd (
1 and
2) and ll (
4 and
5) diketopiperazines was activated in the mutant together with the new compound
3 and the known compound
6 by introduction of the acquired resistance to neomycin. An exploration of the regulatory mechanisms for their activated production requires further investigations into the affected pathways.
A literature survey showed that
3 was a new compound, and all of
1–
6 were also not yet found in the secondary metabolites of other
Aspergillus versicolor strains, although quite a number of metabolites, including many new compounds with novel skeletal structures, have been reported from
Aspergillus versicolor so far. Diketopiperazines consisting of
ll-amino acid residues, such as
4 and
5, are generally seen in the secondary metabolites of a wide range of common microbial species, but those derived from
dd- or
dl-amino acid residues were rarely reported from natural sources. To our knowledge, the present study is the first time that the isolation of cyclo(
d-Pro-
d-Phe) (
1) and cyclo(
d-Pro-
d-Tyr) (
2) from fungal metabolites has been reported, although several
dd- or
dl-diketopiperazines have so far been reported from marine bacteria [
50,
51,
63] and yeast [
64] or marine sponges [
65], including
1 from marine bacteria [
50]. Further, in the present study, this is also the first time that
2 has been isolated from a natural source, although a chemical preparation of
2 has been recorded in a patent application publication [
66].
Compound
1 was initially identified for antibacterial activity on
Vibrio anguillarum (MIC 0.03 µg/mL) [
50] and
2 for the plant disease controlling and plant growth promoting effects by inducing plant pathogen-resistant genes PR-1 and PDF1.2 as signal transducing molecules in plant [
66], but no other bioactivities were reported for both compounds. Compound
5 has been reported to show a weak inhibitory effect on the release of β-glucuronidase from rat polymorphonuclear leukocytes induced by platelet-activating factor, though the similar compound
4 did not show the same activity [
67]. At the same time, no significant cytotoxicity was recorded for both
4 and
5 on several human cancer cell lines, including the human cancer BGC-823 cell line, in the literature [
67]. The very weak inhibitory effect of
4 and
5 on the BGC-823 and the other three human cancer cell lines tested in the present bioassay (
Table 4) was consistent with the previous record on BCG-823 and the other four human cancer cell lines in the literature [
67]. On the other hand, the present bioassay results for
6 on the human cancer HeLa and the other three human cancer cell lines (
Table 4 and
Table 5) reconfirmed its cytotoxicity, initially identified in the rat hepatoma HTC cells [
68] and HeLa cells [
69] in the literature [
68,
69]. Besides, the present bioassay results for
1–
3 on the four tested human cancer cell lines were the first record of their cytotoxicity assay, although their effects were not so promising.
Both the above-mentioned bioassays and the chemical investigations in the present study demonstrated the effectiveness of the presented ultrasound-mediated approach to activate silent metabolite production by introducing acquired resistance to aminoglycoside antibiotics in fungi. The present results also showed the potential of this approach for discovering new compounds with antitumor activity from silenced fungal metabolic pathways. This approach could be applied to elicit the metabolic potential of other fungal isolates, to discover new compounds from cryptic secondary metabolites.



 +10.9 (c 0.14, MeOH), gave a pseudo-molecular ion peak at m/z 245 [M + H]+ in positive ESI-MS. The 1H and 13C NMR data of 1 in CDCl3 were identical with those of cyclo(d-Pro-d-Phe) [50] and cyclo(l-Pro-l-Phe) [51]; however, the positive sign of optical rotation in MeOH was identical with the sign of the former in EtOH [50], but not the negative sign of the latter in MeOH [51]. Thus, 1 was identified as cyclo(d-Pro-d-Phe). Compound 2,
+10.9 (c 0.14, MeOH), gave a pseudo-molecular ion peak at m/z 245 [M + H]+ in positive ESI-MS. The 1H and 13C NMR data of 1 in CDCl3 were identical with those of cyclo(d-Pro-d-Phe) [50] and cyclo(l-Pro-l-Phe) [51]; however, the positive sign of optical rotation in MeOH was identical with the sign of the former in EtOH [50], but not the negative sign of the latter in MeOH [51]. Thus, 1 was identified as cyclo(d-Pro-d-Phe). Compound 2,  +11.9 (c 0.32, MeOH), giving a pseudo-molecular ion peak at m/z 261 [M + H]+ in positive ESI-MS, showed 1H and 13C NMR signals identical with those of cyclo(l-Tyr-l-Pro) in CDCl3 [52], but the sign of the optical rotation in MeOH was opposite of the negative sign of cyclo(l-Tyr-l-Pro) in MeOH [52]. Thus, 2 was identified as cyclo(d-Tyr-d-Pro). Compounds 4,
+11.9 (c 0.32, MeOH), giving a pseudo-molecular ion peak at m/z 261 [M + H]+ in positive ESI-MS, showed 1H and 13C NMR signals identical with those of cyclo(l-Tyr-l-Pro) in CDCl3 [52], but the sign of the optical rotation in MeOH was opposite of the negative sign of cyclo(l-Tyr-l-Pro) in MeOH [52]. Thus, 2 was identified as cyclo(d-Tyr-d-Pro). Compounds 4,  −59.1 (c 0.46, MeOH), and 5,
−59.1 (c 0.46, MeOH), and 5,  −105.8 (c 0.77, MeOH), giving the same pseudo-molecular ion peak at m/z 211 [M + H]+ in positive ESI-MS, were identified as cyclo(l-Ile-l-Pro) [53] and cyclo(l-Leu-l-Pro) [54], respectively, by comparison of their physicochemical and spectroscopic data with those in the literature [53,54].
−105.8 (c 0.77, MeOH), giving the same pseudo-molecular ion peak at m/z 211 [M + H]+ in positive ESI-MS, were identified as cyclo(l-Ile-l-Pro) [53] and cyclo(l-Leu-l-Pro) [54], respectively, by comparison of their physicochemical and spectroscopic data with those in the literature [53,54]. −37 (c 0.22, CHCl3), was identified as 3β,5α,9α-trihydroxy-(22E,24R)-ergosta-7,22-dien-6-one on the basis of physicochemical and spectroscopic data [57].
−37 (c 0.22, CHCl3), was identified as 3β,5α,9α-trihydroxy-(22E,24R)-ergosta-7,22-dien-6-one on the basis of physicochemical and spectroscopic data [57]. −29.2 (c 0.21, MeOH), was assigned the molecular formula C13H15NO3 by HRESIMS (measured 234.1122 [M + H]+, calculated for C13H16NO3 [M + H]+ 234.1130). The 1H and 13C NMR spectra of 3 in CD3OD showed signals from a mono-alkylated benzene ring and several methylene/methine groups together with the two carbonyl carbon signals in the 13C NMR spectrum (Table 3). Analysis of the 1H-1H COSY, HMQC and HMBC spectra (Table 3) established the planar structure of phenethyl 5-oxoprolinate for 3. Then, the absolute configuration at C-5 in 3 was assigned to be S according to the negative sign of its optical rotation, by comparison with those of 5-oxo-l-proline ([α]D −11.2 (c 2.84, H2O) in the literature [58]) and methyl 5-oxo-l-prolinate ([α]D −5.6 (c 2.8, H2O) in the literature [58]). The 5S configuration of 3 was also determined further by the Marfey’s method [55], using d- and l-glutamic acid (Glu) as reference standards, by detecting the l-Glu (2S-Glu) in the hydrolysate of 3, as described above for 1, 2, 4 and 5, except for the hydrolysis of 3 with 6 N HCl at 110 °C for 12 h. Thus, the structure of 3 could be determined to be phenethyl 5-oxo-l-prolinate with 5S absolute configuration. A literature survey showed that although three patent application publications covered the same planar structure with common structures [59,60,61], only two of them [59,60] really dealt a little with the compound, and no more data were provided, except for the melting point in the literature [60] and the melting point, IR (1735 and 1680 cm−1) and 1H NMR data in the literature [59]. Although the literature [59] recorded the 1H NMR data for the compound that they prepared chemically, these data were quite different from the 1H NMR data of 3 (Table 3). Therefore, we report herein 3 as a new compound.
−29.2 (c 0.21, MeOH), was assigned the molecular formula C13H15NO3 by HRESIMS (measured 234.1122 [M + H]+, calculated for C13H16NO3 [M + H]+ 234.1130). The 1H and 13C NMR spectra of 3 in CD3OD showed signals from a mono-alkylated benzene ring and several methylene/methine groups together with the two carbonyl carbon signals in the 13C NMR spectrum (Table 3). Analysis of the 1H-1H COSY, HMQC and HMBC spectra (Table 3) established the planar structure of phenethyl 5-oxoprolinate for 3. Then, the absolute configuration at C-5 in 3 was assigned to be S according to the negative sign of its optical rotation, by comparison with those of 5-oxo-l-proline ([α]D −11.2 (c 2.84, H2O) in the literature [58]) and methyl 5-oxo-l-prolinate ([α]D −5.6 (c 2.8, H2O) in the literature [58]). The 5S configuration of 3 was also determined further by the Marfey’s method [55], using d- and l-glutamic acid (Glu) as reference standards, by detecting the l-Glu (2S-Glu) in the hydrolysate of 3, as described above for 1, 2, 4 and 5, except for the hydrolysis of 3 with 6 N HCl at 110 °C for 12 h. Thus, the structure of 3 could be determined to be phenethyl 5-oxo-l-prolinate with 5S absolute configuration. A literature survey showed that although three patent application publications covered the same planar structure with common structures [59,60,61], only two of them [59,60] really dealt a little with the compound, and no more data were provided, except for the melting point in the literature [60] and the melting point, IR (1735 and 1680 cm−1) and 1H NMR data in the literature [59]. Although the literature [59] recorded the 1H NMR data for the compound that they prepared chemically, these data were quite different from the 1H NMR data of 3 (Table 3). Therefore, we report herein 3 as a new compound. −29.2 (c 0.21, MeOH). Positive ion ESIMS m/z: 234 [M + H]+, 256 [M + Na]+, 272 [M + K]+. Positive HRESIMS m/z: measured 234.1122 [M + H]+, calculated for C13H16NO3 [M + H]+ 234.1130; measured 256.0943 [M + Na]+, calculated for C13H15NO3Na [M + Na]+ 256.0950; measured 272.0681 [M + K]+, calculated for C13H15NO3K [M + K]+ 272.0689; measured 489.1986 [2M + Na]+, calculated for C26H30N2O6Na [M + Na]+ 489.2002. 1H and 13C NMR: Table 3.
−29.2 (c 0.21, MeOH). Positive ion ESIMS m/z: 234 [M + H]+, 256 [M + Na]+, 272 [M + K]+. Positive HRESIMS m/z: measured 234.1122 [M + H]+, calculated for C13H16NO3 [M + H]+ 234.1130; measured 256.0943 [M + Na]+, calculated for C13H15NO3Na [M + Na]+ 256.0950; measured 272.0681 [M + K]+, calculated for C13H15NO3K [M + K]+ 272.0689; measured 489.1986 [2M + Na]+, calculated for C26H30N2O6Na [M + Na]+ 489.2002. 1H and 13C NMR: Table 3.