Biochemical Characteristics and Potential Biomedical Applications of Hydrolyzed Carrageenans
Abstract
:1. Introduction
2. Results and Discussion
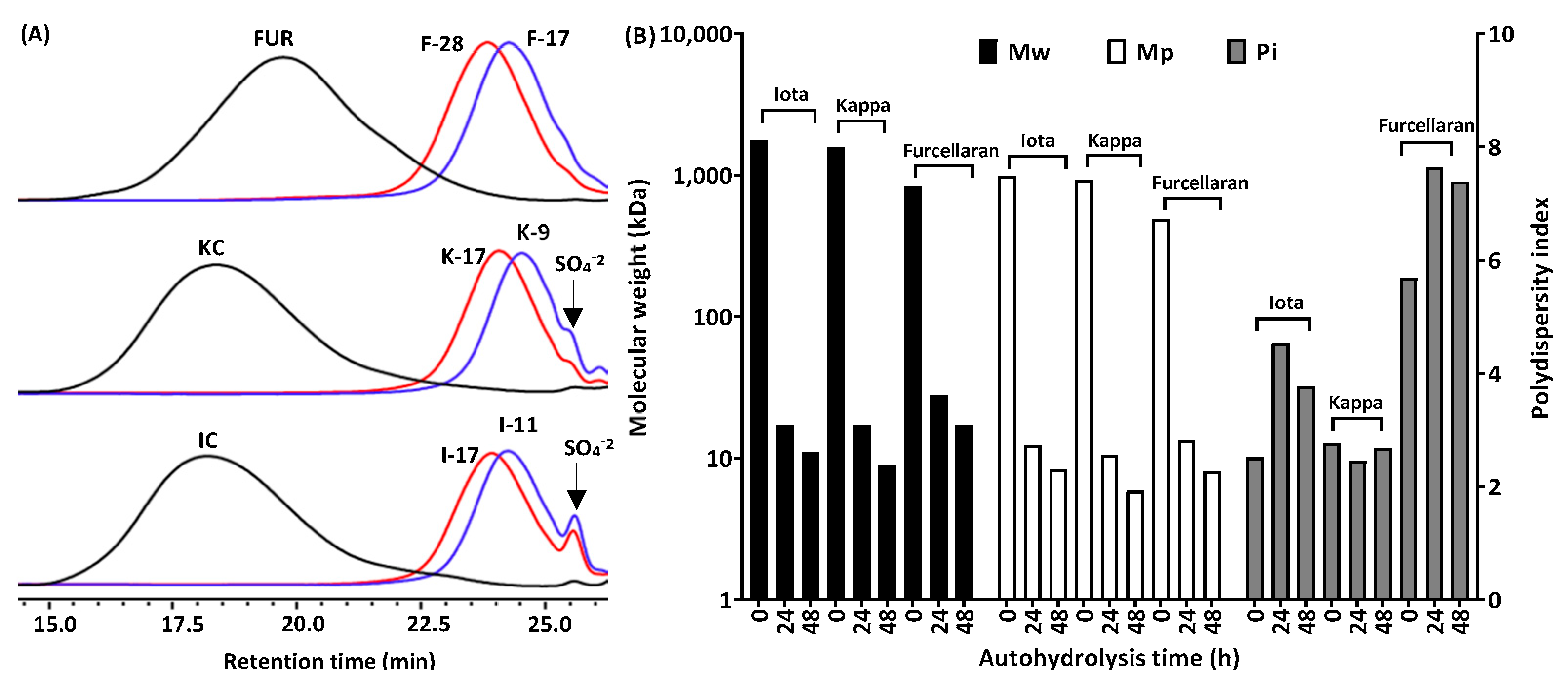
2.1. Molecular Weight Analysis, Sulfate Release and Chemical Composition
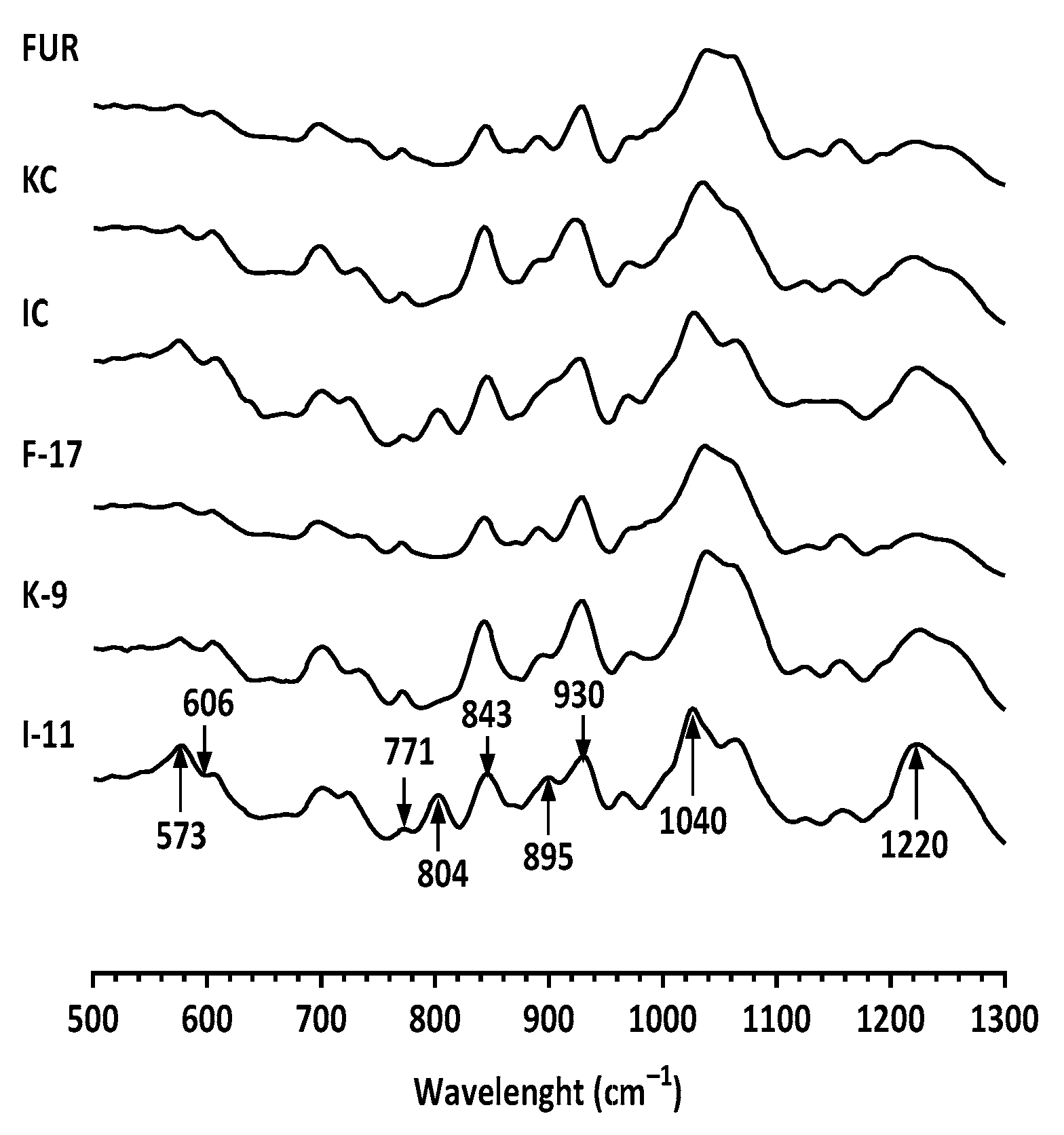
2.2. Vibrational Spectroscopy
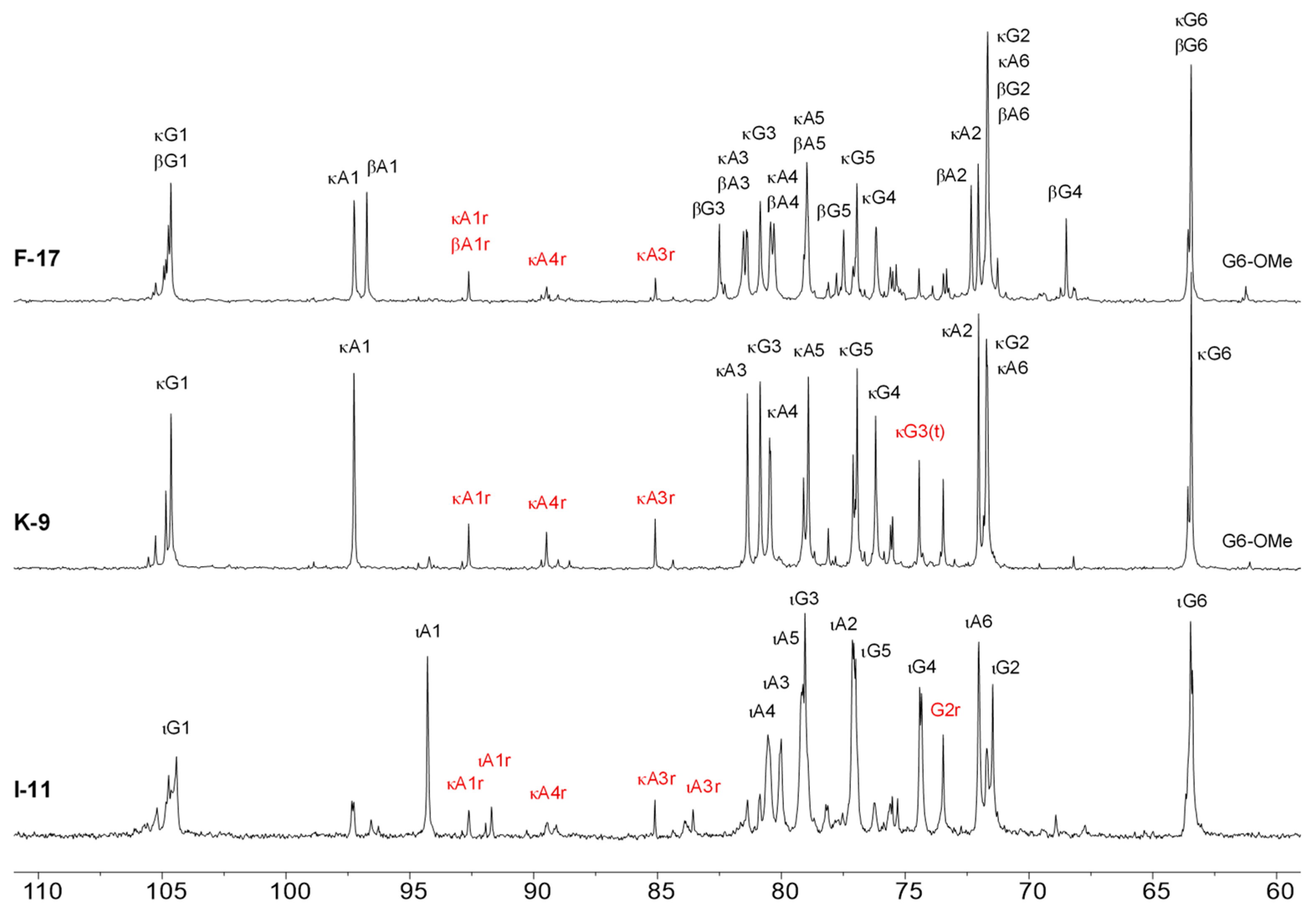
2.3. 1H-NMR Spectroscopy
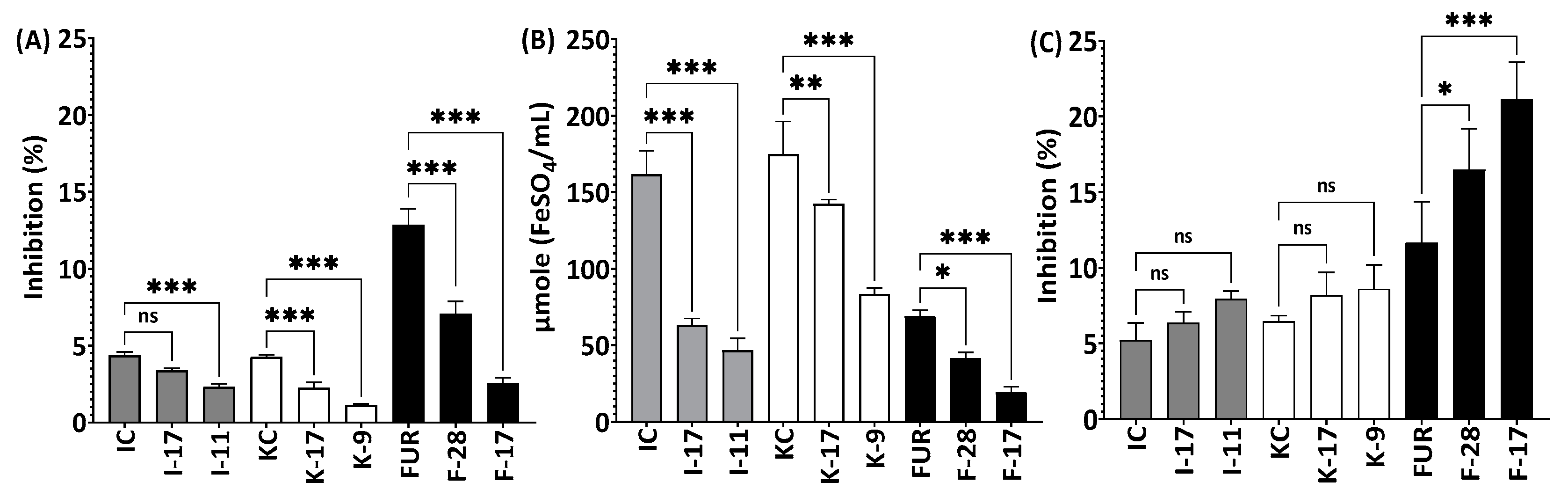
2.4. Antioxidant Activities
2.5. Anticoagulant Activity
2.6. Tyrosinase Inhibition Activity
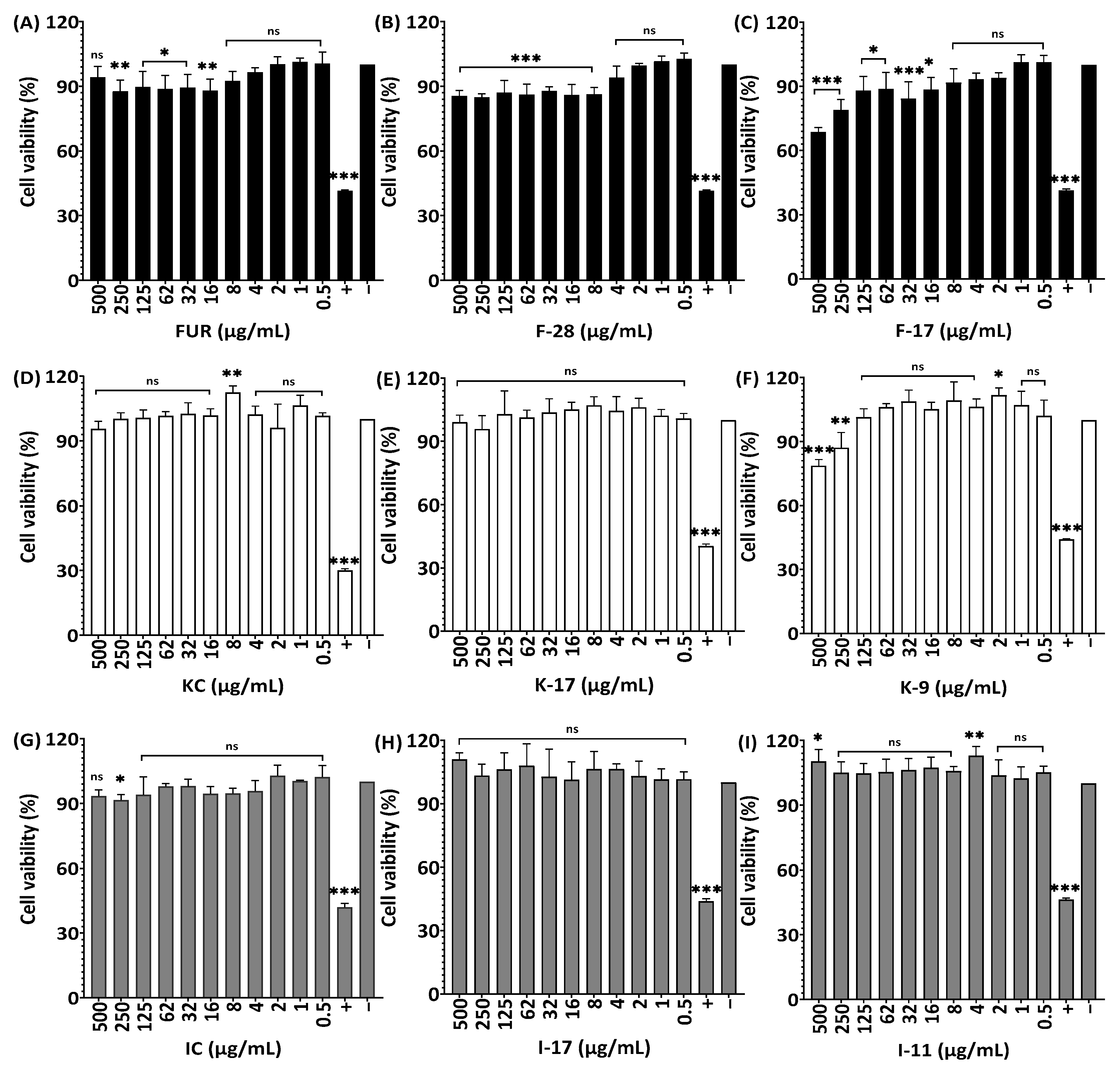
2.7. Cell viability of RAW264.7, HaCaT and HDF Cells
2.8. Effects on RAW264.7, HaCaT and HDF Cell Proliferation
2.9. Inhibition of Nitric Oxide Production
3. Materials and Methods
3.1. Materials
3.2. Sample Preparation
3.3. Estimation of Molecular Weight and Chemical Composition
3.4. Spectroscopic Methods
3.4.1. FTIR Spectroscopic Analysis
3.4.2. 1H-NMR Spectroscopic Analysis
3.5. Antioxidant Assays
3.5.1. Ferric-Reducing Antioxidant Power (FRAP) Assay
3.5.2. ABTS Radical Scavenging Capacity Assay
3.5.3. Hydroxyl Radical Scavenging Capacity Assay
3.6. Anticoagulant Activity
3.7. Mushroom Tyrosinase Inhibition Assay
3.8. Cell Experiments
3.8.1. Cell Culture
3.8.2. Cellular Viability Test
3.8.3. Cell Proliferation Assay
3.8.4. Measurement of Nitric Oxide Production
3.9. Statistical Analysis
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Jiao, G.; Yu, G.; Zhang, J.; Ewart, H.S. Chemical structures and bioactivities of sulfated polysaccharides from marine algae. Mar. Drugs 2011, 9, 196–223. [Google Scholar] [CrossRef] [PubMed]
- Cunha, L.; Grenha, A. Sulfated seaweed polysaccharides as multifunctional materials in drug delivery applications. Mar. Drugs 2016, 14, 42. [Google Scholar] [CrossRef] [PubMed]
- Du, L.; Brenner, T.; Xie, J.; Matsukawa, S. A study on phase separation behavior in kappa/iota carrageenan mixtures by micro DSC, rheological measurements and simulating water and cations migration between phases. Food Hydrocoll. 2016, 55, 81–88. [Google Scholar] [CrossRef]
- BeMiller, J. 5-Polysaccharides: Properties Carbohydrate Chemistry for Food, 2nd ed.; Scientists; Elsevier Inc.: Amsterdam, The Netherlands, 2019. [Google Scholar]
- Estevez, J.M.; Ciancia, M.; Cerezo, A.S. The system of low-molecular-weight carrageenans and agaroids from the room-temperature-extracted fraction of Kappaphycus alvarezii. Carbohydr. Res. 2000, 325, 287–299. [Google Scholar] [CrossRef] [PubMed]
- Laos, K.; Ring, S.G. Note: Characterisation of furcellaran samples from Estonian Furcellaria lumbricalis (Rhodophyta). J. Appl. Phycol. 2005, 17, 461–464. [Google Scholar] [CrossRef]
- van de Velde, F.; Peppelman, H.A.; Rollema, H.S.; Tromp, R.H. On the structure of κ/ι-hybrid carrageenans. Carbohydr. Res. 2001, 331, 271–283. [Google Scholar] [CrossRef]
- McKim, J.M. Food additive carrageenan: Part I: A critical review of carrageenan in vitro studies, potential pitfalls, and implications for human health and safety. Crit. Rev. Toxicol. 2014, 44, 211–243. [Google Scholar] [CrossRef]
- Lomartire, S.; Marques, J.C.; Gonçalves, A.M. An overview to the health benefits of seaweeds consumption. Mar. Drugs 2021, 19, 341. [Google Scholar] [CrossRef]
- de Jesus Raposo, M.F.; De Morais, A.M.B.; De Morais, R.M.S.C. Marine polysaccharides from algae with potential biomedical applications. Mar. Drugs 2015, 13, 2967–3028. [Google Scholar] [CrossRef]
- Fleita, D.; El-Sayed, M.; Rifaat, D. Evaluation of the antioxidant activity of enzymatically-hydrolyzed sulfated polysaccharides extracted from red algae; Pterocladia capillacea. LWT-Food Sci. Technol. 2015, 63, 1236–1244. [Google Scholar] [CrossRef]
- Hamed, I.; Özogul, F.; Özogul, Y.; Regenstein, J.M. Marine bioactive compounds and their health benefits: A review. Compr. Rev. Food Sci. Food Saf. 2015, 14, 446–465. [Google Scholar] [CrossRef]
- Xu, S.-Y.; Huang, X.; Cheong, K.-L. Recent advances in marine algae polysaccharides: Isolation, structure, and activities. Mar. Drugs 2017, 15, 388. [Google Scholar] [CrossRef] [PubMed]
- Priyan Shanura Fernando, I.; Kim, K.-N.; Kim, D.; Jeon, Y.-J. Algal polysaccharides: Potential bioactive substances for cosmeceutical applications. Crit. Rev. Biotechnol. 2019, 39, 99–113. [Google Scholar] [CrossRef]
- Hu, D.; Cheong, K.; Zhao, J.; Li, S. Chromatography in characterization of polysaccharides from medicinal plants and fungi. J. Sep. Sci. 2013, 36, 1–19. [Google Scholar] [CrossRef]
- Fidelis, G.P.; Camara, R.B.G.; Queiroz, M.F.; Costa, M.S.S.P.; Santos, P.C.; Rocha, H.A.O.; Costa, L.S. Proteolysis, NaOH and ultrasound-enhanced extraction of anticoagulant and antioxidant sulfated polysaccharides from the edible seaweed, Gracilaria birdiae. Molecules 2014, 19, 18511–18526. [Google Scholar] [CrossRef] [PubMed]
- Abad, L.V.; Aurigue, F.B.; Montefalcon, D.R.V.; Manguiat, P.H.; Carandang, F.F.; Mabborang, S.A.; Hizon, M.G.S.; Abella, M.E.S. Effect of radiation-modified kappa-carrageenan as plant growth promoter on peanut (Arachis hypogaea L.). Radiat. Phys. Chem. 2018, 153, 239–244. [Google Scholar] [CrossRef]
- Leiter, A.; Mailänder, J.; Wefers, D.; Bunzel, M.; Gaukel, V. Influence of acid hydrolysis and dialysis of κ-carrageenan on its ice recrystallization inhibition activity. J. Food Eng. 2017, 209, 26–35. [Google Scholar] [CrossRef]
- Muzyed, S.; Howlader, M.M.; Tuvikene, R. Fermentation optimization, purification and biochemical characterization of ι-carrageenase from marine bacterium Cellulophaga baltica. Int. J. Biol. Macromol. 2021, 166, 789–797. [Google Scholar] [CrossRef]
- Saluri, K.; Tuvikene, R. Anticoagulant and antioxidant activity of lambda-and theta-carrageenans of different molecular weights. Bioact. Carbohydr. Diet. Fibre 2020, 24, 100243. [Google Scholar] [CrossRef]
- Iranmahboob, J.; Nadim, F.; Monemi, S. Optimizing acid-hydrolysis: A critical step for production of ethanol from mixed wood chips. Biomass Bioenergy 2002, 22, 401–404. [Google Scholar] [CrossRef]
- Kang, E.-k.; Lee, B.M.; Hwang, H.A.; Kim, J.-H. Analysis of mono-sugars obtained by acid hydrolysis of algae-based polysaccharides. J. Ind. Eng. Chem. 2012, 18, 1366–1369. [Google Scholar] [CrossRef]
- de Moura, F.A.; Macagnan, F.T.; da Silva, L.P. Oligosaccharide production by hydrolysis of polysaccharides: A review. Int. J. Food Sci. Technol. 2015, 50, 275–281. [Google Scholar] [CrossRef]
- Vázquez, M.; Alonso, J.; Domínguez, H.; Parajó, J. Enhancing the potential of oligosaccharides from corncob autohydrolysis as prebiotic food ingredients. Ind. Crops Prod. 2006, 24, 152–159. [Google Scholar] [CrossRef]
- Martínez, M.; Gullón, B.; Yáñez, R.; Alonso, J.L.; Parajó, J.C. Kinetic assessment on the autohydrolysis of pectin-rich by-products. Chem. Eng. J. 2010, 162, 480–486. [Google Scholar] [CrossRef]
- Athukorala, Y.; Jung, W.-K.; Vasanthan, T.; Jeon, Y.-J. An anticoagulative polysaccharide from an enzymatic hydrolysate of Ecklonia cava. Carbohydr. Polym. 2006, 66, 184–191. [Google Scholar] [CrossRef]
- Huang, L.; Shen, M.; Morris, G.A.; Xie, J. Sulfated polysaccharides: Immunomodulation and signaling mechanisms. Trends Food Sci. Technol. 2019, 92, 1–11. [Google Scholar] [CrossRef]
- Liu, H.-L.; Kao, T.-H.; Shiau, C.-Y.; Chen, B.-H. Functional components in Scutellaria barbata D. Don with anti-inflammatory activity on RAW264.7 cells. J. Food Drug Anal. 2018, 26, 31–40. [Google Scholar] [CrossRef]
- Xi, Y.; Ge, J.; Wang, M.; Chen, M.; Niu, W.; Cheng, W.; Xue, Y.; Lin, C.; Lei, B. Bioactive anti-inflammatory, antibacterial, antioxidative silicon-based nanofibrous dressing enables cutaneous tumor photothermo-chemo therapy and infection-induced wound healing. ACS Nano 2020, 14, 2904–2916. [Google Scholar] [CrossRef]
- Sanjeewa, K.K.A.; Fernando, I.P.S.; Kim, E.-A.; Ahn, G.; Jee, Y.; Jeon, Y.-J. Anti-inflammatory activity of a sulfated polysaccharide isolated from an enzymatic digest of brown seaweed Sargassum horneri in RAW264.7 cells. Nutr. Res. Pract. 2017, 11, 3–10. [Google Scholar] [CrossRef]
- Tuvikene, R.; Robal, M.; Fujita, D.; Saluri, K.; Truus, K.; Tashiro, Y.; Ogawa, H.; Matsukawa, S. Funorans from Gloiopeltis species. Part I. Extraction and structural characteristics. Food Hydrocoll. 2015, 43, 481–492. [Google Scholar] [CrossRef]
- Gómez-Ordóñez, E.; Rupérez, P. FTIR-ATR spectroscopy as a tool for polysaccharide identification in edible brown and red seaweeds. Food Hydrocoll. 2011, 25, 1514–1520. [Google Scholar] [CrossRef]
- Tuvikene, R.; Truus, K.; Robal, M.; Pehk, T.; Kailas, T.; Vaher, M.; Paalme, T. Structure and thermal stability of pyruvated carrageenans from the red alga Coccotylus truncatus. Carbohydr. Res. 2009, 344, 788–794. [Google Scholar] [CrossRef] [PubMed]
- Freile-Pelegrín, Y.; Robledo, D. Carrageenan of eucheuma isiforme (Solieriaceae, rhodophyta) from Nicaragua. J. Appl. Phycol. 2008, 20, 537–541. [Google Scholar] [CrossRef]
- Pereira, L.; Sousa, A.; Coelho, H.; Amado, A.M.; Ribeiro-Claro, P.J. Use of FTIR, FT-Raman and 13C-NMR spectroscopy for identification of some seaweed phycocolloids. Biomol. Eng. 2003, 20, 223–228. [Google Scholar] [CrossRef] [PubMed]
- Sekkal, M.; Legrand, P. A spectroscopic investigation of the carrageenans and agar in the 1500–100 cm−1 spectral range. Spectrochim. Acta Part A Mol. Spectrosc. 1993, 49, 209–221. [Google Scholar] [CrossRef]
- Calvo, G.H.; Cosenza, V.A.; Sáenz, D.A.; Navarro, D.A.; Stortz, C.A.; Céspedes, M.A.; Mamone, L.A.; Casas, A.G.; Di Venosa, G.M. Disaccharides obtained from carrageenans as potential antitumor agents. Sci. Rep. 2019, 9, 6654. [Google Scholar] [CrossRef]
- Huang, L.; Huang, M.; Shen, M.; Wen, P.; Wu, T.; Hong, Y.; Yu, Q.; Chen, Y.; Xie, J. Sulfated modification enhanced the antioxidant activity of Mesona chinensis Benth polysaccharide and its protective effect on cellular oxidative stress. Int. J. Biol. Macromol. 2019, 136, 1000–1006. [Google Scholar] [CrossRef]
- Yang, Y.; Liu, D.; Wu, J.; Chen, Y.; Wang, S. In vitro antioxidant activities of sulfated polysaccharide fractions extracted from Corallina officinalis. Int. J. Biol. Macromol. 2011, 49, 1031–1037. [Google Scholar] [CrossRef]
- Hu, T.; Liu, D.; Chen, Y.; Wu, J.; Wang, S. Antioxidant activity of sulfated polysaccharide fractions extracted from Undaria pinnitafida in vitro. Int. J. Biol. Macromol. 2010, 46, 193–198. [Google Scholar] [CrossRef]
- Qi, H.; Zhao, T.; Zhang, Q.; Li, Z.; Zhao, Z.; Xing, R. Antioxidant activity of different molecular weight sulfated polysaccharides from Ulva pertusa Kjellm (Chlorophyta). J. Appl. Phycol. 2005, 17, 527–534. [Google Scholar] [CrossRef]
- Rodriguez-Jasso, R.M.; Mussatto, S.I.; Pastrana, L.; Aguilar, C.N.; Teixeira, J.A. Microwave-assisted extraction of sulfated polysaccharides (fucoidan) from brown seaweed. Carbohydr. Polym. 2011, 86, 1137–1144. [Google Scholar] [CrossRef]
- Alban, S.; Schauerte, A.; Franz, G. Anticoagulant sulfated polysaccharides: Part I. Synthesis and structure–activity relationships of new pullulan sulfates. Carbohydr. Polym. 2002, 47, 267–276. [Google Scholar] [CrossRef]
- Hou, N.; Zhang, M.; Xu, Y.; Sun, Z.; Wang, J.; Zhang, L.; Zhang, Q. Polysaccharides and their depolymerized fragments from Costaria costata: Molecular weight and sulfation-dependent anticoagulant and FGF/FGFR signal activating activities. Int. J. Biol. Macromol. 2017, 105, 1511–1518. [Google Scholar] [CrossRef]
- Li, H.; Mao, W.; Hou, Y.; Gao, Y.; Qi, X.; Zhao, C.; Chen, Y.; Chen, Y.; Li, N.; Wang, C. Preparation, structure and anticoagulant activity of a low molecular weight fraction produced by mild acid hydrolysis of sulfated rhamnan from Monostroma latissimum. Bioresour. Technol. 2012, 114, 414–418. [Google Scholar] [CrossRef]
- Liang, W.; Mao, X.; Peng, X.; Tang, S. Effects of sulfate group in red seaweed polysaccharides on anticoagulant activity and cytotoxicity. Carbohydr. Polym. 2014, 101, 776–785. [Google Scholar] [CrossRef]
- Yang, J.; Liu, W.; Yi, M.; Song, S.; Ai, C.; Zhao, J. Depolymerization of heparin by dielectric barrier discharge: Effect of operating modes and anticoagulant potential analysis of low-molecular-weight products. Chem. Phys. Lett. 2018, 708, 194–200. [Google Scholar] [CrossRef]
- Haroun-Bouhedja, F.; Ellouali, M.; Sinquin, C.; Boisson-Vidal, C. Relationship between sulfate groups and biological activities of fucans. Thromb. Res. 2000, 100, 453–459. [Google Scholar] [CrossRef]
- Pereira, M.G.; Benevides, N.M.; Melo, M.R.; Valente, A.P.; Melo, F.R.; Mourão, P.A. Structure and anticoagulant activity of a sulfated galactan from the red alga, Gelidium crinale. Is there a specific structural requirement for the anticoagulant action? Carbohydr. Res. 2005, 340, 2015–2023. [Google Scholar] [CrossRef] [PubMed]
- Olivares, C.; Solano, F. New insights into the active site structure and catalytic mechanism of tyrosinase and its related proteins. Pigment. Cell Melanoma Res. 2009, 22, 750–760. [Google Scholar]
- Huang, K.-F.; Chen, Y.-W.; Chang, C.-T.; Chou, S.-T. Studies on the inhibitory effect of Graptopetalum paraguayense E. Walther extracts on mushroom tyrosinase. Food Chem. 2005, 89, 583–587. [Google Scholar] [CrossRef]
- Park, E.-J.; Choi, J.-I. Melanogenesis inhibitory effect of low molecular weight fucoidan from Undaria pinnatifida. J. Appl. Phycol. 2017, 29, 2213–2217. [Google Scholar] [CrossRef]
- Chen, B.-J.; Shi, M.-J.; Cui, S.; Hao, S.-X.; Hider, R.C.; Zhou, T. Improved antioxidant and anti-tyrosinase activity of polysaccharide from Sargassum fusiforme by degradation. Int. J. Biol. Macromol. 2016, 92, 715–722. [Google Scholar] [CrossRef]
- Kubo, I.; Chen, Q.-X.; Nihei, K.-i. Molecular design of antibrowning agents: Antioxidative tyrosinase inhibitors. Food Chem. 2003, 81, 241–247. [Google Scholar] [CrossRef]
- Yang, B.; Zhao, M.; Jiang, Y. Optimization of tyrosinase inhibition activity of ultrasonic-extracted polysaccharides from longan fruit pericarp. Food Chem. 2008, 110, 294–300. [Google Scholar] [CrossRef]
- Zang, J.; Chen, Z.; Ye, S.; Si, Y.; Li, C. Study on inhibition effect of Sargassum fusiforme polysaccharide extracts on activity of tyrosinase. Sci. Technol. Food Indust. 2012, 20, 27. [Google Scholar]
- Wang, L.; Cui, Y.R.; Yang, H.-W.; Lee, H.G.; Ko, J.-Y.; Jeon, Y.-J. A mixture of seaweed extracts and glycosaminoglycans from sea squirts inhibits α-MSH-induced melanogenesis in B16F10 melanoma cells. Fish. Aquat. Sci. 2019, 22, 11. [Google Scholar] [CrossRef]
- Hendriks, J.; Willem Visser, C.; Henke, S.; Leijten, J.; Saris, D.B.; Sun, C.; Lohse, D.; Karperien, M. Optimizing cell viability in droplet-based cell deposition. Sci. Rep. 2015, 5, 1–10. [Google Scholar] [CrossRef]
- Bielefeld, K.A.; Amini-Nik, S.; Alman, B.A. Cutaneous wound healing: Recruiting developmental pathways for regeneration. Cell. Mol. Life Sci. 2013, 70, 2059–2081. [Google Scholar] [CrossRef]
- Athukorala, Y.; Kim, K.-N.; Jeon, Y.-J. Antiproliferative and antioxidant properties of an enzymatic hydrolysate from brown alga, Ecklonia cava. Food Chem. Toxicol. 2006, 44, 1065–1074. [Google Scholar] [CrossRef]
- Wang, C.; Levis, G.B.S.; Lee, E.B.; Levis, W.R.; Lee, D.W.; Kim, B.S.; Park, S.Y.; Park, E. Platycodin D and D3 isolated from the root of Platycodon grandiflorum modulate the production of nitric oxide and secretion of TNF-α in activated RAW264.7 cells. Int. Immunopharmacol. 2004, 4, 1039–1049. [Google Scholar] [CrossRef]
- Ravipati, A.S.; Zhang, L.; Koyyalamudi, S.R.; Jeong, S.C.; Reddy, N.; Bartlett, J.; Smith, P.T.; Shanmugam, K.; Münch, G.; Wu, M.J. Antioxidant and anti-inflammatory activities of selected Chinese medicinal plants and their relation with antioxidant content. BMC Complement. Altern. Med. 2012, 12, 173. [Google Scholar] [CrossRef] [PubMed]
- Diaz, P.; Jeong, S.C.; Lee, S.; Khoo, C.; Koyyalamudi, S.R. Antioxidant and anti-inflammatory activities of selected medicinal plants and fungi containing phenolic and flavonoid compounds. Chin. Med. 2012, 7, 26. [Google Scholar] [CrossRef] [PubMed]
- Jiang, F.; Ding, Y.; Tian, Y.; Yang, R.; Quan, M.; Tong, Z.; Zhang, X.; Luo, D.; Chi, Z.; Liu, C. Hydrolyzed low-molecular-weight polysaccharide from Enteromorpha prolifera exhibits high anti-inflammatory activity and promotes wound healing. Biomater. Adv. 2022, 133, 112637. [Google Scholar] [CrossRef] [PubMed]
- Cao, R.-A.; Lee, S.-H.; You, S. Structural effects of sulfated-glycoproteins from Stichopus japonicus on the nitric oxide secretion ability of RAW264.7 cells. Prev. Nutr. Food Sci. 2014, 19, 307. [Google Scholar] [CrossRef]
- Wijesinghe, W.; Jeon, Y.-J. Biological activities and potential industrial applications of fucose rich sulfated polysaccharides and fucoidans isolated from brown seaweeds: A review. Carbohydr. Polym. 2012, 88, 13–20. [Google Scholar] [CrossRef]
- Park, H.Y.; Han, M.H.; Park, C.; Jin, C.-Y.; Kim, G.-Y.; Choi, I.-W.; Kim, N.D.; Nam, T.-J.; Kwon, T.K.; Choi, Y.H. Anti-inflammatory effects of fucoidan through inhibition of NF-κB, MAPK and Akt activation in lipopolysaccharide-induced BV2 microglia cells. Food Chem. Toxicol. 2011, 49, 1745–1752. [Google Scholar] [CrossRef]
- Wu, G.-J.; Shiu, S.-M.; Hsieh, M.-C.; Tsai, G.-J. Anti-inflammatory activity of a sulfated polysaccharide from the brown alga Sargassum cristaefolium. Food Hydrocoll. 2016, 53, 16–23. [Google Scholar] [CrossRef]
- Gao, J.; Li, Q.; Liu, Y.; Yang, B.; Sadiqb, F.A.; Li, X.; Mi, S.; Sang, Y. Immunoregulatory effect of Lactobacillus paracasei VL8 exopolysaccharide on RAW264. 7 cells by NF-κB and MAPK pathways. J. Funct. Foods 2022, 95, 105166. [Google Scholar] [CrossRef]
- Cui, M.; Wu, J.; Wang, S.; Shu, H.; Zhang, M.; Liu, K.; Liu, K. Characterization and anti-inflammatory effects of sulfated polysaccharide from the red seaweed Gelidium pacificum Okamura. Int. J. Biol. Macromol. 2019, 129, 377–385. [Google Scholar] [CrossRef]
- Posadas, I.; Terencio, M.C.; Guillén, I.; Ferrándiz, M.L.; Coloma, J.; Payá, M.; Alcaraz, M.J. Co-regulation between cyclo-oxygenase-2 and inducible nitric oxide synthase expression in the time-course of murine inflammation. Naunyn-Schmiedeberg’s Arch. Pharmacol. 2000, 361, 98–106. [Google Scholar] [CrossRef]
- Attur, M.; Patel, R.; Thakker, G.; Vyas, P.; Levartovsky, D.; Patel, P.; Naqvi, S.; Raza, R.; Patel, K.; Abramson, D. Differential anti-inflammatory effects of immunosuppressive drugs: Cyclosporin, rapamycin and FK-506 on inducible nitric oxide synthase, nitric oxide, cyclooxygenase-2 and PGE 2 production. Inflamm. Res. 2000, 49, 20–26. [Google Scholar] [CrossRef] [PubMed]
- Dodgson, K.; Price, R. A note on the determination of the ester sulphate content of sulphated polysaccharides. Biochem. J. 1962, 84, 106. [Google Scholar] [CrossRef]
- Dubois, M.; Gilles, K.; Hamilton, J.; Rebers, P.; Smith, F. A colorimetric method for the determination of sugars. Nature 1951, 168, 167. [Google Scholar] [CrossRef] [PubMed]
- Robal, M.; Brenner, T.; Matsukawa, S.; Ogawa, H.; Truus, K.; Rudolph, B.; Tuvikene, R. Monocationic salts of carrageenans: Preparation and physico-chemical properties. Food Hydrocoll. 2017, 63, 656–667. [Google Scholar] [CrossRef]
- Benzie, I.F.; Strain, J.J. The ferric reducing ability of plasma (FRAP) as a measure of “antioxidant power”: The FRAP assay. Anal. Biochem. 1996, 239, 70–76. [Google Scholar] [CrossRef]
- Wang, Z.; Liu, X.; Bao, Y.; Wang, X.; Zhai, J.; Zhan, X.; Zhang, H. Characterization and anti-inflammation of a polysaccharide produced by Chaetomium globosum CGMCC 6882 on LPS-induced RAW264.7 cells. Carbohydr. Polym. 2021, 251, 117129. [Google Scholar] [CrossRef]
- Chan, Y.; Kim, K.; Cheah, S. Inhibitory effects of Sargassum polycystum on tyrosinase activity and melanin formation in B16F10 murine melanoma cells. J. Ethnopharmacol. 2011, 137, 1183–1188. [Google Scholar] [CrossRef]








| Sample | Measured Values | Theoretical Values * | |||
|---|---|---|---|---|---|
| Total Sugar (%) | Sulfate (%) | Total Sugar (%) | Sulfate (%) | Sodium (%) | |
| FUR | 72.9 ± 1.1 | 21 ± 2.2 | 85.4 ** | 11.8 ** | 2.8 ** |
| KC | 67.7 ± 3.0 | 26 ± 1.6 | 70.9 | 23.5 | 5.6 |
| IC | 48.1 ± 3.5 | 37 ± 3.0 | 53.4 | 37.6 | 9.0 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Humayun, S.; Premarathna, A.D.; Rjabovs, V.; Howlader, M.M.; Darko, C.N.S.; Mok, I.-K.; Tuvikene, R. Biochemical Characteristics and Potential Biomedical Applications of Hydrolyzed Carrageenans. Mar. Drugs 2023, 21, 269. https://doi.org/10.3390/md21050269
Humayun S, Premarathna AD, Rjabovs V, Howlader MM, Darko CNS, Mok I-K, Tuvikene R. Biochemical Characteristics and Potential Biomedical Applications of Hydrolyzed Carrageenans. Marine Drugs. 2023; 21(5):269. https://doi.org/10.3390/md21050269
Chicago/Turabian StyleHumayun, Sanjida, Amal D. Premarathna, Vitalijs Rjabovs, Md Musa Howlader, Clarisa Naa Shormeh Darko, Il-Kyoon Mok, and Rando Tuvikene. 2023. "Biochemical Characteristics and Potential Biomedical Applications of Hydrolyzed Carrageenans" Marine Drugs 21, no. 5: 269. https://doi.org/10.3390/md21050269
APA StyleHumayun, S., Premarathna, A. D., Rjabovs, V., Howlader, M. M., Darko, C. N. S., Mok, I. -K., & Tuvikene, R. (2023). Biochemical Characteristics and Potential Biomedical Applications of Hydrolyzed Carrageenans. Marine Drugs, 21(5), 269. https://doi.org/10.3390/md21050269









