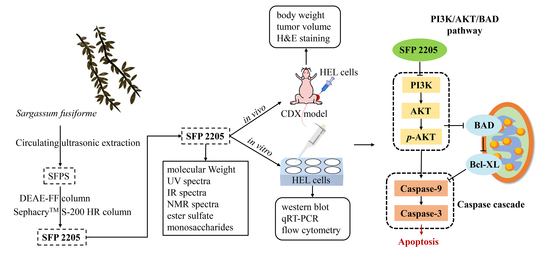
Anti-Leukemia Activity of Polysaccharide from Sargassum fusiforme via the PI3K/AKT/BAD Pathway In Vivo and In Vitro
Abstract
:1. Introduction
2. Results
2.1. Molecular Weight of SFP 2205
2.2. UV and Infrared Spectroscopic Analysis of SFP 2205
2.3. SFP 2205 Monosaccharide Composition and Sulfate Group Content
2.4. NMR Analysis of SFP 2205
2.5. Effect of SFP 2205 Treatment on Tumor Volume and Weight
2.6. Effects of SFP 2205 on Main Organ and Tumor Morphology in HEL Tumor-Bearing Mice
2.7. Effects of SFP 2205 on Expression of Apoptotic Pathway-Related Genes and Proteins
2.8. SFP 2205 Regulated Activity of the PI3K/AKT-Mediated Signaling Pathway in HEL Cells
2.9. PI3K/AKT/Bad/Bcl-xL Axis Contributed to SFP 2205-Induced Apoptosis
3. Discussion
4. Materials and Methods
4.1. Preparation and Purification of S. fusiforme Polysaccharide
4.2. SFP 2205 Molecular Weight
4.3. Ultraviolet (UV) and Infrared (IR) Analysis of SFP 2205
4.4. Preliminary Characterization of SFP 2205
4.5. Nuclear Magnetic Resonance (NMR) Spectroscopy
4.6. Animal Handling and Establishing the HEL-Bearing Mouse Model
4.7. Paraffin Sectioning and Hematoxylin and Eosin (H&E) Staining
4.8. Cell Lines and Cultures
4.9. Analysis of Apoptosis
4.10. Cell Cycle Analysis via Flow Cytometry
4.11. Quantitative Real-Time-PCR (qRT-PCR) for Gene Expression
4.12. Western Blot Assays
4.13. Statistical Analysis
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Bray, F.; Laversanne, M.; Weiderpass, E.; Soerjomataram, I. The ever-increasing importance of cancer as a leading cause of premature death worldwide. Cancer 2021, 127, 3029–3030. [Google Scholar] [CrossRef] [PubMed]
- Xia, C.F.; Dong, X.S.; Li, H.; Cao, M.M.; Sun, D.Q.; He, S.Y.; Yang, F.; Yan, X.X.; Zhang, S.L.; Li, N.; et al. Cancer statistics in China and United States, 2022: Profiles, trends, and determinants. Chin. Med. J. 2022, 135, 584–590. [Google Scholar] [CrossRef] [PubMed]
- Khatoon, E.; Banik, K.; Harsha, C.; Sailo, B.L.; Thakur, K.K.; Khwairakpam, A.D.; Vikkurthi, R.; Devi, T.B.; Gupta, S.C.; Kunnumakkara, A.B. Phytochemicals in cancer cell chemosensitization: Current knowledge and future perspectives. Semin. Cancer Biol. 2022, 80, 306–339. [Google Scholar] [CrossRef] [PubMed]
- Siegel, R.L.; Miller, K.D.; Fuchs, H.E.; Jemal, A. Cancer statistics, 2022. CA Cancer J. Clin. 2022, 72, 7–33. [Google Scholar] [CrossRef] [PubMed]
- Birsen, R.; Larrue, C.; Decroocq, J.; Johnson, N.; Guiraud, N.; Gotanegre, M.; Cantero Aguilar, L.; Grignano, E.; Huynh, T.; Fontenay, M.; et al. APR-246 induces early cell death by ferroptosis in acute myeloid leukemia. Haematologica 2022, 107, 403–416. [Google Scholar] [CrossRef] [PubMed]
- Melenhorst, J.J.; Chen, G.M.; Wang, M.; Porter, D.L.; Chen, C.Y.; Collins, M.A.; Gao, P.; Bandyopadhyay, S.; Sun, H.X.; Zhao, Z.R.; et al. Decade-long leukaemia remissions with persistence of CD4+ CAR T cells. Nature 2022, 602, 503–509. [Google Scholar] [CrossRef]
- Chen, H.L.; Zhang, L.; Long, X.G.; Li, P.F.; Chen, S.C.; Kuang, W.; Guo, J.M. Sargassum fusiforme polysaccharides inhibit VEGF-A-related angiogenesis and proliferation of lung cancer in vitro and in vivo. Biomed. Pharmacother. 2017, 85, 22–27. [Google Scholar] [CrossRef]
- Zuo, Y.; Zhang, C.Z.; Ren, Q.; Chen, Y.; Li, X.; Yang, J.R.; Li, H.R.; Tang, W.T.; Ho, H.M.; Sun, C.; et al. Activation of mitochondrial-associated apoptosis signaling pathway and inhibition of PI3K/AKT/mTOR signaling pathway by voacamine suppress breast cancer progression. Phytomedicine 2022, 99, 154015. [Google Scholar] [CrossRef]
- Cermeño, M.; Bascón, C.; Amigo Benavent, M.; Felix, M.; FitzGerald, R.J. Identification of peptides from edible silkworm pupae (Bombyx mori) protein hydrolysates with antioxidant activity. J. Funct. Foods 2022, 92, 105052. [Google Scholar] [CrossRef]
- Chen, S.T.; Wang, M.; Veeraperumal, S.; Teng, B.; Li, R.; Qian, Z.M.; Chen, J.P.; Zhong, S.Y.; Cheong, K.L. Antioxidative and protective effect of Morchella esculenta against dextran sulfate sodium-induced alterations in liver. Foods 2023, 12, 1115. [Google Scholar] [CrossRef]
- Ding, H.M.; Chen, X.J.; Chen, H.M.; Wang, C.S.; Qian, G.Y. Effect of Sargassum fusiforme polysaccharide on apoptosis and its possible mechanism in human erythroleukemia cells. Chin. J. Nat. Med. 2020, 18, 749–759. [Google Scholar] [CrossRef] [PubMed]
- Fan, S.R.; Yu, G.Q.; Nie, W.J.; Jin, J.; Chen, L.A.; Chen, X.M. Antitumor activity and underlying mechanism of Sargassum fusiforme polysaccharides in CNE-bearing mice. Int. J. Biol. Macromol. 2018, 112, 516–522. [Google Scholar] [CrossRef]
- Chen, L.J.; Chen, P.C.; Liu, J.; Hu, C.X.; Yang, S.S.; He, D.; Yu, P.; Wu, M.J.; Zhang, X. Sargassum fusiforme polysaccharide SFP-F2 activates the NF-kappaB signaling pathway via CD14/IKK and P38 axes in RAW264.7 cells. Mar. Drugs 2018, 16, 264. [Google Scholar] [CrossRef] [PubMed]
- Yu, M.; Ji, Y.B.; Qi, Z.; Cui, D.; Xin, G.S.; Wang, B.; Cao, Y.; Wang, D.D. Anti-tumor activity of sulfated polysaccharides from Sargassum fusiforme. Saudi Pharm. J. 2017, 25, 464–468. [Google Scholar] [CrossRef] [PubMed]
- Zhao, T.X.; Dong, Q.Y.; Zhou, H.B.; Yang, H.L. Drying kinetics, physicochemical properties, antioxidant activity and antidiabetic potential of Sargassum fusiforme processed under four drying techniques. LWT-Food Sci. Technol. 2022, 163, 113578. [Google Scholar] [CrossRef]
- Wang, L.; Cui, Y.R.; Lee, H.G.; Fu, X.T.; Wang, K.Q.; Xu, J.C.; Gao, X.; Jeon, Y.J. Fucoidan isolated from fermented Sargassum fusiforme suppresses oxidative stress through stimulating the expression of superoxidase dismutase and catalase by regulating Nrf2 signaling pathway. Int. J. Biol. Macromol. 2022, 209, 935–941. [Google Scholar] [CrossRef]
- Zhang, R.; Zhang, X.X.; Tang, Y.Y.; Mao, J.L. Composition, isolation, purification and biological activities of Sargassum fusiforme polysaccharides: A review. Carbohydr. Polym. 2020, 228, 115381. [Google Scholar] [CrossRef]
- Ye, Y.H.; Ji, D.S.; You, L.J.; Zhou, L.; Zhao, Z.G.; Brennan, C. Structural properties and protective effect of Sargassum fusiforme polysaccharides against ultraviolet B radiation in hairless Kun Ming mice. J. Funct. Foods 2018, 43, 8–16. [Google Scholar] [CrossRef]
- Green, D.R. Caspase activation and inhibition. CSH Perspect. Biol. 2022, 14, a041020. [Google Scholar] [CrossRef]
- Simpson, D.S.; Pang, J.Y.; Weir, A.; Kong, I.Y.; Fritsch, M.; Rashidi, M.; Cooney, J.P.; Davidson, K.C.; Speir, M.; Djajawi, T.M.; et al. Interferon-γ primes macrophages for pathogen ligand-induced killing via a Caspase-8 and mitochondrial cell death pathway. Immunity 2022, 55, 423–441. [Google Scholar] [CrossRef]
- Eskandari, E.; Eaves, C.J. Paradoxical roles of Caspase-3 in regulating cell survival, proliferation, and tumorigenesis. J. Cell Biol. 2022, 221, e202201159. [Google Scholar] [PubMed]
- Khan, T.; Date, A.; Chawda, H.; Patel, K. Polysaccharides as potential anticancer agents-A review of their progress. Carbohydr. Polym. 2019, 210, 412–428. [Google Scholar] [CrossRef] [PubMed]
- Wu, S.Y.; Liu, J.; Zhang, Y.; Song, J.X.; Zhang, Z.S.; Yang, Y.; Wu, M.J.; Tong, H.B. Structural characterization and antagonistic effect against P-selectin-mediated function of SFF-32, a fucoidan fraction from Sargassum fusiforme. J. Ethnopharmacol. 2022, 295, 115408. [Google Scholar] [CrossRef] [PubMed]
- Hu, J.H.; Yao, W.Z.; Chang, S.Y.; You, L.J.; Zhao, M.M.; Chi-Keung Cheung, P.; Hileuskaya, K. Structural characterization and anti-photoaging activity of a polysaccharide from Sargassum fusiforme. Food Res. Int. 2022, 157, 111267. [Google Scholar] [CrossRef]
- Jin, W.H.; Tang, H.; Zhang, J.M.; Wei, B.; Sun, J.D.; Zhang, W.J.; Zhang, F.M.; Wang, H.; Linhardt, R.J.; Zhong, W.H. Structural analysis of a novel sulfated galacto-fuco-xylo-glucurono-mannan from Sargassum fusiforme and its anti-lung cancer activity. Int. J. Biol. Macromol. 2020, 149, 450–458. [Google Scholar] [CrossRef]
- Wei, X.Q.; Cai, L.Q.; Liu, H.L.; Tu, H.L.; Xu, X.J.; Zhou, F.L.; Zhang, L.N. Chain conformation and biological activities of hyperbranched fucoidan derived from brown algae and its desulfated derivative. Carbohydr. Polym. 2019, 208, 86–96. [Google Scholar] [CrossRef]
- Jin, J.O.; Chauhan, P.S.; Arukha, A.P.; Chavda, V.; Dubey, A.; Yadav, D. The therapeutic potential of the anticancer activity of fucoidan: Current advances and hurdles. Mar. Drugs 2021, 19, 265. [Google Scholar] [CrossRef]
- van Weelden, G.; Bobinski, M.; Okla, K.; van Weelden, W.J.; Romano, A.; Pijnenborg, J.M.A. Fucoidan structure and activity in relation to anti-cancer mechanisms. Mar. Drugs 2019, 17, 32. [Google Scholar] [CrossRef]
- Cong, Q.F.; Chen, H.J.; Liao, W.F.; Xiao, F.; Wang, P.P.; Qin, Y.; Dong, Q.; Ding, K. Structural characterization and effect on anti-angiogenic activity of a fucoidan from Sargassum fusiforme. Carbohydr. Polym. 2016, 136, 899–907. [Google Scholar] [CrossRef]
- Li, Y.P.; Zheng, Y.T.; Zhang, Y.; Yang, Y.Y.; Wang, P.Y.; Imre, B.; Wong, A.C.Y.; Hsieh, Y.S.Y.; Wang, D.M. Brown algae carbohydrates: Structures, pharmaceutical properties, and research challenges. Mar. Drugs 2021, 19, 620. [Google Scholar] [CrossRef]
- Kale, J.; Osterlund, E.J.; Andrews, D.W. BCL-2 family proteins: Changing partners in the dance towards death. Cell Death Differ. 2018, 25, 65–80. [Google Scholar] [CrossRef] [PubMed]
- McArthur, K.; Whitehead, L.W.; Heddleston, J.M.; Li, L.; Padman, B.S.; Oorschot, V.; Geoghegan, N.D.; Chappaz, S.; Davidson, S.; San Chin, H.; et al. BAK/BAX macropores facilitate mitochondrial herniation and mtDNA efflux during apoptosis. Science 2018, 359, eaao6047. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Yang, X.; Ge, X.H.; Zhang, F.Y. Puerarin attenuates neurological deficits via Bcl-2/Bax/cleaved Caspase-3 and Sirt3/SOD2 apoptotic pathways in subarachnoid hemorrhage mice. Biomed. Pharmacother. 2019, 109, 726–733. [Google Scholar] [CrossRef] [PubMed]
- Li, F.; Jiao, X.; Zhao, J.; Liao, X.J.; Wei, Y.L.; Li, Q.H. Antitumor mechanisms of an exopolysaccharide from Lactobacillus fermentum on HT-29 cells and HT-29 tumor-bearing mice. Int. J. Biol. Macromol. 2022, 209, 552–562. [Google Scholar] [CrossRef]
- Yosef, R.; Pilpel, N.; Tokarsky-Amiel, R.; Biran, A.; Ovadya, Y.; Cohen, S.; Vadai, E.; Dassa, L.; Shahar, E.; Condiotti, R.; et al. Directed elimination of senescent cells by inhibition of BCL-W and Bcl-xL. Nat. Commun. 2016, 7, 11190. [Google Scholar] [CrossRef] [PubMed]
- Ashkenazi, A.; Fairbrother, W.J.; Leverson, J.D.; Souers, A.J. From basic apoptosis discoveries to advanced selective BCL-2 family inhibitors. Nat. Rev. Drug Discov. 2017, 16, 273–284. [Google Scholar] [CrossRef]
- Lei, W.; Jae Young, O.; Jin, H.; Jae Young, K.; You Joung, J.; Bo Mi, R. In vitro and in vivo antioxidant activities of polysaccharides isolated from celluclast-assisted extract of an edible brown seaweed, Sargassum fulvellum. Antioxidants 2019, 8, 493. [Google Scholar]
- Fernando, I.P.S.; Sanjeewa, K.K.A.; Lee, H.G.; Kim, H.S.; Vaas, A.P.J.P.; De Silva, H.I.C.; Nanayakkara, C.M.; Abeytunga, D.T.U.; Lee, D.S.; Lee, J.S.; et al. Fucoidan purified from Sargassum polycystum induces apoptosis through mitochondria-mediated pathway in HL-60 and MCF-7 cells. Mar. Drugs 2020, 18, 196. [Google Scholar] [CrossRef]
- Tewari, D.; Patni, P.; Bishayee, A.; Sah, A.N.; Bishayee, A. Natural products targeting the PI3K-AKT-mTOR signaling pathway in cancer: A novel therapeutic strategy. Semin. Cancer Biol. 2022, 80, 1–17. [Google Scholar] [CrossRef]
- Shorning, B.Y.; Dass, M.S.; Smalley, M.J.; Pearson, H.B. The PI3K-AKT-mTOR pathway and prostate cancer: At the crossroads of AR, MAPK, and WNT signaling. Int. J. Mol. Sci. 2020, 21, 4507. [Google Scholar] [CrossRef]
- Xu, F.; Na, L.X.; Li, Y.F.; Chen, L.J. Roles of the PI3K/AKT/mTOR signalling pathways in neurodegenerative diseases and tumours. Cell Biosci. 2020, 10, 54. [Google Scholar] [CrossRef] [PubMed]
- Ediriweera, M.K.; Tennekoon, K.H.; Samarakoon, S.R. Role of the PI3K/AKT/mTOR signaling pathway in ovarian cancer: Biological and therapeutic significance. Semin. Cancer Biol. 2019, 59, 147–160. [Google Scholar] [CrossRef]
- Feng, F.B.; Qiu, H.Y. Effects of Artesunate on chondrocyte proliferation, apoptosis and autophagy through the PI3K/AKT/mTOR signaling pathway in rat models with rheumatoid arthritis. Biomed. Pharmacother. 2018, 102, 1209–1220. [Google Scholar] [CrossRef] [PubMed]
- Xu, Z.R.; Han, X.; Ou, D.M.; Liu, T.; Li, Z.X.; Jiang, G.M.; Liu, J.; Zhang, J. Targeting PI3K/AKT/mTOR-mediated autophagy for tumor therapy. Appl. Microbiol. Biotechnol. 2020, 104, 575–587. [Google Scholar] [CrossRef] [PubMed]
- Guo, J.M.; Xing, H.J.; Cai, J.Z.; Zhang, H.F.; Xu, S.W. H2S exposure-induced oxidative stress promotes LPS-mediated hepatocyte autophagy through the PI3K/AKT/TOR pathway. Ecotoxicol. Environ. Saf. 2021, 209, 111801. [Google Scholar] [CrossRef]
- Rajendran, P.; Ammar, R.B.; Al Saeedi, F.J.; Mohamed, M.E.; ElNaggar, M.A.; Al Ramadan, S.Y.; Bekhet, G.M.; Soliman, A.M. Kaempferol inhibits zearalenone-induced oxidative stress and apoptosis via the PI3K/AKT-Mediated Nrf2 signaling pathway: In vitro and in vivo studies. Int. J. Mol. Sci. 2020, 22, 217. [Google Scholar] [CrossRef] [PubMed]
- Zhang, C.Y.; Lin, T.J.; Nie, G.H.; Hu, R.M.; Pi, S.X.; Wei, Z.J.; Wang, C.; Xing, C.H.; Hu, G.L. Cadmium and molybdenum co-induce pyroptosis via ROS/PTEN/PI3K/AKT axis in duck renal tubular epithelial cells. Environ. Pollut. 2021, 272, 116403. [Google Scholar] [CrossRef]
- Liu, M.M.; Ma, R.H.; Ni, Z.J.; Thakur, K.; Cespedes Acuna, C.L.; Jiang, L.; Wei, Z.J. Apigenin 7-O-glucoside promotes cell apoptosis through the PTEN/PI3K/AKT pathway and inhibits cell migration in cervical cancer HeLa cells. Food Chem. Toxicol. 2020, 146, 111843. [Google Scholar] [CrossRef]
- Reddy, D.; Kumavath, R.; Tan, T.Z.; Ampasala, D.R.; Kumar, A.P. Peruvoside targets apoptosis and autophagy through MAPK Wnt/beta-catenin and PI3K/AKT/mTOR signaling pathways in human cancers. Life Sci. 2020, 241, 117147. [Google Scholar] [CrossRef]
- Zhu, M.; Shi, X.P.; Gong, Z.Y.; Su, Q.; Yu, R.Z.; Wang, B.; Yang, T.F.; Dai, B.L.; Zhan, Y.Z.; Zhang, D.D.; et al. Cantharidin treatment inhibits hepatocellular carcinoma development by regulating the JAK2/STAT3 and PI3K/AKT pathways in an EphB4-dependent manner. Pharmacol. Res. 2020, 158, 104868. [Google Scholar] [CrossRef]
- Ran, L.W.; Chen, F.; Zhang, J.; Mi, J.; Lu, L.; Yan, Y.M.; Cao, Y.L. Antitumor effects of pollen polysaccharides from Chinese wolfberry on DU145 cells via the PI3K/AKT pathway in vitro and in vivo. Int. J. Biol. Macromol. 2020, 152, 1164–1173. [Google Scholar] [CrossRef] [PubMed]
- Tao, H.; Chen, X.; Du, Z.Y.; Ding, K. Corn silk crude polysaccharide exerts anti-pancreatic cancer activity by blocking the EGFR/PI3K/AKT/CREB signaling pathway. Food Funct. 2020, 11, 6961–6970. [Google Scholar] [CrossRef] [PubMed]
- Wang, B.; Li, J.Y. Piceatannol suppresses the proliferation and induce apoptosis of osteosarcoma cells through PI3K/AKT/mTOR pathway. Cancer Manag. Res. 2020, 12, 2631–2640. [Google Scholar] [CrossRef] [PubMed]
- Ding, H.M.; Sun, T.; Xia, P.K.; Tang, Q.; Wang, Z.H.; Wang, C.S.; Chen, H.M.; Qian, G.Y. Inhibition of polysaccharide fraction of Sargassum fusiforme on the α-glucosidase. J. Nucl. Agric. Sci. 2019, 33, 0297–0304. [Google Scholar]
- Yu, S.S.; Ji, H.Y.; Dong, X.D.; Liu, A.J.; Yu, J. FAS/FAS-L-mediated apoptosis and autophagy of SPC-A-1 cells induced by water-soluble polysaccharide from Polygala tenuifolia. Int. J. Biol. Macromol. 2020, 150, 449–458. [Google Scholar] [CrossRef] [PubMed]
- Liu, W.J.; Li, W.Y.; Sui, Y.; Li, X.Q.; Liu, C.Q.; Jing, H.; Zhang, H.M.; Cao, W. Structure characterization and anti-leukemia activity of a novel polysaccharide from Angelica sinensis (Oliv.) Diels. Int. J. Biol. Macromol. 2019, 121, 161–172. [Google Scholar] [CrossRef]









| Genes | Sense Primers (5′-3′) | Antisense Primers (3′-5′) |
|---|---|---|
| Caspase-3 | TCCACGAGCAGAGTCAAA | ACACACTTGAACCAACCG |
| Caspase-9 | CCTGTATCATCCCCACCCT | CACAAGGTTCCAGAGCCG |
| Bcl-xL | GGAGCTGGTGGTTGACTTTCT | CCGGAAGAGTTCATTCACTAC |
| Bad | CCCAGAGTTTGAGCCGAGTG | ATCCCTTCGTCCTCCGT |
| AKT | CGAGGAGGAGGTGTATCA | ATGGCTTGCACGGAAATGGC |
| PI3K | AACGAGAACGTGTGCCATTTG | AAGCTGTCGTACGGTTAGAGA |
| β-actin | CCTGGCACCCAGCACAAT | GGGCCGGACTCGTCATAC |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Du, H.; Jin, X.; Jin, S.; Zhang, D.; Chen, Q.; Jin, X.; Wang, C.; Qian, G.; Ding, H. Anti-Leukemia Activity of Polysaccharide from Sargassum fusiforme via the PI3K/AKT/BAD Pathway In Vivo and In Vitro. Mar. Drugs 2023, 21, 289. https://doi.org/10.3390/md21050289
Du H, Jin X, Jin S, Zhang D, Chen Q, Jin X, Wang C, Qian G, Ding H. Anti-Leukemia Activity of Polysaccharide from Sargassum fusiforme via the PI3K/AKT/BAD Pathway In Vivo and In Vitro. Marine Drugs. 2023; 21(5):289. https://doi.org/10.3390/md21050289
Chicago/Turabian StyleDu, Haofei, Xudong Jin, Sizhou Jin, Donglei Zhang, Qiande Chen, Xuanan Jin, Caisheng Wang, Guoying Qian, and Haomiao Ding. 2023. "Anti-Leukemia Activity of Polysaccharide from Sargassum fusiforme via the PI3K/AKT/BAD Pathway In Vivo and In Vitro" Marine Drugs 21, no. 5: 289. https://doi.org/10.3390/md21050289
APA StyleDu, H., Jin, X., Jin, S., Zhang, D., Chen, Q., Jin, X., Wang, C., Qian, G., & Ding, H. (2023). Anti-Leukemia Activity of Polysaccharide from Sargassum fusiforme via the PI3K/AKT/BAD Pathway In Vivo and In Vitro. Marine Drugs, 21(5), 289. https://doi.org/10.3390/md21050289