Molecular Detection and Characterization of the First Cowpox Virus Isolate Derived from a Bank Vole
Abstract
:1. Introduction
2. Materials and Methods
2.1. Rodent Trapping
2.2. OPV DNA Screening
2.3. Cell Lines and Virus Isolation
2.4. Sequencing, Genome Assembly, and Annotation of the CPXV Isolate
2.5. Phylogenetic Analysis
2.6. Phenotypic Analysis Using Chorioallantoic Membrane Culture
2.7. Virus Replication Kinetics in Different Cell Lines
2.8. Data Availability
3. Results
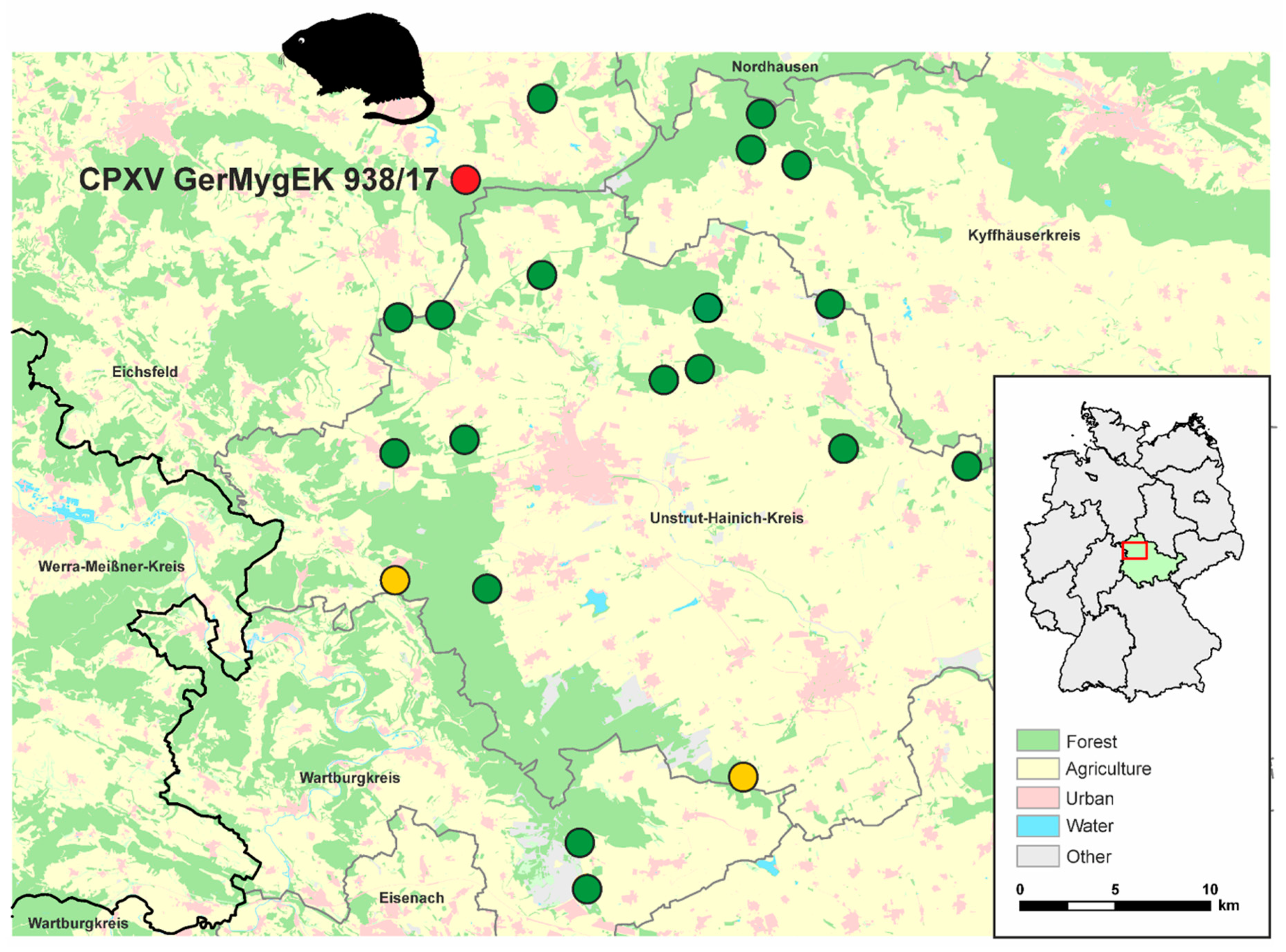
3.1. Rodent Trapping and Detection of OPV DNA
3.2. Virus Isolation, Sequence Determination, and Genome Characterization
3.3. Phylogenetic Analysis
3.4. Phenotypic Characterization on the CAM
3.5. Virus Replication Kinetics in Different Cell Lines
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Moss, B. Poxvirus DNA replication. Cold Spring Harb. Perspect. Biol. 2013, 5, a010199. [Google Scholar] [CrossRef] [PubMed]
- ICTV. International Committee on Taxonomy of Viruses—Taxonomy. Available online: https://talk.ictvonline.org/taxonomy/w/ictv-taxonomy (accessed on 17 September 2018).
- Haller, S.L.; Peng, C.; McFadden, G.; Rothenburg, S. Poxviruses and the evolution of host range and virulence. Infect. Genet. Evol. J. Mol. Epidemiol. Evol. Genet. Infect. Dis. 2014, 21, 15–40. [Google Scholar] [CrossRef] [PubMed]
- Reynolds, M.G.; Guagliardo, S.A.J.; Nakazawa, Y.J.; Doty, J.B.; Mauldin, M.R. Understanding orthopoxvirus host range and evolution: From the enigmatic to the usual suspects. Curr. Opin. Virol. 2018, 28, 108–115. [Google Scholar] [CrossRef] [PubMed]
- Essbauer, S.; Hartnack, S.; Misztela, K.; Kiessling-Tsalos, J.; Baumler, W.; Pfeffer, M. Patterns of orthopox virus wild rodent hosts in south Germany. Vector Borne Zoonotic Dis. 2009, 9, 301–311. [Google Scholar] [CrossRef] [PubMed]
- Oliveira, G.P.; Rodrigues, R.A.L.; Lima, M.T.; Drumond, B.P.; Abrahao, J.S. Poxvirus host range genes and virus-host spectrum: A critical review. Viruses 2017, 9, 331. [Google Scholar] [CrossRef]
- Bratke, K.A.; McLysaght, A.; Rothenburg, S. A survey of host range genes in poxvirus genomes. Infect. Genet. Evol. J. Mol. Epidemiol. Evol. Genet. Infect. Dis. 2013, 14, 406–425. [Google Scholar] [CrossRef]
- Alzhanova, D.; Früh, K. Modulation of the host immune response by cowpox virus. Microbes Infect 2010, 12, 900–909. [Google Scholar] [CrossRef]
- Gazzani, P.; Gach, J.E.; Colmenero, I.; Martin, J.; Morton, H.; Brown, K.; Milford, D.V. Fatal disseminated cowpox virus infection in an adolescent renal transplant recipient. Pediatric Nephrol. 2017, 32, 533–536. [Google Scholar] [CrossRef]
- Fassbender, P.; Zange, S.; Ibrahim, S.; Zoeller, G.; Herbstreit, F.; Meyer, H. Generalized cowpox virus infection in a patient with hiv, Germany, 2012. Emerg. Infect. Dis. 2016, 22, 553–555. [Google Scholar] [CrossRef]
- Shchelkunov, S.N. An increasing danger of zoonotic orthopoxvirus infections. PLoS Pathog. 2013, 9, e1003756. [Google Scholar] [CrossRef]
- Downie, A. A study of the lesions produced experimentally by cowpox virus. J. Pathol. Bacteriol. 1939, 48, 361–379. [Google Scholar] [CrossRef]
- Essbauer, S.; Meyer, H. Genus orthopoxvirus: Cowpox virus. In Poxviruses; Springer: Birkhäuser Basel, Switzerland, 2007; pp. 75–88. [Google Scholar]
- Baxby, D. The natural history of cowpox: The substance of the jenner lecture, read 17th may 1982 in Bristol royal infirmary under the auspices of the jenner trust. Bristol Med. Chir. J. 1982, 97, 12–16. [Google Scholar] [PubMed]
- Archard, L.C.; Mackett, M. Restriction endonuclease analysis of red cowpox virus and its white pock variant. J. Gen. Virol. 1979, 45, 51–63. [Google Scholar] [CrossRef] [PubMed]
- Franke, A.; Pfaff, F.; Jenckel, M.; Hoffmann, B.; Höper, D.; Antwerpen, M.; Meyer, H.; Beer, M.; Hoffmann, D. Classification of cowpox viruses into several distinct clades and identification of a novel lineage. Viruses 2017, 9, 142. [Google Scholar] [CrossRef] [PubMed]
- Antwerpen, M.H.; Georgi, E.; Nikolic, A.; Zoeller, G.; Wohlsein, P.; Baumgartner, W.; Peyrefitte, C.; Charrel, R.; Meyer, H. Use of next generation sequencing to study two cowpox virus outbreaks. PeerJ 2019, 7, e6561. [Google Scholar] [CrossRef] [PubMed]
- Mauldin, M.R.; Antwerpen, M.; Emerson, G.L.; Li, Y.; Zoeller, G.; Carroll, D.S.; Meyer, H. Cowpox virus: What’s in a name? Viruses 2017, 9, 101. [Google Scholar] [CrossRef]
- Chantrey, J.; Meyer, H.; Baxby, D.; Begon, M.; Bown, K.; Hazel, S.; Jones, T.; Montgomery, W.; Bennett, M. Cowpox: Reservoir hosts and geographic range. Epidemiol. Infect. 1999, 122, 455–460. [Google Scholar] [CrossRef]
- Prkno, A.; Hoffmann, D.; Goerigk, D.; Kaiser, M.; van Maanen, A.C.F.; Jeske, K.; Jenckel, M.; Pfaff, F.; Vahlenkamp, T.W.; Beer, M.; et al. Epidemiological investigations of four cowpox virus outbreaks in alpaca herds, Germany. Viruses 2017, 9, 344. [Google Scholar] [CrossRef]
- Wilson, D.E.; Lacher, T.E.; Mittermeier, R.A. (Eds.) Handbook of the Mammals of the World: Rodents II; Lynx Edicions: Barcelona, Spain, 2017. [Google Scholar]
- Kotlik, P.; Deffontaine, V.; Mascheretti, S.; Zima, J.; Michaux, J.R.; Searle, J.B. A northern glacial refugium for bank voles (Clethrionomys glareolus). Proc. Natl. Acad. Sci. USA 2006, 103, 14860–14864. [Google Scholar] [CrossRef]
- Kaplan, C.; Healing, T.D.; Evans, N.; Healing, L.; Prior, A. Evidence of infection by viruses in small British field rodents. Epidemiol. Infect. 1980, 84, 285–294. [Google Scholar] [CrossRef]
- Crouch, A.C.; Baxby, D.; McCracken, C.M.; Gaskell, R.M.; Bennett, M. Serological evidence for the reservoir hosts of cowpox virus in British wildlife. Epidemiol. Infect. 1995, 115, 185–191. [Google Scholar] [CrossRef] [PubMed]
- Begon, M.; Hazel, S.M.; Baxby, D.; Bown, K.; Cavanagh, R.; Chantrey, J.; Jones, T.; Bennett, M. Transmission dynamics of a zoonotic pathogen within and between wildlife host species. Proc. Biol. Sci. 1999, 266, 1939–1945. [Google Scholar] [CrossRef] [PubMed]
- Hazel, S.; Bennett, M.; Chantrey, J.; Bown, K.; Cavanagh, R.; Jones, T.; Baxby, D.; Begon, M. A longitudinal study of an endemic disease in its wildlife reservoir: Cowpox and wild rodents. Epidemiol. Infect. 2000, 124, 551–562. [Google Scholar] [CrossRef] [PubMed]
- Telfer, S.; Bennett, M.; Bown, K.; Carslake, D.; Cavanagh, R.; Hazel, S.; Jones, T.; Begon, M. Infection with cowpox virus decreases female maturation rates in wild populations of woodland rodents. Oikos 2005, 109, 317–322. [Google Scholar] [CrossRef]
- Boulanger, D.; Crouch, A.; Brochier, B.; Bennett, M.; Clement, J.; Gaskell, R.M.; Baxby, D.; Pastoret, P.P. Serological survey for orthopoxvirus infection of wild mammals in areas where a recombinant rabies virus is used to vaccinate foxes. Vet. Rec. 1996, 138, 247–249. [Google Scholar] [CrossRef] [PubMed]
- Sandvik, T.; Tryland, M.; Hansen, H.; Mehl, R.; Moens, U.; Olsvik, Ø.; Traavik, T. Naturally occurring orthopoxviruses: Potential for recombination with vaccine vectors. J. Clin. Microbiol. 1998, 36, 2542–2547. [Google Scholar]
- Tryland, M.; Sandvik, T.; Mehl, R.; Bennett, M.; Traavik, T.; Olsvik, O. Serosurvey for orthopoxviruses in rodents and shrews from Norway. J. Wildl. Dis. 1998, 34, 240–250. [Google Scholar] [CrossRef]
- Kinnunen, P.M.; Henttonen, H.; Hoffmann, B.; Kallio, E.R.; Korthase, C.; Laakkonen, J.; Niemimaa, J.; Palva, A.; Schlegel, M.; Ali, H.S.; et al. Orthopox virus infections in Eurasian wild rodents. Vector Borne Zoonotic Dis. 2011, 11, 1133–1140. [Google Scholar] [CrossRef]
- Oldal, M.; Sironen, T.; Henttonen, H.; Vapalahti, O.; Madai, M.; Horvath, G.; Dallos, B.; Kutas, A.; Foldes, F.; Kemenesi, G.; et al. Serologic survey of orthopoxvirus infection among rodents in Hungary. Vector Borne Zoonotic Dis. 2015, 15, 317–322. [Google Scholar] [CrossRef]
- Pelkonen, P.M.; Tarvainen, K.; Hynninen, A.; Kallio, E.R.; Henttonen, K.; Palva, A.; Vaheri, A.; Vapalahti, O. Cowpox with severe generalized eruption, Finland. Emerg. Infect. Dis. 2003, 9, 1458–1461. [Google Scholar] [CrossRef]
- Essbauer, S.; Pfeffer, M.; Meyer, H. Zoonotic poxviruses. Vet. Microbiol. 2010, 140, 229–236. [Google Scholar] [CrossRef] [PubMed]
- Forbes, K.M.; Voutilainen, L.; Jaaskelainen, A.; Sironen, T.; Kinnunen, P.M.; Stuart, P.; Vapalahti, O.; Henttonen, H.; Huitu, O. Serological survey of rodent-borne viruses in Finnish field voles. Vector Borne Zoonotic Dis. 2014, 14, 278–283. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Stagegaard, J.; Kurth, A.; Stern, D.; Dabrowski, P.W.; Pocknell, A.; Nitsche, A.; Schrick, L. Seasonal recurrence of cowpox virus outbreaks in captive cheetahs (Acinonyx jubatus). PLoS ONE 2017, 12, e0187089. [Google Scholar] [CrossRef] [PubMed]
- Hoffmann, D.; Franke, A.; Jenckel, M.; Tamosiunaite, A.; Schluckebier, J.; Granzow, H.; Hoffmann, B.; Fischer, S.; Ulrich, R.G.; Höper, D.; et al. Out of the reservoir: Phenotypic and genotypic characterization of a novel cowpox virus isolated from a common vole. J. Virol. 2015, 89, 10959–10969. [Google Scholar] [CrossRef] [Green Version]
- Franke, A.; Ulrich, R.G.; Weber, S.; Osterrieder, N.; Keller, M.; Hoffmann, D.; Beer, M. Experimental cowpox virus (CPXV) infections of bank voles: Exceptional clinical resistance and variable reservoir competence. Viruses 2017, 9, 391. [Google Scholar] [CrossRef] [Green Version]
- Reil, D.; Rosenfeld, U.M.; Imholt, C.; Schmidt, S.; Ulrich, R.G.; Eccard, J.A.; Jacob, J. Puumala hantavirus infections in bank vole populations: Host and virus dynamics in central Europe. BMC Ecol. 2017, 17, 9. [Google Scholar] [CrossRef] [Green Version]
- Fischer, S.; Mayer-Scholl, A.; Imholt, C.; Spierling, N.G.; Heuser, E.; Schmidt, S.; Reil, D.; Rosenfeld, U.M.; Jacob, J.; Nockler, K.; et al. Leptospira genomospecies and sequence type prevalence in small mammal populations in Germany. Vector Borne Zoonotic Dis. 2018, 18, 188–199. [Google Scholar] [CrossRef]
- Scaramozzino, N.; Ferrier-Rembert, A.; Favier, A.L.; Rothlisberger, C.; Richard, S.; Crance, J.M.; Meyer, H.; Garin, D. Real-time PCR to identify variola virus or other human pathogenic orthopox viruses. Clin. Chem. 2007, 53, 606–613. [Google Scholar] [CrossRef]
- Langmead, B.; Salzberg, S.L. Fast gapped-read alignment with bowtie 2. Nat. Methods 2012, 9, 357–359. [Google Scholar] [CrossRef] [Green Version]
- Kalthoff, D.; Bock, W.I.; Huhn, F.; Beer, M.; Hoffmann, B. Fatal cowpox virus infection in cotton-top tamarins (Saguinus oedipus) in Germany. Vector Borne Zoonotic Dis. 2014, 14, 303–305. [Google Scholar] [CrossRef]
- Capella-Gutierrez, S.; Silla-Martinez, J.M.; Gabaldon, T. Trimal: A tool for automated alignment trimming in large-scale phylogenetic analyses. Bioinformatics 2009, 25, 1972–1973. [Google Scholar] [CrossRef] [PubMed]
- Sanchez, R.; Serra, F.; Tarraga, J.; Medina, I.; Carbonell, J.; Pulido, L.; de Maria, A.; Capella-Gutierrez, S.; Huerta-Cepas, J.; Gabaldon, T.; et al. Phylemon 2.0: A suite of web-tools for molecular evolution, phylogenetics, phylogenomics and hypotheses testing. Nucleic Acids Res. 2011, 39, W470–W474. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Nguyen, L.T.; Schmidt, H.A.; von Haeseler, A.; Minh, B.Q. IQ-TREE: A fast and effective stochastic algorithm for estimating maximum-likelihood phylogenies. Mol. Biol. Evol. 2015, 32, 268–274. [Google Scholar] [CrossRef] [PubMed]
- Rambaut, A.; Drummond, A. Figtree Version 1.4.0. Available online: http://tree.bio.ed.ac.uk/software/figtree/ (accessed on 15 November 2019).
- Kumar, S.; Stecher, G.; Tamura, K. Mega7: Molecular evolutionary genetics analysis version 7.0 for bigger datasets. Mol. Biol. Evol. 2016, 33, 1870–1874. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- RStudioTeam (2019). RStudio: Integrated Development for R; RStudio, Inc.: Boston, MA, USA, 2015. Available online: http://www.Rstudio.Com/ (accessed on 15 November 2019).
- Cunningham, C. A Laboratory Guide in Virology, 6th ed.; Burgess: Minneapolis, MN, USA, 1966. [Google Scholar]
- Essbauer, S.S.; Krautkramer, E.; Herzog, S.; Pfeffer, M. A new permanent cell line derived from the bank vole (Myodes glareolus) as cell culture model for zoonotic viruses. Virol. J. 2011, 8, 339. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Binder, F.; Lenk, M.; Weber, S.; Stoek, F.; Dill, V.; Reiche, S.; Riebe, R.; Wernike, K.; Hoffmann, D.; Ziegler, U.; et al. Common vole (Microtus arvalis) and bank vole (Myodes glareolus) derived permanent cell lines differ in their susceptibility and replication kinetics of animal and zoonotic viruses. J. Virol. Methods 2019, 274, 113729. [Google Scholar] [CrossRef] [Green Version]
- Spearman, C. The method of ‘right and wrong cases’ (‘constant stimuli’) without Gauss’s formulae. Br. J. Psychol. 1904–1920 1908, 2, 227–242. [Google Scholar] [CrossRef]
- Kärber, G. Beitrag zur kollektiven Behandlung pharmakologischer Reihenversuche. Naunyn-Schmiedebergs Arch. Für Exp. Pathol. Und Pharmakol. 1931, 162, 480–483. [Google Scholar]
- Cardeti, G.; Gruber, C.E.M.; Eleni, C.; Carletti, F.; Castilletti, C.; Manna, G.; Rosone, F.; Giombini, E.; Selleri, M.; Lapa, D.; et al. Fatal outbreak in tonkean macaques caused by possibly novel orthopoxvirus, Italy, january 2015 (1). Emerg. Infect. Dis. 2017, 23, 1941–1949. [Google Scholar] [CrossRef] [Green Version]
- Gruber, C.E.M.; Giombini, E.; Selleri, M.; Tausch, S.H.; Andrusch, A.; Tyshaieva, A.; Cardeti, G.; Lorenzetti, R.; De Marco, L.; Carletti, F.; et al. Whole genome characterization of orthopoxvirus (OPV) Abatino, a zoonotic virus representing a putative novel clade of old world orthopoxviruses. Viruses 2018, 10, 546. [Google Scholar] [CrossRef] [Green Version]
- Feore, S.M.; Bennett, M.; Chantrey, J.; Jones, T.; Baxby, D.; Begon, M. The effect of cowpox virus infection on fecundity in bank voles and wood mice. Proc. R. Soc. Lond. Ser. B Biol. Sci. 1997, 264, 1457–1461. [Google Scholar] [CrossRef] [PubMed]
- Bennett, M.; Crouch, A.J.; Begon, M.; Duffy, B.; Feore, S.; Gaskell, R.M.; Kelly, D.F.; McCracken, C.M.; Vicary, L.; Baxby, D. Cowpox inBritish voles and mice. J. Comp. Pathol. 1997, 116, 35–44. [Google Scholar] [CrossRef]
- Mendez-Rios, J.D.; Martens, C.A.; Bruno, D.P.; Porcella, S.F.; Zheng, Z.M.; Moss, B. Genome sequence of erythromelalgia-related poxvirus identifies it as an ectromelia virus strain. PLoS ONE 2012, 7, e34604. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Chen, N.; Danila, M.I.; Feng, Z.; Buller, R.M.; Wang, C.; Han, X.; Lefkowitz, E.J.; Upton, C. The genomic sequence of ectromelia virus, the causative agent of mousepox. Virology 2003, 317, 165–186. [Google Scholar] [CrossRef] [Green Version]
- Esteban, D.J.; Buller, R.M. Ectromelia virus: The causative agent of mousepox. J. Gen. Virol. 2005, 86, 2645–2659. [Google Scholar] [CrossRef]
- Xu, Z.; Zikos, D.; Tamosiunaite, A.; Klopfleisch, R.; Osterrieder, N.; Tischer, B.K. Identification of 10 cowpox virus proteins that are necessary for induction of hemorrhagic lesions (red pocks) on chorioallantoic membranes. J. Virol. 2014, 88, 8615–8628. [Google Scholar] [CrossRef] [Green Version]
- Lanave, G.; Dowgier, G.; Decaro, N.; Albanese, F.; Brogi, E.; Parisi, A.; Losurdo, M.; Lavazza, A.; Martella, V.; Buonavoglia, C.; et al. Novel orthopoxvirus and lethal disease in cat, Italy. Emerg. Infect. Dis. 2018, 24, 1665–1673. [Google Scholar] [CrossRef] [Green Version]


© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Jeske, K.; Weber, S.; Pfaff, F.; Imholt, C.; Jacob, J.; Beer, M.; Ulrich, R.G.; Hoffmann, D. Molecular Detection and Characterization of the First Cowpox Virus Isolate Derived from a Bank Vole. Viruses 2019, 11, 1075. https://doi.org/10.3390/v11111075
Jeske K, Weber S, Pfaff F, Imholt C, Jacob J, Beer M, Ulrich RG, Hoffmann D. Molecular Detection and Characterization of the First Cowpox Virus Isolate Derived from a Bank Vole. Viruses. 2019; 11(11):1075. https://doi.org/10.3390/v11111075
Chicago/Turabian StyleJeske, Kathrin, Saskia Weber, Florian Pfaff, Christian Imholt, Jens Jacob, Martin Beer, Rainer G. Ulrich, and Donata Hoffmann. 2019. "Molecular Detection and Characterization of the First Cowpox Virus Isolate Derived from a Bank Vole" Viruses 11, no. 11: 1075. https://doi.org/10.3390/v11111075





