DEF Cell-Derived Exosomal miR-148a-5p Promotes DTMUV Replication by Negative Regulating TLR3 Expression
Abstract
1. Introduction
2. Materials and Methods
2.1. Ethics Approval and Consent to Participate
2.2. Reagents
2.3. Cell Lines, Birds, Virus, and Infection
2.4. RNA and cDNA Preparation
2.5. Quantitative Real-Time PCR (qRT-PCR)
2.6. Recombinant Plasmid Construction
2.7. TLR3-shRNA, MDA5-shRNA, and Generation of shRNA-Based Knockdown Cell Lines
2.8. Western Blot Analysis of TLR3, MDA5, and Exosome Proteins
2.9. Exosome isolation
2.10. PBMC Preparation
2.11. Interaction of Exosomes with PBMCs
2.12. Enzyme-Linked Immunosorbent Assay (ELISA) to Detect Duck TLR3 Protein
2.13. Dual-Luciferase Reporter Assay
2.14. Statistical Analysis
3. Results
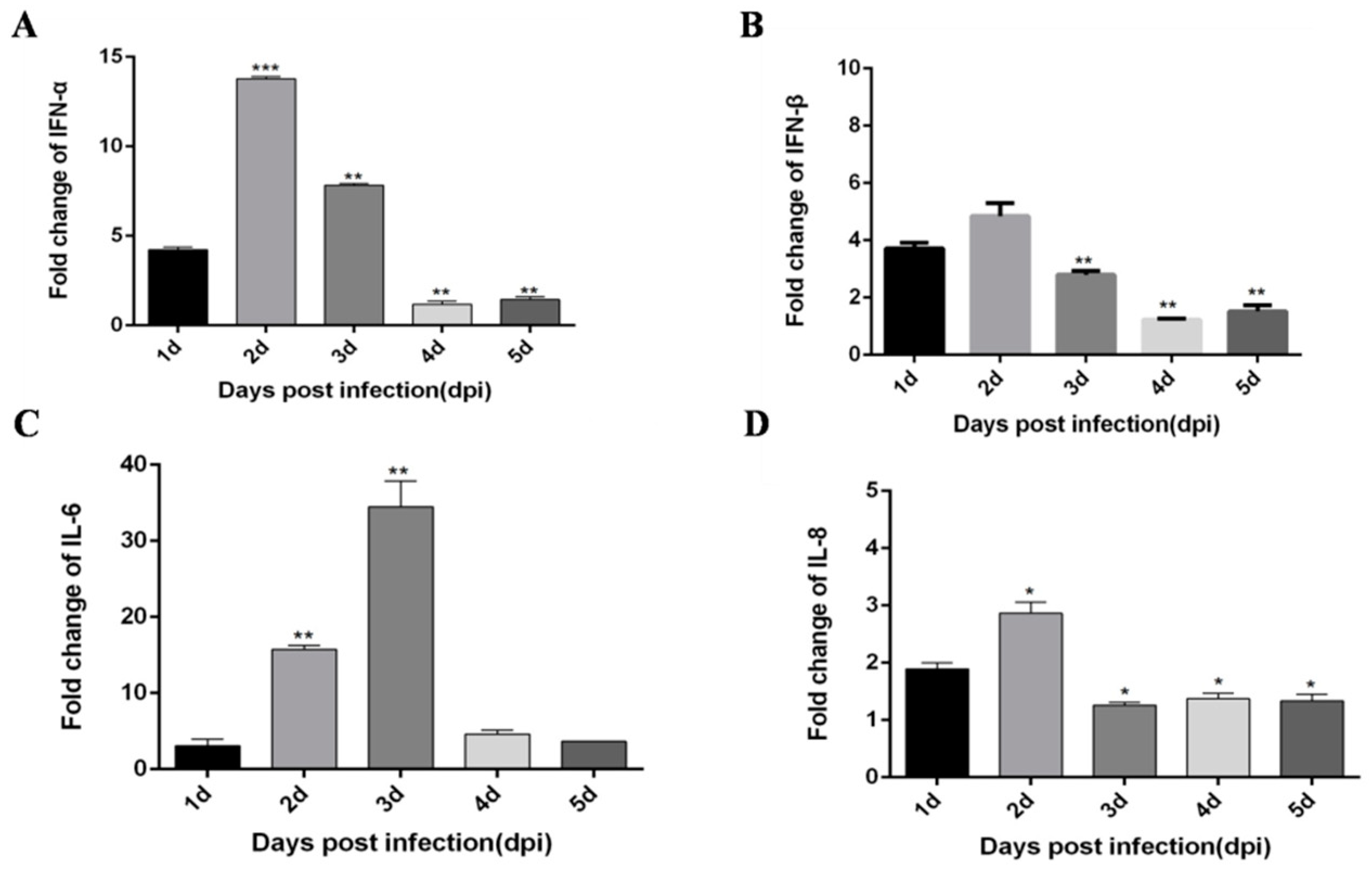
3.1. DTMUV-Infected Ducks Show Up-Regulated the Expression of TLR3, MDA5, and Inflammatory Cytokines
3.2. TLR3 and MDA5 Play Major Immunity Roles in DTMUV-Infected DEF Cells
3.3. MDA5 and TLR3-Dependent Signaling Pathways have Important Roles in Immunity During DTMUV Infection
3.4. Type I IFN Significantly Impairs DTMUV Replication in Dose- and Time-Dependent Manners
3.5. Morphological Characterization and Identification of Exosomes
3.6. DTMUV-Infected DEF Cells and DEF-Derived Exosomes Show Decreased Levels of miR-148a-5p
3.7. miR-148a-5p Targets TLR3
3.8. miR-148a-5p and Exo-miR-148a-5p Negatively Regulate TLR3 and IFN-β Expression in PBMCs
3.9. TLR3 May Be One of the Reasons for the Decrease of miR-148a-5p
4. Discussion
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Liu, P.; Lu, H.; Li, S.; Wu, Y.; Gao, G.F.; Su, J. Duck egg drop syndrome virus: An emerging Tembusu-related flavivirus in China. China Life Sci. 2013, 56, 701–710. [Google Scholar] [CrossRef]
- Cao, Z.; Zhang, C.; Liu, Y.; Liu, Y.; Ye, W.; Han, J.; Ma, G.; Zhang, D.; Xu, F.; Gao, X.; et al. Tembusu virus in ducks, China. Emerg. Infect. Dis. 2011, 17, 1873–1875. [Google Scholar] [CrossRef]
- Su, J.; Li, S.; Hu, X.; Yu, X.; Wang, Y.; Liu, P.; Lu, X.; Zhang, G.; Hu, X.; Liu, D.; et al. Duck egg-drop syndrome caused by BYD virus, a new Tembusu-related flavivirus. PLoS ONE 2011, 6, e18106. [Google Scholar]
- Yan, P.; Zhao, Y.; Zhang, X.; Xu, D.; Dai, X.; Teng, Q.; Yan, L.; Zhou, J.; Ji, X.; Zhang, S.; et al. An infectious disease of ducks caused by a newly emerged Tembusu virus strain in mainland China. Virology 2011, 417, 1–8. [Google Scholar] [CrossRef] [PubMed]
- He, Y.; Wang, A.; Chen, S.; Wu, Z.; Zhang, J.; Wang, M.; Jia, R.; Zhu, D.; Liu, M.; Yang, Q.; et al. Differential immune-related gene expression in the spleens of duck Tembusu virus-infected goslings. Vet. Microbiol. 2017, 212, 39–47. [Google Scholar] [CrossRef] [PubMed]
- Fu, G.; Huang, Y.; Cheng, L.; Wan, C.; Shi, S.; Fu, Q.; Chen, H.; Lin, J.; Lin, F. Genome sequence and phylogenetic analysis of Tembusu viruses isolated from chicken. Chin. J. Vet. Sci. 2014, 34, 1418–1441. [Google Scholar]
- Tang, Y.; Diao, Y.; Yu, C.; Gao, X.; Ju, X.; Xue, C.; Liu, X.; Ge, P.; Qu, J.; Zhang, D.; et al. Characterization of a Tembusu virus isolated from naturally infected house sparrows (Passer domesticus) in Northern China. Transbound. Emerg. Dis. 2013, 60, 152–158. [Google Scholar] [CrossRef]
- Liu, P.; Lu, H.; Li, S.; Moureau, G.; Deng, Y.Q.; Wang, Y.; Zhang, L.; Jiang, T.; de Lamballerie, X.; Qin, C.F.; et al. Genomic and antigenic characterization of the newly emerging Chinese duck egg-drop syndrome flavivirus: Genomic comparison with Tembusu and Sitiawan viruses. J. Gen. Virol. 2012, 93, 2158–2170. [Google Scholar] [CrossRef]
- Pétrilli, V.; Dostert, C.; Muruve, D.A.; Tschopp, J. The inflammasome: A danger sensing complex triggering innate immunity. Curr. Opin. Immunol. 2007, 19, 615–622. [Google Scholar] [CrossRef]
- Brownlie, R.; Allan, B. Avian toll-like receptors. Cell Tissue Res. 2011, 343, 121–130. [Google Scholar] [CrossRef]
- Nasirudeen, A.M.; Wong, H.H.; Thien, P.; Xu, S.; Lam, K.P.; Liu, D.X. RIG-I, MDA5, and TLR3 synergistically play an important role in the restriction of dengue virus infection. PLoS Negl. Trop. Dis. 2011, 5, e926. [Google Scholar] [CrossRef] [PubMed]
- Han, Y.W.; Choi, J.Y.; Uyangaa, E.; Kim, S.B.; Kim, J.H.; Kim, B.S.; Kim, K.; Eo, S.K. Distinct dictation of Japanese encephalitis virus-induced neuroinflammation and lethality via triggering TLR3 and TLR4 signal pathways. Cytokine 2014, 70, 38. [Google Scholar] [CrossRef] [PubMed]
- Dang, J.; Tiwari, S.K.; Lichinchi, G.; Yue, Q.; Patil, V.; Eroshkin, A.; Rana, T.M. Zikavirus depletes neural progenitors in human cerebral organoids through activation of the innate immune receptor TLR3. Cell Stem Cell 2016, 19, 258–265. [Google Scholar] [CrossRef] [PubMed]
- Kawai, T.; Akira, S. The role of pattern-recognition receptors in innate immunity: Update on Toll-like receptors. Nat. Immunol. 2010, 11, 373–384. [Google Scholar] [CrossRef]
- Temperley, N.D.; Berlin, S.; Paton, I.R.; Griffin, D.K.; Burt, D.W. Evolution of the chicken Toll-like receptor gene family: A story of gene gain and gene loss. BMC Genom. 2008, 9, 62. [Google Scholar] [CrossRef]
- Häcker, H.; Karin, M. Regulation and function of IKK and IKK-related kinases. Sci. STKE 2006, 357, 13. [Google Scholar] [CrossRef]
- Häcker, H.; Redecke, V.; Blagoev, B.; Kratchmarova, I.; Hsu, L.C.; Wang, G.G.; Kamps, M.P.; Raz, E.; Wagner, H.; Häcker, G.; et al. Specificity in Toll-like receptor signaling through distinct effector functions of TRAF3 and TRAF6. Nature 2006, 439, 204–207. [Google Scholar] [CrossRef]
- Bhoj, V.G.; Chen, Z.J. Ubiquitylation in innate and adaptive immunity. Nature 2009, 458, 430–437. [Google Scholar] [CrossRef]
- Kawagoe, T.; Sato, S.; Matsushita, K.; Kato, H.; Matsui, K.; Kumagai, Y.; Saitoh, T.; Kawai, T.; Takeuchi, O.; Akira, S. Sequential control of Toll-like receptor-dependent responses by IRAK1 and IRAK2. Nat. Immunol. 2008, 9, 684–691. [Google Scholar] [CrossRef]
- Kawai, T.; Takahashi, K.; Sato, S.; Coban, C.; Kumar, H.; Kato, H.; Ishii, K.J.; Takeuchi, O.; Akira, S. IPS-1, an adaptor triggering RIG-I- and Mda5-mediated type I interferon induction. Nat. Immunol. 2005, 6, 981–988. [Google Scholar] [CrossRef]
- Chen, S.; Luo, G.; Yang, Z.; Lin, S.; Chen, S.; Wang, S.; Goraya, M.U.; Chi, X.; Zeng, X.; Chen, J.-L. Avian Tembusu virus infection effectively triggers host innate immune response through MDA5 and TLR3-dependent signaling pathways. Vet. Res. 2016, 47, 74. [Google Scholar] [CrossRef] [PubMed]
- Yu, G.; Lin, Y.; Tang, Y.; Diao, Y. Comparative transcriptomic analysis of immune-related gene expression in duck embryo fibroblasts following duck Tembusu virus infection. Int. J. Mol. Sci. 2018, 19, 2328. [Google Scholar] [CrossRef]
- Vlassov, A.V.; Magdaleno, S.; Setterquist, R.; Conrad, R. Exosomes: Current knowledge of their composition, biological functions, and diagnostic and therapeutic potentials. Biochim. Biophys. Acta 2012, 1820, 940–948. [Google Scholar] [CrossRef] [PubMed]
- Brinton, L.T.; Sloane, H.S.; Kester, M.; Kelly, K.A. Formation and role of exosomes in cancer. Cell. Mol. Life Sci. 2015, 72, 659–671. [Google Scholar] [CrossRef] [PubMed]
- Chen, J.; Chopp, M. Exosome therapy for stroke. Stroke 2018, 49, 1083–1090. [Google Scholar] [CrossRef] [PubMed]
- Yu, L.; Yang, F.; Jiang, L.; Chen, Y.; Wang, K.; Xu, F.; Wei, Y.; Cao, X.; Wang, J.; Cai, Z. Exosomes with membrane-associated TGF-β1 from gene-modified dendritic cells inhibit murine EAE independently of MHC restriction. Eur. J. Immunol. 2013, 43, 2461–2472. [Google Scholar] [CrossRef]
- Cai, J.; Han, Y.; Ren, H.; Chen, C.; He, D.; Zhou, L.; Eisner, G.M.; Asico, L.D.; Jose, P.A.; Zeng, C. Extracellular vesicle-mediated transfer of donor genomic DNA to recipient cells is a novel mechanism for genetic influence between cells. J. Mol. Cell. Biol. 2013, 5, 227–238. [Google Scholar] [CrossRef]
- Valencia, K.; Luis-Ravelo, D.; Bovy, N.; Antón, I.; Martínez-Canarias, S.; Zandueta, C.; Ormazábal, C.; Struman, I.; Tabruyn, S.; Rebmann, V.; et al. miRNA cargo within exosome-like vesicle transfer influences metastatic bone colonization. Mol. Oncol. 2014, 8, 689–703. [Google Scholar] [CrossRef]
- Andre, F.; Schartz, N.E.; Movassagh, M.; Flament, C.; Pautier, P.; Morice, P.; Pomel, C.; Lhomme, C.; Escudier, B.; Le Chevalier, T.; et al. Malignant effusions and immunogenic tumour-derived exosomes. Lancet 2002, 360, 295–305. [Google Scholar] [CrossRef]
- Théry, C.; Zitvogel, L.; Amigorena, S. Exosomes: Composition, biogenesis, and function. Nat. Rev. Immunol. 2002, 2, 569–579. [Google Scholar] [CrossRef]
- Wang, K.; Liu, H.; He, Y.; Chen, T.; Yang, Y.; Niu, Y.; Chen, H.; Chen, Y.; Liu, J.; Ye, F.; et al. Correlation of TLR1-10 expression in peripheral blood mononuclear cells with chronic hepatitis B and chronic hepatitis B-related liver failure. Hum. Immunol. 2010, 71, 950–956. [Google Scholar] [CrossRef] [PubMed]
- Wang, L.; Li, G.; Yao, Z.Q.; Moorman, J.P.; Ning, S. MicroRNA regulation of viral immunity, latency, and carcinogenesis of selected tumor viruses and HIV. Rev. Med. Virol. 2015, 25, 320–341. [Google Scholar] [CrossRef] [PubMed]
- Zhu, K.; Huang, J.; Jia, R.; Zhang, B.; Wang, M.; Zhu, D.; Chen, S.; Liu, M.; Yin, Z.; Cheng, A. Identification and molecular characterization of a novel duck Tembusu virus isolate from Southwest China. Arch. Virol. 2015, 160, 2781–2790. [Google Scholar] [CrossRef] [PubMed]
- Li, R.; Li, N.; Zhang, J.; Wang, Y.; Liu, J.; Cai, Y.; Chai, T.; Wei, L. Expression of immune-related genes of ducks infected with Avian Pathogenic Escherichia coli (APEC). Front. Microbiol. 2016, 7, 637. [Google Scholar] [CrossRef]
- Song, K.; Chen, Z.; Huang, Y.; Li, C.; Meng, C.; Li, D.; Zhang, M.; Liu, G. Development of real-time fluorescence quantitative PCR assay for detection of duck Toll-like receptor 3. Chin. J. Anim. Infect. Dis. 2015, 3, 42–49. [Google Scholar]
- Li, N.; Wang, Y.; Li, R.; Liu, J.; Zhang, J.; Cai, Y.; Liu, S.; Chai, T.; Wei, L. Immune responses of ducks infected with duck Tembusu virus. Front. Microbiol. 2015, 6, 425. [Google Scholar] [CrossRef]
- Chen, S.; Wang, T.; Liu, P.; Yang, C.; Wang, M.; Jia, R.; Zhu, D.; Liu, M.; Yang, Q.; Wu, Y.; et al. Duck interferon regulatory factor 7 (IRF7) can control duck Tembusu virus (DTMUV) infection by triggering type I interferon production and its signal transduction pathway. Cytokine 2019, 113, 31–38. [Google Scholar] [CrossRef]
- Cui, M.; Jia, R.; Huang, J.; Wu, X.; Hu, Z.; Zhang, X.; Wang, M.; Zhu, D.; Chen, S.; Liu, M.; et al. Analysis of the microRNA expression profiles in DEF cells infected with duck Tembusu virus. Infect. Genet. Evol. 2018, 63, 126–134. [Google Scholar] [CrossRef]
- Palma-Ocampo, H.K.; Flores-Alonso, J.C.; Vallejo-Ruiz, V.; Reyes-Leyva, J.; Flores-Mendoza, L.; Herrera-Camacho, I.; Rosas-Murrieta, N.H.; Santos-López, G. Interferon lambda inhibits dengue virus replication in epithelial cells. Virol. J. 2015, 12, 150. [Google Scholar] [CrossRef]
- Pinto, S.; Cunha, C.; Barbosa, M.; Vaz, A.R.; Brites, D. Exosomes from NSC-34 cells transfected with hSOD1-G93A are enriched in miR-124 and drive alterations in microglia phenotype. Front. Neurosci. 2017, 11, 273. [Google Scholar] [CrossRef]
- Zhang, B.; Wang, A.; Xia, C.; Lin, Q.; Chen, C. A single nucleotide polymorphism in primary-microRNA-146a reduces the expression of mature microRNA-146a in patients with Alzheimer’s disease and is associated with the pathogenesis of Alzheimer’s disease. Mol. Med. Rep. 2015, 12, 4037–4042. [Google Scholar] [CrossRef] [PubMed]
- Chen, H.W.; King, K.; Tu, J.; Luster, A.D.; Shresta, S. The roles of IRF-3 and IRF-7 in innate antiviral immunity against dengue virus. J. Immunol. 2013, 191, 4194–4201. [Google Scholar] [CrossRef] [PubMed]
- Tsai, Y.T.; Chang, S.Y.; Lee, C.N.; Kao, C.L. Human TLR3 recognizes dengue virus and modulates viral replication in vitro. Cell. Microbiol. 2009, 11, 604–615. [Google Scholar] [CrossRef] [PubMed]
- Xia, J.; Winkelmann, E.R.; Gorder, S.R.; Mason, P.W.; Milligan, G.N. TLR3-and MyD88-dependent signaling differentially influences the development of West Nile virus-specific B cell responses in mice following immunization with RepliVAX WN, a single-cycle flavivirus vaccine candidate. J. Virol. 2013, 87, 12090–12101. [Google Scholar] [CrossRef] [PubMed]
- Dalrymple, N.A.; Cimica, V.; Mackow, E.R. Dengue virus NS proteins inhibit RIG-I/MAVS signaling by blocking TBK1/IRF3 phosphorylation: Dengue virus serotype 1 NS4A Is a unique interferon-regulating virulence determinant. mBio 2015, 6. [Google Scholar] [CrossRef]
- Chen, S.; Wu, Z.; Zhang, J.; Wang, M.; Jia, R.; Zhu, D.; Liu, M.; Sun, K.; Yang, Q.; Wu, Y.; et al. Duck stimulator of interferon genes plays an important role in host anti-duck plague virus infection through an IFN-dependent signalling pathway. Cytokine 2017, 102, 191–199. [Google Scholar] [CrossRef]
- Zhou, M.; Chen, J.; Zhou, L.; Chen, W.; Ding, G.; Cao, L. Pancreatic cancer derived exosomes regulate the expression of TLR4 in dendritic cells via miR-203. Cell. Immunol. 2014, 292, 65–69. [Google Scholar] [CrossRef]
- Paschon, V.; Takada, S.H.; Sousa, E.; Raeisossadati, R.; Ulrich, H.; Kihara, A.H. Interplay between exosomes, microRNAs and Toll-like receptors in brain disorders. Mol. Neurobiol. 2016, 53, 1–13. [Google Scholar] [CrossRef]
- Li, X.; Wang, S.; Zhu, R.; Li, H.; Han, Q.; Zhao, R.C. Lung tumor exosomes induce a pro-inflammatory phenotype in mesenchymal stem cells via NFκB-TLR signaling pathway. J. Hematol. Oncol. 2016, 9, 42. [Google Scholar] [CrossRef]
- Wang, J.; Lei, C.Q.; Ji, Y.; Zhou, H.; Ren, Y.; Peng, Q.; Zeng, Y.; Jia, Y.; Ge, J.; Zhong, B.; et al. Duck Tembusu Virus Nonstructural Protein 1 Antagonizes IFN-β Signaling Pathways by Targeting VISA. J. Immunol. 2016, 197, 4704–4713. [Google Scholar] [CrossRef]
- Wu, Z.; Zhang, W.; Wu, Y.Y.; Wang, T.; Wu, S.; Wang, M.; Jia, R.; Zhu, D.; Liu, M.; Zhao, X.; et al. Binding of the Duck Tembusu Virus Protease to STING Is Mediated by NS2B and Is Crucial for STING Cleavage and for Impaired Induction of IFN-β. J. Immunol. 2019, 203, 3374–3385. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Zhang, S.; Tang, Y.; Diao, Y. Screening of Duck Tembusu Virus NS3 Interacting Host Proteins and Identification of Its Specific Interplay Domains. Viruses 2019, 11, 740. [Google Scholar] [CrossRef] [PubMed]















| Primer Name | Oligonucleotide Sequence (5′→3′) | Length | References |
|---|---|---|---|
| TLR2 | F1: TCCTTCATTCAGCACCAGGC R2: GAAAAACACAGCGCAGATCA | 171 bp | [34] |
| TLR3 | F2: ATGTCATGCAAACCTGACCA R2: CCAGGGTCTTGAAAGGATCA | 239 bp | [35] |
| TLR4 | F3: ATCTTTCAAGGTGCCACATCC R3: ACTGACCTACCGATTGGACAC | 194 bp | |
| MyD88 | F4: TTTACAGCATGAATCCCTTGGC R4: TGGGAGTGTAAAATCCTGGTGT | 184 bp | |
| IFNα | F5: TCAGCACCACATCCACCACCTT R5: GGTTCTGGAGGAAGTGTTGGAT | 131 bp | |
| IFNβ | F6: TCAGCAGTCCAAGCATCCCT R6: GGAAGTGTTGGATGCTCCTGAAGTA | 186 bp | |
| IL-6 | F7: GGTCCAGAACAACCTCAACCTCC R7: CGTTGCCAGATGCTTTGTGC | 202 bp | |
| IL-8 | F8: CGGCATCGGTGTTCTTATCT R8: CTGTCCAGTGCCTTCAGTTT | 147 bp | |
| MDA5 | F9: GCTACAGAAGATAGAAGTGTCA R9: CAGGATCAGATCTGGTTCAG | 120 bp | [36] |
| IPS-1 | F10: TGCGACCGCCTACAAATTCTAT | 139 bp | |
| R10: AGGGGTTTGGTAGAGGTCGTAG | |||
| NF-κB | F11: ATCAACCCTTTTAACGTGCCT | 142 bp | |
| R11: GGTTGGAAATCAAAGGAGGC | |||
| TRIF | F12: TCTACTCACTGCTGGCAAAGG | 129 bp | |
| R12: CAGCCAGGACGCAGTTTTGTG | |||
| IRF7 | F13: ACAACGCCAGGAAGGATGT | 120 bp | |
| R13: AGCGAAAGTTGGTCTTCCACT | |||
| RIG-I | F14: GCGGATAGAGGCAACAAT | 133 bp | |
| R14: AGTTATGCCTGCTGCTTT | |||
| DTMUV envelope | F15: AATGGCTGTGGCTTGTTTGG | 207 bp | [37] |
| R15: GGGCGTTATCACGAATCTA | |||
| U6 | F16: CTCGCTTCGGCACGACA | 73 bp | [38] |
| R16: GCGTGTCATCCTTGCGC | |||
| β-action | F17: CCCCATTGAACACGGTATTGTC R17: GGCTACATACATGGCTGGGG | 199 bp | [35] |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Guo, H.; Cheng, A.; Zhang, X.; Pan, Y.; Wang, M.; Huang, J.; Zhu, D.; Chen, S.; Liu, M.; Zhao, X.; et al. DEF Cell-Derived Exosomal miR-148a-5p Promotes DTMUV Replication by Negative Regulating TLR3 Expression. Viruses 2020, 12, 94. https://doi.org/10.3390/v12010094
Guo H, Cheng A, Zhang X, Pan Y, Wang M, Huang J, Zhu D, Chen S, Liu M, Zhao X, et al. DEF Cell-Derived Exosomal miR-148a-5p Promotes DTMUV Replication by Negative Regulating TLR3 Expression. Viruses. 2020; 12(1):94. https://doi.org/10.3390/v12010094
Chicago/Turabian StyleGuo, Hongyan, Anchun Cheng, Xingcui Zhang, YuHong Pan, Mingshu Wang, Juan Huang, Dekang Zhu, Shun Chen, Mafeng Liu, Xinxin Zhao, and et al. 2020. "DEF Cell-Derived Exosomal miR-148a-5p Promotes DTMUV Replication by Negative Regulating TLR3 Expression" Viruses 12, no. 1: 94. https://doi.org/10.3390/v12010094
APA StyleGuo, H., Cheng, A., Zhang, X., Pan, Y., Wang, M., Huang, J., Zhu, D., Chen, S., Liu, M., Zhao, X., Wu, Y., Yang, Q., Zhang, S., Yu, Y., Pan, L., Tian, B., Rehman, M. U., Chen, X., Liu, Y., ... Jia, R. (2020). DEF Cell-Derived Exosomal miR-148a-5p Promotes DTMUV Replication by Negative Regulating TLR3 Expression. Viruses, 12(1), 94. https://doi.org/10.3390/v12010094






