Canine Adipose-Derived Mesenchymal Stem Cells (cAdMSCs) as a “Trojan Horse” in Vaccinia Virus Mediated Oncolytic Therapy against Canine Soft Tissue Sarcomas
Abstract
:1. Introduction
2. Materials and Methods
2.1. Ethics Statement
2.2. Viruses, Cells, and Cell Lines
2.3. Adipose-Derived Stem Cells Isolation and Culture
2.4. Viral Replication
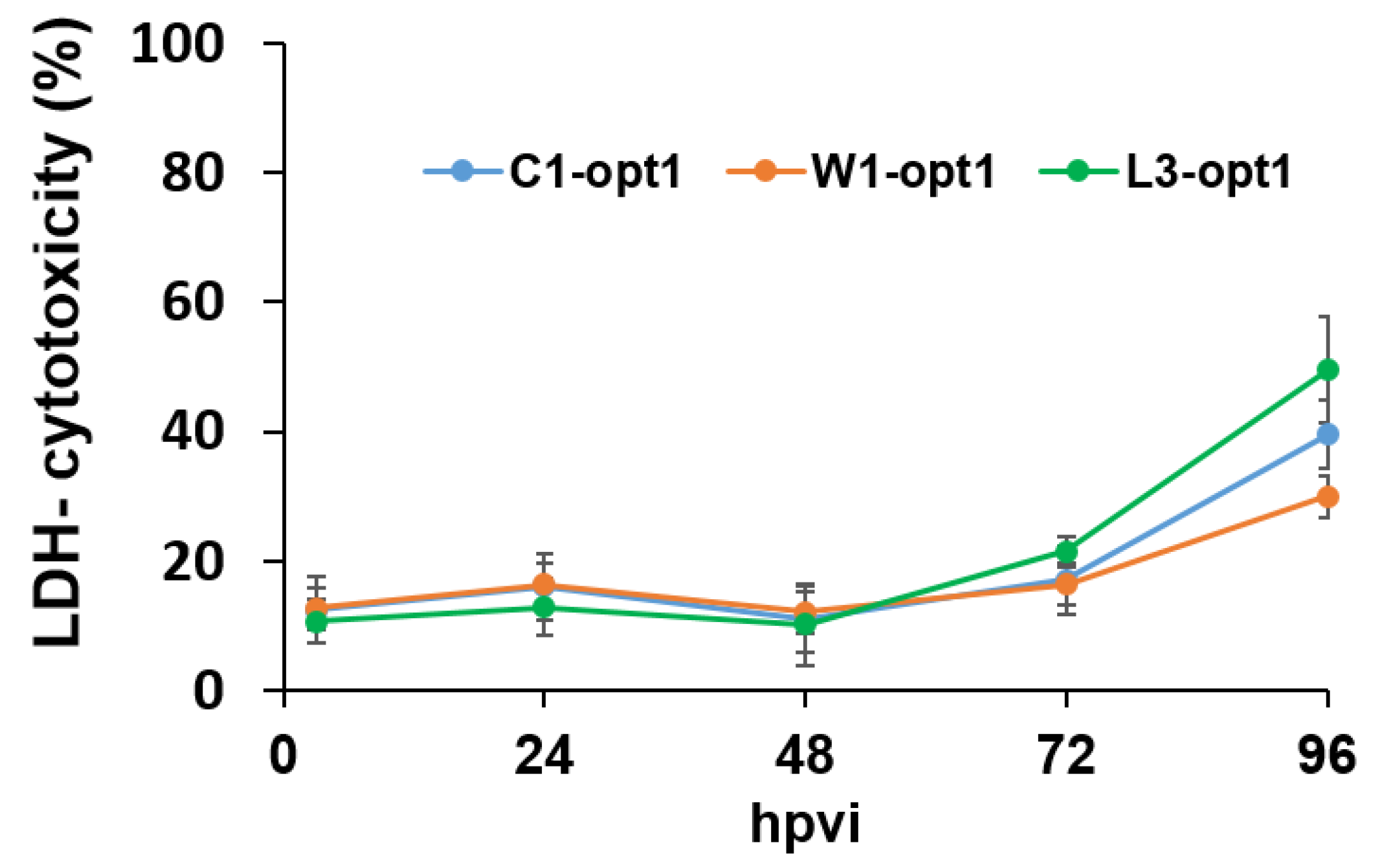
2.5. Lactate Dehydrogenase (LDH) Assay
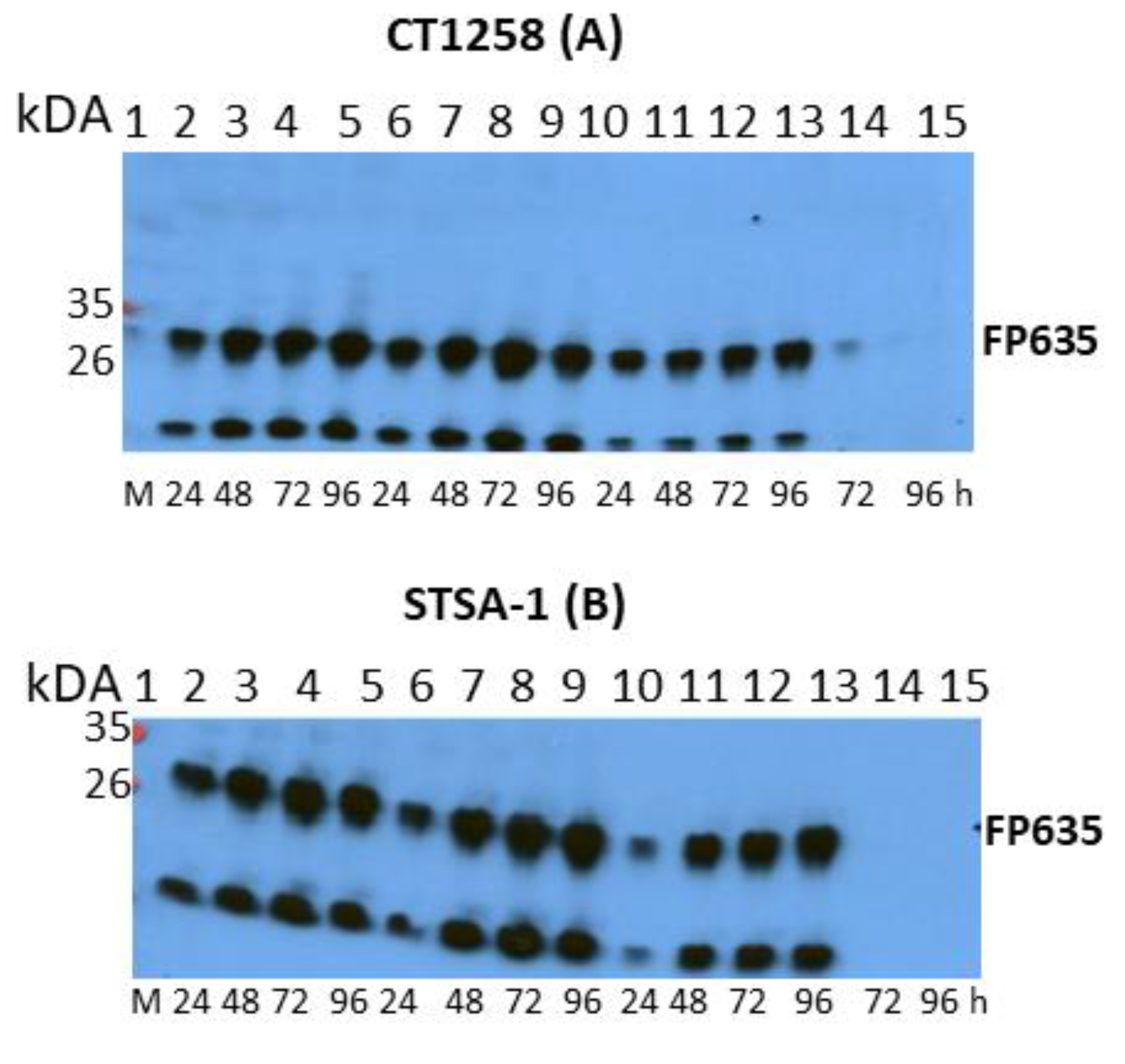
2.6. Western Blot Analysis
2.7. Animal Experiments
2.8. Histological Analysis of Tumors
2.9. PCR Detection of Vaccinia Virus in Serum of Virus-Injected Mice
3. Results
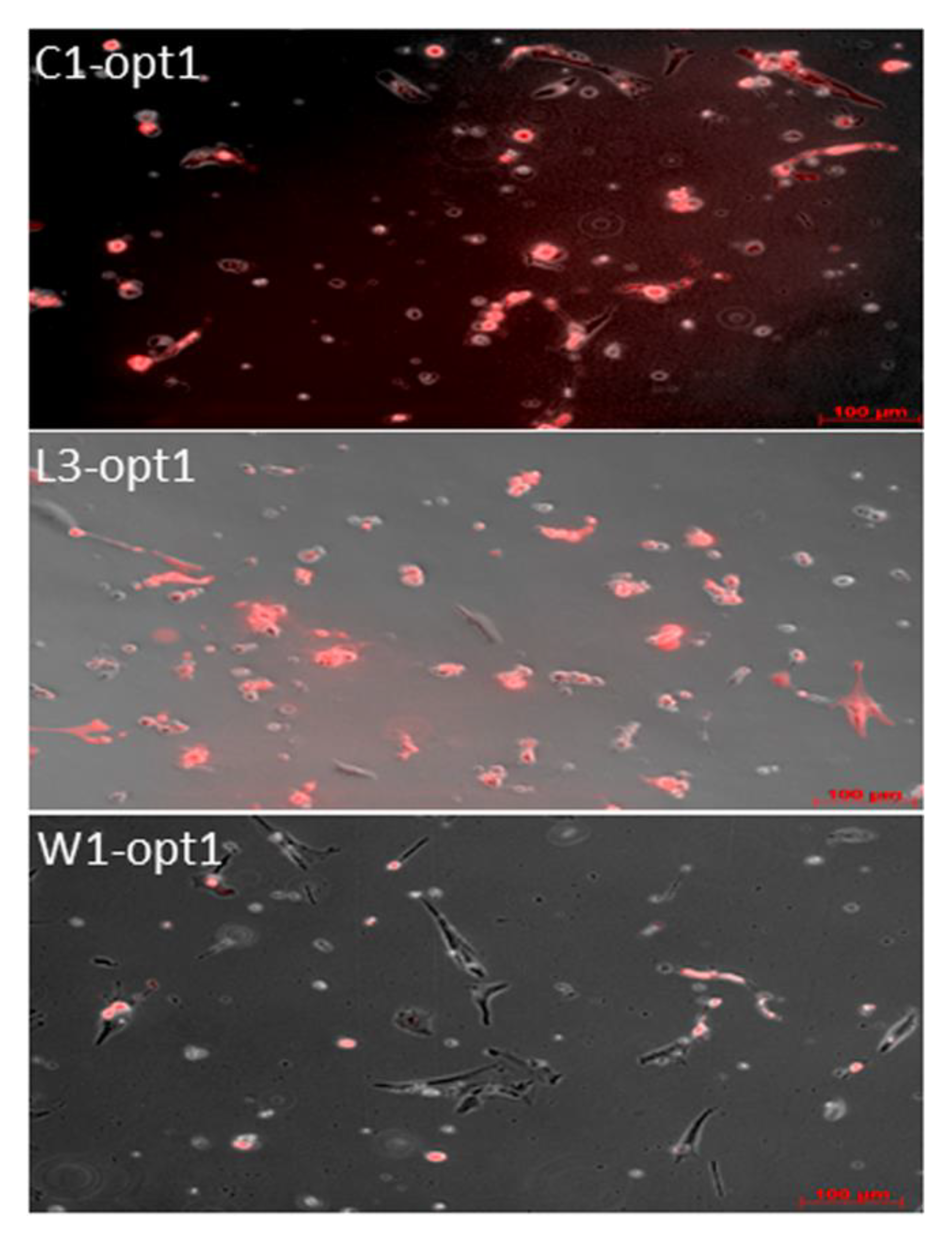
3.1. Analysis of the Oncolytic Potential of C1-opt1, W1-opt1, and L3-opt1 Vaccinia Virus Strains against Canine Cancer Cells
3.2. Canine AdMSCs Provide Potent Amplification of Vaccinia Virus
3.3. A Single Systemic Application of C1-opt1 Vaccinia Virus Alone or in Combination with cAdMSCs Causes Significant Inhibition of Tumor Growth in STSA-1 Xenografts
3.4. Virus Distribution in STSA-1 Tumor-Bearing Nude Mice after Virus Injection
4. Discussion
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Patil, S.S.; Gentschev, I.; Nolte, I.; Ogilvie, G.; Szalay, A.A. Oncolytic virotherapy in veterinary medicine: Current status and future prospects for canine patients. J. Transl. Med. 2012, 10, 3. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gentschev, I.; Patil, S.S.; Petrov, I.; Cappello, J.; Adelfinger, M.; Szalay, A.A. Oncolytic virotherapy of canine and feline cancer. Viruses 2014, 6, 2122–2137. [Google Scholar] [CrossRef] [PubMed]
- MacNeill, A.L. On the potential of oncolytic virotherapy for the treatment of canine cancers. Oncolytic Virother. 2015, 4, 95–107. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sanchez, D.; Cesarman-Maus, G.; Amador-Molina, A.; Lizano, M. Oncolytic viruses for canine cancer treatment. Cancers (Basel) 2018, 10, 404. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ong, H.T.; Hasegawa, K.; Dietz, A.B.; Russell, S.J.; Peng, K.W. Evaluation of t cells as carriers for systemic measles virotherapy in the presence of antiviral antibodies. Gene Ther. 2007, 14, 324–333. [Google Scholar] [CrossRef] [PubMed]
- Komarova, S.; Kawakami, Y.; Stoff-Khalili, M.A.; Curiel, D.T.; Pereboeva, L. Mesenchymal progenitor cells as cellular vehicles for delivery of oncolytic adenoviruses. Mol. Cancer Ther. 2006, 5, 755–766. [Google Scholar] [CrossRef] [Green Version]
- Moreno, R.; Fajardo, C.A.; Farrera-Sal, M.; Perise-Barrios, A.J.; Morales-Molina, A.; Al-Zaher, A.A.; Garcia-Castro, J.; Alemany, R. Enhanced antitumor efficacy of oncolytic adenovirus-loaded menstrual blood-derived mesenchymal stem cells in combination with peripheral blood mononuclear cells. Mol. Cancer Ther. 2019, 18, 127–138. [Google Scholar] [CrossRef] [Green Version]
- Cejalvo, T.; Perise-Barrios, A.J.; Del Portillo, I.; Laborda, E.; Rodriguez-Milla, M.A.; Cubillo, I.; Vazquez, F.; Sardon, D.; Ramirez, M.; Alemany, R.; et al. Remission of spontaneous canine tumors after systemic cellular viroimmunotherapy. Cancer Res. 2018, 78, 4891–4901. [Google Scholar] [CrossRef] [Green Version]
- Power, A.T.; Wang, J.; Falls, T.J.; Paterson, J.M.; Parato, K.A.; Lichty, B.D.; Stojdl, D.F.; Forsyth, P.A.; Atkins, H.; Bell, J.C. Carrier cell-based delivery of an oncolytic virus circumvents antiviral immunity. Mol. Ther. 2007, 15, 123–130. [Google Scholar] [CrossRef] [PubMed]
- Greish, K.; Frandsen, J.; Scharff, S.; Gustafson, J.; Cappello, J.; Li, D.; O’Malley, B.W., Jr.; Ghandehari, H. Silk-elastinlike protein polymers improve the efficacy of adenovirus thymidine kinase enzyme prodrug therapy of head and neck tumors. J. Gene Med. 2010, 12, 572–579. [Google Scholar] [CrossRef] [Green Version]
- Fujiwara, S.; Nawa, A.; Luo, C.; Kamakura, M.; Goshima, F.; Kondo, C.; Kiyono, T.; Kikkawa, F.; Nishiyama, Y. Carrier cell-based delivery of replication-competent hsv-1 mutants enhances antitumor effect for ovarian cancer. Cancer Gene Ther. 2011, 18, 77–86. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ikeda, K.; Ichikawa, T.; Wakimoto, H.; Silver, J.S.; Deisboeck, T.S.; Finkelstein, D.; Harsh, G.R.t.; Louis, D.N.; Bartus, R.T.; Hochberg, F.H.; et al. Oncolytic virus therapy of multiple tumors in the brain requires suppression of innate and elicited antiviral responses. Nat. Med. 1999, 5, 881–887. [Google Scholar] [CrossRef]
- Fisher, K.D.; Seymour, L.W. Hpma copolymers for masking and retargeting of therapeutic viruses. Adv. Drug Deliv. Rev. 2010, 62, 240–245. [Google Scholar] [CrossRef] [PubMed]
- Russell, K.A.; Chow, N.H.; Dukoff, D.; Gibson, T.W.; LaMarre, J.; Betts, D.H.; Koch, T.G. Characterization and immunomodulatory effects of canine adipose tissue- and bone marrow-derived mesenchymal stromal cells. PLoS ONE 2016, 11, e0167442. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Reich, C.M.; Raabe, O.; Wenisch, S.; Bridger, P.S.; Kramer, M.; Arnhold, S. Isolation, culture and chondrogenic differentiation of canine adipose tissue- and bone marrow-derived mesenchymal stem cells--a comparative study. Vet. Res. Commun. 2012, 36, 139–148. [Google Scholar] [CrossRef] [PubMed]
- Fritz, V.; Jorgensen, C. Mesenchymal stem cells: An emerging tool for cancer targeting and therapy. Curr. Stem Cell Res. Ther. 2008, 3, 32–42. [Google Scholar] [CrossRef]
- Scioli, M.G.; Storti, G.; D’Amico, F.; Gentile, P.; Kim, B.S.; Cervelli, V.; Orlandi, A. Adipose-derived stem cells in cancer progression: New perspectives and opportunities. Int. J. Mol. Sci. 2019, 20, 3296. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Draganov, D.D.; Santidrian, A.F.; Minev, I.; Nguyen, D.; Kilinc, M.O.; Petrov, I.; Vyalkova, A.; Lander, E.; Berman, M.; Minev, B.; et al. Delivery of oncolytic vaccinia virus by matched allogeneic stem cells overcomes critical innate and adaptive immune barriers. J. Transl. Med. 2019, 17, 100. [Google Scholar] [CrossRef] [PubMed]
- Gentschev, I.; Adelfinger, M.; Josupeit, R.; Rudolph, S.; Ehrig, K.; Donat, U.; Weibel, S.; Chen, N.G.; Yu, Y.A.; Zhang, Q.; et al. Preclinical evaluation of oncolytic vaccinia virus for therapy of canine soft tissue sarcoma. PLoS ONE 2012, 7, e37239. [Google Scholar] [CrossRef]
- Fork, M.A.; Murua Escobar, H.; Soller, J.T.; Sterenczak, K.A.; Willenbrock, S.; Winkler, S.; Dorsch, M.; Reimann-Berg, N.; Hedrich, H.J.; Bullerdiek, J.; et al. Establishing an in vivo model of canine prostate carcinoma using the new cell line ct1258. BMC Cancer 2008, 8, 240. [Google Scholar] [CrossRef] [Green Version]
- Muller, M.; Raabe, O.; Addicks, K.; Wenisch, S.; Arnhold, S. Effects of non-steroidal anti-inflammatory drugs on proliferation, differentiation and migration in equine mesenchymal stem cells. Cell Biol. Int. 2011, 35, 235–248. [Google Scholar] [CrossRef]
- Gentschev, I.; Patil, S.S.; Adelfinger, M.; Weibel, S.; Geissinger, U.; Frentzen, A.; Chen, N.G.; Yu, Y.A.; Zhang, Q.; Ogilvie, G.; et al. Characterization and evaluation of a new oncolytic vaccinia virus strain livp6.1.1 for canine cancer therapy. Bioengineered 2013, 4, 84–89. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Fischer, U.M.; Harting, M.T.; Jimenez, F.; Monzon-Posadas, W.O.; Xue, H.; Savitz, S.I.; Laine, G.A.; Cox, C.S., Jr. Pulmonary passage is a major obstacle for intravenous stem cell delivery: The pulmonary first-pass effect. Stem Cells Dev. 2009, 18, 683–692. [Google Scholar] [CrossRef]
- Schrepfer, S.; Deuse, T.; Reichenspurner, H.; Fischbein, M.P.; Robbins, R.C.; Pelletier, M.P. Stem cell transplantation: The lung barrier. Transpl. Proc. 2007, 39, 573–576. [Google Scholar] [CrossRef] [PubMed]
- Donat, U.; Weibel, S.; Hess, M.; Stritzker, J.; Hartl, B.; Sturm, J.B.; Chen, N.G.; Gentschev, I.; Szalay, A.A. Preferential colonization of metastases by oncolytic vaccinia virus strain glv-1h68 in a human pc-3 prostate cancer model in nude mice. PLoS ONE 2012, 7, e45942. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Adelfinger, M.; Bessler, S.; Frentzen, A.; Cecil, A.; Langbein-Laugwitz, J.; Gentschev, I.; Szalay, A.A. Preclinical testing oncolytic vaccinia virus strain glv-5b451 expressing an anti-vegf single-chain antibody for canine cancer therapy. Viruses 2015, 7, 4075–4092. [Google Scholar] [CrossRef] [PubMed] [Green Version]






| Antibody/Fluorochrome | Dilution | Vendor/Cat. No./Clone |
|---|---|---|
| CD45 mouse IgG2a | 1:100 | Antikoerper-online.de, ABIN2664726, UCHL1 |
| MHC II mouse IgG1 | 1:200 | Bio-Rad, MCA1085GA, CVS20 |
| CD44 rat IgG2b | 1:400 | BD Biosciences, 553131, IM7 |
| CD90 rat IgG2b | 1:50 | Bio-Rad, MCA1036G, YKIX337.217 |
| Anti-mouse polyclonal PE | 1:200 | Antikoerper-online.de, ABIN376443 |
| Anti-rat polyclonal APC | 1:600 | BD Biosciences, 551019 |
| Mouse IgG1 Isotype Control | 1:200 | Thermo Fisher Scientific, 02-6100 |
| Mouse IgG2a Isotype Control | 1:200 | Thermo Fisher Scientific, 02-6200 |
| Rat IgG2b Isotype Control | 1:800 | Thermo Fisher Scientific, 02-9288 |
| PFU/per Gram (g) of Organ | STSA-1 Xenografts Treated with C1-opt1 Alone | STSA-1 Xenografts Treated with cAdMSCs/C1-opt1 | ||||
|---|---|---|---|---|---|---|
| Mouse No | 78 | 88 | 89 | 80 | 83 | 86 |
| Tumor | 4.80 × 104 | 1.54 × 105 | 5.78 × 105 | 2.50 × 106 | 3.83 × 105 | 2.37 × 106 |
| Lung | n.d. | n.d. | n.d. | 3.45 × 102 | 3.45 × 102 | n.d |
| Liver | n.d. | n.d. | n.d. | n.d. | n.d. | n.d |
| Spleen | n.d. | n.d. | n.d. | n.d. | n.d. | n.d |
| Kidney | n.d. | n.d. | n.d. | n.d. | n.d. | n.d |
| Serum | n.d. | n.d. | n.d. | n.d. | n.d. | n.d |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Petrov, I.; Gentschev, I.; Vyalkova, A.; Elashry, M.I.; Klymiuk, M.C.; Arnhold, S.; Szalay, A.A. Canine Adipose-Derived Mesenchymal Stem Cells (cAdMSCs) as a “Trojan Horse” in Vaccinia Virus Mediated Oncolytic Therapy against Canine Soft Tissue Sarcomas. Viruses 2020, 12, 750. https://doi.org/10.3390/v12070750
Petrov I, Gentschev I, Vyalkova A, Elashry MI, Klymiuk MC, Arnhold S, Szalay AA. Canine Adipose-Derived Mesenchymal Stem Cells (cAdMSCs) as a “Trojan Horse” in Vaccinia Virus Mediated Oncolytic Therapy against Canine Soft Tissue Sarcomas. Viruses. 2020; 12(7):750. https://doi.org/10.3390/v12070750
Chicago/Turabian StylePetrov, Ivan, Ivaylo Gentschev, Anna Vyalkova, Mohamed I. Elashry, Michele C. Klymiuk, Stefan Arnhold, and Aladar A. Szalay. 2020. "Canine Adipose-Derived Mesenchymal Stem Cells (cAdMSCs) as a “Trojan Horse” in Vaccinia Virus Mediated Oncolytic Therapy against Canine Soft Tissue Sarcomas" Viruses 12, no. 7: 750. https://doi.org/10.3390/v12070750





