Prognosis of Indolent Adult T-Cell Leukemia/Lymphoma
Abstract
:1. Introduction
2. Materials and Methods
2.1. Patients
2.2. Statistical Analysis
3. Results
3.1. Patient Characteristics
3.2. Disease Progression and Therapy
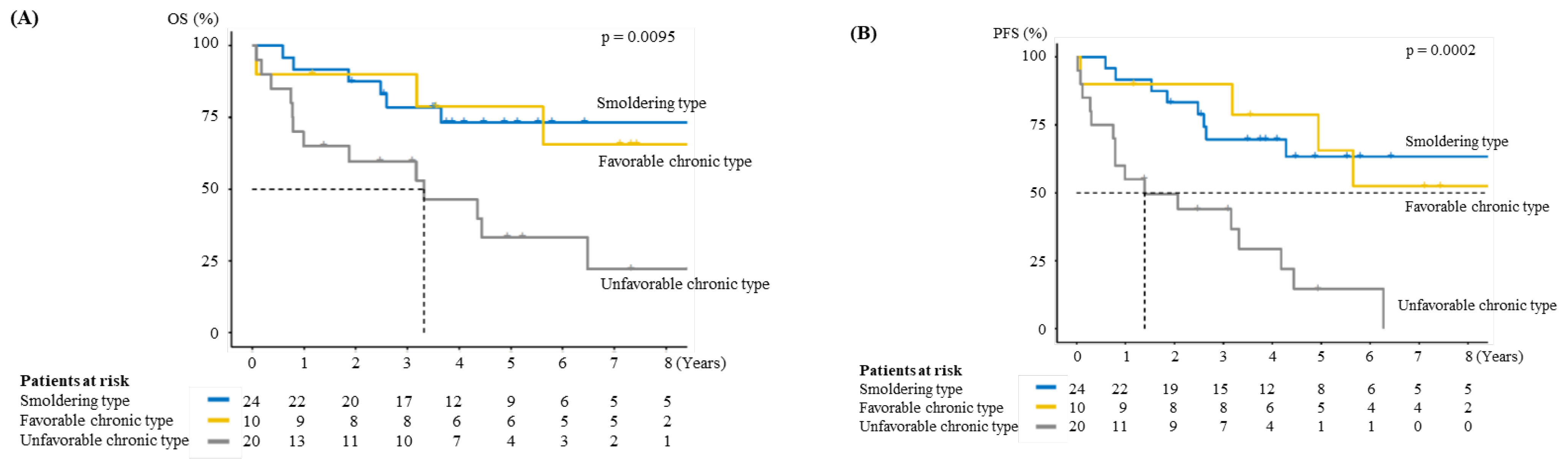
3.3. Prognosis
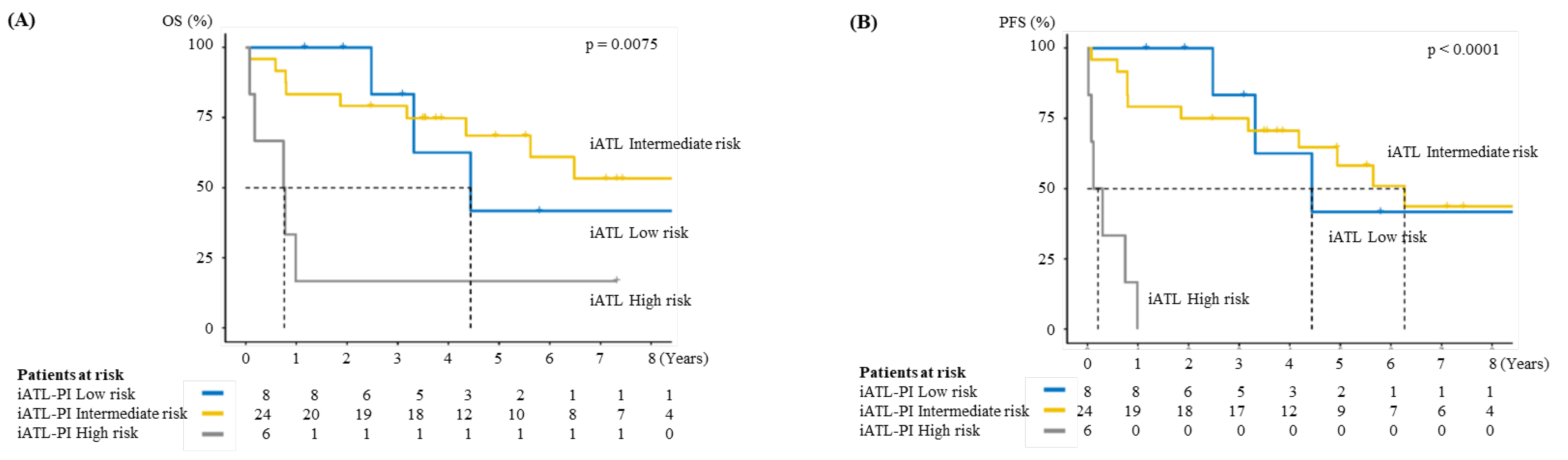
3.4. Validation of Indolent ATL-Prognostic Index (iATL-PI)
4. Discussion
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Uchiyama, T.; Yodoi, J.; Sagawa, K.; Takatsuki, K.; Uchino, H. Adult T-cell leukemia: Clinical and hematologic features of 16 cases. Blood 1977, 50, 481–492. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Vose, J.; Armitage, J.; Weisenburger, D.; International, T.C.L.P. International peripheral T-cell and natural killer/T-cell lymphoma study: Pathology findings and clinical outcomes. J. Clin. Oncol. 2008, 26, 4124–4130. [Google Scholar] [CrossRef] [PubMed]
- Swerdlow, S.H.; Campo, E.; Harris, N.L.; Jaffe, E.S.; Pileri, S.A.; Stein, H.; Thiele, J.; Vardiman, J.W. (Eds.) WHO Classification of Tumors of Haematopoietic and Lymphoid Tissues. In Adult T-Cell Leukaemia/Lymphoma; IARC Press: Lyon, France, 2008; pp. 281–284. [Google Scholar]
- Shimoyama, M. Diagnostic criteria and classification of clinical subtypes of adult T-cell leukaemia-lymphoma. A report from the Lymphoma Study Group (1984–1987). Br. J. Haematol. 1991, 79, 428–437. [Google Scholar] [CrossRef] [PubMed]
- Shimoyama, M. Chemotherapy of ATL. In Adult T-Cell Leukemia; Takatsuki, K., Ed.; Oxford University Press: Oxford, UK, 1994; pp. 221–227. [Google Scholar]
- Katsuya, H.; Shimokawa, M.; Ishitsuka, K.; Kawai, K.; Amano, M.; Utsunomiya, A.; Hino, R.; Hanada, S.; Jo, T.; Tsukasaki, K.; et al. Prognostic index for chronic- and smoldering-type adult T-cell leukemia-lymphoma. Blood 2017, 130, 39–47. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Imaizumi, Y.; Iwanaga, M.; Nosaka, K.; Ishitsuka, K.; Ishizawa, K.; Ito, S.; Amano, M.; Ishida, T.; Uike, N.; Utsunomiya, A.; et al. Prognosis of patients with adult T-cell leukemia/lymphoma in Japan: A nationwide hospital-based study. Cancer Sci. 2020, 111, 4567–4580. [Google Scholar] [CrossRef] [PubMed]
- Takasaki, Y.; Iwanaga, M.; Imaizumi, Y.; Tawara, M.; Joh, T.; Kohno, T.; Yamada, Y.; Kamihira, S.; Ikeda, S.; Miyazaki, Y.; et al. Long-term study of indolent adult T-cell leukemia-lymphoma. Blood 2010, 115, 4337–4343. [Google Scholar] [CrossRef] [PubMed]
- Sekine, M.; Kameda, T.; Shide, K.; Maeda, K.; Toyama, T.; Kawano, N.; Takeuchi, M.; Kawano, H.; Sato, S.; Ishizaki, J.; et al. Higher average chemotherapy dose intensity improves prognosis in patients with aggressive adult T-cell leukemia/lymphoma. Eur. J. Haematol. 2021, 106, 398–407. [Google Scholar] [CrossRef]
- Katsuya, H.; Ishitsuka, K.; Utsunomiya, A.; Hanada, S.; Eto, T.; Moriuchi, Y.; Saburi, Y.; Miyahara, M.; Sueoka, E.; Uike, N.; et al. Treatment and survival among 1594 patients with ATL. Blood 2015, 126, 2570–2577. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Katsuya, H.; Yamanaka, T.; Ishitsuka, K.; Utsunomiya, A.; Sasaki, H.; Hanada, S.; Eto, T.; Moriuchi, Y.; Saburi, Y.; Miyahara, M.; et al. Prognostic index for acute- and lymphoma-type adult T-cell leukemia/lymphoma. J. Clin. Oncol. 2012, 30, 1635–1640. [Google Scholar] [CrossRef] [PubMed]
- Tsukasaki, K.; Marcais, A.; Nasr, R.; Kato, K.; Fukuda, T.; Hermine, O.; Bazarbachi, A. Diagnostic Approaches and Established Treatments for Adult T Cell Leukemia Lymphoma. Front. Microbiol. 2020, 11, 1207. [Google Scholar] [CrossRef] [PubMed]
| Variable | Smoldering Type | Chronic Type | p |
|---|---|---|---|
| n | 24 | 30 | |
| Age (median [IQR]) | 75.5 (65.5, 80.5) | 73.0 [66.0, 81.0) | 0.882 |
| Sex, Female/Male, no (%) | 8/16 (33.3/66.7) | 18/12 (60.0/40.0) | 0.094 |
| ECOG PS, no (%) | 0.219 | ||
| 0 | 15 (62.5) | 15 (50.0) | |
| 1 | 6 (25.0) | 7 (23.3) | |
| 2 | 3 (12.5) | 2 (6.7) | |
| 3 | 0 (0.0) | 5 (16.7) | |
| 4 | 0 (0.0) | 1 (3.3) | |
| Skin lesion, absent/present, no (%) | 17/7 (70.8/29.2) | 21/9 (70.0/30.0) | 1 |
| Lung involvement, absent/present, no (%) | 23/1 (95.8/4.2) | 29/1 (96.7/3.3) | 1 |
| WBC count, ×109/L, median [IQR] | 6.73 (5.74, 7.48) | 12.70 (9.43, 15.19) | <0.001 |
| Neutrophil count, ×109/L, median [IQR] | 3.56 (3.01, 4.82) | 3.64 (2.72, 4.22) | 0.657 |
| Lymphocyte count, ×109/L, median [IQR] | 1.63 (1.20, 2.54) | 3.22 (1.92, 4.59) | 0.006 |
| Abnormal lymphocyte proportion, %, (median [IQR]) | 5.25 (3.00, 8.62) | 31.3 (13.3, 53.5) | <0.001 |
| Abnormal lymphocyte count, ×109/L, median [IQR] | 0.37 (0.15, 0.60) | 3.21 (2.49, 7.08) | <0.001 |
| Hemoglobin level, g/dL, median [IQR] | 12.8 (11.7, 14.3) | 12.4 (11.5, 14.0) | 0.662 |
| Platelet count, ×109/L, median [IQR] | 20.0 (17.0, 24.5) | 17.5 (13.0, 22.0) | 0.055 |
| Serum TP, g/dL, median [IQR] | 6.95 (6.68, 7.53) | 6.84 (6.30, 7.27) | 0.388 |
| Serum Alb, g/dL, median [IQR] | 3.95 (3.70, 4.15) | 4.00 (3.70, 4.21) | 0.752 |
| BUN, mg/dL, median [IQR] | 15.1 (11.0, 17.3) | 15.2 (12.6, 19.9) | 0.329 |
| Cre, mg/dL, median [IQR] | 0.80 (0.64, 1.00) | 0.70 (0.60, 0.83) | 0.251 |
| Ca, mg/dL, median [IQR] | 9.15 (9.00, 9.53) | 9.30 (9.03, 9.40) | 0.6 |
| T-Bil, mg/dL, median [IQR] | 0.60 (0.50, 0.70) | 0.70 (0.60, 0.90) | 0.056 |
| ALT, IU/L, median [IQR] | 17.5 (13.8, 24.3) | 20.0 (12.0, 27.8) | 0.889 |
| LDH, IU/L, median [IQR] | 199 (183,220) | 228 (198,291) | 0.119 |
| CRP, mg/dL, median [IQR] | 0.46 (0.10, 1.53) | 0.10 (0.04, 0.40) | 0.028 |
| sIL-2R, U/mL, median [IQR] | 1180 (930,1730) | 2190 (1300, 5040) | 0.015 |
| iATL-PI, no (%) | 0.121 | ||
| Low | 4 (30.8) | 4 (16.0) | |
| Intermediate | 9 (69.2) | 15 (60.0) | |
| High | 0 (0.0) | 6 (24.0) |
| Variable | Favorable Chronic Type | Unfavorable Chronic Type | p |
|---|---|---|---|
| n | 10 | 20 | |
| Age (median [IQR]) | 70.5 (64.0, 79.0) | 74.5 (68.0, 81.0) | 0.441 |
| Sex, Female/Male, no (%) | 8/2 (80.0/20.0) | 10/10 (50.0/50.0) | 0.236 |
| ECOG PS, no (%) | 0.312 | ||
| 0 | 7 (70.0) | 8 (40.0) | |
| 1 | 2 (20.0) | 5 (25.0) | |
| 2 | 1 (10.0) | 1 (5.0) | |
| 3 | 0 (0.0) | 5 (25.0) | |
| 4 | 0 (0.0) | 1 (5.0) | |
| Skin lesion, absent/present, no (%) | 9/1 (90.0/10.0) | 12/8 (60.0/40.0) | 0.205 |
| Lung involvement, absent/present, no (%) | 10/0 (100/0) | 19/1 (95.0/5.0) | 1 |
| WBC count, ×109/L, median [IQR] | 10.9 (8.75, 17.8) | 13.5 (9.88, 14.7) | 0.792 |
| Neutrophil count, ×109/L, median [IQR] | 3.14 (2.72, 3.99) | 3.74 (2.96, 4.37) | 0.481 |
| Lymphocyte count, ×109/L, median [IQR] | 3.84 (2.30, 5.09) | 3.03 (1.39, 4.53) | 0.455 |
| Abnormal lymphocyte proportion, %, (median [IQR]) | 23.0 (8.6, 31.6) | 40.0 (19.3, 58.0) | 0.179 |
| Abnormal lymphocyte count, ×109/L, median [IQR] | 2.72 (1.12, 4.01) | 3.83 (2.50, 7.29) | 0.356 |
| Hemoglobin level, g/dL, median [IQR] | 13.8 (11.8, 14.2) | 12.0 (11.6, 13.2) | 0.185 |
| Platelet count, ×109/L, median [IQR] | 20.0 (17.0, 22.0) | 14.5 (12.7, 20.0) | 0.333 |
| Serum TP, g/dL, median [IQR] | 7.20 (6.93, 7.38) | 6.70 (6.20, 7.03) | 0.025 |
| Serum Alb, g/dL, median [IQR] | 4.20 (4.00, 4.57) | 3.80 (3.55, 4.03) | 0.012 |
| BUN, mg/dL, median [IQR] | 12.7 (10.2, 15.3) | 18.5 (13.0, 22.9) | 0.014 |
| Cre, mg/dL, median [IQR] | 0.60 (0.52, 0.70) | 0.80 (0.67, 0.86) | 0.039 |
| Ca, mg/dL, median [IQR] | 9.25 (9.00, 9.30) | 9.30 (9.10, 9.43) | 0.58 |
| T-Bil, mg/dL, median [IQR] | 0.64 (0.60, 0.78) | 0.70 (0.65, 0.92) | 0.34 |
| ALT, IU/L, median [IQR] | 14.0 (12.0, 26.0) | 20.5 (13.5, 28.5) | 0.481 |
| LDH, IU/L, median [IQR] | 187 (161,202) | 260 (231,326) | <0.001 |
| CRP, mg/dL, median [IQR] | 0.04 (0.01, 0.06) | 0.13 (0.10, 0.57) | 0.021 |
| sIL-2R, U/mL, median [IQR] | 1595 (1308, 2020) | 4840 (1735, 9570) | 0.052 |
| iATL-PI, no (%) | 0.03 | ||
| Low | 1 (10.0) | 3 (20.0) | |
| Intermediate | 9 (90.0) | 6 (40.0) | |
| High | 0 (0.0) | 6 (40.0) |
| Variable | Univariate | Multivariate | ||
|---|---|---|---|---|
| HR (95% CI) | p | HR (95% CI) | p | |
| Age | 1.05 (0.99–1.12) | 0.110 | ||
| Sex, male vs. female | 1.02 (0.39–2.66) | 0.968 | ||
| Subtype, chronic vs. smoldering | 2.65 (0.76–9.23) | 0.126 | ||
| ECOG PS | 1.47 (1.02–2.14) | 0.041 | 1.03 (0.63–1.70) | 0.898 |
| Skin lesion, present vs. absent | 0.59 (0.19–1.82) | 0.356 | ||
| WBC count, ×109/L # | 1.06 (0.97–1.15) | 0.203 | ||
| Neutrophil count, ×109/L # | 1.08 (0.93–1.25) | 0.325 | ||
| Lymphocyte count, ×109/L # | 0.94 (0.78–1.13) | 0.509 | ||
| Abnormal lymphocyte count, ×109/L # | 1.12 (1.00–1.25) | 0.050 | 1.07 (0.90–1.27) | 0.434 |
| Hemoglobin level, g/dL | 0.89 (0.69–1.15) | 0.370 | ||
| Platelet count, ×109/L # | 1.02 (0.96–1.07) | 0.562 | ||
| Serum TP, g/dL | 0.64 (0.31–1.34) | 0.238 | ||
| Serum Alb, g/dL | 0.45 (0.18–1.14) | 0.093 | 0.42 (0.13–1.44) | 0.168 |
| BUN, mg/dL | 1.02 (0.95–1.10) | 0.586 | ||
| Cre, mg/dL | 0.91 (0.32–2.61) | 0.861 | ||
| Ca, mg/dL | 0.93 (0.40–2.18) | 0.866 | ||
| T-Bil, mg/dL | 0.72 (0.11–4.52) | 0.723 | ||
| ALT, IU/L | 1.02 (0.99–1.05) | 0.277 | ||
| LDH, ×102 IU/L # | 1.50 (0.93–2.42) | 0.101 | ||
| sIL2-R, ×103 U/mL # | 1.07 (1.03–1.11) | 0.001 | 1.06 (1.01–1.11) | 0.010 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kameda, T.; Shide, K.; Tahira, Y.; Sekine, M.; Sato, S.; Ishizaki, J.; Takeuchi, M.; Akizuki, K.; Kamiunten, A.; Shimoda, H.; et al. Prognosis of Indolent Adult T-Cell Leukemia/Lymphoma. Viruses 2022, 14, 710. https://doi.org/10.3390/v14040710
Kameda T, Shide K, Tahira Y, Sekine M, Sato S, Ishizaki J, Takeuchi M, Akizuki K, Kamiunten A, Shimoda H, et al. Prognosis of Indolent Adult T-Cell Leukemia/Lymphoma. Viruses. 2022; 14(4):710. https://doi.org/10.3390/v14040710
Chicago/Turabian StyleKameda, Takuro, Kotaro Shide, Yuki Tahira, Masaaki Sekine, Seiichi Sato, Junzo Ishizaki, Masanori Takeuchi, Keiichi Akizuki, Ayako Kamiunten, Haruko Shimoda, and et al. 2022. "Prognosis of Indolent Adult T-Cell Leukemia/Lymphoma" Viruses 14, no. 4: 710. https://doi.org/10.3390/v14040710






