Treating COVID-19 with Medicinal Plants: Is It Even Conceivable? A Comprehensive Review
Abstract
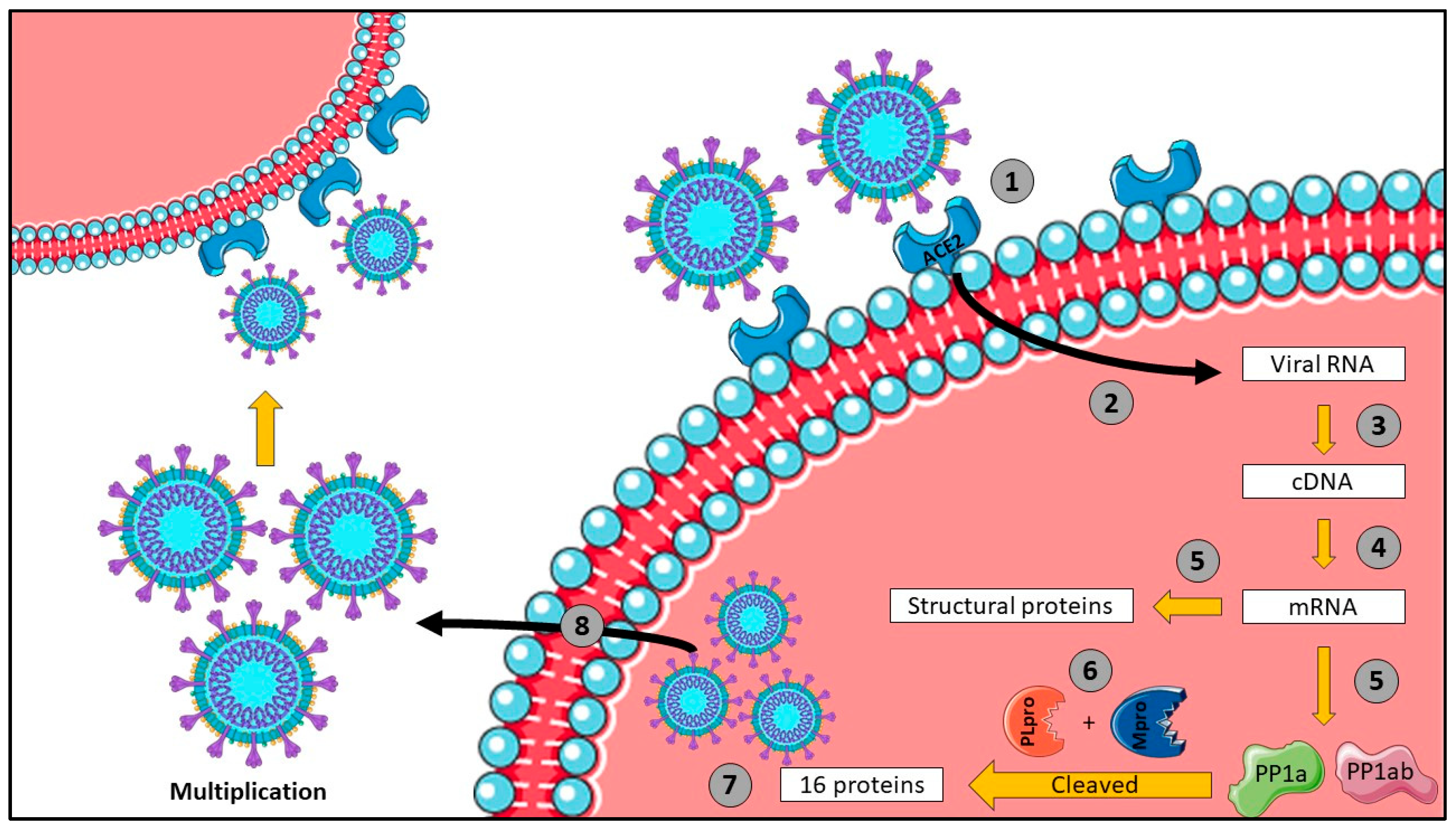
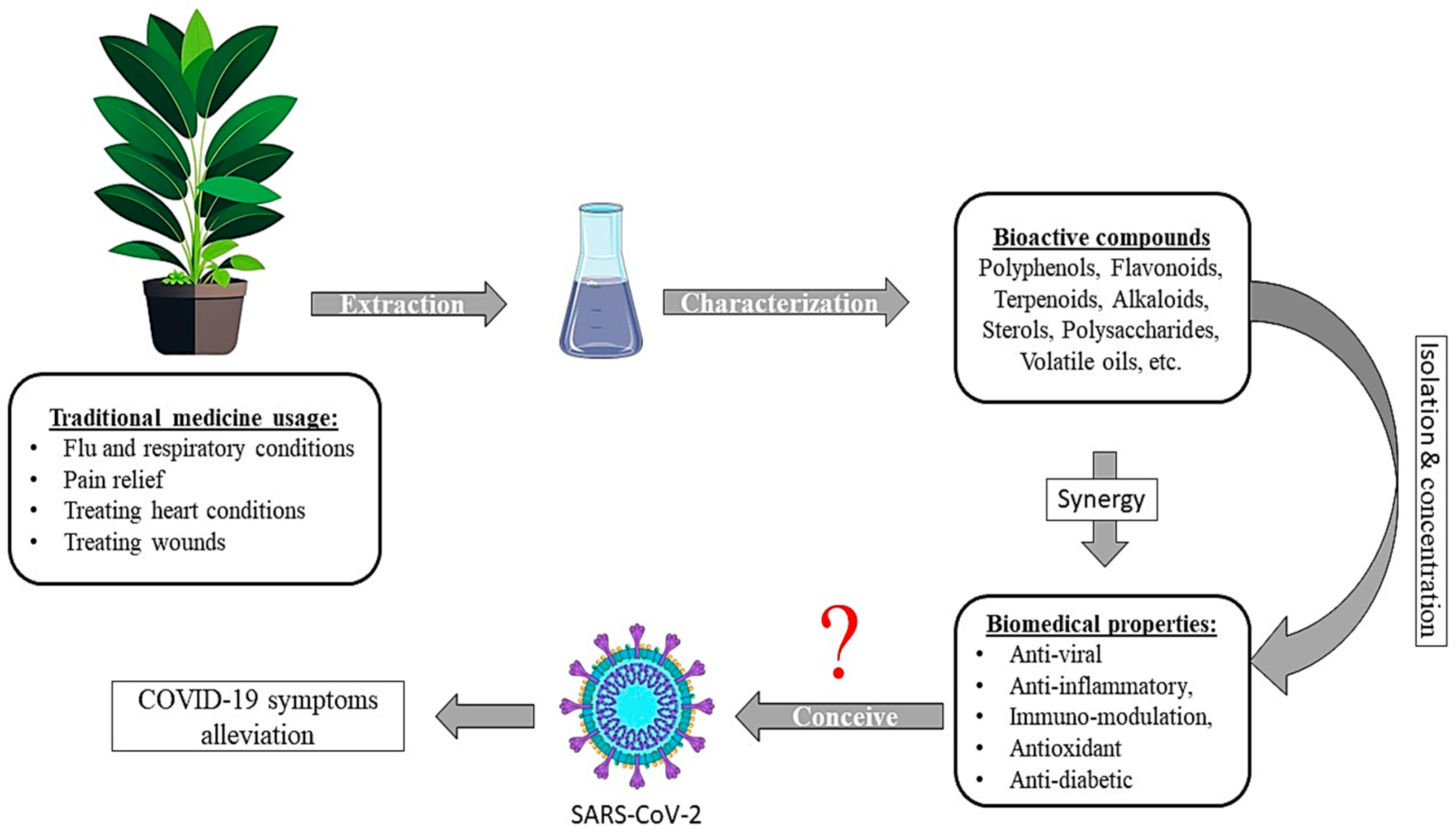
1. Introduction
2. Ginkgo biloba (G. biloba)
2.1. Inhibition of Viral Replication
2.2. Hypotensive Effect
2.3. Anti-Inflammatory Effect
3. Curcuma longa (C. longa, turmeric)
3.1. Anti-Inflammatory Activity
3.2. Anti-Coagulant Activity
3.3. Anti-Viral Properties
4. Artemisia annua (A. annua)
4.1. Anti-Malarial Activity
4.2. Anti-Viral Activity
4.3. Anti-Inflammatory and Pro-Immunogenic Properties
5. Nigella sativa (N. sativa)
5.1. Anti-Oxidant Activity
5.2. Anti-Inflammatory and Immune-Modulating Activity
5.3. Anti-Viral Properties
6. Zingiber officinale (Z. officinale)
6.1. Anti-Oxidant Activity
6.2. Anti-Viral Properties
6.3. Anti-Inflammatory and Anti-Thrombotic Activity
7. Allium sativum (A. sativum)
7.1. Anti-Inflammatory and Immuno-Modulatory Activities
7.2. Anti-Viral Activity
8. Cinnamomum verum (C. verum)
8.1. Anti-Viral Activity
8.2. Anti-Diabetic and Anti-Atherosclerotic Activity
8.3. Anti-Depressive Activity
9. Rosmarinus officinale (R. officinale)
9.1. Anti-Oxidant Activity
9.2. Anti-Viral Activity
9.3. Anti-Inflammatory and Analgesic Properties
10. Taraxacum officinale (T. officinale)
10.1. Antioxidant Activity
10.2. Antiviral Activity
11. Origanum vulgare (O. vulagre)
11.1. Antioxidant Activity
11.2. Antiviral Activity
12. Discussion
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Rahman, S.; Montero, M.T.V.; Rowe, K.; Kirton, R.; Kunik, F. Epidemiology, pathogenesis, clinical presentations, diagnosis and treatment of COVID-19: A review of current evidence. Expert Rev. Clin. Pharmacol. 2021, 14, 601–621. [Google Scholar] [CrossRef]
- Chilamakuri, R.; Agarwal, S. COVID-19: Characteristics and Therapeutics. Cells 2021, 10, 206. [Google Scholar] [CrossRef]
- Liu, Y.; Liang, C.; Xin, L.; Ren, X.; Tian, L.; Ju, X.; Li, H.; Wang, Y.; Zhao, Q.; Liu, H.; et al. The development of Coronavirus 3C-Like protease (3CLpro) inhibitors from 2010 to 2020. Eur. J. Med. Chem. 2020, 206, 112711. [Google Scholar] [CrossRef] [PubMed]
- Mousavizadeh, L.; Ghasemi, S. Genotype and phenotype of COVID-19: Their roles in pathogenesis. J. Microbiol. Immunol. Infect. 2021, 54, 159–163. [Google Scholar] [CrossRef] [PubMed]
- Li, F.; Li, W.; Farzan, M.; Harrison, S.C. Structure of SARS Coronavirus Spike Receptor-Binding Domain Complexed with Receptor. Science 2005, 309, 1864–1868. [Google Scholar] [CrossRef] [PubMed]
- Hoffmann, M.; Kleine-Weber, H.; Schroeder, S.; Krüger, N.; Herrler, T.; Erichsen, S.; Schiergens, T.S.; Herrler, G.; Wu, N.-H.; Nitsche, A.; et al. SARS-CoV-2 Cell Entry Depends on ACE2 and TMPRSS2 and Is Blocked by a Clinically Proven Protease Inhibitor. Cell 2020, 181, 271–280.e8. [Google Scholar] [CrossRef] [PubMed]
- Kim, D.; Lee, J.-Y.; Yang, J.-S.; Kim, J.W.; Kim, V.N.; Chang, H. The Architecture of SARS-CoV-2 Transcriptome. Cell 2020, 181, 914–921.e10. [Google Scholar] [CrossRef] [PubMed]
- Hamming, I.; Timens, W.; Bulthuis, M.; Lely, A.; Navis, G.; van Goor, H. Tissue distribution of ACE2 protein, the functional receptor for SARS coronavirus. A first step in understanding SARS pathogenesis. J. Pathol. 2004, 203, 631–637. [Google Scholar] [CrossRef] [PubMed]
- Guo, Y.-R.; Cao, Q.-D.; Hong, Z.-S.; Tan, Y.-Y.; Chen, S.-D.; Jin, H.-J.; Tan, K.-S.; Wang, D.-Y.; Yan, Y. The origin, transmission and clinical therapies on coronavirus disease 2019 (COVID-19) outbreak—An update on the status. Mil. Med. Res. 2020, 7, 11. [Google Scholar] [CrossRef] [PubMed]
- Acter, T.; Uddin, N.; Das, J.; Akhter, A.; Choudhury, T.R.; Kim, S. Evolution of severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) as coronavirus disease 2019 (COVID-19) pandemic: A global health emergency. Sci. Total Environ. 2020, 730, 138996. [Google Scholar] [CrossRef]
- Sazed, S.A.; Kibria, M.G.; Zamil, M.F.; Hossain, M.S.; Khan, J.Z.; Juthi, R.T.; Hossain, M.E.; Ahmed, D.; Noor, Z.; Haque, R.; et al. Direct Nasal Swab for Rapid Test and Saliva as an Alternative Biological Sample for RT-PCR in COVID-19 Diagnosis. Microbiol. Spectr. 2022, 10, e01998-22. [Google Scholar] [CrossRef]
- Sun, K.S.; Lau, T.S.M.; Yeoh, E.K.; Chung, V.C.H.; Leung, Y.S.; Yam, C.H.K.; Hung, C.T. Effectiveness of different types and levels of social distancing measures: A scoping review of global evidence from earlier stage of COVID-19 pandemic. BMJ Open 2022, 12, e053938. [Google Scholar] [CrossRef] [PubMed]
- Ndwandwe, D.; Wiysonge, C.S. COVID-19 vaccines. Curr. Opin. Immunol. 2021, 71, 111–116. [Google Scholar] [CrossRef]
- Kaur, R.J.; Dutta, S.; Bhardwaj, P.; Charan, J.; Dhingra, S.; Mitra, P.; Singh, K.; Yadav, D.; Sharma, P.; Misra, S. Adverse Events Reported From COVID-19 Vaccine Trials: A Systematic Review. Indian J. Clin. Biochem. 2021, 36, 427–439. [Google Scholar] [CrossRef]
- Sharifian-Dorche, M.; Bahmanyar, M.; Sharifian-Dorche, A.; Mohammadi, P.; Nomovi, M.; Mowla, A. Vaccine-induced immune thrombotic thrombocytopenia and cerebral venous sinus thrombosis post COVID-19 vaccination; a systematic review. J. Neurol. Sci. 2021, 428, 117607. [Google Scholar] [CrossRef]
- Blair, H.A. Remdesivir: A Review in COVID-19. Drugs 2023, 83, 1215–1237. [Google Scholar] [CrossRef] [PubMed]
- Marzolini, C.; Kuritzkes, D.R.; Marra, F.; Boyle, A.; Gibbons, S.; Flexner, C.; Pozniak, A.; Boffito, M.; Waters, L.; Burger, D.; et al. Recommendations for the Management of Drug–Drug Interactions between the COVID-19 Antiviral Nirmatrelvir/Ritonavir (Paxlovid) and Comedications. Clin Pharma Ther. 2022, 112, 1191–1200. [Google Scholar] [CrossRef] [PubMed]
- Jahan, I.; Onay, A. Potentials of plant-based substance to inhabit and probable cure for the COVID-19. Turk. J. Biol. 2020, 44, 228–241. [Google Scholar] [CrossRef]
- Al-kuraishy, H.M.; Al-Gareeb, A.I.; Kaushik, A.; Kujawska, M.; Batiha, G.E.-S. Ginkgo biloba in the management of the COVID-19 severity. Arch. Der Pharm. 2022, 355, 2200188. [Google Scholar] [CrossRef]
- Fuzimoto, A.D. An overview of the anti-SARS-CoV-2 properties of Artemisia annua, its antiviral action, protein-associated mechanisms, and repurposing for COVID-19 treatment. J. Integr. Med. 2021, 19, 375–388. [Google Scholar] [CrossRef]
- Saifulazmi, N.F.; Rohani, E.R.; Harun, S.; Bunawan, H.; Hamezah, H.S.; Nor Muhammad, N.A.; Azizan, K.A.; Ahmed, Q.U.; Fakurazi, S.; Mediani, A.; et al. A Review with Updated Perspectives on the Antiviral Potentials of Traditional Medicinal Plants and Their Prospects in Antiviral Therapy. Life 2022, 12, 1287. [Google Scholar] [CrossRef]
- Alagarasu, K.; Patil, P.; Kaushik, M.; Chowdhury, D.; Joshi, R.K.; Hegde, H.V.; Kakade, M.B.; Hoti, S.L.; Cherian, S.; Parashar, D. In Vitro Antiviral Activity of Potential Medicinal Plant Extracts Against Dengue and Chikungunya Viruses. Front. Cell. Infect. Microbiol. 2022, 12, 866452. [Google Scholar] [CrossRef]
- Yang, F.; Jiang, X.; Tariq, A.; Sadia, S.; Ahmed, Z.; Sardans, J.; Aleem, M.; Ullah, R.; Bussmann, R.W. Potential medicinal plants involved in inhibiting 3CLpro activity: A practical alternate approach to combating COVID-19. J. Integr. Med. 2022, 20, 488–496. [Google Scholar] [CrossRef] [PubMed]
- De Oliveira, J.R.; Antunes, B.S.; Do Nascimento, G.O.; Kawall, J.C.D.S.; Oliveira, J.V.B.; Silva, K.G.D.S.; Costa, M.A.D.T.; Oliveira, C.R. Antiviral activity of medicinal plant-derived products against SARS-CoV-2. Exp. Biol. Med. 2022, 247, 1797–1809. [Google Scholar] [CrossRef]
- Zrig, A. The Effect of Phytocompounds of Medicinal Plants on Coronavirus (2019-NCOV) Infection. Pharm. Chem. J. 2022, 55, 1080–1084. [Google Scholar] [CrossRef] [PubMed]
- Ćavar Zeljković, S.; Schadich, E.; Džubák, P.; Hajdúch, M.; Tarkowski, P. Antiviral Activity of Selected Lamiaceae Essential Oils and Their Monoterpenes Against SARS-CoV-2. Front. Pharmacol. 2022, 13, 893634. [Google Scholar] [CrossRef] [PubMed]
- Le-Trilling, V.T.K.; Mennerich, D.; Schuler, C.; Sakson, R.; Lill, J.K.; Kasarla, S.S.; Kopczynski, D.; Loroch, S.; Flores-Martinez, Y.; Katschinski, B.; et al. Identification of herbal teas and their compounds eliciting antiviral activity against SARS-CoV-2 in vitro. BMC Biol. 2022, 20, 264. [Google Scholar] [CrossRef]
- Das, K. Herbal plants as immunity modulators against COVID-19: A primary preventive measure during home quarantine. J. Herb. Med. 2022, 32, 100501. [Google Scholar] [CrossRef] [PubMed]
- Liu, L.; Wang, Y.; Zhang, J.; Wang, S. Advances in the chemical constituents and chemical analysis of Ginkgo biloba leaf, extract, and phytopharmaceuticals. J. Pharm. Biomed. Anal. 2021, 193, 113704. [Google Scholar] [CrossRef] [PubMed]
- van Beek, T.A. Chemical analysis of Ginkgo biloba leaves and extracts. J. Chromatogr. A 2002, 967, 21–55. [Google Scholar] [CrossRef]
- Borenstein, R.; Hanson, B.A.; Markosyan, R.M.; Gallo, E.S.; Narasipura, S.D.; Bhutta, M.; Shechter, O.; Lurain, N.S.; Cohen, F.S.; Al-Harthi, L.; et al. Ginkgolic acid inhibits fusion of enveloped viruses. Sci. Rep. 2020, 10, 4746. [Google Scholar] [CrossRef]
- Haruyama, T.; Nagata, K. Anti-influenza virus activity of Ginkgo biloba leaf extracts. J. Nat. Med. 2013, 67, 636–642. [Google Scholar] [CrossRef]
- Xiong, Y.; Zhu, G.-H.; Wang, H.-N.; Hu, Q.; Chen, L.-L.; Guan, X.-Q.; Li, H.-L.; Chen, H.-Z.; Tang, H.; Ge, G.-B. Discovery of naturally occurring inhibitors against SARS-CoV-2 3CLpro from Ginkgo biloba leaves via large-scale screening. Fitoterapia 2021, 152, 104909. [Google Scholar] [CrossRef]
- Chen, Z.; Cui, Q.; Cooper, L.; Zhang, P.; Lee, H.; Chen, Z.; Wang, Y.; Liu, X.; Rong, L.; Du, R. Ginkgolic acid and anacardic acid are specific covalent inhibitors of SARS-CoV-2 cysteine proteases. Cell Biosci. 2021, 11, 45. [Google Scholar] [CrossRef]
- He, J.; Hu, L.; Huang, X.; Wang, C.; Zhang, Z.; Wang, Y.; Zhang, D.; Ye, W. Potential of coronavirus 3C-like protease inhibitors for the development of new anti-SARS-CoV-2 drugs: Insights from structures of protease and inhibitors. Int. J. Antimicrob. Agents 2020, 56, 106055. [Google Scholar] [CrossRef]
- Cherrak, S.A.; Merzouk, H.; Mokhtari-Soulimane, N. Potential bioactive glycosylated flavonoids as SARS-CoV-2 main protease inhibitors: A molecular docking and simulation studies. PLoS ONE 2020, 15, e0240653. [Google Scholar] [CrossRef]
- Chen, Y.; Guo, Y.; Pan, Y.; Zhao, Z.J. Structure analysis of the receptor binding of 2019-nCoV. Biochem. Biophys. Res. Commun. 2020, 525, 135–140. [Google Scholar] [CrossRef] [PubMed]
- Derosa, G.; Maffioli, P.; D’Angelo, A.; Di Pierro, F. A role for quercetin in coronavirus disease 2019 (COVID-19). Phytother. Res. 2021, 35, 1230–1236. [Google Scholar] [CrossRef] [PubMed]
- Khazdair, M.; Anaeigoudari, A.; Agbor, G. Anti-viral and anti-inflammatory effects of kaempferol and quercetin and COVID-2019: A scoping review. Asian Pac. J. Trop. Biomed. 2021, 11, 327. [Google Scholar] [CrossRef]
- El-Arif, G.; Khazaal, S.; Farhat, A.; Harb, J.; Annweiler, C.; Wu, Y.; Cao, Z.; Kovacic, H.; Abi Khattar, Z.; Fajloun, Z.; et al. Angiotensin II Type I Receptor (AT1R): The Gate towards COVID-19-Associated Diseases. Molecules 2022, 27, 2048. [Google Scholar] [CrossRef] [PubMed]
- Al-kuraishy, H.M.; Al-Gareeb, A.I.; Alblihed, M.; Guerreiro, S.G.; Cruz-Martins, N.; Batiha, G.E.-S. COVID-19 in Relation to Hyperglycemia and Diabetes Mellitus. Front. Cardiovasc. Med. 2021, 8, 644095. [Google Scholar] [CrossRef]
- South, A.M.; Brady, T.M.; Flynn, J.T. ACE2 (Angiotensin-Converting Enzyme 2), COVID-19, and ACE Inhibitor and Ang II (Angiotensin II) Receptor Blocker Use During the Pandemic: The Pediatric Perspective. Hypertension 2020, 76, 16–22. [Google Scholar] [CrossRef]
- Jiang, H.; Qu, P. Effects of Ginkgo biloba leaf extract on local renin-angiotensin system through TLR4/NF-κB pathway in cardiac myocyte. Exp. Ther. Med. 2017, 14, 5857–5862. [Google Scholar] [CrossRef][Green Version]
- Liskova, A.; Samec, M.; Koklesova, L.; Samuel, S.M.; Zhai, K.; Al-Ishaq, R.K.; Abotaleb, M.; Nosal, V.; Kajo, K.; Ashrafizadeh, M.; et al. Flavonoids against the SARS-CoV-2 induced inflammatory storm. Biomed. Pharmacother. 2021, 138, 111430. [Google Scholar] [CrossRef] [PubMed]
- Freeman, T.L.; Swartz, T.H. Targeting the NLRP3 Inflammasome in Severe COVID-19. Front. Immunol. 2020, 11, 1518. [Google Scholar] [CrossRef]
- Saeedi-Boroujeni, A.; Mahmoudian-Sani, M.-R. Anti-inflammatory potential of Quercetin in COVID-19 treatment. J. Inflamm. 2021, 18, 3. [Google Scholar] [CrossRef] [PubMed]
- Thibane, V.S.; Ndhlala, A.R.; Finnie, J.F.; Van Staden, J. Modulation of the enzyme activity of secretory phospholipase A2, lipoxygenase and cyclooxygenase involved in inflammation and disease by extracts from some medicinal plants used for skincare and beauty. S. Afr. J. Bot. 2019, 120, 198–203. [Google Scholar] [CrossRef]
- Shah, P.M.; Priya, V.V.; Gayathri, R. Quercetin-a flavonoid: A systematic review. J. Pharm. Sci. Res. 2016, 8, 878. [Google Scholar]
- Colunga Biancatelli RM, L.; Berrill, M.; Catravas, J.D.; Marik, P.E. Quercetin and Vitamin C: An Experimental, Synergistic Therapy for the Prevention and Treatment of SARS-CoV-2 Related Disease (COVID-19). Front. Immunol. 2020, 11, 1451. [Google Scholar] [CrossRef]
- Rajagopal, K.; Varakumar, P.; Baliwada, A.; Byran, G. Activity of phytochemical constituents of Curcuma longa (turmeric) and Andrographis paniculata against coronavirus (COVID-19): An in silico approach. Futur. J. Pharm. Sci. 2020, 6, 104. [Google Scholar] [CrossRef]
- Suresh, M.V.; Francis, S.; Aktay, S.; Kralovich, G.; Raghavendran, K. Therapeutic potential of curcumin in ARDS and COVID-19. Clin. Exp. Pharmacol. Physiol. 2023, 50, 267–276. [Google Scholar] [CrossRef]
- Hewlings, S.; Kalman, D. Curcumin: A Review of Its Effects on Human Health. Foods 2017, 6, 92. [Google Scholar] [CrossRef]
- Pawar, K.S.; Mastud, R.N.; Pawar, S.K.; Pawar, S.S.; Bhoite, R.R.; Bhoite, R.R.; Kulkarni, M.V.; Deshpande, A.R. Oral Curcumin with Piperine as Adjuvant Therapy for the Treatment of COVID-19: A Randomized Clinical Trial. Front. Pharmacol. 2021, 12, 669362. [Google Scholar] [CrossRef]
- Suresh, M.V.; Wagner, M.C.; Rosania, G.R.; Stringer, K.A.; Min, K.A.; Risler, L.; Shen, D.D.; Georges, G.E.; Reddy, A.T.; Parkkinen, J.; et al. Pulmonary Administration of a Water-Soluble Curcumin Complex Reduces Severity of Acute Lung Injury. Am. J. Respir. Cell Mol. Biol. 2012, 47, 280–287. [Google Scholar] [CrossRef]
- Liu, Z.; Ying, Y. The Inhibitory Effect of Curcumin on Virus-Induced Cytokine Storm and Its Potential Use in the Associated Severe Pneumonia. Front. Cell Dev. Biol. 2020, 8, 479. [Google Scholar] [CrossRef] [PubMed]
- Vahedian-Azimi, A.; Abbasifard, M.; Rahimi-Bashar, F.; Guest, P.C.; Majeed, M.; Mohammadi, A.; Banach, M.; Jamialahmadi, T.; Sahebkar, A. Effectiveness of Curcumin on Outcomes of Hospitalized COVID-19 Patients: A Systematic Review of Clinical Trials. Nutrients 2022, 14, 256. [Google Scholar] [CrossRef] [PubMed]
- Thimmulappa, R.K.; Mudnakudu-Nagaraju, K.K.; Shivamallu, C.; Subramaniam, K.J.T.; Radhakrishnan, A.; Bhojraj, S.; Kuppusamy, G. Antiviral and immunomodulatory activity of curcumin: A case for prophylactic therapy for COVID-19. Heliyon 2021, 7, e06350. [Google Scholar] [CrossRef] [PubMed]
- Muchtaridi, M.; Amirah, S.R.; Harmonis, J.A.; Ikram, E.H.K. Role of Nuclear Factor Erythroid 2 (Nrf2) in the Recovery of Long COVID-19 Using Natural Antioxidants: A Systematic Review. Antioxidants 2022, 11, 1551. [Google Scholar] [CrossRef] [PubMed]
- Saber-Moghaddam, N.; Salari, S.; Hejazi, S.; Amini, M.; Taherzadeh, Z.; Eslami, S.; Rezayat, S.M.; Jaafari, M.R.; Elyasi, S. Oral nano-curcumin formulation efficacy in management of mild to moderate hospitalized coronavirus disease-19 patients: An open label nonrandomized clinical trial. Phytother. Res. 2021, 35, 2616–2623. [Google Scholar] [CrossRef]
- Keihanian, F.; Saeidinia, A.; Bagheri, R.K.; Johnston, T.P.; Sahebkar, A. Curcumin, hemostasis, thrombosis, and coagulation. J. Cell. Physiol. 2018, 233, 4497–4511. [Google Scholar] [CrossRef]
- Kumar, G.; Kumar, D.; Singh, N.P. Therapeutic Approach against 2019-nCoV by Inhibition of ACE-2 Receptor. Drug Res. 2021, 71, 213–218. [Google Scholar] [CrossRef]
- Patel, A.; Rajendran, M.; Shah, A.; Patel, H.; Pakala, S.B.; Karyala, P. Virtual screening of curcumin and its analogs against the spike surface glycoprotein of SARS-CoV-2 and SARS-CoV. J. Biomol. Struct. Dyn. 2022, 40, 5138–5146. [Google Scholar] [CrossRef]
- Shanmugarajan, D.; Prabitha, P.; Kumar, B.P.; Suresh, B. Curcumin to inhibit binding of spike glycoprotein to ACE2 receptors: Computational modelling, simulations, and ADMET studies to explore curcuminoids against novel SARS-CoV-2 targets. RSC Adv. 2020, 10, 31385–31399. [Google Scholar] [CrossRef] [PubMed]
- Dhar, S.; Bhattacharjee, P. Promising role of curcumin against viral diseases emphasizing COVID-19 management: A review on the mechanistic insights with reference to host-pathogen interaction and immunomodulation. J. Funct. Foods 2021, 82, 104503. [Google Scholar] [CrossRef]
- Zhang, H.; Penninger, J.M.; Li, Y.; Zhong, N.; Slutsky, A.S. Angiotensin-converting enzyme 2 (ACE2) as a SARS-CoV-2 receptor: Molecular mechanisms and potential therapeutic target. Intensive Care Med. 2020, 46, 586–590. [Google Scholar] [CrossRef]
- Wrapp, D.; Wang, N.; Corbett, K.S.; Goldsmith, J.A.; Hsieh, C.-L.; Abiona, O.; Graham, B.S.; McLellan, J.S. Cryo-EM structure of the 2019-nCoV spike in the prefusion conformation. Science 2020, 367, 1260–1263. [Google Scholar] [CrossRef]
- Abodunrin, O.P.; Onifade, O.F.; Adegboyega, A.E. Therapeutic capability of five active compounds in typical African medicinal plants against main proteases of SARS-CoV-2 by computational approach. Inform. Med. Unlocked 2022, 31, 100964. [Google Scholar] [CrossRef] [PubMed]
- Septembre-Malaterre, A.; Lalarizo Rakoto, M.; Marodon, C.; Bedoui, Y.; Nakab, J.; Simon, E.; Hoarau, L.; Savriama, S.; Strasberg, D.; Guiraud, P.; et al. Artemisia annua, a Traditional Plant Brought to Light. Int. J. Mol. Sci. 2020, 21, 4986. [Google Scholar] [CrossRef] [PubMed]
- Sehailia, M.; Chemat, S. Antimalarial-agent artemisinin and derivatives portray more potent binding to Lys353 and Lys31-binding hotspots of SARS-CoV-2 spike protein than hydroxychloroquine: Potential repurposing of artenimol for COVID-19. J. Biomol. Struct. Dyn. 2021, 39, 6184–6194. [Google Scholar] [CrossRef]
- Cao, R.; Hu, H.; Li, Y.; Wang, X.; Xu, M.; Liu, J.; Zhang, H.; Yan, Y.; Zhao, L.; Li, W.; et al. Anti-SARS-CoV-2 Potential of Artemisinins In Vitro. ACS Infect. Dis. 2020, 6, 2524–2531. [Google Scholar] [CrossRef]
- Nair, M.S.; Huang, Y.; Fidock, D.A.; Polyak, S.J.; Wagoner, J.; Towler, M.J.; Weathers, P.J. Artemisia annua L. extracts inhibit the in vitro replication of SARS-CoV-2 and two of its variants. J. Ethnopharmacol. 2021, 274, 114016. [Google Scholar] [CrossRef]
- Efferth, T.; Romero, M.R.; Wolf, D.G.; Stamminger, T.; Marin, J.J.G.; Marschall, M. The Antiviral Activities of Artemisinin and Artesunate. Clin. Infect. Dis. 2008, 47, 804–811. [Google Scholar] [CrossRef]
- Das, S.; Sarmah, S.; Lyndem, S.; Singha Roy, A. An investigation into the identification of potential inhibitors of SARS-CoV-2 main protease using molecular docking study. J. Biomol. Struct. Dyn. 2021, 39, 3347–3357. [Google Scholar] [CrossRef]
- Sharma, S.; Deep, S. In-silico drug repurposing for targeting SARS-CoV-2 main protease (Mpro). J. Biomol. Struct. Dyn. 2022, 40, 3003–3010. [Google Scholar] [CrossRef] [PubMed]
- Orege, J.I.; Adeyemi, S.B.; Tiamiyu, B.B.; Akinyemi, T.O.; Ibrahim, Y.A.; Orege, O.B. Artemisia and Artemisia-based products for COVID-19 management: Current state and future perspective. Adv. Tradit. Med. 2023, 23, 85–96. [Google Scholar] [CrossRef]
- Maideen, N.M.P. Prophetic Medicine-Nigella Sativa (Black cumin seeds)—Potential herb for COVID-19? J. Pharmacopunct. 2020, 23, 62–70. [Google Scholar] [CrossRef]
- Akram Khan, M.; Afzal, M. Chemical composition of Nigella sativa Linn: Part 2 Recent advances. Inflammopharmacology 2016, 24, 67–79. [Google Scholar] [CrossRef] [PubMed]
- Delgado-Roche, L.; Mesta, F. Oxidative Stress as Key Player in Severe Acute Respiratory Syndrome Coronavirus (SARS-CoV) Infection. Arch. Med. Res. 2020, 51, 384–387. [Google Scholar] [CrossRef] [PubMed]
- Namazi, N.; Mahdavi, R.; Alizadeh, M.; Farajnia, S. Oxidative Stress Responses to Nigella sativa Oil Concurrent with a Low-Calorie Diet in Obese Women: A Randomized, Double-Blind Controlled Clinical Trial. Phytother. Res. 2015, 29, 1722–1728. [Google Scholar] [CrossRef]
- Burits, M.; Bucar, F. Antioxidant activity of Nigella sativa essential oil. Phytother. Res. 2000, 14, 323–328. [Google Scholar] [CrossRef]
- Mashayekhi-Sardoo, H.; Rezaee, R.; Karimi, G. An overview of in vivo toxicological profile of thymoquinone. Toxin Rev. 2020, 39, 115–122. [Google Scholar] [CrossRef]
- Khazdair, M.R.; Ghafari, S.; Sadeghi, M. Possible therapeutic effects of Nigella sativa and its thymoquinone on COVID-19. Pharm. Biol. 2021, 59, 694–701. [Google Scholar] [CrossRef] [PubMed]
- Mahdavi, R.; Namazi, N.; Alizadeh, M.; Farajnia, S. Nigella sativa oil with a calorie-restricted diet can improve biomarkers of systemic inflammation in obese women: A randomized double-blind, placebo-controlled clinical trial. J. Clin. Lipidol. 2016, 10, 1203–1211. [Google Scholar] [CrossRef] [PubMed]
- Hajhashemi, V.; Ghannadi, A.; Jafarabadi, H. Black cumin seed essential oil, as a potent analgesic and antiinflammatory drug. Phytother. Res. 2004, 18, 195–199. [Google Scholar] [CrossRef] [PubMed]
- V’kovski, P.; Gerber, M.; Kelly, J.; Pfaender, S.; Ebert, N.; Braga Lagache, S.; Simillion, C.; Portmann, J.; Stalder, H.; Gaschen, V.; et al. Determination of host proteins composing the microenvironment of coronavirus replicase complexes by proximity-labeling. eLife 2019, 8, e42037. [Google Scholar] [CrossRef] [PubMed]
- Frieman, M.; Yount, B.; Agnihothram, S.; Page, C.; Donaldson, E.; Roberts, A.; Vogel, L.; Woodruff, B.; Scorpio, D.; Subbarao, K.; et al. Molecular Determinants of Severe Acute Respiratory Syndrome Coronavirus Pathogenesis and Virulence in Young and Aged Mouse Models of Human Disease. J. Virol. 2012, 86, 884–897. [Google Scholar] [CrossRef] [PubMed]
- Deng, X.; Hackbart, M.; Mettelman, R.C.; O’Brien, A.; Mielech, A.M.; Yi, G.; Kao, C.C.; Baker, S.C. Coronavirus nonstructural protein 15 mediates evasion of dsRNA sensors and limits apoptosis in macrophages. Proc. Natl. Acad. Sci. USA 2017, 114, E4251–E4260. [Google Scholar] [CrossRef]
- Baig, A.; Srinivasan, H. SARS-CoV-2 Inhibitors from Nigella sativa. Appl. Biochem. Biotechnol. 2022, 194, 1051–1090. [Google Scholar] [CrossRef]
- Abdelli, I.; Hassani, F.; Bekkel Brikci, S.; Ghalem, S. In silico study the inhibition of angiotensin converting enzyme 2 receptor of COVID-19 by Ammoides verticillata components harvested from Western Algeria. J. Biomol. Struct. Dyn. 2021, 39, 3263–3276. [Google Scholar] [CrossRef]
- Kulkarni, S.A.; Nagarajan, S.K.; Ramesh, V.; Palaniyandi, V.; Selvam, S.P.; Madhavan, T. Computational evaluation of major components from plant essential oils as potent inhibitors of SARS-CoV-2 spike protein. J. Mol. Struct. 2020, 1221, 128823. [Google Scholar] [CrossRef]
- Elfiky, A.A. Natural products may interfere with SARS-CoV-2 attachment to the host cell. J. Biomol. Struct. Dyn. 2021, 39, 3194–3203. [Google Scholar] [CrossRef]
- Ahmad, S.; Abbasi, H.W.; Shahid, S.; Gul, S.; Abbasi, S.W. Molecular docking, simulation and MM-PBSA studies of nigella sativa compounds: A computational quest to identify potential natural antiviral for COVID-19 treatment. J. Biomol. Struct. Dyn. 2021, 39, 4225–4233. [Google Scholar] [CrossRef]
- Jakhmola Mani, R.; Sehgal, N.; Dogra, N.; Saxena, S.; Pande Katare, D. Deciphering underlying mechanism of SARS-CoV-2 infection in humans and revealing the therapeutic potential of bioactive constituents from Nigella sativa to combat COVID19: In-silico study. J. Biomol. Struct. Dyn. 2022, 40, 2417–2429. [Google Scholar] [CrossRef]
- Koshak, D.A.E.; Koshak, P.E.A. Nigella sativa L as a potential phytotherapy for coronavirus disease 2019: A mini review of in silico studies. Curr. Ther. Res. 2020, 93, 100602. [Google Scholar] [CrossRef]
- Kiyama, R. Nutritional implications of ginger: Chemistry, biological activities and signaling pathways. J. Nutr. Biochem. 2020, 86, 108486. [Google Scholar] [CrossRef] [PubMed]
- Jafarzadeh, A.; Jafarzadeh, S.; Nemati, M. Therapeutic potential of ginger against COVID-19: Is there enough evidence? J. Tradit. Chin. Med. Sci. 2021, 8, 267–279. [Google Scholar] [CrossRef]
- Ma, R.-H.; Ni, Z.-J.; Zhu, Y.-Y.; Thakur, K.; Zhang, F.; Zhang, Y.-Y.; Hu, F.; Zhang, J.-G.; Wei, Z.-J. A recent update on the multifaceted health benefits associated with ginger and its bioactive components. Food Funct. 2021, 12, 519–542. [Google Scholar] [CrossRef] [PubMed]
- Tanweer, S.; Mehmood, T.; Zainab, S.; Ahmad, Z.; Shehzad, A. Comparison and HPLC quantification of antioxidant profiling of ginger rhizome, leaves and flower extracts. Clin. Phytoscience 2020, 6, 12. [Google Scholar] [CrossRef]
- Mošovská, S.; Nováková, D.; Kaliňák, M. Antioxidant activity of ginger extract and identification of its active components. Acta Chim. Slovaca 2015, 8, 115–119. [Google Scholar] [CrossRef]
- Guo, J.; Wu, H.; Du, L.; Zhang, W.; Yang, J. Comparative Antioxidant Properties of Some Gingerols and Shogaols, and the Relationship of Their Contents with the Antioxidant Potencies of Fresh and Dried Ginger (Zingiber officinale Roscoe). J. Agric. Sci. Technol. 2014, 16, 1063–1072. [Google Scholar]
- Shanmugam, K.R.; Mallikarjuna, K.; Kesireddy, N.; Sathyavelu Reddy, K. Neuroprotective effect of ginger on anti-oxidant enzymes in streptozotocin-induced diabetic rats. Food Chem. Toxicol. 2011, 49, 893–897. [Google Scholar] [CrossRef] [PubMed]
- Hassan, S.M. Assessment of Toxicological Effect of Shogaol in Albino Mice. Pak. Vet. J. 2018, 38, 377–383. [Google Scholar] [CrossRef]
- DE Flora, S.; Balansky, R.; LA Maestra, S. Anti-oxidants and COVID-19. J. Prev. Med. Hyg. 2021, 62 (Suppl. S3), E34–E45. [Google Scholar] [CrossRef]
- Jalali, M.; Mahmoodi, M.; Moosavian, S.P.; Jalali, R.; Ferns, G.; Mosallanezhad, A.; Imanieh, M.H.; Mosallanezhad, Z. The effects of ginger supplementation on markers of inflammatory and oxidative stress: A systematic review and meta-analysis of clinical trials. Phytother. Res. 2020, 34, 1723–1733. [Google Scholar] [CrossRef]
- Goswami, D.; Kumar, M.; Ghosh, S.K.; Das, A. Natural Product Compounds in Alpinia officinarum and Ginger are Potent SARS-CoV-2 Papain-like Protease Inhibitors. ChemRxiv. 2020. [Google Scholar] [CrossRef]
- Al-Sanea, M.M.; Abelyan, N.; Abdelgawad, M.A.; Musa, A.; Ghoneim, M.M.; Al-Warhi, T.; Aljaeed, N.; Alotaibi, O.J.; Alnusaire, T.S.; Abdelwahab, S.F.; et al. Strawberry and Ginger Silver Nanoparticles as Potential Inhibitors for SARS-CoV-2 Assisted by In Silico Modeling and Metabolic Profiling. Antibiotics 2021, 10, 824. [Google Scholar] [CrossRef] [PubMed]
- Ahkam, A.H.; Hermanto, F.E.; Alamsyah, A.; Aliyyah, I.H.; Fatchiyah, F. Virtual prediction of antiviral potential of ginger (Zingiber officinale) bioactive compounds against spike and MPro of SARS-CoV-2 protein. Berk. Penelit. Hayati 2020, 25, 52–57. [Google Scholar] [CrossRef]
- Suherman, M.; Maulidya, S.A.I. In Silico Study: Secondary Metabolites from Red Ginger Rhizome (Zingiber officinale Var. Rubrum) as potential inhibitors OF3CLpro AND PLpro OF SARS-CoV-2. Med. Sains J. Ilm Kefarmasian 2023, 8, 1249–1262. [Google Scholar] [CrossRef]
- Rabie, A.M. New Potential Inhibitors of Coronaviral Main Protease (CoV-Mpro): Strychnine Bush, Pineapple, and Ginger could be Natural Enemies of COVID-19. Int. J. New Chem. 2022, 9, 225–237. [Google Scholar]
- Zubair, M.; Maulana, S.; Widodo, A.; Pitopang, R.; Arba, M.; Hariono, M. GC-MS, LC-MS/MS, Docking and Molecular Dynamics Approaches to Identify Potential SARS-CoV-2 3-Chymotrypsin-Like Protease Inhibitors from Zingiber officinale Roscoe. Molecules 2021, 26, 5230. [Google Scholar] [CrossRef]
- Thomson, M.; Al-Qattan, K.K.; Al-Sawan, S.M.; Alnaqeeb, M.A.; Khan, I.; Ali, M. The use of ginger (Zingiber officinale Rosc.) as a potential anti-inflammatory and antithrombotic agent. Prostaglandins Leukot. Essent. Fat. Acids 2002, 67, 475–478. [Google Scholar] [CrossRef]
- Jing, Y.; Cheng, W.; Li, M.; Zhang, Y.; Pang, X.; Qiu, X.; Zheng, Y.; Zhang, D.; Wu, L. Structural Characterization, Rheological Properties, Antioxidant and Anti-Inflammatory Activities of Polysaccharides from Zingiber officinale. Plant Foods Hum. Nutr. 2023, 78, 160–165. [Google Scholar] [CrossRef]
- Adrianta, K.A.; Somantara, I.G. The Curcumin and Gingerol Combination as an Immune Regulator and Anti-Inflammatory Agent of SARS-CoV Infection According to a Nutrigenomic Approach: A Mini-Review. Nat. Prod. J. 2023, 13, 3–12. [Google Scholar] [CrossRef]
- Utami, A.T.; Abdullah Qarrah, A.G. Benefits of Ginger as Medicine for COVID-19: Literature Study. J. Biomed. Res. Environ. Sci. 2022, 3, 1208–1223. [Google Scholar] [CrossRef]
- Kuete, V. (Ed.) Medicinal Spices and Vegetables from Africa: Therapeutic Potential against Metabolic, Inflammatory, Infectious and Systemic Diseases; Academic Press: Amsterdam, The Netherlands, 2017. [Google Scholar]
- Singh, P.; Singh, J.; Singh, S.; Singh, B.R. Medicinal values of Garlic (Allium sativum L.) in Human Life: An Overview. Greener J. Agric. Sci. 2014, 4, 265–280. [Google Scholar] [CrossRef]
- Nafissa, B.; Abdelkrim, M.; Abderrahmane, R.; Abdelkrim, K.; Fatma, G. Etude préliminaire de l’Effet de l’ail (Allium sativum L.) chez des malades atteints du SARS-CoV-2. Alger. J. Health Sci. 2021, 3, 9–14. [Google Scholar]
- Jeong, Y.; Ryu, J.; Shin, J.-H.; Kang, M.; Kang, J.; Han, J.; Kang, D. Comparison of Anti-Oxidant and Anti-Inflammatory Effects between Fresh and Aged Black Garlic Extracts. Molecules 2016, 21, 430. [Google Scholar] [CrossRef]
- Batiha, G.E.-S.; Beshbishy, A.M.; Wasef, L.G.; Elewa, Y.H.A.; Al-Sagan, A.A.; El-Hack, M.E.A.; Taha, A.E.; Abd-Elhakim, Y.M.; Devkota, H.P. Chemical Constituents and Pharmacological Activities of Garlic (Allium sativum L.): A Review. Nutrients 2020, 12, 872. [Google Scholar] [CrossRef] [PubMed]
- Moutia, M.; Habti, N.; Badou, A. In Vitro and In Vivo Immunomodulator Activities of Allium sativum L. Evid.-Based Complement. Altern. Med. 2018, 2018, 4984659. [Google Scholar] [CrossRef]
- Rouf, R.; Uddin, S.J.; Sarker, D.K.; Islam, M.T.; Ali, E.S.; Shilpi, J.A.; Nahar, L.; Tiralongo, E.; Sarker, S.D. Antiviral potential of garlic (Allium sativum) and its organosulfur compounds: A systematic update of pre-clinical and clinical data. Trends Food Sci. Technol. 2020, 104, 219–234. [Google Scholar] [CrossRef]
- Khubber, S.; Hashemifesharaki, R.; Mohammadi, M.; Gharibzahedi, S.M.T. Garlic (Allium sativum L.): A potential unique therapeutic food rich in organosulfur and flavonoid compounds to fight with COVID-19. Nutr. J. 2020, 19, 124. [Google Scholar] [CrossRef] [PubMed]
- Listiyani, P.; Kharisma, V.D.; Ansori, A.N.M.; Widyananda, M.H.; Probojati, R.T.; Murtadlo, A.A.A.; Turista, D.D.R.; Ullah, M.E.; Jakhmola, V.; Zainul, R. In Silico Phytochemical Compounds Screening of Allium sativum Targeting the Mpro of SARS-CoV-2. Pharmacogn. J. 2022, 14, 604–609. [Google Scholar] [CrossRef]
- Atoum, M.F.; Padma, K.R.; Don, K.R. Paving New Roads Using Allium sativum as a Repurposed Drug and Analyzing its Antiviral Action Using Artificial Intelligence Technology. Iran. J. Pharm. Res. 2022, 21, e131577. [Google Scholar] [CrossRef] [PubMed]
- Pathak, R.; Sharma, H. A Review on Medicinal Uses of Cinnamomum verum (Cinnamon). J. Drug Deliv. Ther. 2021, 11, 161–166. [Google Scholar] [CrossRef]
- Reichling, J.; Schnitzler, P.; Suschke, U.; Saller, R. Essential Oils of Aromatic Plants with Antibacterial, Antifungal, Antiviral, and Cytotoxic Properties—An Overview. Complement. Med. Res. 2009, 16, 79–90. [Google Scholar] [CrossRef]
- Yakhchali, M.; Taghipour, Z.; Mirabzadeh Ardakani, M.; Alizadeh Vaghasloo, M.; Vazirian, M.; Sadrai, S. Cinnamon and its possible impact on COVID-19: The viewpoint of traditional and conventional medicine. Biomed. Pharmacother. 2021, 143, 112221. [Google Scholar] [CrossRef] [PubMed]
- Leka, K.; Hamann, C.; Desdemoustier, P.; Frédérich, M.; Garigliany, M.; Ledoux, A. In vitro antiviral activity against SARS-CoV-2 of common herbal medicinal extracts and their bioactive compounds. Phytother. Res. 2022, 36, 3013–3015. [Google Scholar] [CrossRef]
- Xie, Z.; Li, Y.; Liu, Z.; Zeng, M.; Moore, J.C.; Gao, B.; Wu, X.; Sun, J.; Wang, T.T.Y.; Pehrsson, P.; et al. Bioactive Compositions of Cinnamon (Cinnamomum verum J. Presl) Extracts and Their Capacities in Suppressing SARS-CoV-2 Spike Protein Binding to ACE2, Inhibiting ACE2, and Scavenging Free Radicals. J. Agric. Food Chem. 2023, 71, 4890–4900. [Google Scholar] [CrossRef]
- Ranasinghe, P.; Pigera, S.; Premakumara, G.S.; Galappaththy, P.; Constantine, G.R.; Katulanda, P. Medicinal properties of ‘true’ cinnamon (Cinnamomum zeylanicum): A systematic review. BMC Complement. Altern. Med. 2013, 13, 275. [Google Scholar] [CrossRef]
- Gazzaz, Z.J. Diabetes and COVID-19. Open Life Sci. 2021, 16, 297–302. [Google Scholar] [CrossRef]
- Vuorio, A.; Strandberg, T.E.; Raal, F.; Santos, R.D.; Kovanen, P.T. Familial hypercholesterolemia and COVID-19: A menacing but treatable vasculopathic condition. Atheroscler. Plus 2021, 43, 3–6. [Google Scholar] [CrossRef]
- Okdeh, N.; Mahfouz, G.; Harb, J.; Sabatier, J.-M.; Roufayel, R.; Gazo Hanna, E.; Kovacic, H.; Fajloun, Z. Protective Role and Functional Engineering of Neuropeptides in Depression and Anxiety: An Overview. Bioengineering 2023, 10, 258. [Google Scholar] [CrossRef]
- Sohrabi, R.; Pazgoohan, N.; Rezaei Seresht, H.; Amin, B. Repeated systemic administration of the cinnamon essential oil possesses anti-anxiety and anti-depressant activities in mice. Iran. J. Basic Med. Sci. 2017, 20, 708–714. [Google Scholar] [CrossRef]
- Renaud-Charest, O.; Lui, L.M.W.; Eskander, S.; Ceban, F.; Ho, R.; Di Vincenzo, J.D.; Rosenblat, J.D.; Lee, Y.; Subramaniapillai, M.; McIntyre, R.S. Onset and frequency of depression in post-COVID-19 syndrome: A systematic review. J. Psychiatr. Res. 2021, 144, 129–137. [Google Scholar] [CrossRef]
- Andrade, J.M.; Faustino, C.; Garcia, C.; Ladeiras, D.; Reis, C.P.; Rijo, P. Rosmarinus officinalis L.: An update review of its phytochemistry and biological activity. Future Sci. OA 2018, 4, FSO283. [Google Scholar] [CrossRef] [PubMed]
- Shin, H.-B.; Choi, M.-S.; Ryu, B.; Lee, N.-R.; Kim, H.-I.; Choi, H.-E.; Chang, J.; Lee, K.-T.; Jang, D.S.; Inn, K.-S. Antiviral activity of carnosic acid against respiratory syncytial virus. Virol. J. 2013, 10, 303. [Google Scholar] [CrossRef] [PubMed]
- Kontogianni, V.G.; Tomic, G.; Nikolic, I.; Nerantzaki, A.A.; Sayyad, N.; Stosic-Grujicic, S.; Stojanovic, I.; Gerothanassis, I.P.; Tzakos, A.G. Phytochemical profile of Rosmarinus officinalis and Salvia officinalis extracts and correlation to their antioxidant and anti-proliferative activity. Food Chem. 2013, 136, 120–129. [Google Scholar] [CrossRef] [PubMed]
- Suhail, S.; Zajac, J.; Fossum, C.; Lowater, H.; McCracken, C.; Severson, N.; Laatsch, B.; Narkiewicz-Jodko, A.; Johnson, B.; Liebau, J.; et al. Role of Oxidative Stress on SARS-CoV (SARS) and SARS-CoV-2 (COVID-19) Infection: A Review. Protein J. 2020, 39, 644–656. [Google Scholar] [CrossRef] [PubMed]
- Khazaal, S.; Harb, J.; Rima, M.; Annweiler, C.; Wu, Y.; Cao, Z.; Abi Khattar, Z.; Legros, C.; Kovacic, H.; Fajloun, Z.; et al. The Pathophysiology of Long COVID throughout the Renin-Angiotensin System. Molecules 2022, 27, 2903. [Google Scholar] [CrossRef] [PubMed]
- Patel, U.; Desai, K.; Dabhi, R.C.; Maru, J.J.; Shrivastav, P.S. Bioprospecting phytochemicals of Rosmarinus officinalis L. for targeting SARS-CoV-2 main protease (Mpro): A computational study. J. Mol. Model. 2023, 29, 161. [Google Scholar] [CrossRef] [PubMed]
- Kılınç, N.; Açar, M.; Tuncay, S.; Karasakal, Ö.F. Identification of Potential Inhibitors for Severe Acute Respiratory Syndrome-related Coronavirus 2 (SARS-CoV-2) Angiotensin-converting Enzyme 2 and the Main Protease from Anatolian Traditional Plants. Lett. Drug Des. Discov. 2022, 19, 996–1006. [Google Scholar] [CrossRef]
- Rocha, J.; Eduardo-Figueira, M.; Barateiro, A.; Fernandes, A.; Brites, D.; Bronze, R.; Duarte, C.M.; Serra, A.T.; Pinto, R.; Freitas, M. Anti-inflammatory effect of rosmarinic acid and an extract of Rosmarinus officinalis in rat models of local and systemic inflammation. Basic Clin. Pharmacol. Toxicol. 2015, 116, 398–413. [Google Scholar] [CrossRef]
- Karadağ, A.E.; Demirci, B.; Çaşkurlu, A.; Demirci, F.; Okur, M.E.; Orak, D.; Sipahi, H.; Başer, K.H.C. In vitro antibacterial, antioxidant, anti-inflammatory and analgesic evaluation of Rosmarinus officinalis L. flower extract fractions. S. Afr. J. Bot. 2019, 125, 214–220. [Google Scholar] [CrossRef]
- Amer, N.S.; El-Refaiy, A.I. Ameliorative Effect of Dandelion Leaves Extract Against Liver Injury Induced by Schistosomiasis in Mice. J. Pharm. Negat. Results 2022, 13, 2554–2569. [Google Scholar] [CrossRef]
- Di Napoli, A.; Zucchetti, P. A comprehensive review of the benefits of Taraxacum officinale on human health. Bull. Natl. Res. Cent. 2021, 45, 110. [Google Scholar] [CrossRef]
- González-Castejón, M.; Visioli, F.; Rodriguez-Casado, A. Diverse biological activities of dandelion. Nutr. Rev. 2012, 70, 534–547. [Google Scholar] [CrossRef] [PubMed]
- Petkova, N.T.; Ivanov, I.; Topchieva, S.; Denev, P.; Pavlov, A. Biologically active substances and in vitro antioxidant activity of different extracts from dandelion (Taraxacum officinale) roots. Sci. Bull. Ser. F. Biotechnol. 2015, 19, 190–197. [Google Scholar] [CrossRef]
- Ivanov, I.G. Polyphenols Content and Antioxidant Activities of Taraxacum officinale F.H. Wigg (Dandelion) Leaves. Int. J. Pharmacogn. Phytochem. Res. 2014, 6, 889–893. [Google Scholar]
- Li, J.; Zhao, C.; Wei, L.; Li, X.; Liu, F.; Zhang, M.; Liu, X.; Wang, Y. Preservation of Cichoric Acid Antioxidant Properties Loaded in Heat Treated Lactoferrin Nanoparticles. Molecules 2018, 23, 2678. [Google Scholar] [CrossRef] [PubMed]
- Cecchini, R.; Cecchini, A.L. SARS-CoV-2 infection pathogenesis is related to oxidative stress as a response to aggression. Med. Hypotheses 2020, 143, 110102. [Google Scholar] [CrossRef] [PubMed]
- He, W.; Han, H.; Wang, W.; Gao, B. Anti-influenza virus effect of aqueous extracts from dandelion. Virol. J. 2011, 8, 538. [Google Scholar] [CrossRef]
- Tran, H.T.T.; Le, N.P.K.; Gigl, M.; Dawid, C.; Lamy, E. Common dandelion (Taraxacum officinale) efficiently blocks the interaction between ACE2 cell surface receptor and SARS-CoV-2 spike protein D614, mutants D614G, N501Y, K417N and E484K in vitro. Microbiology 2021. [Google Scholar] [CrossRef]
- Tran, H.T.T.; Gigl, M.; Dawid, C.; Lamy, E. Common dandelion (Taraxacum officinale) leaf extract efficiently inhibits SARS-CoV-2 Omicron infection in vitro. Microbiology 2022. [Google Scholar] [CrossRef]
- Ali, A.A.; Maher, F.T.; Al-Bajari, S.A. Green Biosynthesis of Silver Nanoparticles from Taraxacum officinale Roots Plant and Studying Its Antiviral Properties to Coronavirus (SARS-CoV-2) Infected Lung Cells. J. Hyg. Eng. Des. 2023, 42, 361–369. [Google Scholar]
- Oniga, I.; Pușcaș, C.; Silaghi-Dumitrescu, R.; Olah, N.-K.; Sevastre, B.; Marica, R.; Marcus, I.; Sevastre-Berghian, A.; Benedec, D.; Pop, C.; et al. Origanum vulgare ssp. vulgare: Chemical Composition and Biological Studies. Molecules 2018, 23, 2077. [Google Scholar] [CrossRef]
- Zhang, X.-L.; Guo, Y.-S.; Wang, C.-H.; Li, G.-Q.; Xu, J.-J.; Chung, H.Y.; Ye, W.-C.; Li, Y.-L.; Wang, G.-C. Phenolic compounds from Origanum vulgare and their antioxidant and antiviral activities. Food Chem. 2014, 152, 300–306. [Google Scholar] [CrossRef]
- Ozdemir, N.; Ozgen, Y.; Kiralan, M.; Bayrak, A.; Arslan, N.; Ramadan, M.F. Effect of different drying methods on the essential oil yield, composition and antioxidant activity of Origanum vulgare L. and Origanum onites L. Food Measure 2018, 12, 820–825. [Google Scholar] [CrossRef]
- Valdivieso-Ugarte, M.; Gomez-Llorente, C.; Plaza-Díaz, J.; Gil, Á. Antimicrobial, Antioxidant, and Immunomodulatory Properties of Essential Oils: A Systematic Review. Nutrients 2019, 11, 2786. [Google Scholar] [CrossRef]
- Husain, I.; Ahmad, R.; Siddiqui, S.; Chandra, A.; Misra, A.; Srivastava, A.; Ahamad, T.; Khan, M.F.; Siddiqi, Z.; Trivedi, A.; et al. Structural interactions of phytoconstituent(s) from cinnamon, bay leaf, oregano, and parsley with SARS-CoV-2 nucleocapsid protein: A comparative assessment for development of potential antiviral nutraceuticals. J. Food Biochem. 2022, 46, e14262. [Google Scholar] [CrossRef] [PubMed]
- Demirci, F.; Teralı, K.; Karadağ, A.E.; Biltekin, S.N.; Sakallı, E.A.; Demirci, B.; Koşar, M.; Başer, K.H.C. In vitro and in silico evaluation of ACE2 and LOX inhibitory activity of Origanum essential oils and Carvacrol. Planta Medica 2023, 89, 790–799. [Google Scholar] [CrossRef] [PubMed]
- Mohammed, A.; Kalle, A.M.; Reddanna, P. Managing SARS-CoV-2 Infections Through Resolution of Inflammation by Eicosanoids: A Review. J. Inflamm. Res. 2022, 15, 4349–4358. [Google Scholar] [CrossRef]
- Al-Sehemi, A.G.; Olotu, F.A.; Dev, S.; Pannipara, M.; Soliman, M.E.; Carradori, S.; Mathew, B. Natural Products Database Screening for the Discovery of Naturally Occurring SARS-CoV-2 Spike Glycoprotein Blockers. ChemistrySelect 2020, 5, 13309–13317. [Google Scholar] [CrossRef] [PubMed]
- Martin, K.W. Herbal medicines for treatment of bacterial infections: A review of controlled clinical trials. J. Antimicrob. Chemother. 2003, 51, 241–246. [Google Scholar] [CrossRef] [PubMed]
- Parveen, A.; Parveen, B.; Parveen, R.; Ahmad, S. Challenges and guidelines for clinical trial of herbal drugs. J. Pharm. Bioallied Sci. 2015, 7, 329. [Google Scholar]
- Ameh, S.J.; Obodozie, O.O.; Babalola, P.C.; Gamaniel, K.S. Medical Herbalism and Herbal Clinical Research: A Global Perspective. Br. J. Pharm. Res. 2011, 1, 99. [Google Scholar] [CrossRef]
- Izzo, A.A.; Ernst, E. Interactions between Herbal Medicines and Prescribed Drugs: A Systematic Review. Drugs 2001, 61, 2163–2175. [Google Scholar] [CrossRef] [PubMed]
- Izzo, A.A.; Ernst, E. Interactions between Herbal Medicines and Prescribed Drugs: An Updated Systematic Review. Drugs 2009, 69, 1777–1798. [Google Scholar] [CrossRef] [PubMed]
- Vaou, N.; Stavropoulou, E.; Voidarou, C.; Tsakris, Z.; Rozos, G.; Tsigalou, C.; Bezirtzoglou, E. Interactions between Medical Plant-Derived Bioactive Compounds: Focus on Antimicrobial Combination Effects. Antibiotics 2022, 11, 1014. [Google Scholar] [CrossRef]
- Gupta, A.; Naraniwal, M.; Kothari, V. Modern extraction methods for preparation of bioactive plant extracts. Int. J. Appl. Nat. Sci. 2012, 1, 8–26. [Google Scholar]
| Plant | Bioactive Compounds | Mechanisms against SARS-CoV-2 |
|---|---|---|
| Ginkgo biloba |
| |
| Curcuma longa |
| |
| Artemisia annua |
| |
| Nigella sativa |
| |
| Zingiber officinale |
| |
| Allium sativum |
| |
| Cinnamomum verum |
| |
| Rosmarinus officinale |
| |
| Taraxacum officinale |
| |
| Origanum vulgare |
|
|
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Al-Jamal, H.; Idriss, S.; Roufayel, R.; Abi Khattar, Z.; Fajloun, Z.; Sabatier, J.-M. Treating COVID-19 with Medicinal Plants: Is It Even Conceivable? A Comprehensive Review. Viruses 2024, 16, 320. https://doi.org/10.3390/v16030320
Al-Jamal H, Idriss S, Roufayel R, Abi Khattar Z, Fajloun Z, Sabatier J-M. Treating COVID-19 with Medicinal Plants: Is It Even Conceivable? A Comprehensive Review. Viruses. 2024; 16(3):320. https://doi.org/10.3390/v16030320
Chicago/Turabian StyleAl-Jamal, Hadi, Sara Idriss, Rabih Roufayel, Ziad Abi Khattar, Ziad Fajloun, and Jean-Marc Sabatier. 2024. "Treating COVID-19 with Medicinal Plants: Is It Even Conceivable? A Comprehensive Review" Viruses 16, no. 3: 320. https://doi.org/10.3390/v16030320
APA StyleAl-Jamal, H., Idriss, S., Roufayel, R., Abi Khattar, Z., Fajloun, Z., & Sabatier, J.-M. (2024). Treating COVID-19 with Medicinal Plants: Is It Even Conceivable? A Comprehensive Review. Viruses, 16(3), 320. https://doi.org/10.3390/v16030320







