The Effect of Pluronic-Coated Gold Nanoparticles in Hearing Preservation Following Cochlear Implantation-Pilot Study
Abstract
:1. Introduction
2. Materials and Methods
2.1. Animals
2.2. Chemicals
2.3. Nanoparticles Preparation and Loading with Dexamethasone
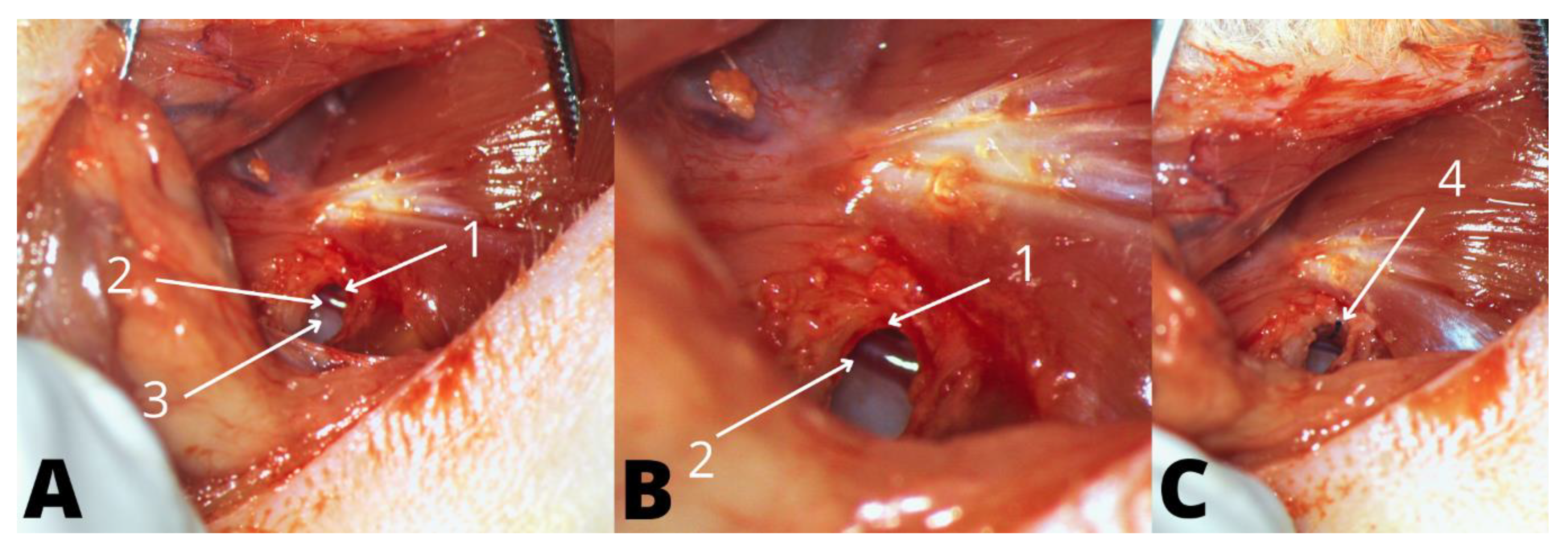
2.4. Anesthesia, Surgical Preparation, and Approach
2.5. Dummy Electrode
2.6. Groups
2.7. Hearing Threshold Measurement: Auditory Brainstem Response (ABR)
2.8. Statistics
3. Results
3.1. Spectral Characterization of Dex-Plu-AuNPs
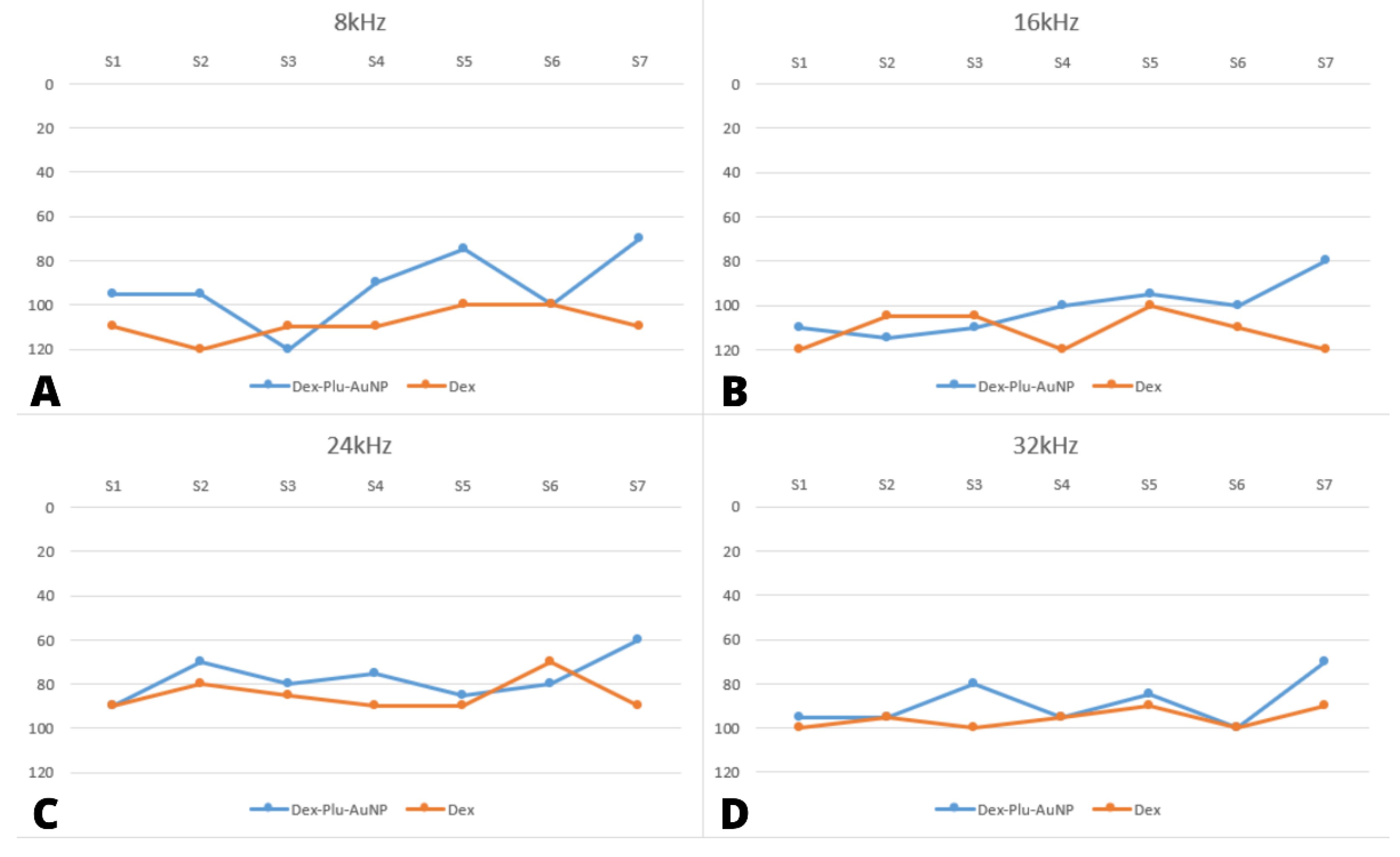
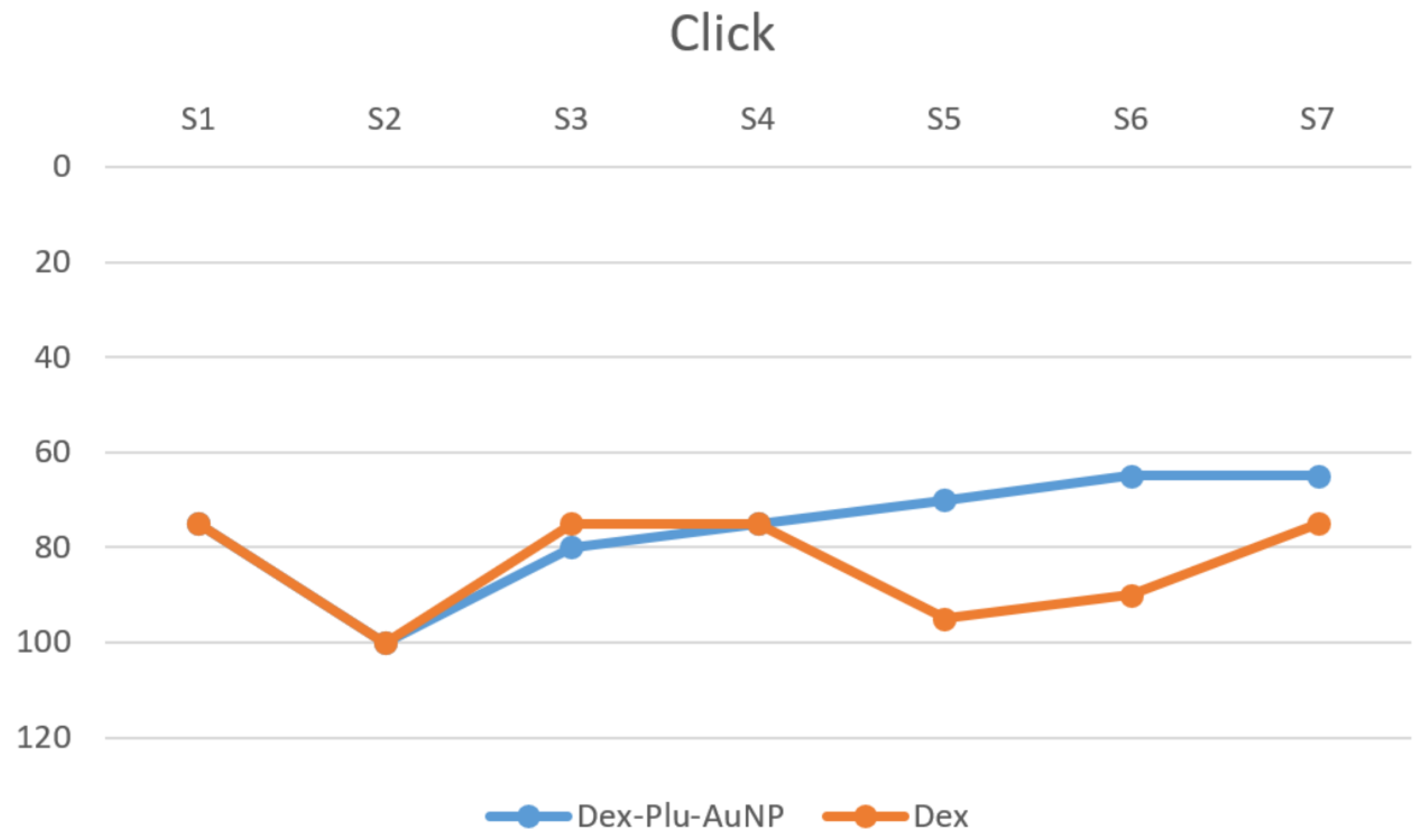
3.2. Audiological Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Blebea, C.M.; Ujvary, L.P.; Necula, V.; Dindelegan, M.G.; Perde-Schrepler, M.; Stamate, M.C.; Cosgarea, M.; Maniu, A.A. Current Concepts and Future Trends in Increasing the Benefits of Cochlear Implantation: A Narrative Review. Medicina 2022, 58, 747. [Google Scholar] [CrossRef]
- Park, L.R.; Teagle, H.F.B.; Gagnon, E.; Woodard, J.; Brown, K.D. Electric-Acoustic Stimulation Outcomes in Children. Ear Hear. 2019, 40, 849–857. [Google Scholar] [CrossRef]
- Rader, T.; Bohnert, A.; Matthias, C.; Koutsimpelas, D.; Kainz, M.A.; Strieth, S. Hearing Preservation in Children with Electric-Acoustic Stimulation after Cochlear Implantation: Outcome after Electrode Insertion with Minimal Insertion Trauma. HNO 2018, 66, 56–62. [Google Scholar] [CrossRef]
- Rajan, G.; Tavora-Vieira, D.; Baumgartner, W.D.; Godey, B.; Müller, J.; O’Driscoll, M.; Skarzynski, H.; Skarzynski, P.; Usami, S.I.; Adunka, O.; et al. Hearing Preservation Cochlear Implantation in Children: The HEARRING Group Consensus and Practice Guide. Cochlear Implant. Int. 2017, 19, 1–13. [Google Scholar] [CrossRef]
- Matin, F.; Artukarslan, E.N.; Illg, A.; Lesinski-Schiedat, A.; Lenarz, T.; Suhling, M.C. Cochlear Implantation in Elderly Patients with Residual Hearing. J. Clin. Med. 2021, 10, 4305. [Google Scholar] [CrossRef]
- Skarzynski, H.; Van De Heyning, P.; Agrawal, S.; Arauz, S.L.; Atlas, M.; Baumgartner, W.; Caversaccio, M.; De Bodt, M.; Gavilan, J.; Godey, B.; et al. Towards a Consensus on a Hearing Preservation Classification System. Acta Oto-Laryngol. 2013, 133, 3–13. [Google Scholar] [CrossRef] [PubMed]
- Foggia, M.J.; Quevedo, R.V.; Hansen, M.R. Intracochlear Fibrosis and the Foreign Body Response to Cochlear Implant Biomaterials. Laryngoscope Investig. Otolaryngol. 2019, 4, 678–683. [Google Scholar] [CrossRef] [PubMed]
- Khater, A.; El-Anwar, M.W. Methods of Hearing Preservation during Cochlear Implantation. Int. Arch. Otorhinolaryngol. 2017, 21, 297–301. [Google Scholar] [CrossRef] [PubMed]
- Xu, X.; Zheng, J.; He, Y.; Lin, K.; Li, S.; Zhang, Y.; Song, P.; Zhou, Y.; Chen, X. Nanocarriers for Inner Ear Disease Therapy. Front. Cell. Neurosci. 2021, 15, 791573. [Google Scholar] [CrossRef] [PubMed]
- Dindelegan, M.G.; Blebea, C.; Perde-Schrepler, M.; Buzoianu, A.D.; Maniu, A.A. Recent Advances and Future Research Directions for Hearing Loss Treatment Based on Nanoparticles. J. Nanomater. 2022, 2022, 7794384. [Google Scholar] [CrossRef]
- Mittal, R.; Pena, S.A.; Zhu, A.; Eshraghi, N.; Fesharaki, A.; Horesh, E.J.; Mittal, J.; Eshraghi, A.A. Nanoparticle-Based Drug Delivery in the Inner Ear: Current Challenges, Limitations and Opportunities. Artif. Cells Nanomed. Biotechnol. 2019, 47, 1312–1320. [Google Scholar] [CrossRef] [PubMed]
- Lin, Y.C.; Shih, C.P.; Chen, H.C.; Chou, Y.L.; Sytwu, H.K.; Fang, M.C.; Lin, Y.Y.; Kuo, C.Y.; Su, H.H.; Hung, C.L.; et al. Ultrasound Microbubble–Facilitated Inner Ear Delivery of Gold Nanoparticles Involves Transient Disruption of the Tight Junction Barrier in the Round Window Membrane. Front. Pharmacol. 2021, 12, 689032. [Google Scholar] [CrossRef] [PubMed]
- Mahdi, W.A.; Hussain, A.; Ramzan, M.; Faruk, A.; Bukhari, S.I.; Dev, A. Pluronic-Coated Biogenic Gold Nanoparticles for Colon Delivery of 5-Fluorouracil: In Vitro and Ex Vivo Studies. AAPS PharmSciTech 2021, 22, 64. [Google Scholar] [CrossRef] [PubMed]
- Fenwick, N.; Griffin, G.; Gauthier, C. The Welfare of Animals Used in Science: How the “Three Rs” Ethic Guides Improvements. Can. Vet. J. 2009, 50, 523–530. [Google Scholar] [PubMed]
- Frens, G. Controlled Nucleation for the Regulation of the Particle Size in Monodisperse Gold Suspensions. Nat. Phys. Sci. 1973, 241, 20–22. [Google Scholar] [CrossRef]
- Ruebhausen, M.R.; Brozoski, T.J.; Bauer, C.A. A Comparison of the Effects of Isoflurane and Ketamine Anesthesia on Auditory Brainstem Response (ABR) Thresholds in Rats. Hear. Res. 2012, 287, 25–29. [Google Scholar] [CrossRef]
- Tamames, I.; King, C.; Bas, E.; Dietrich, W.D.; Telischi, F.; Rajguru, S.M. A Cool Approach to Reducing Electrode-Induced Trauma: Localized Therapeutic Hypothermia Conserves Residual Hearing in Cochlear Implantation. Hear. Res. 2016, 339, 32–39. [Google Scholar] [CrossRef]
- Gur, H.; Alimoglu, Y.; Duzenli, U.; Korkmaz, S.; Inan, S.; Olgun, L. The Effect of Local Application of Insulin-like Growth Factor for Prevention of Inner-Ear Damage Caused by Electrode Trauma. J. Laryngol. Otol. 2017, 131, 245–252. [Google Scholar] [CrossRef]
- Parys, Q.A.; Van Bulck, P.; Loos, E.; Verhaert, N. Inner Ear Pharmacotherapy for Residual Hearing Preservation in Cochlear Implant Surgery: A Systematic Review. Biomolecules 2022, 12, 529. [Google Scholar] [CrossRef]
- Lee, M.Y.; Kim, Y.C.; Jang, J.; Jung, J.Y.; Choi, H.; Jang, J.H.; Choung, Y.H. Dexamethasone Delivery for Hearing Preservation in Animal Cochlear Implant Model: Continuity, Long-Term Release, and Fast Release Rate. Acta Oto-Laryngol. 2020, 140, 705–714. [Google Scholar] [CrossRef] [PubMed]
- O’Leary, S.J.; Choi, J.; Brady, K.; Matthews, S.; Ozdowska, K.B.; Payne, M.; McLean, T.; Rousset, A.; Lo, J.; Creber, N.; et al. Systemic Methylprednisolone for Hearing Preservation during Cochlear Implant Surgery: A Double Blinded Placebo-Controlled Trial. Hear. Res. 2021, 404, 108224. [Google Scholar] [CrossRef] [PubMed]
- Skarżyńska, M.B.; Skarżyński, P.H.; Król, B.; Kozieł, M.; Osińska, K.; Gos, E.; Skarżyński, H. Preservation of Hearing Following Cochlear Implantation Using Different Steroid Therapy Regimens: A Prospective Clinical Study. Med. Sci. Monit. 2018, 24, 2437–2445. [Google Scholar] [CrossRef] [PubMed]
- Skarzynska, M.B.; Kolodziejak, A.; Gos, E.; Skarzynski, P.H. The Clinical Effects of Steroids Therapy in the Preserving Residual Hearing after Cochlear Implantation with the OTICON Neuro Zti EVO. J. Clin. Med. 2021, 10, 2868. [Google Scholar] [CrossRef]
- Skarzynska, M.B.; Kolodziejak, A.; Gos, E.; Skarzynski, P.H.; Lorens, A.; Walkowiak, A. The Clinical Effect of Steroid Therapy on Preserving Residual Hearing after Cochlear Implantation with the Advanced Bionics HiRes Ultra 3D Cochlear Implant System. Life 2022, 12, 486. [Google Scholar] [CrossRef] [PubMed]
- Dizaj, S.M.; Lotfipour, F.; Barzegar-Jalali, M.; Zarrintan, M.H.; Adibkia, K. Antimicrobial Activity of the Metals and Metal Oxide Nanoparticles. Mater. Sci. Eng. C 2014, 44, 278–284. [Google Scholar] [CrossRef] [PubMed]
- Kayyali, M.; Brake, L.; Ramsey, A.; Wright, A.; O’Malley, B.; Li, D.D. A Novel Nano-Approach for Targeted Inner Ear Imaging. J. Nanomed. Nanotechnol. 2017, 8, 456. [Google Scholar] [CrossRef] [PubMed]
- Eastwood, H.; Pinder, D.; James, D.; Chang, A.; Galloway, S.; Richardson, R.; O’Leary, S. Permanent and Transient Effects of Locally Delivered N-Acetyl Cysteine in a Guinea Pig Model of Cochlear Implantation. Hear. Res. 2010, 259, 24–30. [Google Scholar] [CrossRef] [PubMed]

| Stimuli | Dex-Plu-AuNPs (n = 7) | Dex (n = 7) | |||
|---|---|---|---|---|---|
| Threshold Mean (dB SPL) | SD | Threshold Mean (dB SPL) | SD | p Value (t-Test) | |
| Click | 20 | 0 | 20 | 0 | - |
| 8 kHz | 20 | 0 | 20.7 | 1.8 | 0.35 |
| 16 kHz | 20 | 0 | 20.7 | 1.8 | 0.35 |
| 24 kHz | 20 | 0 | 20 | 0 | - |
| 32 kHz | 21.4 | 10.6 | 22.85 | 5.6 | 0.59 |
| Stimuli | Group | N | Mean dB SPL | Std. Deviation | Std. Error Mean |
|---|---|---|---|---|---|
| clickpost | Dex-Plu-AuNPs | 7 | 63.57 | 28.536 | 10.785 |
| Dex | 7 | 74.29 | 21.876 | 8.268 | |
| post8 kHz | Dex-Plu-AuNPs | 7 | 87.86 | 23.954 | 9.054 |
| Dex | 7 | 108.57 | 6.901 | 2.608 | |
| post16 kHz | Dex-Plu-AuNPs | 7 | 97.14 | 18.225 | 6.888 |
| Dex | 7 | 108.57 | 14.351 | 5.424 | |
| post24 kHz | Dex-Plu-AuNPs | 7 | 70.71 | 16.439 | 6.213 |
| Dex | 7 | 79.29 | 14.840 | 5.609 | |
| post32 kHz | Dex-Plu-AuNPs | 7 | 87.14 | 11.852 | 4.480 |
| Dex | 7 | 92.86 | 10.746 | 4.062 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Blebea, C.M.; Necula, V.; Potara, M.; Dindelegan, M.G.; Ujvary, L.P.; Botan, E.C.; Maniu, A.A.; Cosgarea, M. The Effect of Pluronic-Coated Gold Nanoparticles in Hearing Preservation Following Cochlear Implantation-Pilot Study. Audiol. Res. 2022, 12, 466-475. https://doi.org/10.3390/audiolres12050047
Blebea CM, Necula V, Potara M, Dindelegan MG, Ujvary LP, Botan EC, Maniu AA, Cosgarea M. The Effect of Pluronic-Coated Gold Nanoparticles in Hearing Preservation Following Cochlear Implantation-Pilot Study. Audiology Research. 2022; 12(5):466-475. https://doi.org/10.3390/audiolres12050047
Chicago/Turabian StyleBlebea, Cristina Maria, Violeta Necula, Monica Potara, Maximilian George Dindelegan, Laszlo Peter Ujvary, Emil Claudiu Botan, Alma Aurelia Maniu, and Marcel Cosgarea. 2022. "The Effect of Pluronic-Coated Gold Nanoparticles in Hearing Preservation Following Cochlear Implantation-Pilot Study" Audiology Research 12, no. 5: 466-475. https://doi.org/10.3390/audiolres12050047
APA StyleBlebea, C. M., Necula, V., Potara, M., Dindelegan, M. G., Ujvary, L. P., Botan, E. C., Maniu, A. A., & Cosgarea, M. (2022). The Effect of Pluronic-Coated Gold Nanoparticles in Hearing Preservation Following Cochlear Implantation-Pilot Study. Audiology Research, 12(5), 466-475. https://doi.org/10.3390/audiolres12050047






