1. Introduction
Corn is the primary crops in the world and used as a raw material of food, feed, and industrial products. However, corn is highly susceptible to the fungus
Aspergillus flavus, thus resulting in aflatoxin formation [
1]. Aflatoxins are secondary metabolites of
Aspergillus flavus, and aflatoxin contamination in food and grains is a great concern all over the world. According to the United Food and Agriculture Organization of the United Nations (FAO), aflatoxin contamination in food and feed results in a huge worldwide economic loss each year. Aflatoxin B
1(AFB
1) is one type of aflatoxin, which has been shown to be an extremely potent liver carcinogen [
2]. Due to the toxicity of AFB
1, it has been classified as group 1 by the International Agency for Research on Cancer [
3]. This makes the seeking of a proper method that will effectively reduce AFB
1 in corn become a significant job. Over the last several decades, many physical, chemical, and biological methods for aflatoxins reduction have been reported. For instance, Conway [
4] reported that the AFB
1 in corn could be reduced 40–81% by roasting at 145–165 °C. Aquino [
5] reported that AFB
1 in corn could be partially and completely degradation by gamma rays at doses of 2 kGy and 10 kGy, respectively. Luo [
6] reported that the AFB
1 in corn with a moisture content of 20.37% could be reduced 72.4% after 40 min of treatment with 90 mg/L ozone. Sangare [
7] reported that AFB
1 could be degraded 82.6% by
Pseudomonas strain N17-1 after incubation in Nutrient Broth (NB) medium at 37 °C for 72 h. Different methods have their advantages and disadvantages. For a successful detoxification method, the process must be economical, leave no harmful residues, and have little impaired to commodity quality [
8].
Microwave has been widely used in the food industry because of its high efficiency of heating. Several studies have been reported that microwave heating was useful for the reduction of aflatoxin in peanut and corn [
9,
10]. In these researches, the degradation of AFB
1 mainly depended on the high temperature produced by microwave heating, and the temperature of 130–150 °C or higher was required for achieving an effective reduction of AFB
1. However, in practice, it had some major drawbacks when relying on high temperatures to reduce AFB
1 in corn. For instance, the rapid heating of microwave usually significantly increased the crack rate and heat-damaged kernels of corn because of the volumetric heat generation, and possible textural damage due to difficulty to control the final product temperature [
11]. Moreover, the large penetration depth of microwave often formed a higher temperature inside the grain kernel compared with the surface, which causes unnecessary heat damage to the inner tissue of maize. Because the AFB
1 mainly distributed on the surface of the grain [
12]. Besides this, it was easy to become popcorn because of the heating characteristics of the microwave for the full corn kernels with the approximately spherical shape, whereas AFB
1 in corn was mostly distributed in broken kernels rather than full kernels. These greatly limited the use of microwaves to degrade AFB
1 in corn.
At the same time, the relationship between degradation of AFB
1 and water had been reported in many studies [
13,
14]. According to these reports, AFB
1 was stable up to their melting points with dry heating and the thermal degradation temperature of AFB
1 was up to 300 °C [
15]. However, the presence of water molecules helped in opening the lactone ring in AFB
1 to form a terminal carboxylic acid [
16] and lowered the decomposition temperature of AFB
1 [
13]. Hu [
17] reported that AFB
1 standard substance in water could be degraded at 80 °C with microwave heating while a temperature above 200 °C was required for AFB
1 degradation in edible oils [
13]. Similar results were reported by Mobeen [
18]. It might be a valuable research to used water as microwave heating mediums to reduce AFB
1 in corn. In the literature, there were some researchers reported that the combination of water and microwaves had high potential as a postharvest disinfestation treatment [
19]. However, the information on the combined effects of water and microwaves on AFB
1 reduction of corn was lacking.
In the present study, the corn kernels containing AFB1 were submerged in water and then treated in a microwave oven. We named this method water-assisted microwave treatment (WMT). The effect of WMT on the reduction of AFB1-contaminated corn at different processing times, microwave power levels, and initial AFB1 contamination of corn were preliminary researched. The purposes of these studies were to evaluate the feasibility of WMT to reduce AFB1, study whether WMT could avoid producing heat-damaged kernels in corn, and analyze the role of water during the reduction of AFB1 by WMT. Meanwhile, the main quality changes of corn during the processing were also studied. This study could provide basic and valuable information for the reduction of AFB1 in corn.
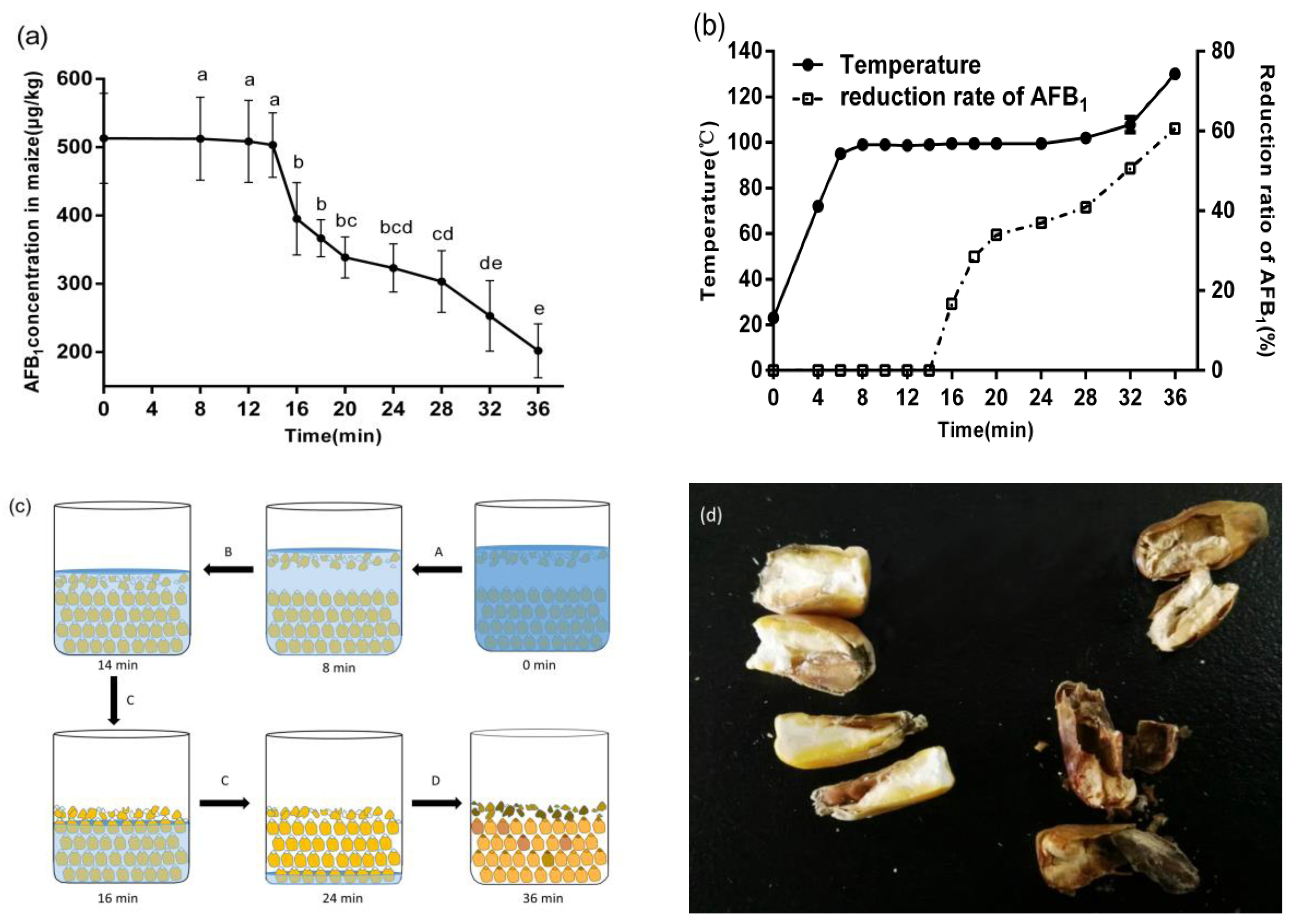
3. Discussion
In our study, WMT was effective in reducing AFB
1 in corn. Different from the reduction of AFB
1 by previous microwave roasting, the WMT process involves a lot of water, which led to the unique characteristics of the degradation of AFB
1. The reduction of AFB
1 in corn by WMT could be divided into four stages (A, B, C, and D) according to the changes in temperature, moisture content, and AFB
1 content with processing time. The characteristics of each stage are described in
Section 2.1.1.
Stage A could be considered as a process of temperature rising by WMT. There was no reduction of AFB
1 in corn because of the lower temperature of the whole system. Stage B had the same temperature with stage C, but there was no reduction of AFB
1 in stage B. One possible reason was that the corn was immersed in water completely at this stage. The water acted as a barrier, so most of the microwave energy was absorbed by the water rather than by the corn. For instance, the water was usually used as a barrier to prevent the leakage of microwave energy from instruments to avoid harm to the human body in the microwave industry [
20]. On the other hand, it suggested that short-time cooking with excess water could not effectively reduce AFB
1 in corn. The same result was reported in rice [
21]. It also means that microwave extraction could not remove AFB
1 from corn kernels. Stage C was the most meaningful stage of AFB
1 reduction during the WMT. It was that the kernels began to emerge from the water and exposed to microwave radiation directly due to the evaporation of water. However, the rich moisture of corn (30~40%) absorbed at the stages A and B, ensuring that the temperature of the corn was maintained at 100 °C rather than further increased at microwave heating processing. By the end of stage C, the degradation rate of AFB
1 reached 38%. In Mann’s study [
15], 34% reduction of aflatoxins in a peanut meal (30% moisture content) was found when it was heated at 100 °C for 2 h. However, a similar AFB
1 reduction rate by WMT (100 °C) only needed 10 min in our study. It reflected the advantages of microwave in the thermal degradation of AFB
1, because microwave heating could speed up the chemical reaction process and shorten the reaction time compared with conventional heating [
22,
23]. After entering the stage D (24 min~36 min), the water in the beaker had evaporated basically, and the moisture content of corn continually decreased to 12.13%, because of the microwave dehydration. At the end of this stage, the process of corn exposed to the microwave field appears to be similar to that of microwave roasting. With the increase in temperature, the reduction rate of AFB
1 increased [
10].
The effect of microwave power on the AFB
1 reduction rate was studied. Microwave power did not affect the trend of the whole cooking process, but affected the duration of the whole process. For instance, the whole cooking process only lasted for 14 min at high microwave power (700 W), while it would take 60 min at low power (200 W). Compared with low power treatment, higher microwave power could make the cooking process enter stage C and D earlier and achieve more reduction of AFB
1 at the same time. However, higher microwave power would shorten the stage C, which was the most important stage in the reduction of AFB
1. Moreover, the higher power would make the dehydration rate of corn too fast, thus significantly increasing the crack rate of corn [
24]. Therefore, the matching of different cooking stages with appropriate power may be the next stage of the research objective.
The initial concentration of AFB1 in corn had little effect on the AFB
1 reduction rate. These results were in agreement with the result of Pluyer [
9], who used naturally contaminated peanuts to study aflatoxin destruction by microwave roasting, and their results also showed no correlation between aflatoxin initial concentration and the percentage of destruction. However, there were also some different reports. For instance, Lee [
25] reported that the more the initial aflatoxin level increases, the more the aflatoxin reduction increases. Arzandeh [
26] reported that an increase in initial aflatoxin concentration causes an increase in aflatoxin reduction, whereas whenever the initial aflatoxin concentration exceeded 200 ng/g, there was a decrease in aflatoxin reduction. The reason for these results might be related to the different matrices and initial AFB
1 concentration used by these researchers and the current study. The different analytical techniques used to quantify the aflatoxins, the different extraction methods and the different moisture contents of the samples could partially account for the different results [
9].
In addition, it was important that water played an important role in the reduction of AFB
1 during WMT. The participation of water had the following three unique advantages: (1) With the help of water, the corn was automatically graded. The moth-eaten and mechanical damage kernels would be floated above the water surface because of its relatively low density (
Figure 1c). Previous studies showed that the content of AFB
1 in this damaged kernels was more than 80% of the total AFB
1 in corn [
27]. Those highly toxic kernels, which float above the water surface, would be exposed to microwave radiation firstly. This might help to explain the rapid increase in AFB
1 reduction at the beginning of stage C. (2) A large amount of moisture in corn ensured that the temperature of corn under microwave irradiation does not exceed 120 °C during the whole cooking process, except at the end of stage D. Therefore, the color of the corn did not darken due to high temperature, and effectively inhibited the appearance of heat-damaged kernels and burnt smell. That was an important advantage of WMT in reducing AFB
1 compared with microwave roasting. (3) Due to the strong absorption of microwaves by water molecules, the water molecules in corn were like a guide in the microwave electromagnetic field, which direct microwave energy to attack some parts of the corn with high AFB
1 concentration. It was because the areas of high AFB
1 concentration (embryo tissue and broken grain) in corn kernel were also the areas with high water content. There were three reasons for this phenomenon: Firstly, due to the relatively short cooking time of corn (stage A, B, and C), the tissues of the corn kernel did not fully saturate with water. Secondly, the corn embryo was rich in protein, which has stronger hydrophilicity than starch. Thirdly, the specific surface area increased after the kernels were broken, and the inner structure was exposed to the outside at the same time, which was also favorable for water entering into the kernel. Because the microwaves were strongly absorbed by water, these areas would receive more microwave energy. The sectional drawing of corn kernels at the end of WMT (36 min) was a strong proof, i.e., the degree of microwave heating among corn kernels was different (
Figure 1d). The full kernels still had the loose structure of the Chalky endosperm and the white color of the starch, suggesting that the microwave energy did not penetrate too much into the kernel. Otherwise, the interior of the kernel would be heated more because of the greater penetration depth of the microwave. On the other hand, corn embryos and irregularly shaped damaged seeds were blackened and charring obviously. It proved that these areas absorbed most of the microwave energy. These features reflected the potential advantages of WMT in reducing AFB
1 in corn.
Besides that, the microwave could kill microorganisms and dehydrate corn efficiently, which brought another advantage of WMT. For instance, the moisture content of corn decreased continuously in the later period of WMT, and finally decreased to less than safe moisture condition (14.0%). Almost all the molds were killed at the early WMT because of the higher ability of microwave sterilization, including AFB
1-producing
Aspergillus flavus [
28]. These two results can effectively avoid the risk of mold growth and mycotoxin production again after being processed, which is of great significance for safe storage.
At last, the changes in free fatty acid value and pasting properties of corn were researched during WMT. Higher temperatures usually caused the increase of the free fatty acid value of corn in the processing of heat treatment [
29]. However, due to the fungal infection, the corn samples had severe rancidity and the free fatty acid content had been up to 200 (KOH)/(mg/100 g). This might limit further increases in free fatty acid values. As for pasting properties of corn powder, all the paste properties were reduced and the pasting temperature was elevated for starch after WMT, which indicated that the WMT could change the pasting properties of corn significantly. The result might be related to an alteration of the starch granules, because microwave irradiation could reduce the crystallinity, solubility, and swelling characteristics of corn starch [
30]. Another explanation might be the production of large amounts of resistant starch after WMT. This is because thermal treatment and high moisture content and cooling processes were the important factors that affect the production of resistant starch [
31]. The same results appeared in David’s experiments [
32]. Therefore, it might be an important application to use the corn treated by WMT to produce resistant starch. In addition, microwave irradiation as a method for starch modification had been reported in many researches [
33]. It might be meaningful to take into account both the reduction of AFB
1 in corn and the modification of corn starch by WMT. That is because it helps to expand the use of corn contaminated by AFB
1 and reduce the economic loss caused by corn contaminated by AFB
1.
5. Materials and Methods
5.1. Chemicals and Reagents
Aflatoxin B1 was purchased from J&K Chemical Ltd. (Shanghai, China). The purity of these materials was 99.5%. AFB1 standard was dissolved in Acetonitrile and diluted to a concentration of 1 mg·L−1, and the AFB1 standard solution was stored at −20 °C in a freezer before conducting the experiment. acetonitrile and methanol were purchased from Thermo Fisher Scientific (Shanghai, China). Corn samples (local variety) were brought from the Local farmers (Zhengzhou city, Henan Province, China).
For the UHPLC-MS analysis, ultrapure water (18.2 MΩ cm−1 resistivity) was obtained from a Milli-Q SP Reagent Water system (Millipore, Bedford, MA, USA) and prefiltered through a 0.2-μm membrane.
5.2. Aspergillus Flavus
Aspergillus flavus was isolated from mildew corn, which was obtained from farmland of Zhengzhou city, Henan Province, China.
A. flavus was grown on potato-dextrose-agar (PDA) slant for 7 days at 28 °C. Spores were harvest by adding sterilized water, filtering through eight layers of sterilized cheesecloth, washing several times with sterilized distilled water, and resuspending in sterilized distilled water [
10].
5.3. Infection of Corn
The spores were scraped by adding sterile distilled water to the surface of the PDA slant and the resulting spore suspension was added to corn samples. Then, the corn was incubated at 30 °C with the moisture content of 19% for 4, 6, 8, and 10 days to allow
Aspergillus flavus growth and AFB
1 production [
10]. AFB
1 concentration produced by
Aspergillus flavus increased gradually with the extension of storage time. Finally, the corn was transferred to plastic bags and stored at −20 °C.
5.4. Water-Assisted Microwave Treatment
The 200 g of corn kernels (containing AFB
1) and 200 mL of distilled water (electrical conductance ≤ 10 μS/cm) were added into a glass beaker (1000 mL) successively. Most of the corn kernels were completely submerged by water, and the water surface was 1 cm approximately above the top of the corn pile. A small part of broken corn kernels floated on the water. Then, they were placed in a type XH-8000 Plus multi-function microwave synthesizer microwave oven (
Figure 5). The microwave oven was manufactured by Beijing Xianghu Science and Technology Development Co., Ltd. (Beijing, China). During the microwave working, the temperature was detected with a fiber optic sensor inserted in the vessel, and shown on the control panel. The status of the reactor was observed using the monitor with a connected camera inside the oven.
The corn samples were treated by WMT for various periods (8, 12, 14, 16, 20, 24, 28, 32, and 36 min). The microwave power was changed from 300 W to 700 W (300 W, 400 W, 500 W, 600 W, and 700 W). When the processing procedure was finished, the glass beaker was moved out of the microwave oven and then the corn kernels were removed. The water on the surface of corn kernels was absorbed with absorbent paper. After cooling at room temperature, the processed corn kernels were moved into airtight bags and stored at room temperature for no more than 24 h before grinding. Then, the processed corn kernels were grinded to determine the AFB1 concentration and corn quality. If corn kernels had high moisture and could not be ground, they needed to be air-dried first.
5.5. UHPLC-MS Analysis
A portion of 5 g of corn powder was weighed in an Erlenmeyer flask. AFB1 was extracted with 20 mL of a mixture of acetonitrile/water/glacial acetic acid (79:20:1, v/v/v). The slurry solution was shaken on a rotary shaker for 20 min at 100 rpm and then centrifuged at 4000 rpm for 20 min (25 °C). A volume of 1 mL of the supernatant obtained from previous centrifugation was diluted in 1 mL of Milli-Q water and then centrifuged at 12,000 rpm for 15 min (4 °C).
The supernatant of the extracts was then transferred to an Eppendorf tube (2.0 mL) for UHPLC-MS (mass spectrometry) analysis. UHPLC-MS data on detection of AFB1 was obtained using a Q-Orbitrap instrument system (Thermo Fisher Scientific, Waltham, MA, USA) equipped with a binary solvent delivery system and an autosampler. The pump used in our UHPLC was a bivariate gradient pump. Chromatography was performed using a 2.1 × 100 mm, 1.7 µm particle C18 column (Thermo Fisher). The injection volume was 5 µL and the flow rate was 300 µL/min. The mobile stage was a gradient prepared from the water with 0.1% formic acid and 5 mol/L ammonium acetate (component A) and methanol (component B). The gradient elution started with 25% B for 1 min, then B was increased linearly to 95% in 1 min, and kept isocratic for 2 min. The proportion of B was then decreased back to 25% and kept isocratic for 2 min. The total run time was 6 min. The MS was run with positive electrospray ionization (ESI), and the data was collected over the range of 150–600 m/z.
5.6. Color and Moisture Content Analysis
The CIELAB method was used to analyze the corn color after WMT treatment. Corn samples were ground so that more than 90% of the corn power could pass through a 0.25 mm aperture sieve. Then, the on-sieve and under-sieve were mixed evenly for color determination. The color reflectance spectrometry measurements of lightness (L*), redness (a*), and yellowness (b*) values were determined at ambient temperature using a Minolta CR410 chroma meter (Konica Minolta Sensing, Inc., Osaka, Japan). The L* value represents lightness, and the +a* and −a* values represent redness and greenness, respectively. The +b* and −b* values represent yellowness and blueness, respectively [
34]. Three repeat measurements were taken for each corn sample, and the mean values were calculated and recorded.
The moisture content of corn was tested according to the AACC (American Association of Cereal Chemists) Method 44–15 A. Corn kernels were cooled to room temperature after WMT processing and the water on the surface of corn kernels was absorbed with absorbent paper. These corn samples were placed in a glass dish (15 cm in diameter) and their initial weight (m
1) was recorded. Then, they were dried for 12–24 h at 45°C in an oven, so that the moisture content of the corn kernels was less than 10%. We weighed these corn kernels after they cooled to room temperature (m
2). These corn kernels were ground with a laboratory mill (equipped with a 20-mesh screen) and we transferred immediately a 4- to 5-g portion to each of three tared moisture dishes; We covered and weighed dishes at once and subtracted tare weights and recorded weight of the sample (m
3). Then, we uncovered the dishes and placed them on the shelf of an oven. Oven (mechanical-convection) temperature was maintained at 105 °C (±1 °C) for 3 or 6 h. The dishes were removed from the oven and covered rapidly (using rubber finger insulators), and then they were transferred to a desiccator as quickly as possible. The corn powder in dishes (subtract the weight of each dish from the total weight) were weighed after they cooled to room temperature (m
4). Replicate determinations were checked within 0.2% moisture. The calculation method of moisture content was performed using Equation (1):
5.7. Microbiological Analysis of Samples
The identification of mold counts and microfloral species from corn samples were conducted according to the following method [
35]: 25 g of each corn sample was withdrawn and added to 225 mL of sterile distilled water in a 1-L flask bottle. Then, they were shaken for 30 min on a flatbed shaker at 300 excursions per minute to completely dissolve the microorganism in the corn samples. The sample solution was diluted 10
2–10
6 times, and 1 mL of each diluted solution was plated on modified Czapek’s medium (3% sucrose, 2% agar powder, 0.05% KCl, 0.05%, MgSO
4 × 7H
2O, 0.1% K
2HPO
4, 0.3% NaNO
3, and 6% NaCl) and incubated at 28 °C for 7 days. The mold count was calculated at day 5 and the microfloral species were identified using a microscope at day 7.
5.8. Free Fatty Acid Analysis of Samples
The determination of free fatty acid of corn samples was conducted according to the following method [
36]. A test portion was extracted by petroleum ether for 10 min, filtrated, and the filtrate was added into 50% ethanol solution; then, it was titrated with a standard volumetric sodium hydroxide solution using phenolphthalein as indicator until the end-point was indicated by a sharp change of color from neutral to pink.
5.9. Analysis of Pasting Properties
Microwaved corn starch pasting properties were analyzed using a Rapid Visco Analyser (RVA-4, Foss North America, Eden Prairie, MN, USA), using a method previously used for starch suspensions [
37]. Viscosity profiles of starches from corn were recorded using starch suspensions (6%,
w/
w; 28 g total weight). A programmed heating and cooling cycle were used, where the samples were held at 50 °C for 1 min, heated to 95 °C at 6 °C/min, held at 95 °C for 2.7 min, before cooling from 95 °C to 50 °C at 6 °C/min and holding at 50 °C for 2 min. Parameters recorded were pasting temperature, peak viscosity, trough viscosity (minimum viscosity at 95 °C), final viscosity (viscosity at 50 °C), breakdown viscosity (peak-trough viscosity), and setback viscosity (final-trough viscosity). All measurements were replicated thrice.
5.10. Statistical Analysis
All experiments were independently performed a minimum of three times and data was expressed as mean ± standard deviation (SD). Statistics were analyzed with SPSS 16.0 (SPSS Institute, Chicago, IL, USA). The statistical significance of the data was analyzed using Duncan’s multiple range post hoc tests (p < 0.05).