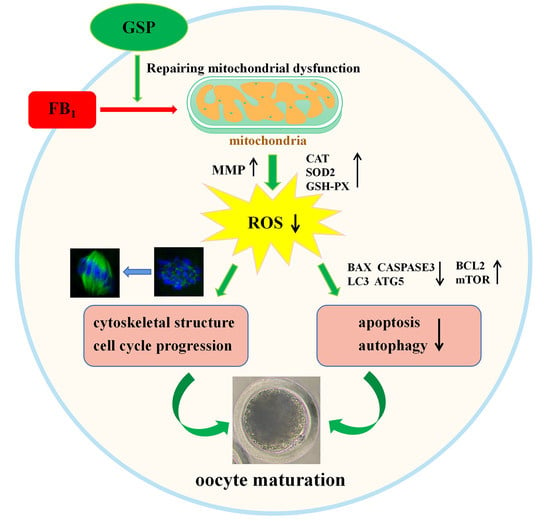
Grape Seed Proanthocyanidin Ameliorates FB1-Induced Meiotic Defects in Porcine Oocytes
Abstract
1. Introduction
2. Results
2.1. GSP Ameliorated Meiotic Maturation Defects in FB1-Exposed Porcine Oocytes
2.2. GSP Protected the Cell Cycle Progression of FB1-Exposed Porcine Oocytes
2.3. GSP Alleviated the Defects of Spindle Assembly and Actin Distribution in FB1-Exposed Oocytes
2.4. GSP Protected Mitochondrial Function in FB1-Exposed Oocytes
2.5. GSP Alleviated Oxidative Stress in FB1-Exposed Oocytes
2.6. GSP Reduced Early Apoptosis in FB1-Exposed Oocytes
2.7. GSP Decreased Autophagy Levels in FB1-Exposed Oocytes
3. Discussion
4. Conclusions
5. Materials and Methods
5.1. Antibodies and Chemicals
5.2. Ethics Statement and Porcine Oocyte In Vitro Maturation
5.3. FB1 Exposure and GSP Treatment
5.4. Oocyte Parthenogenetic Activation and Early Embryo Culture In Vitro
5.5. Immunofluorescence Staining
5.6. Mitochondrial Function Evaluation
5.7. Measurement of ROS
5.8. Annexin-V Staining
5.9. Fluorescence Intensity Analysis
5.10. Western Blotting
5.11. Quantitative Real-Time PCR (qRT-PCR)
5.12. Experimental Design
5.12.1. Effects of GSP on the Meiotic Maturation of FB1-Exposed Porcine Oocytes
5.12.2. Effects of GSP on the Embryo Developmental Potential of FB1-Exposed Porcine Oocytes
5.12.3. Effects of GSP on the Cell Cycle Progression of FB1-Exposed Porcine Oocytes
5.12.4. Effects of GSP on Spindle Assembly and Actin Distribution in FB1-Exposed Porcine Oocytes
5.12.5. Effects of GSP on Mitochondrial Function in FB1-Exposed Porcine Oocytes
5.12.6. Effects of GSP on Oxidative Stress in FB1-Exposed Porcine Oocytes
5.12.7. Effects of GSP on Apoptosis in FB1-Exposed Porcine Oocytes
5.12.8. Effects of GSP on Autophagy Levels in FB1-Exposed Porcine Oocytes
5.13. Statistical Analyses
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Kamle, M.; Mahato, D.K.; Devi, S.; Lee, K.E.; Kang, S.G.; Kumar, P. Fumonisins: Impact on agriculture, food, and human health and their management strategies. Toxins 2019, 11, 328. [Google Scholar] [CrossRef]
- Ponce-Garcia, N.; Serna-Saldivar, S.O.; Garcia-Lara, S. Fumonisins and their analogues in contaminated corn and its processed foods—A review. Food Addit. Contam. Part A Chem. Anal. Control Expo. Risk Assess. 2018, 35, 2183–2203. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Jiang, Y.; Li, R.; Pang, M.; Liu, Y.; Dong, J. Natural occurrence of fumonisins B1 and B2 in maize from eight provinces of China in 2014. Food Addit. Contam. Part B Surveill. 2017, 10, 113–117. [Google Scholar] [CrossRef]
- Régnier, M.; Polizzi, A.; Lukowicz, C.; Smati, S.; Lasserre, F.; Lippi, Y.; Naylies, C.; Laffitte, J.; Bétoulières, C.; Montagner, A.; et al. The protective role of liver X receptor (LXR) during fumonisin B1-induced hepato-toxicity. Arch. Toxicol. 2019, 93, 505–517. [Google Scholar] [CrossRef]
- Müller, S.; Dekant, W.; Mally, A. Fumonisin B1 and the kidney: Modes of action for renal tumor formation by fumonisin B1 in rodents. Food Chem. Toxicol. 2012, 50, 3833–3846. [Google Scholar] [CrossRef]
- Domijan, A.M. Fumonisin B (1): A neurotoxic mycotoxin. Arch. Ind. Hyg. Toxicol. 2012, 63, 531. [Google Scholar] [CrossRef]
- Li, Y.; Fan, Y.; Xia, B.; Xiao, Q.; Wang, Q.; Sun, W.; Zhang, H.; He, C. The immunosuppressive characteristics of FB1 by inhibition of maturation and function of BMDCs. Int. Immunopharmacol. 2017, 47, 206–211. [Google Scholar] [CrossRef] [PubMed]
- IARC. Some traditional herbal medicines, some mycotoxins, naphthalene and styrene. IARC Monogr. Eval. Carcinog. Risks Hum. 2002, 82, 1–556. [Google Scholar]
- Desai, K.; Sullards, M.C.; Allegood, J.; Wang, E.; Schmelz, E.M.; Hartl, M.; Humpf, H.U.; Liotta, D.C.; Peng, Q.; Jr, M.A.H. Fumonisins and fumonisin analogs as inhibitors of ceramide synthase and inducers of apoptosis. Biochim. Biophys. Acta 2002, 1585, 188–192. [Google Scholar] [CrossRef]
- Liu, X.; Fan, L.; Yin, S.; Chen, H.; Hu, H. Molecular mechanisms of fumonisin B1-induced toxicities and its applications in the mechanism-based interventions. Toxicon 2019, 167, 1–5. [Google Scholar] [CrossRef] [PubMed]
- Cortinovis, C.; Pizzo, F.; Spicer, L.J.; Caloni, F. Fusarium mycotoxins: Effects on reproductive function in domestic animals--a review. Theriogenology 2013, 80, 557–564. [Google Scholar] [CrossRef] [PubMed]
- Lumsangkul, C.; Chiang, H.I.; Lo, N.W.; Fan, Y.K.; Ju, J.C. Developmental toxicity of mycotoxin fumonisin B1 in animal embryogenesis: An overview. Toxins 2019, 11, 114. [Google Scholar] [CrossRef] [PubMed]
- Gelineau-van Waes, J.; Starr, L.; Maddox, J.; Aleman, F.; Voss, K.A.; Wilberding, J.; Riley, R.T. Maternal fumonisin exposure and risk for neural tube defects: Mechanisms in an in vivo mouse model. Birth Defects Res. A Clin. Mol. Teratol. 2005, 73, 487–497. [Google Scholar] [CrossRef]
- Ewuola, E.O.; Egbunike, G.N. Gonadal and extra-gonadal sperm reserves and sperm production of pubertal rabbits fed dietary fumonisin B1. Anim. Reprod. Sci. 2010, 119, 282–286. [Google Scholar] [CrossRef] [PubMed]
- Gbore, F.A.; Egbunike, G.N. Testicular and epididymal sperm reserves and sperm production of pubertal boars fed dietary fumonisin B(1). Anim. Reprod. Sci. 2008, 105, 392–397. [Google Scholar] [CrossRef] [PubMed]
- Cortinovis, C.; Caloni, F.; Schreiber, N.B.; Spicer, L.J. Effects of fumonisin B1 alone and combined with deoxynivalenol or zearalenone on porcine granulosa cell proliferation and steroid production. Theriogenology 2014, 81, 1042–1049. [Google Scholar] [CrossRef] [PubMed]
- Brunet, S.; Maro, B. Cytoskeleton and cell cycle control during meiotic maturation of the mouse oocyte: Integrating time and space. Reproduction 2005, 130, 801–811. [Google Scholar] [CrossRef]
- Luo, Y.; Che, M.; Liu, C.; Liu, H.; Fu, X.; Hou, Y. Toxicity and related mechanisms of dihydroartemisinin on porcine oocyte maturation in vitro. Toxicol. Appl. Pharmacol. 2018, 341, 8–15. [Google Scholar] [CrossRef]
- Tarazona, A.M.; Rodriguez, J.I.; Restrepo, L.F.; Olivera-Angel, M. Mitochondrial activity, distribution and segregation in bovine oocytes and in embryos produced in vitro. Reprod. Domest. Anim. 2006, 41, 5–11. [Google Scholar] [CrossRef]
- Abate, M.; Festa, A.; Falco, M.; Lombardi, A.; Luce, A.; Grimaldi, A.; Zappavigna, S.; Sperlongano, P.; Irace, C.; Caraglia, M.; et al. Mitochondria as playmakers of apoptosis, autophagy and senescence. Semin. Cell Dev. Biol. 2020, 98, 139–153. [Google Scholar] [CrossRef] [PubMed]
- Ding, Z.M.; Ahmad, M.J.; Meng, F.; Chen, F.; Wang, Y.S.; Zhao, X.Z.; Zhang, S.X.; Miao, Y.L.; Xiong, J.J.; Huo, L.J. Triclocarban exposure affects mouse oocyte in vitro maturation through inducing mitochondrial dysfunction and oxidative stress. Environ. Pollut. 2020, 262, 114271. [Google Scholar] [CrossRef] [PubMed]
- Li, W.; Zhao, H.; Zhuang, R.; Wang, Y.; Cao, W.; He, Y.; Jiang, Y.; Rui, R.; Ju, S.J.T. Fumonisin B exposure adversely affects porcine oocyte maturation in vitro by inducing mitochondrial dysfunction and oxidative stress. Theriogenology 2021, 164, 1–11. [Google Scholar] [CrossRef]
- Zhang, L.; Wang, Y.; Li, D.; Ho, C.T.; Li, J.; Wan, X. The absorption, distribution, metabolism and excretion of procyanidins. Food Funct. 2016, 7, 1273–1281. [Google Scholar] [CrossRef] [PubMed]
- Rodríguez-Pérez, C.; García-Villanova, B.; Guerra-Hernández, E.; Verardo, V. Grape seeds proanthocyanidins: An overview of in vivo bioactivity in animal models. Nutrients 2019, 11, 2435. [Google Scholar] [CrossRef] [PubMed]
- Zeng, Y.X.; Wang, S.; Wei, L.; Cui, Y.Y.; Chen, Y.H. Proanthocyanidins: Components, pharmacokinetics and biomedical properties. Am. J. Chin. Med. 2020, 48, 813–869. [Google Scholar] [CrossRef]
- Bagchi, D.; Swaroop, A.; Preuss, H.G.; Bagchi, M. Free radical scavenging, antioxidant and cancer chemoprevention by grape seed proanthocyanidin: An overview. Mutat. Res. 2014, 768, 69–73. [Google Scholar] [CrossRef] [PubMed]
- Bladé, C.; Aragonès, G.; Arola-Arnal, A.; Muguerza, B.; Bravo, F.I.; Salvadó, M.J.; Arola, L.; Suárez, M. Proanthocyanidins in health and disease. BioFactors 2016, 42, 5–12. [Google Scholar] [CrossRef] [PubMed]
- Rajput, S.A.; Zhang, C.; Feng, Y.; Wei, X.T.; Khalil, M.M.; Rajput, I.R.; Baloch, D.M.; Shaukat, A.; Rajput, N.; Qamar, H.; et al. Proanthocyanidins alleviates aflatoxinB1-induced oxidative stress and apoptosis through mitochondrial pathway in the bursa of fabricius of broilers. Toxins 2019, 11, 157. [Google Scholar] [CrossRef] [PubMed]
- Zhao, Y.; Gao, L.P.; Zhang, H.L.; Guo, J.X.; Guo, P.P. Grape seed proanthocyanidin extract prevents DDP-induced testicular toxicity in rats. Food Funct. 2014, 5, 605–611. [Google Scholar] [CrossRef] [PubMed]
- Su, L.; Deng, Y.; Zhang, Y.; Li, C.; Zhang, R.; Sun, Y.; Zhang, K.; Li, J.; Yao, S. Protective effects of grape seed pro-cyanidin extract against nickel sulfate-induced apoptosis and oxidative stress in rat testes. Toxicol. Mech. Methods 2011, 21, 487–494. [Google Scholar] [CrossRef]
- Bashir, N.; Shagirtha, K.; Manoharan, V.; Miltonprabu, S. The molecular and biochemical insight view of grape seed proan-thocyanidins in ameliorating cadmium-induced testes-toxicity in rat model: Implication of PI3K/Akt/Nrf-2 signaling. Biosci. Rep. 2019, 39, BSR20180515. [Google Scholar] [CrossRef] [PubMed]
- Abdel-Kawi, S.H.; Hashem, K.S.; Abd-Allah, S. Mechanism of diethylhexylphthalate (DEHP) induced testicular damage and of grape seed extract-induced protection in the rat. Food Chem. Toxicol. 2016, 90, 64–75. [Google Scholar] [CrossRef] [PubMed]
- Liu, X.; Lin, X.; Mi, Y.; Li, J.; Zhang, C. Grape seed proanthocyanidin extract prevents ovarian aging by inhibiting ox-idative stress in the hens. Oxid. Med. Cell. Longev. 2018, 2018, 9390810. [Google Scholar] [CrossRef] [PubMed]
- El-Ashmawy, I.M.; Bayad, A.E. Folic acid and grape seed extract prevent azathioprine-induced fetal malformations and renal toxicity in rats. Phytother. Res. PTR 2016, 30, 2027–2035. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J.; Gao, B.; Wang, J.; Ren, Q.; Chen, J.; Ma, Q.; Zhang, Z.; Xing, B. Critical role of foxO1 in granulosa cell apoptosis caused by oxidative stress and protective effects of grape seed procyanidin B2. Oxid. Med. Cell. Longev. 2016, 2016, 6147345. [Google Scholar] [CrossRef]
- Prasain, J.K.; Peng, N.; Dai, Y.; Moore, R.; Arabshahi, A.; Wilson, L.; Barnes, S.; Michael Wyss, J.; Kim, H.; Watts, R.L. Liquid chromatography tandem mass spectrometry identification of proanthocyanidins in rat plasma after oral administration of grape seed extract. Phytomedicine 2009, 16, 233–243. [Google Scholar] [CrossRef] [PubMed]
- Sharma, A.; Tiwari, M.; Gupta, A.; Pandey, A.N.; Yadav, P.K.; Chaube, S.K. Journey of oocyte from metaphase-I to metaphase-II stage in mammals. J. Cell. Physiol. 2018, 233, 5530–5536. [Google Scholar] [CrossRef]
- Hou, F.; Xiao, M.; Li, J.; Cook, D.; Zeng, W.; Zhang, C.; Mi, Y. Ameliorative effect of grape seed proanthocyanidin extract on cadmium-induced meiosis inhibition during oogenesis in chicken embryos. Anat. Rec. 2016, 299, 450–460. [Google Scholar] [CrossRef]
- Marin, D.E.; Gouze, M.E.; Taranu, I.; Oswald, I.P. Fumonisin B1 alters cell cycle progression and interleukin-2 synthesis in swine peripheral blood mononuclear cells. Mol. Nutr. Food Res. 2007, 51, 1406–1412. [Google Scholar] [CrossRef]
- Zhao, X.; Wang, Y.; Liu, J.L.; Zhang, J.H.; Zhang, S.C.; Ouyang, Y.; Huang, J.T.; Peng, X.Y.; Zeng, Z.; Hu, Z.Q. Fumonisin B1 affects the biophysical properties, migration and cytoskeletal structure of human umbilical vein endothelial cells. Cell Biochem. Biophys. 2020, 78, 375–382. [Google Scholar] [CrossRef]
- Hassan, H.A.; Edrees, G.M.; El-Gamel, E.M.; El-Sayed, E.A. Proanthocyanidin and fish oil potent activity against cispla-tin-induced renal cell cycle arrest and apoptosis in rats. Ren. Fail. 2015, 37, 1356–1362. [Google Scholar] [CrossRef] [PubMed]
- Caton, P.W.; Pothecary, M.R.; Lees, D.M.; Khan, N.Q.; Wood, E.G.; Shoji, T.; Kanda, T.; Rull, G.; Corder, R. Regulation of vascular endothelial function by procyanidin-rich foods and beverages. J. Agric. Food Chem. 2010, 58, 4008–4013. [Google Scholar] [CrossRef] [PubMed]
- Romek, M.; Gajda, B.; Rolka, M.; Smorag, Z. Mitochondrial activity and morphology in developing porcine oocytes and pre-implantation non-cultured and cultured embryos. Reprod. Domest. Anim. 2011, 46, 471–480. [Google Scholar] [CrossRef] [PubMed]
- Domijan, A.; Abramov, A. Fumonisin B1 inhibits mitochondrial respiration and deregulates calcium homeosta-sis--implication to mechanism of cell toxicity. J. Biochem. Cell Biol. 2011, 43, 897–904. [Google Scholar] [CrossRef]
- Al-Zubaidi, U.; Liu, J.; Cinar, Q.; Robker, R.L.; Adhikari, D.; Carroll, J. The spatio-temporal dynamics of mitochondrial membrane potential during oocyte maturation. Mol. Hum. Reprod. 2019, 25, 695–705. [Google Scholar] [CrossRef]
- Sun, M.; Li, X.; Xu, Y.; Xu, Y.; Pan, Z.; Sun, S. Citrinin exposure disrupts organelle distribution and functions in mouse oocytes. Environ. Res. 2020, 185, 109476. [Google Scholar] [CrossRef]
- Cerbaro, A.F.; Rodrigues, V.S.B.; Rigotti, M.; Branco, C.S.; Rech, G.; de Oliveira, D.L.; Salvador, M. Grape seed proanthocya-nidins improves mitochondrial function and reduces oxidative stress through an increase in sirtuin 3 expression in EA. hy926 cells in high glucose condition. Mol. Biol. Rep. 2020, 47, 3319–3330. [Google Scholar] [CrossRef]
- Li, L.; Geng, X.; Tian, L.; Wang, D.; Wang, Q. Grape seed proanthocyanidins protect retinal ganglion cells by inhibiting oxidative stress and mitochondrial alteration. Arch. Pharm. Res. 2020, 43, 1056–1066. [Google Scholar] [CrossRef]
- Pajuelo, D.; Quesada, H.; Díaz, S.; Fernández-Iglesias, A.; Arola-Arnal, A.; Bladé, C.; Salvadó, J.; Arola, L. Chronic dietary supplementation of proanthocyanidins corrects the mitochondrial dysfunction of brown adipose tissue caused by diet-induced obesity in Wistar rats. Br. J. Nutr. 2012, 107, 170–178. [Google Scholar] [CrossRef]
- Roth, Z. Symposium review: Reduction in oocyte developmental competence by stress is associated with alterations in mi-tochondrial function. J. Dairy Sci. 2018, 101, 3642–3654. [Google Scholar] [CrossRef] [PubMed]
- Zhang, X.; Zhou, C.; Li, W.; Li, J.; Wu, W.; Tao, J.; Liu, H. Vitamin C protects porcine oocytes from microcystin-LR toxicity during maturation. Front. Cell Dev. Biol. 2020, 8, 582715. [Google Scholar] [CrossRef]
- Prasad, S.; Tiwari, M.; Pandey, A.; Shrivastav, T.; Chaube, S. Impact of stress on oocyte quality and reproductive outcome. J. Biomed. Sci. 2016, 23, 36. [Google Scholar] [CrossRef]
- Stockmann-Juvala, H.; Savolainen, K. A review of the toxic effects and mechanisms of action of fumonisin B1. Hum. Exp. Toxicol. 2008, 27, 799–809. [Google Scholar] [CrossRef]
- Tiwari, M.; Prasad, S.; Tripathi, A.; Pandey, A.N.; Ali, I.; Singh, A.K.; Shrivastav, T.S.; Chaube, S.K. Apoptosis in mammalian oocytes: A review. Apoptosis 2015, 20, 1019–1025. [Google Scholar] [CrossRef]
- Kim, S.H.; Singh, M.P.; Sharma, C.; Kang, S.C. Fumonisin B1 actuates oxidative stress-associated colonic damage via apoptosis and autophagy activation in murine model. J. Biochem. Mol. Toxicol. 2018, e22161. [Google Scholar] [CrossRef]
- Chang, P.; Mo, B.; Cauvi, D.M.; Yu, Y.; Guo, Z.H.; Zhou, J.; Huang, Q.; Yan, Q.; Chen, G.M.; Liu, Z.G. Grape seed proantho-cyanidin extract protects lymphocytes against histone-induced apoptosis. PeerJ 2017, 5, e3108. [Google Scholar] [CrossRef]
- Chen, Y.; McMillan-Ward, E.; Kong, J.; Israels, S.J.; Gibson, S.B. Oxidative stress induces autophagic cell death independent of apoptosis in transformed and cancer cells. Cell Death Differ. 2008, 15, 171–182. [Google Scholar] [CrossRef]
- Parzych, K.; Klionsky, D. An overview of autophagy: Morphology, mechanism, and regulation. Antioxid. Redox Signal. 2014, 20, 460–473. [Google Scholar] [CrossRef]
- Zhou, J.; Peng, X.; Mei, S. Autophagy in ovarian follicular development and atresia. Int. J. Biol. Sci. 2019, 15, 726–737. [Google Scholar] [CrossRef]
- Fulda, S.; Kögel, D. Cell death by autophagy: Emerging molecular mechanisms and implications for cancer therapy. Oncogene 2015, 34, 5105–5113. [Google Scholar] [CrossRef]
- Yin, S.; Guo, X.; Li, J.; Fan, L.; Hu, H. Fumonisin B1 induces autophagic cell death via activation of ERN1-MAPK8/9/10 pathway in monkey kidney MARC-145 cells. Arch. Toxicol. 2016, 90, 985–996. [Google Scholar] [CrossRef] [PubMed]
- Dai, J.P.; Wang, G.F.; Li, W.Z.; Zhang, L.; Yang, J.C.; Zhao, X.F.; Chen, X.X.; Xu, Y.X.; Li, K.S. High-throughput screening for anti-influenza A virus drugs and study of the mechanism of procyanidin on influenza A virus-induced autophagy. J. Biomol. Screen. 2012, 17, 605–617. [Google Scholar] [CrossRef] [PubMed]
- Shi, F.; Li, W.; Zhao, H.; He, Y.; Jiang, Y.; Ni, J.; Abbasi, B.; Rui, R.; Ju, S. Microcystin-LR exposure results in aberrant spindles and induces apoptosis in porcine oocytes. Theriogenology 2020, 158, 358–367. [Google Scholar] [CrossRef] [PubMed]
- Cui, Z.; Yu, L.; Shi Yang, X.; Zhang, Y.; Shi, X.; Li, Y.; Chen, Q.; Xiong, B. Brefeldin A impairs porcine oocyte meiotic maturation via interruption of organelle dynamics. J. Cell. Physiol. 2019, 234, 20111–20117. [Google Scholar] [CrossRef] [PubMed]
- Mantena, S.K.; Katiyar, S.K. Grape seed proanthocyanidins inhibit UV-radiation-induced oxidative stress and activation of MAPK and NF-kappaB signaling in human epidermal keratinocytes. Free Radic. Biol. Med. 2006, 40, 1603–1614. [Google Scholar] [CrossRef] [PubMed]
- Barbe, A.; Ramé, C.; Mellouk, N.; Estienne, A.; Bongrani, A.; Brossaud, A.; Riva, A.; Guérif, F.; Froment, P.; Dupont, J. Effects of grape seed extract and proanthocyanidin b2 on in vitro proliferation, viability, steroidogenesis, oxidative stress, and cell signaling in human granulosa cells. Int. J. Mol. Sci. 2019, 20, 4215. [Google Scholar] [CrossRef] [PubMed]
- Yang, C.; Wang, P.; Liu, S.; Miao, J.; Liu, X.; Miao, Y.; Du, Z. Long noncoding RNA 2193 regulates meiosis through global epigenetic modification and cytoskeleton organization in pig oocytes. J. Cell. Physiol. 2020, 235, 8304–8318. [Google Scholar] [CrossRef]
- Ju, S.; Peng, X.; Yang, X.; Sozar, S.; Muneri, C.; Xu, Y.; Chen, C.; Cui, P.; Xu, W.; Rui, R.J.T. Aurora B inhibitor barasertib prevents meiotic maturation and subsequent embryo development in pig oocytes. Theriogenology 2016, 86, 503–515. [Google Scholar] [CrossRef]
- Almubarak, A.; Kim, E.; Yu, I.; Jeon, Y. Supplementation with Niacin during in vitro maturation improves the quality of porcine embryos. Theriogenology 2021, 169, 36–46. [Google Scholar] [CrossRef]
- Zhang, Y.; Wang, T.; Lan, M.; Zang, X.W.; Li, Y.L.; Cui, X.S.; Kim, N.H.; Sun, S.C. Melatonin protects oocytes from MEHP exposure-induced meiosis defects in porcine. Biol. Reprod. 2018, 98, 286–298. [Google Scholar] [CrossRef]






| Gene | Primer Sequence (5′-3′) | Fragment Size (bp) |
|---|---|---|
| GAPDH | F-5’-CGTCCCTGAGACACGATGGT-3’ R-5’-GCCTTGACTGTGCCGTGGAAT-3’ | 194 |
| CAT | F-5’-AACTGTCCCTTCCGTGCTA-3’ R-5’-CCTGGGTGACATTATCTTCG-3’ | 195 |
| GSH-PX | F-5’-CAAGTCCTTCTACGACCTCA-3’ R-5’-GAAGCCAAGAACCACCAG-3’ | 210 |
| SOD1 | F-5’-ACCTGGGCAATGTGACTG-3’ R-5’-TCCAGCATTTCCCGTCT-3’ | 197 |
| SOD2 | F-5’-GGACAAATCTGAGCCCTAACG-3’ R-5’-CCTTGTTGAAACCGAGCC-3’ | 184 |
| BAX | F-5’-CCAGGATCGAGCAGGGCGAAT-3’ R-5’-CACAGGGCCTTGAGCACCAGTTT-3’ | 285 |
| BCL-2 | F-5’-CAGGGACAGCGTATCAGAGC-3’ R-5’-TTGCGATCCGACTCACCAAT-3 | 156 |
| CASPASE-3 | F-5’-GAACTCTAACTGGCAAACCCAA-3’ R-5’-GCATACAAGAAGTCTGCCTCAA-3’ | 142 |
| LC3 | F-5’-CCGAACCTTCGAACAGAGAG-3’ | 206 |
| R-5’-AGGCTTGGTTAGCATTGAGC-3’ | ||
| LAMP2 | F-5’-GCTTTTGCAGCGTTGTGG-3’ | 169 |
| R-5’-GACGAGGCAGAGCATAAGGAG-3’ | ||
| mTOR | F-5’-GCACAAGGACGGATTCCTAC-3’ | 248 |
| R-5’-CACTTGCGTTGGGAGATC-3’ | ||
| ATG3 | F-5’-CACGACTATGGTTGTTTGGCTATG-3’ | 127 |
| R-5’-GGTGGAAGGTGAGGGTGATTT-3’ | ||
| ATG5 | F-5’-CCTGAAGATGGGGAAAGAAAGA-3’ | 140 |
| R-5’-TCTGTTGGTTGCGGGATG-3’ |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Li, W.; He, Y.; Zhao, H.; Peng, L.; Li, J.; Rui, R.; Ju, S. Grape Seed Proanthocyanidin Ameliorates FB1-Induced Meiotic Defects in Porcine Oocytes. Toxins 2021, 13, 841. https://doi.org/10.3390/toxins13120841
Li W, He Y, Zhao H, Peng L, Li J, Rui R, Ju S. Grape Seed Proanthocyanidin Ameliorates FB1-Induced Meiotic Defects in Porcine Oocytes. Toxins. 2021; 13(12):841. https://doi.org/10.3390/toxins13120841
Chicago/Turabian StyleLi, Wenhui, Yijing He, Hongyu Zhao, Lei Peng, Jia Li, Rong Rui, and Shiqiang Ju. 2021. "Grape Seed Proanthocyanidin Ameliorates FB1-Induced Meiotic Defects in Porcine Oocytes" Toxins 13, no. 12: 841. https://doi.org/10.3390/toxins13120841
APA StyleLi, W., He, Y., Zhao, H., Peng, L., Li, J., Rui, R., & Ju, S. (2021). Grape Seed Proanthocyanidin Ameliorates FB1-Induced Meiotic Defects in Porcine Oocytes. Toxins, 13(12), 841. https://doi.org/10.3390/toxins13120841