Fatty Acids and Oxylipins as Antifungal and Anti-Mycotoxin Agents in Food: A Review
Abstract
:1. Introduction
2. Effects of FAs on the Regulation of Fungal Growth and Mycotoxin Synthesis
3. Roles of Oxylipins in Regulating Fungal Growth and Mycotoxin Production
4. Pathways of Oxylipin Formation
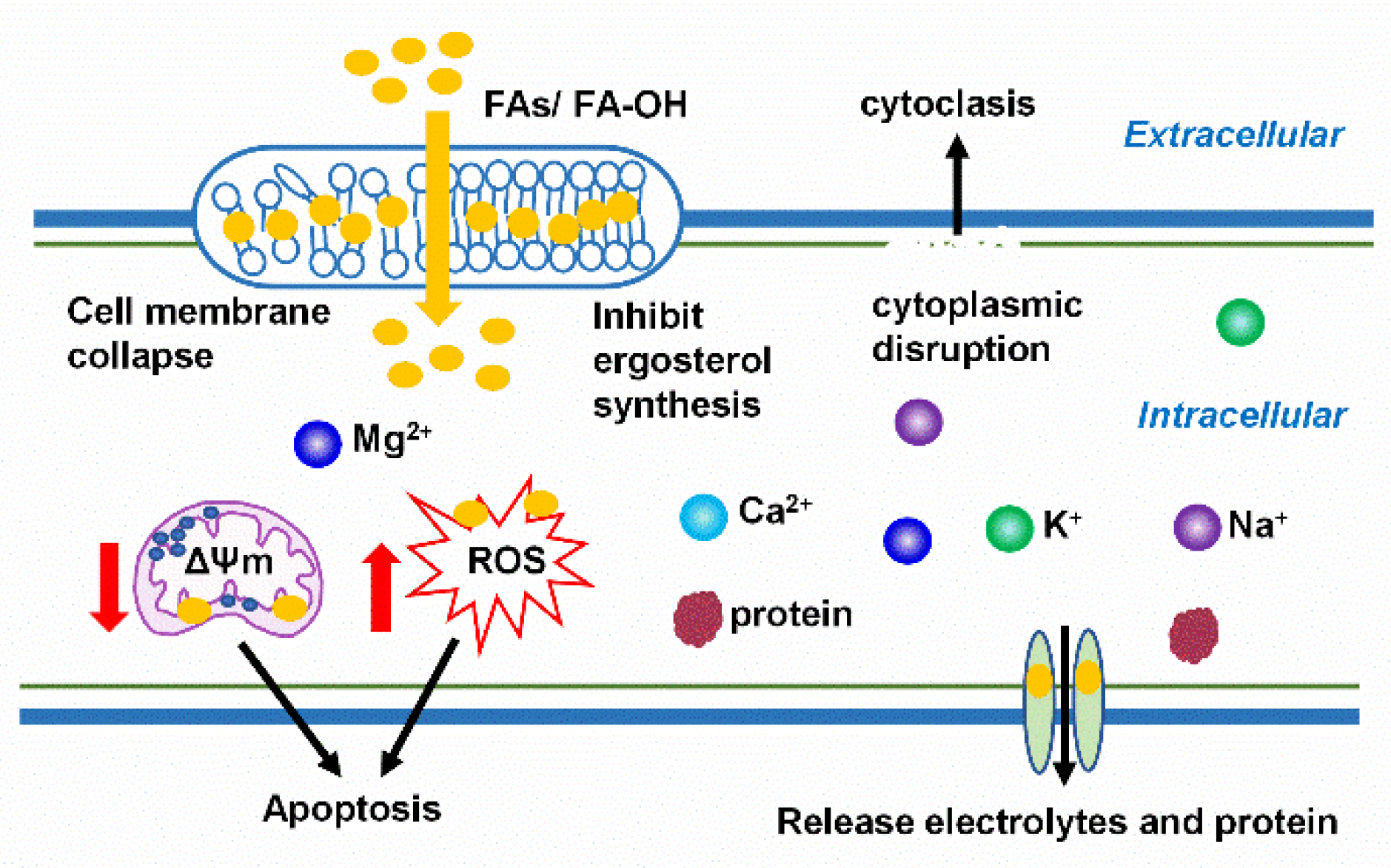
5. Mechanisms Underlying the Antifungal Actions of FAs and Oxylipins
6. Oxylipins in Foods
7. Conclusions and Remarks
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Conflicts of Interest
References
- Garnier, L.; Penland, M.; Thierry, A.; Maillard, M.B.; Jardin, J.; Coton, M.; Leyva-Salas, M.; Coton, E.; Valence, F.; Mounier, J. Antifungal activity of fermented dairy ingredients: Identification of antifungal compounds. Int. J. Food Microbiol. 2020, 322, 108574. [Google Scholar] [CrossRef] [PubMed]
- Faizan, A.S.; Bowen, Y.; Fengwei, T.; Jianxin, Z.; Hao, Z.; Wei, C. Lactic Acid Bacteria as Antifungal and Anti-Mycotoxigenic Agents: A Comprehensive Review. Compr. Rev. Food Sci. Food Saf. 2019, 18, 1403–1436. [Google Scholar] [CrossRef] [Green Version]
- Liu, Y.; Yamdeu, J.G.; Gong, Y.; Caroline, O. A review of postharvest approaches to reduce fungal and mycotoxin contamination of foods. Compr. Rev. Food Sci. Food Saf. 2020, 19, 1521–1560. [Google Scholar] [CrossRef]
- Liao, W.; Badri, W.; Dumas, E.; Ghnimi, S.; Elaissari, A.; Saurel, R.; Gharsallaoui, A. Nanoencapsulation of Essential Oils as Natural Food Antimicrobial Agents: An Overview. Appl. Sci. 2021, 11, 5778. [Google Scholar] [CrossRef]
- Maurya, A.; Prasad, J.; Das, S.; Dwivedy, A.K. Essential Oils and Their Application in Food Safety. Front. Sustain. Food Syst. 2021, 5, 133. [Google Scholar] [CrossRef]
- Chaudhari, A.K.; Singh, V.K.; Das, S.; Dubey, N.K. Nanoencapsulation of essential oils and their bioactive constituents: A novel strategy to control mycotoxin contamination in food system. Food. Chem. Toxicol. 2021, 149, 112019. [Google Scholar] [CrossRef]
- Liu, S.; Ruan, W.; Li, J.; Xu, H.; Wang, J.; Gao, Y.; Wang, J. Biological Control of Phytopathogenic Fungi by Fatty Acids. Mycopathologia 2008, 166, 93–102. [Google Scholar] [CrossRef]
- Christinat, N.; Morin-Rivron, D.; Masoodi, M. High-throughput quantitative lipidomics analysis of nonesterified fatty acids in human plasma. Proteome Res. 2016, 15, 2228–2235. [Google Scholar] [CrossRef]
- Corrêa-Oliveira, R.; Fachi, J.L.; Vieira, A.; Sato, F.T.; Vinolo, M.A.R. Regulation of immune cell function by short-chain fatty acids. Clin. Transl. Immunol. 2016, 5, 73. [Google Scholar] [CrossRef] [PubMed]
- Kimura, I.; Ichimura, A.; Ohue-Kitano, R.; Igarashi, M. Free fatty acid receptors in health and disease. Physiol. Rev. 2020, 100, 171–210. [Google Scholar] [CrossRef]
- Walters, D.; Raynor, L.; Mitchell, A.; Walker, R.; Walker, K. Antifungal activities of four fatty acids against plant pathogenic fungi. Mycopathologia 2004, 157, 87–90. [Google Scholar] [CrossRef]
- Clément, M.; Tremblay, J.; Lange, M.; Thibodeau, J.; Belhumeur, P. Purification and Identification of Bovine Cheese Whey Fatty Acids Exhibiting In Vitro Antifungal Activity. J. Dairy Sci. 2008, 91, 2535–2544. [Google Scholar] [CrossRef] [Green Version]
- Dimitrios, I.T.; Nancy, P.K. Oxylipins as developmental and host-fungal communication signals. Trends Microbiol. 2007, 15, 109–118. [Google Scholar] [CrossRef]
- Gessler, N.N.; Filippovich, Y.S.; Bachurina, G.P.; Kharchenko, E.A.; Groza, N.V.; Belozerskaya, T.A. Oxylipins and Oxylipin Synthesis Pathways in Fungi. Appl. Biochem. Microbiol. 2017, 53, 628–639. [Google Scholar] [CrossRef]
- Gabbs, M.; Leng, S.; Devassy, J.G.; Monirujjaman, M.; Aukema, H.M. Advances in Our Understanding of Oxylipins Derived from Dietary PUFAs. Adv. Nutr. Am. Int. Rev. J. 2015, 6, 513–540. [Google Scholar] [CrossRef] [Green Version]
- Reddy, M.J.; Shetty, H.S.; Fanelli, C.; Lacey, J. Role of seed lipids in Aspergillus parasiticus growth and aflatoxin production. J. Sci. Food Agr. 1992, 59, 177–181. [Google Scholar] [CrossRef]
- Brown, R.L.; Cotty, P.J.; Cleveland, T.E.; Widstrom, N.W. Living maize embryo influences accumulation of aflatoxin in maize kernels. J. Food Protect. 1993, 56, 967–971. [Google Scholar] [CrossRef] [Green Version]
- Keller, N.P.; Butchko, R.A.E.; Sarr, B.; Phillips, T.D. A visual pattern of mycotoxin production in maize kernels by Aspergillus spp. Phytopathology 1994, 84, 483–488. [Google Scholar] [CrossRef]
- Mellon, J.E.; Dowd, M.K.; Cotty, P.J. Substrate Utilization by Aspergillus flavus in Inoculated Whole Corn Kernels and Isolated Tissues. J. Agric. Food Chem. 2005, 53, 2351–2357. [Google Scholar] [CrossRef] [PubMed]
- Rajasekaran, K.; Ford, G.; Sethumadhavan, K.; Carter-Wientjes, C.; Bland, J.; Cao, H.; Bhatnagar, D. Aspergillus flavus growth and aflatoxin production as influenced by total lipid content during growth and development of cottonseed. J. Crop. Improv. 2017, 31, 91–99. [Google Scholar] [CrossRef]
- Gao, J.; Li, C.; Li, K.; Peng, M.; Liang, Z. Effects of Oxylipins on Spore Production and Ochratoxin A Synthesis of Aspergillus ochraceus and Grain Infection. Food Sci. 2019, 40, 116–121, (In Chinese with English abstract). [Google Scholar] [CrossRef]
- Gupta, S.R.; Prasanna, H.R.; Viswanathan, L.; Venkitasubramanian, T.A. Carboxylic acids as carbon sources for aflatoxin production. Experientia 1974, 30, 1244–1246. [Google Scholar] [CrossRef] [PubMed]
- Tiwari, R.P.; Mittal, V.; Singh, G.; Bhalla, T.C.; Saini, S.S.; Vadehra, D.V. Effect of fatty acids on aflatoxin production by Aspergillus parasiticus. Folia Microbiol. 1986, 31, 120–123. [Google Scholar] [CrossRef] [PubMed]
- Řiháková, Z.; Plocková, M.; Filip, V. Antifungal activity of lauric acid derivatives against Aspergillus Niger. Eur. Food Res. Technol. 2001, 213, 488–490. [Google Scholar] [CrossRef]
- Sjögren, J.; Magnusson, J.; Broberg, A.; Schnürer, J.; Kenne, L. Antifungal 3-Hydroxy Fatty acids from Lactobacillus plantarum MiLAB 14. Appl. Environ. Microbio. 2003, 69, 7554–7557. [Google Scholar] [CrossRef] [Green Version]
- Altieri, C.; Cardillo, D.; Bevilacqua, A.; Singaglia, M. Inhibition of Aspergillus spp. and Penicillium spp. by fatty acids and their monoglycerides. J. Food Protect. 2007, 70, 1206–1212. [Google Scholar] [CrossRef]
- Leyva, M.O.; Vicedo, B.; Finiti, I.; Del Amo, G.; Real, M.D.; García-Agustin, P.; González-Bosch, C. Preventative and post-infection control of Botrytis cinerea in tomato plants by hexanoic acid. Plant Physiol. 2008, 57, 1038–1046. [Google Scholar] [CrossRef]
- Yan, S.; Liang, Y.; Zhang, J.; Chen, Z.; Liu, C. Autoxidated linolenic acid inhibits aflatoxin biosynthesis in Aspergillus flavus via oxylipin species. Fungal Genet. Biol. 2015, 81, 229–237. [Google Scholar] [CrossRef]
- Righetti, L.; Lucini, L.; Giorni, P.; Locatelli, S.; Dall’Asta, C.; Battilani, P. Lipids as key markers in maize response to fumonisin accumulation. J. Agric. Food Chem. 2019, 67, 4064–4070. [Google Scholar] [CrossRef]
- Black, B.A.; Zannini, E.; Curtis, J.M.; Gänzle, M.G. Antifungal hydroxy fatty acids produced during sourdough fermentation: Microbial and enzymatic pathways, and antifungal activity in bread. Appl. Environ. Microbiol. 2013, 79, 1866–1873. [Google Scholar] [CrossRef] [Green Version]
- Burrow, G.B.; NesbilttI, T.C.; Dunlap, J.; Keller, N.P. Seed lipoxygenase products modulate Aspergillus mycotoxin biosynthesis. Mol. Plant Microbe Interact. 1997, 10, 380–387. [Google Scholar] [CrossRef] [Green Version]
- Gao, J.; Liang, Z. Effect of oxylipins on ochratoxin A production by Aspergillus ochraceus in soybean culture medium. Microbiol. China. 2020, 47, 1721–1729. [Google Scholar] [CrossRef]
- Liang, N.; Dacko, A.; Tan, A.K.; Xiang, S.; Curtis, J.M.; Gänzle, M.G. Structure-function relationships of antifungal monohydroxy unsaturated fatty acids (HUFA) of plant and bacterial origin. Food Res. Internat. 2020, 134, 109237. [Google Scholar] [CrossRef] [PubMed]
- Fabbri, A.A.; Fanelli, C.; Panfili, G.; Passi, S.; Fasella, P. Lipoperoxidation and aflatoxin biosynthesis by Aspergillus parasiticus and Aspergillus flavus. J. Gen. Appl. Microbiol. 1983, 129, 3447–3452. [Google Scholar] [CrossRef] [Green Version]
- Tsitsigiannis, D.I.; Kunze, S.; Willis, D.K.; Feussner, I.; Keller, N.P. Aspergillus infection inhibits the expression of peanut 13S-HPODE-forming seed lipoxygenases. Mol. Plant Microbe Interact. 2005, 18, 1081–1089. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wilson, R.A.; Gardner, H.W.; Keller, N.P. Cultivar dependent expression of a maize lipoxygenase responsive to seed infesting fungi. Mol. Plant Microbe Interact. 2001, 14, 980–987. [Google Scholar] [CrossRef] [Green Version]
- Gao, X.Q.; Shim, W.B.; Gobel, C.; Kunze, S.; Feussner, I.; Meeley, R.; Balint-Kurti, P.; Kolomiets, M. Disruption of a maize 9-lipoxygenase results in increased resistance to fungal pathogens and reduced levels of contamination with mycotoxin fumonisin. Mol. Plant Microbe Interact. 2007, 20, 922–933. [Google Scholar] [CrossRef] [Green Version]
- Nobili, C.; D’Angeli, S.; Altamura, M.M.; Scala, V.; Fabbri, A.A.; Reverberi, M.; Fanelli, C. ROS and 9-oxylipins are correlated with deoxynivalenol accumulation in the germinating caryopses of Triticum aestivum after Fusarium graminearum infection. Eur. J. Plant Pathol. 2014, 139, 429–444. [Google Scholar] [CrossRef]
- Scala, V.; Camera, E.; Ludovici, M.; Dall’Asta, C.; Cirlini, M.; Giorni, P.; Battilani, P.; Bello, C.; Fabbri, A.A.; Fanelli, C.; et al. Fusarium verticillioides and maize interaction in vitro: Relationship between oxylipin cross-talk and fumonisin synthesis. World Mycotoxin J. 2013, 6, 343–351. [Google Scholar] [CrossRef]
- Chitarra, G.S.; Abee, T.; Rombouts, F.M.; Posthumus, M.A.; Dijksterhuis, J. Germination of Penicillium paneum conidia is regulated by 1-octen-3-ol, a volatile self-inhibitor. Appl. Environ. Microbiol. 2004, 70, 2823–2829. [Google Scholar] [CrossRef] [Green Version]
- Herrero-Garcia, E.; Garzia, A.; Cordobés, S.; Espeso, E.A.; Ugalde, U. Carbon oxylipins inhibit germination and growth, and stimulate aerial conidiation in Aspergillus Nidulans. Fungal Biol. 2011, 115, 393–400. [Google Scholar] [CrossRef] [PubMed]
- Singh, D.; Son, S.Y.; Lee, C.H. Interactions modulating the growth and secondary metabolism. Sci. Rep. 2020, 10, 11116. [Google Scholar] [CrossRef] [PubMed]
- Meimaroglou, D.M.; Galanopoulou, D.; Markaki, P. Study of the effect of methyl jasmonate concentration on aflatoxin B1 biosynthesis by Aspergillus parasiticus in yeast extract sucrose medium. Int. J. Microbiol. 2009, 2009, 842626. [Google Scholar] [CrossRef] [Green Version]
- Vigor, C.; Bertrand-Michel, J.; Pinot, E.; Oger, C.; Vercauteren, J.; Le Faouder, P.; Galano, J.M.; Lee, J.C.Y.; Durand, T. Non-enzymatic lipid oxidation products in biological systems: ASSESSMENT of the metabolites from polyunsaturated fatty acids. J. Chromatogr. B Anal. Technol. Biomed. Life Sci. 2014, 964, 65–78. [Google Scholar]
- Shearer, G.C.; Walker, R.E. An overview of the biologic effects of omega-6 oxylipins in humans. Prostaglandins Leukot. Essent. Fat. Acids 2018, 137, 26–38. [Google Scholar] [CrossRef]
- Spiteller, G. Lipid peroxidation in aging and age-dependent diseases. Exp. Gerontol. 2001, 36, 1425–1457. [Google Scholar] [CrossRef]
- Yuan, Z.-X.; Majchrzak-Hong, S.; Keyes, G.S.; Iadarola, M.J.; Mannes, A.J.; Ramsden, C.E. Lipidomic profiling of targeted oxylipins with ultra-performance liquid chromatography-tandem mass spectrometry. Anal. Bioanal. Chem. 2018, 410, 6009–6029. [Google Scholar] [CrossRef]
- Serhan, C.N.; Dalli, J.; Colas, R.A.; Winkler, J.W.; Chiang, N. Protectins and maresins: New pro-resolving families of mediators in acute inflammation and resolution bioactive metabolome. Biochim. Biophys. Acta 2015, 1851, 397–413. [Google Scholar] [CrossRef] [Green Version]
- Capdevila, J.H.; Falck, J.R. Biochemical and molecular characteristics of the cytochrome P450 arachidonic acid monooxygenase. Prostag. Prostaglandins Other Lipid Mediat. 2000, 62, 271–292. [Google Scholar] [CrossRef]
- Gao, X.; Kolomiets, M.V. Host-derived lipids and oxylipins are crucial signals in modulating mycotoxin production by fungi. Toxin Rev. 2009, 28, 79–88. [Google Scholar] [CrossRef]
- Prost, I.; Dhondt, S.; Rothe, G.; Vicente, J.; Rodriguez, M.J.; Kift, N.; Carbonne, F.; Griffiths, G.; Esquerré-Tugayé, M.-T.; Rosahl, S.; et al. Evaluation of the Antimicrobial Activities of Plant Oxylipins Supports Their Involvement in Defense against Pathogens. Plant Physiol. 2005, 139, 1902–1913. [Google Scholar] [CrossRef] [Green Version]
- Wasternack, C.; Feussner, I. The Oxylipin Pathways: Biochemistry and Function. Annu. Rev. Plant Biol. 2018, 69, 363–386. [Google Scholar] [CrossRef] [PubMed]
- Avis, T.J.; Belanger, R.R. Specificity and Mode of Action of the Antifungal Fatty Acid cis-9-Heptadecenoic Acid Produced by Pseudozyma flocculosa. Appl. Environ. Microbiol. 2001, 67, 956–960. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Thibane, V.S.; Ells, R.; Hugo, A.; Albertyn, J.; van Rensburg, W.J.J.; Van Wyk, P.W.J.; Kock, J.L.F.; Pohl, C.H. Polyunsaturated fatty acids cause apoptosis in C. albicans and C. dubliniensis biofilms. Biochim. Biophys. Acta 2012, 1820, 1463–1468. [Google Scholar] [CrossRef]
- Pohl, C.H.; Kock, J.L.F.; Thibane, V.S. Antifungal free fatty acids: A review. In Science Against Microbial Pathogens: Communicating Current Research and Technological Advances; Méndez-Vilas, A., Ed.; Formatex Research Center: Badajoz, Spain, 2011; pp. 61–71. Available online: https://www.researchgate.net/publication/266463207 (accessed on 10 October 2021).
- Champe, S.P.; El-Zayat, A.A. Isolation of a sexual sporulation hormone from Aspergillus nidulans. J. Bacteriol. 1989, 171, 3982–3988. [Google Scholar] [CrossRef] [Green Version]
- Tsitsigiannis, D.I.; Zarnowski, R.; Keller, N.P. The lipid body protein, PpoA, coordinates sexual and asexual sporulation in Aspergillus nidulans. J. Biol. Chem. 2004, 279, 11344–11353. [Google Scholar] [CrossRef] [Green Version]
- Tsitsigiannis, D.I.; Kowieski, T.M.; Zarnowski, R.; Keller, N.P. Endogenous lipogenic regulators of spore balance in Aspergillus nidulans. Eukaryot. Cell 2004, 3, 1398–1411. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kim, H.S.; Han, K.Y.; Kim, K.J.; Han, D.M.; Jahng, K.Y.; Chae, K.S. The veA gene activates sexual development in Aspergillus nidulans. Fungal Genet. Biol. 2002, 37, 72–80. [Google Scholar] [CrossRef]
- Busch, S.; Eckert, S.E.; Krappmann, S.; Braus, G.H. The COP9 signalosome is an essential regulator of development in the filamentous fungus Aspergillus nidulans. Mol. Microbiol. 2003, 49, 717–730. [Google Scholar] [CrossRef]
- Han, S.; Adams, T.H. Complex control of the developmental regulatory locus brlA in Aspergillus nidulans. Mol. Genet. Genomics. 2001, 266, 260–270. [Google Scholar] [CrossRef]
- Han, K.H.; Han, K.Y.; Yu, J.H.; Chae, K.S.; Jahng, K.Y.; Han, D.M. The nsdD gene encodes a putative GATA-type transcription factor necessary for sexual development of Aspergillus nidulans. Mol. Microbiol. 2001, 41, 299–309. [Google Scholar] [CrossRef] [PubMed]
- Horowitz, B.S.; Zarnowski, R.; Sharpee, W.C.; Keller, N.P. Morphological transitions governed by density dependence and lipoxygenase activity in Aspergillus flavus. Appl. Environ. Microbiol. 2008, 74, 5674–5685. [Google Scholar] [CrossRef] [Green Version]
- Brown, S.H.; Scott, J.B.; Bhaheetharan, J.; Sharpee, W.C.; Milde, L.; Wilson, R.A.; Keller, N.P. Oxygenase coordination is required for morphological transition and the host-fungus interaction of Aspergillus flavus. Mole. Plant Microbe Interact. 2009, 22, 882–894. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Fabbri, A.A. Lipoperoxidation affects ochratoxin A biosynthesis in Aspergillus ochraceus and its interaction with wheat seeds. Appl. Microbiol. Biotechnol. 2010, 85, 1935–1946. [Google Scholar] [CrossRef]
- McDonald, T.; Devi, T.; Shimizu, K.; Sim, S.-C.; Keller, N.P. Signaling events connecting mycotoxin biosynthesis and sporulation in Aspergillus and Fusarium spp. In New Horizon of Mycotoxicology for Assuring Food Safety, Proceedings of International Symposium of Mycotoxicology; Yoshizawa, T., Ed.; Bookish Co.: Takamatsu, Japan, 2003; pp. 139–147. [Google Scholar] [CrossRef] [Green Version]
- Yu, J.H.; Wieser, J.; Adams, T.H. The Aspergillus FlbA RGS domain protein antagonizes G protein signaling to block proliferation and allow development. EMBO J. 1996, 15, 5184–5190. [Google Scholar] [CrossRef]
- Shimizu, K.; Keller, N.P. Genetic involvement of cAMP dependent protein kinase in a G protein signaling pathway regulating morphological and chemical transitions in Aspergillus nidulans. Genetics 2001, 157, 591–600. [Google Scholar] [CrossRef]
- Shimizu, K.; Hicks, J.K.; Huang, T.P.; Keller, N.P. Pka, Ras and RGS protein interactions regulate activity of AflR, a Zn(II)2Cys6 transcription factor in Aspergillus nidulans. Genetics 2003, 165, 1095–1104. [Google Scholar] [CrossRef]
- Affeldt, K.J.; Brodhagen, M.; Keller, N.P. Aspergillus oxylipin signaling and quorum sensing pathways depend on G protein-coupled receptors. Toxins 2012, 4, 695–717. [Google Scholar] [CrossRef] [Green Version]
- de Souza, W.R.; Morais, E.R.; Krohn, N.G.; Savoldi, M.; Goldman, M.H.S.; Rodrigues, F.; Caldana, C.; Semelka, C.; Tikunov, A.P.; Macdonald, J.M.; et al. Identification of metabolic pathways influenced by the G-Protein coupled receptors GprB and GprD in Aspergillus nidulans. PLoS ONE 2013, 8, 62088. [Google Scholar] [CrossRef] [Green Version]
- Affeldt, K.J.; Carrig, J.; Amare, M.; Keller, N.P. Global survey of canonical Aspergillus flavus G protein-coupled receptors. mBio 2014, 5, 01501–01514. [Google Scholar] [CrossRef] [Green Version]
- Lafon, A.; Han, K.H.; Seo, J.A.; Yu, J.H.; d’Enfert, C. G-protein and cAMP-mediated signaling in aspergilli: A genomic perspective. Fungal Genet. Biol. 2006, 43, 490–502. [Google Scholar] [CrossRef] [PubMed]
- Püssa, T.; Raudsepp, P.; Toomik, P.; Pällin, R.; Mäeorg, U.; Kuusik, S.; Soidla, R.; Rei, M. A study of oxidation products of free polyunsaturated fatty acids in mechanically deboned meat. J. Food Compos. Anal. 2009, 22, 307–314. [Google Scholar] [CrossRef]
- Bian, H.; Ma, J.; Geng, Z.; Liu, T.; Sun, C.; Wang, D.; Zhang, M.; Xu, W. Changes of hydroxyl-linoleic acids during Chinese-style sausage processing and their relationships with lipids oxidation. Food Chem. 2019, 296, 63–68. [Google Scholar] [CrossRef] [PubMed]
- Dias, F.F.G.; Augusto-Obara, T.R.; Hennebelle, M.; Chantieng, S.; Ozturk, G.; Taha, A.Y.; de Moura, J.M.L.N. Effects of industrial heat treatments on bovine milk oxylipins and conventional markers of lipid oxidation. Prostag. Leukotr. Ess. 2020, 152, 102040. [Google Scholar] [CrossRef] [PubMed] [Green Version]

| Fatty Acid | Concentration | Fungal Species | Fungal Growth | Mycotoxin Production | Ref. |
|---|---|---|---|---|---|
| lauric acid | 50.00 mM | Aspergillus parasiticus | + | − AF | [23] |
| myristic acid | + | + AF | |||
| palmitic acid | − | − AF | |||
| oleic acid | + | − AF | |||
| linoleic acid | − | + AF | |||
| lauric acid | 2.50 mM | Aspergillus niger | − | u | [24] |
| capric acid | 0.60 mM | Aspergillus fumigatus | − | u | [25] |
| Aspergillus nidulans | − | u | |||
| 0.30 mM | Penicillium commune | − | u | ||
| 0.15 mM | Penicillium roqueforti | − | u | ||
| lauric acid | 0.20 mM | Aspergillus niger | − | u | [26] |
| myristic acid | 0.09 mM | Penicillium glabrum | − | u | |
| 0.13 mM | Aspergillus niger | + | u | ||
| 0.13 mM | Penicillium italicum | − | u | ||
| myristoleic acid | 0.09 mM | Aspergillus niger | − | u | |
| 0.13 mM | Penicillium italicum | − | u | ||
| palmitic acid | 12.00 mM | Botrytis cinerea | − | u | [27] |
| stearic acid | 0.10 mM | Aspergillus flavus | u | + AF | [28] |
| linolenic acid | 1.25 mM | u | − AF |
| Qxylipin | Host Source | Fungal Species | Fungal Growth | Mycotoxin Production | Ref. |
|---|---|---|---|---|---|
| 13S-HPODE | c.a | Aspergillus flavus; Aspergillus parasiticus | u | − AF | [34] |
| peanut | Aspergillus flavus; Fusarium verticillioides | + | + AF | [35] | |
| 13S-HODE | c.a | Aspergillus ochraceus | + | − OTA | [32] |
| 9S-HPODE | soybean | Aspergillus flavus; Aspergillus nidulans | +/− | − AF/ST | [31] |
| corn | Aspergillus flavus; Fusarium verticillioides | + | + AF | [36] | |
| 9S-HOD(T)E | corn | Fusarium verticillioides | n | + AFB1 | [37] |
| 9-HODE | wheat | Fusarium graminearum | u | + DON | [38] |
| 9-HPODE | |||||
| 9S-HODE | c.a | Aspergillus ochraceus | − | + OTA | [21] |
| 9-HODE | maize | Fusarium verticillioides | u | + FB1 | [39] |
| 10-HODE | |||||
| 1-octen-3-ol | c.a | Penicillium paneum | − | u | [40] |
| c.a | Aspergillus nidulans | − | u | [41] | |
| Aspergillus oryzae | Aspergillus flavus | − | + AFB1 | [42] | |
| MeJA | c.a | Aspergillus parasiticus | − | − AF | [43] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Qiu, M.; Wang, Y.; Sun, L.; Deng, Q.; Zhao, J. Fatty Acids and Oxylipins as Antifungal and Anti-Mycotoxin Agents in Food: A Review. Toxins 2021, 13, 852. https://doi.org/10.3390/toxins13120852
Qiu M, Wang Y, Sun L, Deng Q, Zhao J. Fatty Acids and Oxylipins as Antifungal and Anti-Mycotoxin Agents in Food: A Review. Toxins. 2021; 13(12):852. https://doi.org/10.3390/toxins13120852
Chicago/Turabian StyleQiu, Mei, Yaling Wang, Lijun Sun, Qi Deng, and Jian Zhao. 2021. "Fatty Acids and Oxylipins as Antifungal and Anti-Mycotoxin Agents in Food: A Review" Toxins 13, no. 12: 852. https://doi.org/10.3390/toxins13120852




