1. Introduction
Aflatoxins (AFTs) are the most common mycotoxins [
1] produced by
Aspergillus flavus, A.
parasiticus, and A.
nomius [
2,
3,
4] in cereal grains in the entire food chain, from farm to factory, under favorable temperature and humidity conditions [
5]. AFTs include aflatoxin B
1 (AFB
1), aflatoxin B
2, aflatoxin G
1, and aflatoxin G
2. Among these, AFB
1 is the most toxic and has the highest detection frequency [
6,
7,
8]. The AFB
1 contamination of food products is the most common contamination problem in the food chain and is potentially hazardous to humans and animals because it causes high carcinogenicity, mutagenicity, suppression of immunity, and liver damage [
8,
9]. To minimize the human health risk, many countries and regions have set extremely low maximum limits (MLs) for AFB
1 in food. For example, the European Commission established 2–12 μg/kg as the ML of AFB
1 in food, while China established 5–20 μg/kg as the ML of AFB
1 [
10,
11]. Therefore, it is necessary to address the contamination problem by assessing the risk of AFB
1 contamination in the food chain for food safety and human health protection.
Currently, conventional analytical approaches used for AFB
1 monitoring in food include advanced instrumental analysis and fast detection techniques. Instrumental analysis methods mainly rely on chromatography-based techniques, such as high-performance liquid chromatography [
11,
12,
13] and liquid chromatography–tandem mass spectrometry (LC-MS/MS) [
14,
15,
16], because of their high selectivity, excellent accuracy, and reproducibility. However, these analytical procedures have several limitations. Chromatographic methods incur high equipment costs and involve complicated and time-consuming sample operation and analysis procedures, which limit their use to skilled operators [
10]. In addition, chromatography also requires consuming some chromatographic consumables, such as chromatographic columns, etc. These limitations are overcome by fast detection methods, such as thin-layer chromatography [
17,
18,
19,
20,
21], colloidal gold immunochromatography [
22,
23,
24,
25], and enzyme-linked immunosorbent assay [
26,
27,
28]; these are commonly used in the routine monitoring of AFB
1, especially for on-site or field detection, because of their low cost and convenience of operation. However, because limited sample clean-up strategies are used, the fast detection methods are often affected by matrix effects, leading to false-positive/negative results. Hence, a rapid, accurate, sensitive, and robust methodology based on inexpensive and easy-to-operate techniques for high-frequency and precise monitoring of trace AFB
1 must be developed.
Atomic absorption spectroscopy (AAS) is one of the most commonly used techniques for tracing heavy-metal element determination in food, owing to its robustness, accuracy, speed, and simplicity. However, AAS-based methods have received considerably less attention in the analysis of organic contaminants such as mycotoxins primarily because they are typically regarded as “elemental analytical methods [
29].” In recent years, with the continuous development of molecular labeling methods and the high specificity of antigen–antibody reactions, the target signal (organic contaminant signal) to be detected can be successfully converted to metal ion signals, which can be detected by using an elemental analysis method. Wang [
30] and Hansen [
31] et al. completed the conversion of DNA and protein signals to metal ion signals with the help of modified quantum dots, which allowed for the detection of target DNA and proteins by electrochemical means. This suggests that the gap between inorganic and organic analyses can be bridged. In addition, nanomaterials such as quantum dots (QDs) are aggregates of atoms and molecules on the nanometer scale, which can generate a large number of atoms after digestion, thereby improving signal amplification and detection sensitivity [
32,
33]. Furthermore, an AAS-based analysis system has a lower instrument cost than chromatography-based systems. As a necessary instrument for food analysis labs (many end users), the analytical potential of AAS for mycotoxins is worth exploring.
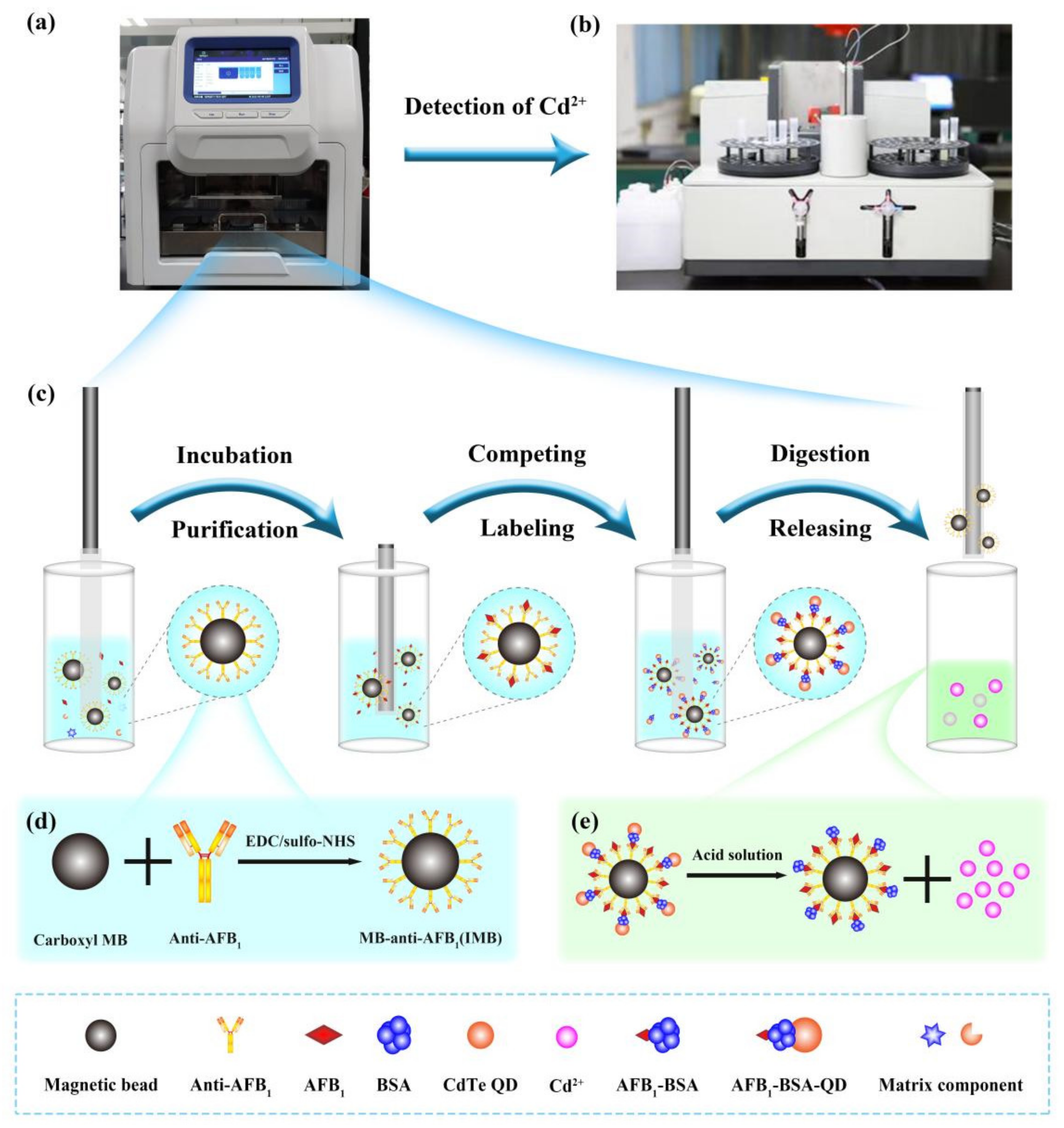
In conventional analysis, several manual steps such as extraction, filtering, and purification are required. Consequently, it is only applicable to high-end laboratories with skilled technicians. Previously, we reported a fully automated pretreatment platform for sample enrichment, purification, and elution based on immunoaffinity magnetic beads (IMBs) [
10,
34,
35,
36], which consumes less time, costs, and labor as well as introduces fewer errors compared to manual processing of mycotoxin assays.
In this study, we proposed an AAS-based analysis method for the specific and sensitive detection of AFB1 in complex food samples by using QDs as labels. To overcome the limitations of cumbersome steps, in this work, we used the IMB-based sample pretreatment platform and the AAS system in combination to generate new automated magneto-controlled analytical approaches for achieving programmable immunoassay operations, including automatic enrichment, purification, QD labeling, release, and analysis. In-house magneto-controlled systems present significant advantages over other immunosensors, which minimize labor and eliminate operational errors. The proposed method overcomes the shortcomings of existing methods and has several advantages such as simple manipulation, high sensitivity, acceptable linear calibration range and reproducibility, low-cost detection, ease of automation, and high analyte throughput. To the best of our knowledge, this study is the first to use AAS to detect AFB1 in different food matrices.
4. Materials and Methods
4.1. Materials
AFB1, dimethyl formamide (DMF), pyridine,O-(carboxymethyl) hydroxylamine hemihydrochloride (CMO), trichloromethane, 1-ethyl-3-(3-dimethylaminopropyl) carbodiimide hydrochloride (EDC), sodium hydroxide, N-hydroxysuccinimide (NHS), hydrochloric acid (HCl), nitric acid (HNO3), bovine serum albumin (BSA), and phosphate buffer (PBS) were purchased from SigmaAldrich (St. Louis, MO, USA). HPLC grade methanol (MeOH) was obtained from Merck (Darmstadt, Germany). Deionized water (H2O) was purchased from Watsons (Hong Kong, China). Blank maize, rice, wheat, husked rice, and peanut oil samples were obtained from a market in China. Certified reference material and reference materials are provided by the Academy of National Food and Strategic Reserves Administration(ASAG) (Beijing, China; GBW(E)100386, JTZK-007, JTZK-002).
4.2. Synthesis of Immunomagnetic Beads (IMB)
First, take 500 μL of NHS-activated carboxyl magnetic beads, discard the supernatant after magnetic separation, add 1 mL of dilute hydrochloric acid pre-cooled at 2–8 °C and mix well. After magnetic separation, the supernatant was discarded, and 500 μL of 2 mg/mL AFB1 antibody was added. After homogeneous, it was placed on a mixer for 2 h at room temperature. Next, the reaction mixture was magnetically separated, the supernatant was discarded, 1 mL of blocking buffer (Tris-HCl) was added and mixed, and then placed on a mixer for 2 h at room temperature to form IMB. Finally, the mixture was magnetically separated, and the supernatant was discarded, washed with 1 mL PBST (2 times) and 1 mL PBS (1 time) IMB, and then added 500 μL PBS to resuspend and stored at 4℃ for later use.
4.3. Synthesis of Aflatoxin Haptens (AFB1-CMO)
Since there is no active group on the surface of the AFB1 molecule that can be coupled with the protein, the AFB1 molecule needs to be derivatized before coupling with BSA. We used O-(carboxymethyl)hydroxylamine hemihydrochloride (CMO) as a derivatizing agent to make AFB1 carry a carboxyl group, which can then react with the amino group on BSA to achieve the coupling effect. The derivatization scheme was as follows: 1 mg of AFB1 was dissolved in 0.6 mL of methanol–water–pyridine (4:1:1) solution, 2 mg of CMO was added, and the reaction was carried out under magnetic stirring in a water bath at 70 °C for 6 h and allowed to stand overnight at room temperature in the dark. The reaction solution was blown dry with nitrogen, and the precipitate was dissolved in 1 mL of chloroform solution and extracted three times with an equal volume of ultrapure water. The organic phase was collected and dried with nitrogen, and the precipitate was dissolved in 200 μL of Dimethyl Formamide (DMF) solution, which was the derivative product.
4.4. Synthesis of Aflatoxin Complete Antigen (AFB1-BSA)
The complete antigen synthesis is based on the hapten synthesis by adding the coupling protein BSA. The coupled protein BSA was first dissolved in carbonate buffer. Then, 1 mg of AFB1-CMO was dissolved in 200 μL of DMF solution, 2.4 mg of EDC and 1.3 mg of NHS were added, and the reaction was conducted under magnetic stirring at room temperature for 12 h. The reaction solution was added dropwise to the carrier protein solution (BSA: 7.54 mg), and the reaction was magnetically stirred at room temperature overnight. The coupled product was ultrafiltered with an ultrafiltration tube, and the retentate was resuspended in PBS and stored at −20 °C for later use. The successful synthesis of AFB1-BSA was characterized by the UV-Vis absorption spectra of BSA and AFB1-BSA. The characteristic absorption peak of BSA is at 278 nm, the characteristic absorption peak of AFB1 is at 265 nm and 360 nm, and the characteristic absorption peak of AFB1-BSA is between 265–278 nm, which is mainly due to the superposition of the characteristic absorption peaks of AFB1 and BSA. The successful synthesis of the complete antigen can be well characterized by the UV-Vis absorption pattern.
4.5. Coupling of Pegylated Quantum Dots with Complete Antigen AFB1-BSA
The cross-linking principle of polyethylene glycol-modified quantum dots and AFB1-BSA is based on the fact that after heat treatment of BSA, a part of the internal hydrophobic structure will be exposed, which can be adsorbed with PEG on the surface of quantum dots. The cross-linking steps are as follows: 22.2 µL of 100 mM PBS was added to 200 µL of 1 µM PEG-modified quantum dot solution to prepare a 10 mM PBS quantum dot solution (pH 7.4). Subsequently, 67 µg of AFB1-BSA was added and mixed, followed by a boiling water bath for 10 min, centrifugation at 25,000 rpm for 15 min, the supernatant was discarded, and the quantum dots were resuspended.
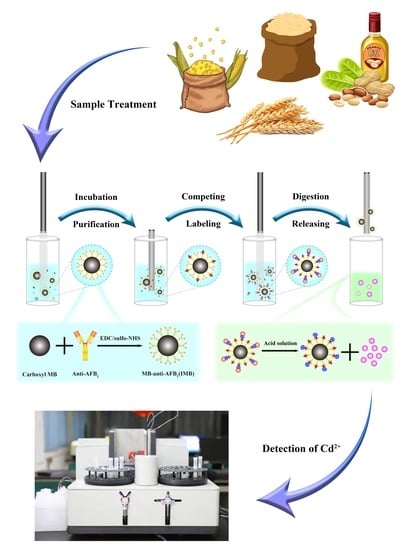
4.6. Sample Automatic Processing
Representative samples were thoroughly ground and homogenized according to the Codex General Guidelines on Sampling from the FAO and WHO with minor modifications [
38].Cereal samples are treated as follows: 3 points are randomly selected for sampling, and the sampling amount of each point is 0.5 kg as the laboratory sample size. All laboratory samples were pulverized with a particle size of 0.5 mm. After fully mixing, 5 g of the sample was weighed for processing. Oil samples are treated as follows: after the peanut oil sample is fully mixed, 5 g of the sample is weighed for processing. Sequentially, 5 g [
37] sample and 20 mL extraction solution were vortexed at 2500 rpm for 20 min in a centrifuge tube (50-mL). Finally, the centrifuge tube was centrifuged at 7000 rpm for 5 min, with the supernatants for further analysis. Proper mixing frequency, mixing amplitude, and sufficient reaction time can ensure sufficient reaction, washing, and elution of the sample. In order to prevent the solvent from splashing out during the mixing process, the mixing range was set to 80%, and other reaction conditions were shown in
Table 5. After the pretreatment, the eluates in 5 wells were collected for detection by atomic absorption spectrometer or LC-MS/MS [
37].
4.7. Atomic Absorption Detection
The determination of cadmium (Cd
2+) was done by using the graphite furnace atomic absorption spectrometer (CPG2S, China). A Cd hollow cathode lamp operating at the 228.8 nm analytical line (4 mA current and a 0.8 nm spectral bandpass) was used for absorbance measurement, and the deuterium lamp was used to correct the background. Other experimental conditions and heating procedures are shown in
Table 6.
4.8. Method Verification
The linear range, limit of detection (LOD), limit of quantification (LOQ), recovery, intra-day relative standard deviation, and inter-day relative standard deviation were determined for the method, and real samples were analyzed together by LC-MS/MS. Where the limit of detection (3 σ/s) and the limit of quantification (10 σ/s) are calculated from the calibration, “σ” is the standard deviation of the 11 blank measurements, and “s” is the slope of the calibration curve.