Insights into the Cardiotoxic Effects of Veratrum Lobelianum Alkaloids: Pilot Study
Abstract
:1. Introduction
2. Results
2.1. Molecular Modeling
2.2. Clinical Study
3. Discussion
4. Conclusions
5. Materials and Methods
5.1. Materials
5.2. LogP and pKa Values Calculations for Transmembrane Permeability Prediction
5.3. Binding Mode and Binding Affinity Predictions by Molecular Docking
5.4. Machine Learning-based SAR Modeling
5.5. Patients
5.6. Ethics Statement
5.7. Standard Sample Preparation
5.8. Sample Preparation
5.9. HPLC-MS/MS Analysis
- Jervine: m/z 426.2 > 114.1/109.1/84.1;
- ProA: m/z 794.2 > 776.1/758.1/658.1;
- ProB: m/z 810.4 > 792.5/676.5/658/5;
- Quinidine: m/z 325.2 > 172.0/160/81.2.
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Srinivasan, P.; Smolke, C.D. Biosynthesis of medicinal tropane alkaloids in yeast. Nature 2020, 585, 614–619. [Google Scholar] [CrossRef] [PubMed]
- Kurek, J. (Ed.) Alkaloids—Their Importance in Nature and Human Life; IntechOpen: London, UK, 2019. [Google Scholar] [CrossRef]
- Bribi, S.; Basu, V.R.; Basha, S.K.; Chandra, K.; Anasuya, D. A Review on Pharmacological Activities of Alkaloids. World J. Curr. Med. Pharm. Res. 2019, 1, 230–234. [Google Scholar] [CrossRef] [Green Version]
- Zong, X.-X.; Yan, G.; Wu, J.-L.; Leung, E.L.-H.; Zhou, H.; Li, N.; Liu, L. New C19-diterpenoid alkaloids from the parent roots of Aconitum carmichaelii. Tetrahedron Lett. 2017, 58, 1622–1626. [Google Scholar] [CrossRef]
- Wong, V.K.-W.; Law, B.Y.-K.; Yao, X.-J.; Chen, X.; Xu, S.W.; Liu, L.; Leung, E.L.-H. Advanced research technology for discovery of new effective compounds from Chinese herbal medicine and their molecular targets. Pharmacol. Res. 2016, 111, 546–555. [Google Scholar] [CrossRef] [PubMed]
- Liu, L.-J.; Leung, K.-H.; Chan, D.S.-H.; Wang, Y.-T.; Ma, D.-L.; Leung, C.-H. Identification of a natural product-like STAT3 dimerization inhibitor by structure-based virtual screening. Cell Death Dis. 2014, 5, e1293. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zhong, H.-J.; Lee, B.R.; Boyle, J.W.; Wang, W.; Ma, D.-L.; Chan, P.W.H.; Leung, C.-H. Structure-based screening and optimization of cytisine derivatives as inhibitors of the menin–MLL interaction. Chem. Commun. 2016, 52, 5788–5791. [Google Scholar] [CrossRef] [PubMed]
- Zhong, Z.; Liu, L.-J.; Dong, Z.-Q.; Lu, L.; Wang, M.; Leung, C.-H.; Ma, D.-L.; Wang, Y. Structure-based discovery of an immunomodulatory inhibitor of TLR1–TLR2 heterodimerization from a natural product-like database. Chem. Commun. 2015, 51, 11178–11181. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Schep, L.J.; Schmierer, D.M.; Fountain, J.S. Veratrum Poisoning. Toxicol. Rev. 2006, 25, 73–78. [Google Scholar] [CrossRef]
- Heretsch, P.; Gianis, A. The Veratrum and Solanum Alkaloids. In The Alkaloids: Chemistry and Biology; Knölker, H.-J., Ed.; Elsevier Inc.: Amsterdam, The Netherlands, 2015; Volume 74, pp. 201–232. [Google Scholar] [CrossRef]
- Plants of the World Online. Available online: https://powo.science.kew.org/taxon/urn:lsid:ipni.org:names:30300236-2 (accessed on 10 May 2022).
- Rozhanets, V.V.; Nuzhny, V.P. About permissibility of veratrum intake for conditioned reflex therapy of alcoholism. Narcology 2003, 4, 45–48. (In Russian) [Google Scholar]
- Vicentini, C.B.; Manfredini, S.; Contini, C. Ancient treatment for lice: A source of suggestions for carriers of other infectious diseases? Infez. Med. 2018, 26, 181–192. [Google Scholar]
- Guarrera, P.M. Traditional antihelmintic, antiparasitic and repellent uses of plants in Central Italy. J. Ethnopharmacol. 1999, 68, 183–192. [Google Scholar] [CrossRef]
- Melnik, E.V.; Belova, M.V.; Tyurin, I.A.; Ramenskaya, G.V. Quantitative Content Parameter in the Standardization of Veratrum Aqua, Veratrum Lobelianum Bernh. Based Drug. Drug Dev. Regist. 2021, 10, 107–113. (In Russian) [Google Scholar] [CrossRef]
- Ministry of Public Health of Russian Federation. State Pharmacopeia of Russian Federation, 14th ed.; Ministry of Public Health of Russian Federation: Moscow, Russia, 2018; Volume 2. (In Russian)
- Zheng, B.; Wang, C.; Song, W.; Ye, X.; Xiang, Z. Pharmacokinetics and enterohepatic circulation of jervine, an antitumor steroidal alkaloid from Veratrum nigrum in rats. J. Pharm. Anal. 2019, 9, 367–372. [Google Scholar] [CrossRef]
- Chen, J.; Wen, B.; Wang, Y.; Wu, S.; Zhang, X.; Gu, Y.; Wang, Z.; Wang, J.; Zhang, W.; Yong, J. Jervine exhibits anticancer effects on nasopharyngeal carcinoma through promoting autophagic apoptosis via the blockage of Hedgehog signaling. Biomed. Pharmacother. 2020, 132, 110898. [Google Scholar] [CrossRef] [PubMed]
- Li, Q.; Yang, K.-X.; Zhao, Y.-L.; Qin, X.-J.; Yang, X.-W.; Liu, L.; Liu, Y.-P.; Luo, X.-D. Potent anti-inflammatory and analgesic steroidal alkaloids from Veratrum taliense. J. Etnopharmacol. 2016, 179, 274–279. [Google Scholar] [CrossRef] [PubMed]
- Yakan, S.; Aydin, T.; Gulmez, C.; Ozden, O.; Erdogan, K.E.; Daglioglu, Y.K.; Andic, F.; Atakisi, O.; Cakir, A. The protective role of jervine against radiation-induced gastrointestinal toxicity. J. Enzyme. Inhib. Med. Chem. 2019, 34, 789–798. [Google Scholar] [CrossRef]
- Plouvier, B.; Beatch, G.N.; Jung, G.L.; Zolotoy, A.; Sheng, T.; Clohs, L.; Barrett, T.D.; Fedida, D.; Wang, W.Q.; Zhu, J.J.; et al. Synthesis and Biological Studies of Novel 2-Aminoalkylethers as Potential Antiarrhythmic Agents for the Conversion of Atrial Fibrillation. J. Med. Chem. 2007, 50, 2818–2841. [Google Scholar] [CrossRef] [PubMed]
- Rokin, S.R.; Sentsov, V.G. Acute veratrin poisoning. In Emergency Clinical Toxicology (Guide for Medical Doctors); Luzhnikov, E.A., Ed.; Medpractika-M: Moscow, Russia, 2007; pp. 582–586. [Google Scholar]
- Madzhidov, T.I.; Rakhimbekova, A.; Afonina, V.A.; Gimadiev, T.R.; Mukhametgaleev, R.N.; Nugmanov, R.I.; Baskin, I.I.; Varnek, A. Machine learning modelling of chemical reaction characteristics: Yesterday, today, tomorrow. Mendeleev Commun. 2021, 31, 769–780. [Google Scholar] [CrossRef]
- Taldaev, A.; Terekhov, R.; Nikitin, I.; Zhevlakova, A.; Selivanova, I. Insights into the Pharmacological Effects of Flavonoids: The Systematic Review of Computer Modeling. Int. J. Mol. Sci. 2022, 23, 6023. [Google Scholar] [CrossRef]
- Ma, D.-L.; Chan, D.S.-H.; Leung, C.-H. Drug repositioning by structure-based virtual screening. Chem. Soc. Rev. 2013, 42, 2130–2141. [Google Scholar] [CrossRef]
- Li, Z.; Jin, X.; Wu, T.; Huang, G.; Wu, K.; Lei, J.; Pan, X.; Yan, N. Structural Basis for Pore Blockade of the Human Cardiac Sodium Channel Nav1.5 by the Antiarrhythmic Drug Quinidine. Angew. Chem. Int. Ed. 2021, 60, 11474–11480. [Google Scholar] [CrossRef] [PubMed]
- Tikhonov, D.S.; Zhorov, B.S. Mechanism of sodium channel block by local anesthetics, antiarrhythmics, and anticonvulsants. J. Gen. Physiol. 2017, 149, 465–481. [Google Scholar] [CrossRef] [PubMed]
- Fozzard, H.A.; Lee, P.J.; Lipkind, G.M. Mechanism of Local Anesthetic Drug Action on Voltage-Gated Sodium Channels. Curr. Pharm. Des. 2005, 11, 2671–2686. [Google Scholar] [CrossRef] [PubMed]
- Malik, O.; Kaye, A.D.; Kaye, A.; Belani, K.; Urman, R.D. Emerging roles of liposomal bupivacaine in anesthesia practice. J. Anaesthesiol. Clin. Pharmacol. 2017, 33, 151–156. [Google Scholar] [CrossRef]
- Bekhit, M.H. Lidocaine for neural blockade. In The Essence of Analgesia and Analgesics; Sinatra, R.S., Jahr, J.S., Watkins-Pitchford, J.M., Eds.; Cambridge University Press: New York, NY, USA, 2011; pp. 279–282. [Google Scholar]
- Forrester, J.D.; Price, J.H.; Holstege, C.P. Intoxication with a Ramp (Allium tricocca) Mimicker. Wilderness Environ. Med. 2010, 21, 61–63. [Google Scholar] [CrossRef] [Green Version]
- Rauber-Lüthy, C.; Halbsguth, U.; Kupferschmidt, H.; König, N.; Mégevand, C.; Zihlmann, K.; Ceschi, A. Low-dose exposure to Veratrum album in children causes mild effects—A case series. Clin. Toxicol. 2010, 48, 234–237. [Google Scholar] [CrossRef] [Green Version]
- Grobosch, T.; Binscheck, T.; Martens, F.; Lampe, D. Accidental Intoxication with Veratrum album. J. Anal. Toxicol. 2008, 32, 768–773. [Google Scholar] [CrossRef] [Green Version]
- Mankad, P.; Kalahasty, G. Antiarrhythmic Drugs—Risks and Benefits. Med. Clin. 2019, 103, 821–834. [Google Scholar] [CrossRef]
- Grube, S.; Langguth, P.; Junginger, H.E.; Kopp, S.; Midha, K.K.; Shah, V.P.; Stavchansky, S.; Dressman, J.B.; Barends, D.M. Biowaiver monographs for immediate release solid oral dosage forms: Quinidine sulfate. J. Pharm. Sci. 2009, 98, 2238–2251. [Google Scholar] [CrossRef]
- Serdoz, L.V.; Rittger, H.; Furlanello, F.; Bastian, D. Quinidine—A legacy within the modern era of antiarrhythmic therapy. Pharmacol. Res. 2019, 144, 257–263. [Google Scholar] [CrossRef]
- Melnik, E.V.; Belova, M.V.; Potskhveriya, M.M.; Simonova, A.Y.; Tyurin, I.A.; Ramenskaya, G.V. Veratrum Alkaloid Determination in Four Cases of Veratrum Aqua Poisonings. J. Anal. Toxicol. 2022, 46, e42–e47. [Google Scholar] [CrossRef] [PubMed]
- Liang, Y.; Wu, J.-L.; Leung, E.L.-H.; Zhou, H.; Liu, Z.; Yan, G.; Liu, Y.; Liu, L.; Li, N. Identification of Oxygenated Fatty Acid as a Side Chain of Lipo-Alkaloids in Aconitum carmichaelii by UHPLC-Q-TOF-MS and a Database. Molecules 2016, 21, 437. [Google Scholar] [CrossRef] [PubMed]
- Gu, W.-Y.; Li, N.; Leung, E.L.-H.; Zhou, H.; Yao, X.-J.; Liu, L.; Wu, J.-L. Rapid identification of new minor chemical constituents from Smilacis Glabrae Rhizoma by combined use of UHPLC-Q-TOF-MS, preparative HPLC and UHPLC-SPE-NMR-MS techniques. Phytochem. Anal. 2015, 26, 428–435. [Google Scholar] [CrossRef] [PubMed]
- Ma, X.-Q.; Leung, A.K.M.; Chan, C.-L.; Su, T.; Li, W.-D.; Li, S.-M.; Fong, D.W.F.; Yu, Z.-L. UHPLC UHD Q-TOF MS/MS analysis of the impact of sulfur fumigation on the chemical profile of Codonopsis Radix (Dangshen). Analyst 2014, 139, 505–516. [Google Scholar] [CrossRef]
- Steiger, B.; Meier, P.J. Pharmacogenetics of drug transporters in the enterohepatic circulation. Pharmacogenomics 2011, 12, 611–631. [Google Scholar] [CrossRef] [Green Version]
- Huang, J.; Bathena, S.P.; Tong, J.; Roth, M.; Hagenbuch, B.; Alnouti, Y. Kinetic analysis of bile acid sulfation by stably expressed human sulfotransferase 2A1 (SULT2A1). Xenobiotica 2010, 40, 184–194. [Google Scholar] [CrossRef] [PubMed]
- Kooistra, B.; Dijkman, B.; Einhorn, T.A.; Bhandari, M. How to Design a Good Case Series. J. Bone Jt. Surg. 2009, 91, 21–26. [Google Scholar] [CrossRef]
- Albrecht, J.; Meves, A.; Bigby, M. Case reports and case series from Lancet had significant impact on medical literature. J. Clin. Epidemiol. 2005, 58, 1227–1232. [Google Scholar] [CrossRef]
- Irwin, J.J.; Shoichet, B.K. ZINC—A Free Database of Commercially Available Compounds for Virtual Screening. J. Chem. Inf. Model. 2005, 45, 177–182. [Google Scholar] [CrossRef] [Green Version]
- Waterhouse, A.; Bertoni, M.; Bienert, S.; Studer, G.; Tauriello, G.; Gumienny, R.; Heer, F.T.; de Beer, T.A.P.; Rempfer, C.; Bordoli, L.; et al. SWISS-MODEL: Homology modelling of protein structures and complexes. Nucleic Acids Res. 2018, 46, W296–W303. [Google Scholar] [CrossRef] [Green Version]
- Abraham, M.J.; Murtola, T.; Schulz, R.; Pall, S.; Smith, J.C.; Hess, B.; Lindahl, E. GROMACS: High performance molecular simulations through multi-level parallelism from laptops to supercomputers. SoftwareX 2015, 1–2, 19–25. [Google Scholar] [CrossRef] [Green Version]
- Forli, S.; Huey, R.; Pique, M.E.; Sanner, M.F.; Goodsell, D.S.; Olson, A.J. Computational protein–ligand docking and virtual drug screening with the AutoDock suite. Nat. Protoc. 2016, 11, 905–919. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Trott, O.; Olson, A.J. AutoDock Vina: Improving the speed and accuracy of docking with a new scoring function, efficient optimization, and multithreading. J. Comput. Chem. 2010, 31, 455–461. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sun, J.; Jeliazkova, N.; Chupakhin, V.; Golib-Dzib, J.-F.; Engkvist, O.; Carlsson, L.; Wegner, J.; Ceulemans, H.; Georgiev, I.; Jeliazkov, V.; et al. ExCAPE-DB: An integrated large scale dataset facilitating Big Data analysis in chemogenomics. J. Cheminform. 2017, 9, 17. [Google Scholar] [CrossRef] [Green Version]
- Veerman, C.C.; Wilde, A.A.M.; Lodder, E.M. The cardiac sodium channel gene SCN5A and its gene product NaV1.5: Role in physiology and pathophysiology. Gene 2015, 573, 177–187. [Google Scholar] [CrossRef]
- Morgan, H.L. The Generation of a Unique Machine Description for Chemical Structures—A Technique Developed at Chemical Abstracts Service. J. Chem. Doc. 1965, 5, 107–113. [Google Scholar] [CrossRef]
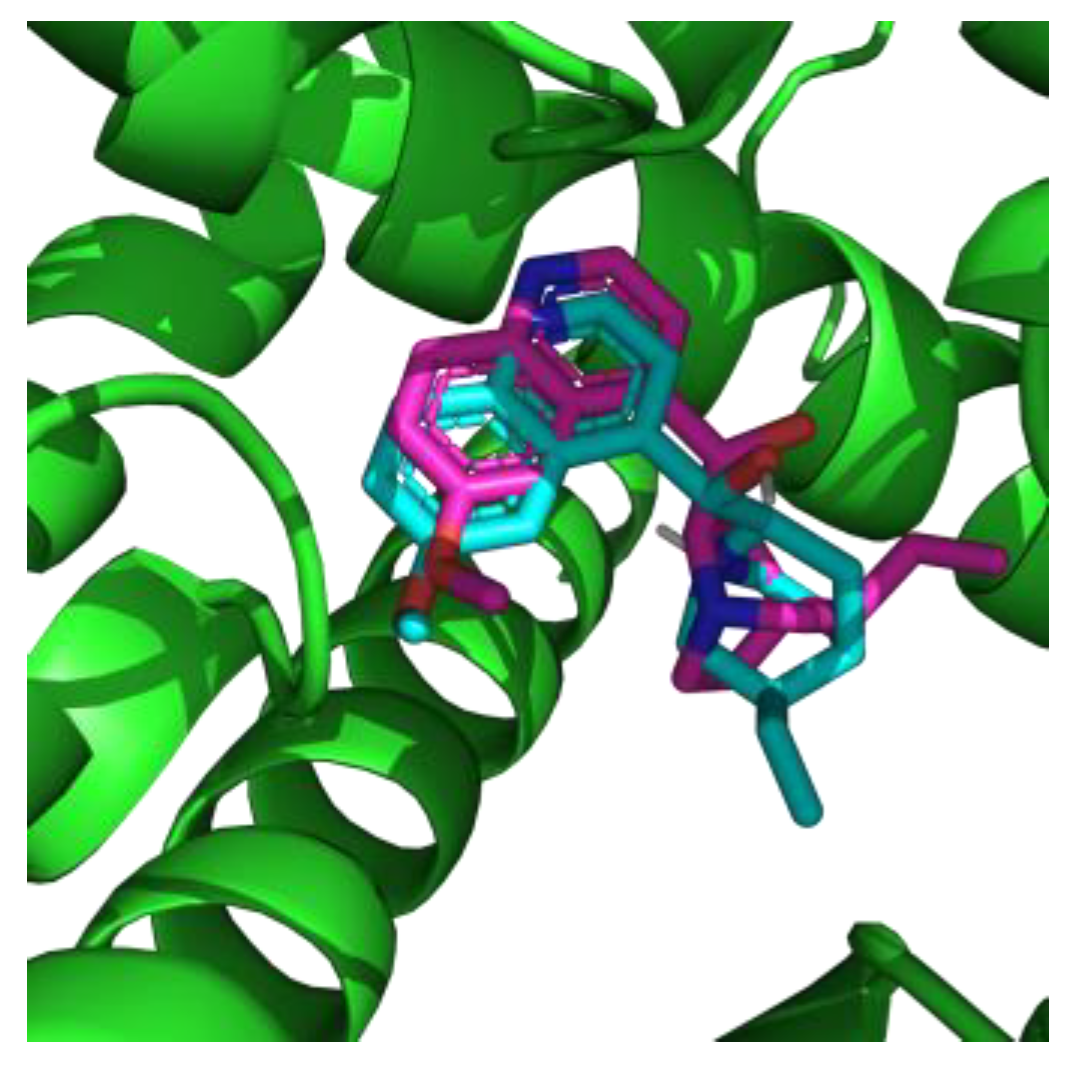
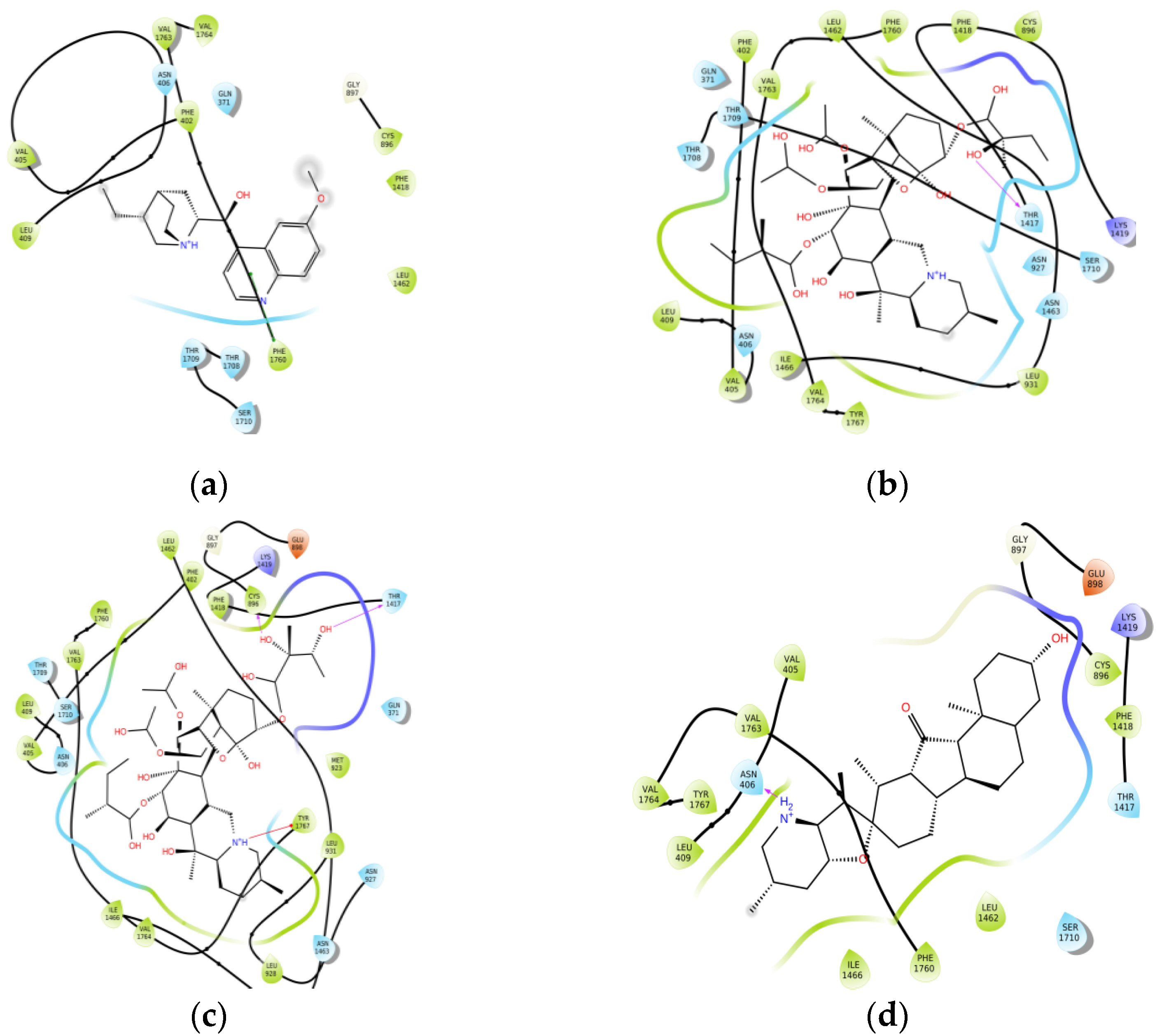

| Compound | Docking Score (kcal/mol) | Predicted IC50 (μmol) | logP | pKa |
|---|---|---|---|---|
| Jervine | −10.8 | 5.00 | 4.188 | 9.64 |
| ProA | −6.8 | 6.18 | 1.607 | 7.28 |
| ProB | −6.6 | 6.20 | 0.578 | 7.28 |
| Quinidine | −7.2 | 7.10 1 | 3.173 | 8.71 |
| Patient | Veratrum Alkaloid | Alkaloid Concentration, ng/mL | ||||
|---|---|---|---|---|---|---|
| 0 h | 24 h | 36 h | 48 h | 60 h | ||
| 1 | Jervine | 0.52 | 0.14 | - | - | - |
| ProA | 0.20 | - | - | - | - | |
| ProB | + | - | - | - | - | |
| 2 | Jervine | 0.35 | 0.10 | - | - | - |
| ProA | 0.11 | - | - | - | - | |
| ProB | + | - | - | - | - | |
| 3 | Jervine | 5.01 | 0.72 | 0.48 | 0.33 | 0.18 |
| ProA | 0.67 | - | - | - | - | |
| ProB | + | + | - | - | - | |
| 4 | Jervine | 0.10 | - | - | - | - |
| ProA | - | - | - | - | - | |
| ProB | - | - | - | - | - | |
| 5 | Jervine | 0.15 | - | - | - | - |
| ProA | 0.10 | - | - | - | - | |
| ProB | - | - | - | - | - | |
| 6 | Jervine | 0.11 | - | - | - | - |
| ProA | - | - | - | - | - | |
| ProB | - | - | - | - | - | |
| 7 | Jervine | 0.13 | - | - | - | - |
| ProA | 0.12 | - | - | - | - | |
| ProB | + | - | - | - | - | |
| Patient | Veratrum Alkaloid | Alkaloid Concentration, ng/mL | ||||
|---|---|---|---|---|---|---|
| 0 h | 24 h | 36 h | 48 h | 60 h | ||
| 1 | Jervine | 0.58 | 9.55 | - | - | - |
| ProA | 37.70 | 1.01 | - | - | - | |
| ProB | + | + | - | - | - | |
| 2 | Jervine | 0.24 | 1.15 | - | - | - |
| ProA | 6.13 | 8.14 | 0.19 | - | - | |
| ProB | + | + | + | - | - | |
| 3 | Jervine | 1.71 | 5.19 | 1.85 | 1.07 | 0.23 |
| ProA | 4.87 | 2.81 | 0.87 | 0.46 | - | |
| ProB | + | + | + | + | + | |
| 4 | Jervine | 0.23 | 0.10 | - | - | - |
| ProA | 0.15 | - | - | - | - | |
| ProB | + | + | - | - | - | |
| 5 | Jervine | 0.21 | 1.47 | - | - | - |
| ProA | 11.96 | 1.66 | - | - | - | |
| ProB | + | - | - | - | - | |
| 6 | Jervine | 0.46 | - | - | - | - |
| ProA | 54.41 | - | - | - | - | |
| ProB | + | - | - | - | - | |
| 7 | Jervine | 1.39 | - | - | - | - |
| ProA | 52.16 | - | - | - | - | |
| ProB | + | - | - | - | - | |
| Time, min | Volume Fraction of Solvent A, % | Volume Fraction of Solvent B, % |
|---|---|---|
| 0:00 | 90 | 10 |
| 1:00 | 90 | 10 |
| 1:10 | 75 | 25 |
| 1:50 | 75 | 25 |
| 9:50 | 50 | 50 |
| 9:60 | 10 | 90 |
| 11:00 | 10 | 90 |
| 11:10 | 90 | 10 |
| 14:00 | 90 | 10 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Taldaev, A.; Terekhov, R.P.; Melnik, E.V.; Belova, M.V.; Kozin, S.V.; Nedorubov, A.A.; Pomerantseva, T.Y.; Ramenskaya, G.V. Insights into the Cardiotoxic Effects of Veratrum Lobelianum Alkaloids: Pilot Study. Toxins 2022, 14, 490. https://doi.org/10.3390/toxins14070490
Taldaev A, Terekhov RP, Melnik EV, Belova MV, Kozin SV, Nedorubov AA, Pomerantseva TY, Ramenskaya GV. Insights into the Cardiotoxic Effects of Veratrum Lobelianum Alkaloids: Pilot Study. Toxins. 2022; 14(7):490. https://doi.org/10.3390/toxins14070490
Chicago/Turabian StyleTaldaev, Amir, Roman P. Terekhov, Elizaveta V. Melnik, Maria V. Belova, Sergey V. Kozin, Andrey A. Nedorubov, Tatyana Ya. Pomerantseva, and Galina V. Ramenskaya. 2022. "Insights into the Cardiotoxic Effects of Veratrum Lobelianum Alkaloids: Pilot Study" Toxins 14, no. 7: 490. https://doi.org/10.3390/toxins14070490
APA StyleTaldaev, A., Terekhov, R. P., Melnik, E. V., Belova, M. V., Kozin, S. V., Nedorubov, A. A., Pomerantseva, T. Y., & Ramenskaya, G. V. (2022). Insights into the Cardiotoxic Effects of Veratrum Lobelianum Alkaloids: Pilot Study. Toxins, 14(7), 490. https://doi.org/10.3390/toxins14070490





