Immobilization of Microbes for Biodegradation of Microcystins: A Mini Review
Abstract
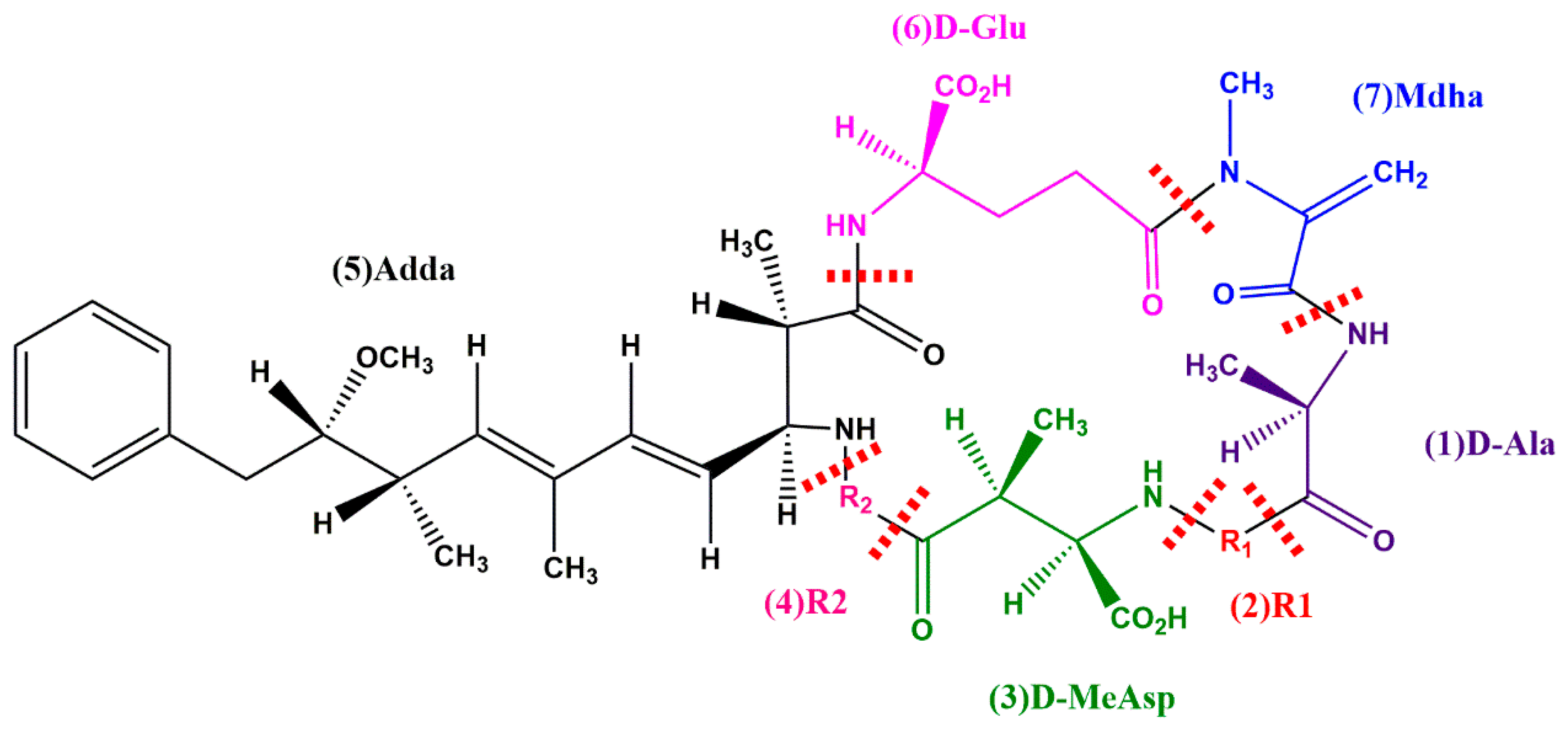
:1. Introduction
2. Biodegradation of MCs
3. Microbial Immobilization and Its Application for MCs Degradation
3.1. Microbial Immobilization
3.2. The Carriers of Microbial Immobilization
3.3. The Methods of Microbial Immobilization
3.4. Present Understanding of Microbial Immobilization for MCs Degradation
4. Summary
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Ho, J.C.; Michalak, A.M.; Pahlevan, N. Widespread global increase in intense lake phytoplankton blooms since the 1980s. Nature 2019, 574, 667–670. [Google Scholar] [CrossRef]
- Svirčev, Z.; Lalić, D.; Bojadžija Savić, G.; Tokodi, N.; Drobac Backović, D.; Chen, L.; Meriluoto, J.; Codd, G.A. Global geographical and historical overview of cyanotoxin distribution and cyanobacterial poisonings. Arch. Toxicol. 2019, 93, 2429–2481. [Google Scholar] [CrossRef] [PubMed]
- Christoffersen, K.; Kaas, H. Toxic cyanobacteria in water. A guide to their public health consequences, monitoring, and management. Limnol. Oceanogr. 2000, 45, 1212. [Google Scholar] [CrossRef]
- Cheung, M.Y.; Liang, S.; Lee, J. Toxin-producing cyanobacteria in freshwater: A review of the problems, impact on drinking water safety, and efforts for protecting public health. J. Microbiol. 2013, 51, 1–10. [Google Scholar] [CrossRef]
- Kadiri, M.O.; Isagba, S.; Ogbebor, J.U.; Omoruyi, O.A.; Unusiotame-Owolagba, T.E.; Lorenzi, A.S.; Bittencourt-Oliveira, M.d.C.; Chia, M.A. The presence of microcystins in the coastal waters of Nigeria, from the Bights of Bonny and Benin, Gulf of Guinea. Environ. Sci. Pollut. Res. 2020, 27, 35284–35293. [Google Scholar] [CrossRef]
- Wan, X.; Steinman, A.D.; Gu, Y.; Zhu, G.; Shu, X.; Xue, Q.; Zou, W.; Xie, L. Occurrence and risk assessment of microcystin and its relationship with environmental factors in lakes of the eastern plain ecoregion, China. Environ. Sci. Pollut. Res. 2020, 27, 45095–45107. [Google Scholar] [CrossRef] [PubMed]
- Wood, S.A.; Puddick, J.; Hawes, I.; Steiner, K.; Dietrich, D.R.; Hamilton, D.P. Variability in microcystin quotas during a Microcystis bloom in a eutrophic lake. PLoS ONE 2021, 16, e0254967. [Google Scholar] [CrossRef]
- Bouaïcha, N.; Miles, C.O.; Beach, D.G.; Labidi, Z.; Djabri, A.; Benayache, N.Y.; Nguyen-Quang, T. Structural diversity, characterization and toxicology of microcystins. Toxins 2019, 11, 714. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Schaefer, A.M.; Yrastorza, L.; Stockley, N.; Harvey, K.; Harris, N.; Grady, R.; Sullivan, J.; Mcfarland, M.; Reif, J.S. Exposure to microcystin among coastal residents during a cyanobacteria bloom in Florida. Harmful Algae 2020, 92, 101769. [Google Scholar] [CrossRef]
- Xiang, L.; Li, Y.W.; Liu, B.L.; Zhao, H.M.; Li, Q.X. High ecological and human health risks from microcystins in vegetable fields in southern China. Environ. Int. 2019, 133, 105142. [Google Scholar] [CrossRef]
- Teixeira Mda, G.; Costa Mda, C.; de Carvalho, V.L.; Pereira Mdos, S.; Hage, E. Gastroenteritis epidemic in the area of the Itaparica Dam, Bahia, Brazil. Bull. Pan. Am. Health Organ. 1993, 27, 244–253. [Google Scholar]
- Carmichael, W.W.; Azevedo, S.M.; An, J.S.; Molica, R.J.; Jochimsen, E.M.; Lau, S.; Rinehart, K.L.; Shaw, G.R.; Eaglesham, G.K. Human fatalities from cyanobacteria: Chemical and biological evidence for cyanotoxins. Environ. Health Persp. 2001, 109, 663–668. [Google Scholar] [CrossRef]
- Greer, B.; Meneely, J.P.; Elliott, C.T. Uptake and accumulation of Microcystin-LR based on exposure through drinking water: An animal model assessing the human health risk. Sci. Rep. 2018, 8, 4913. [Google Scholar] [CrossRef] [Green Version]
- Gu, S.; Yan, M.; Wang, C.; Meng, X.; Xiang, Z.; Qiu, Y.; Han, X. Microcystin-leucine-arginine induces liver fibrosis by activating the Hedgehog pathway in hepatic stellate cells. Biochem. Biophys. Res. Commun. 2020, 533, 770–778. [Google Scholar] [CrossRef]
- Codd, G.; Bell, S.; Kaya, K.; Ward, C.; Beattie, K.; Metcalf, J. Cyanobacterial toxins, exposure routes and human health. Eur. J. Phycol. 1999, 34, 405–415. [Google Scholar] [CrossRef]
- Yi, X.; Xu, S.; Huang, F.; Wen, C.; Zheng, S.; Feng, H.; Guo, J.; Chen, J.; Feng, X.; Yang, F. Effects of chronic exposure to microcystin-LR on kidney in mice. Int. J. Environ. Res. Public Health 2019, 16, 5030. [Google Scholar] [CrossRef] [Green Version]
- Du, C.; Zheng, S.; Yang, Y.; Feng, X.; Chen, J.; Tang, Y.; Wang, H.; Yang, F. Chronic exposure to low concentration of MC-LR caused hepatic lipid metabolism disorder. Ecotox. Environ. Saf. 2022, 239, 113649. [Google Scholar] [CrossRef]
- PR, H. Cyanobacterial toxins and human health. Symp. Ser. Soc. Appl. Microbiol. 1998, 27, 35S–40S. [Google Scholar]
- Clark, S.P.; Davis, M.A.; Ryan, T.P.; Searfoss, G.H.; Hooser, S.B. Hepatic gene expression changes in mice associated with prolonged sublethal microcystin exposure. Toxicol. Pathol. 2007, 35, 594–605. [Google Scholar] [CrossRef]
- Fujiki, H.; Suganuma, M. Tumor promoters--microcystin-LR, nodularin and TNF-α and human cancer development. Anti-Cancer Agents Med. Chem. 2011, 11, 4–18. [Google Scholar] [CrossRef]
- Mackintosh, C.; Beattie, K.A.; Klumpp, S.; Cohen, P.; Codd, G.A. Cyanobacterial microcystin-LR is a potent and specific inhibitor of protein phosphatases 1 and 2A from both mammals and higher plants. FEBS Lett. 1990, 264, 187–192. [Google Scholar] [CrossRef] [Green Version]
- IARC Working Group on the Evaluation of Carcinogenic Risks to Humans. IARC monographs on the evaluation of carcinogenic risks to humans. Ingested nitrate and nitrite, and cyanobacterial peptide toxins. IARC Monogr. Eval. Carcinog. Risks Hum. 2010, 94, v–vii, 1–412. [Google Scholar]
- Zhao, Y.; Yan, Y.; Xie, L.; Wang, L.; He, Y.; Wan, X.; Xue, Q. Long-term environmental exposure to microcystins increases the risk of nonalcoholic fatty liver disease in humans: A combined fisher-based investigation and murine model study. Environ. Int. 2020, 138, 105648. [Google Scholar] [CrossRef] [PubMed]
- WHO. Cyanobacterial Toxins: Microcystin-LR in Drinking-Water Background Document for Development of WHO Guidelines for Drinking-Water Quality; WHO: Geneva, Switzerland, 1998. [Google Scholar]
- Chen, L.; Giesy, J.P.; Adamovsky, O.; Svirčev, Z.; Meriluoto, J.; Codd, G.A.; Mijovic, B.; Shi, T.; Tuo, X.; Li, S.-C.; et al. Challenges of using blooms of Microcystis spp. in animal feeds: A comprehensive review of nutritional, toxicological and microbial health evaluation. Sci. Total Environ. 2021, 764, 142319. [Google Scholar] [CrossRef] [PubMed]
- Cai, D.; Wei, J.; Huang, F.; Feng, H.; Peng, T.; Luo, J.; Yang, F. The detoxification activities and mechanisms of microcystinase towards MC-LR. EcoToxicol. Environ. Saf. 2022, 236, 113436. [Google Scholar] [CrossRef] [PubMed]
- Massey, I.Y.; Peng, T.; Danping, C.; Yang, F. Optimization of biodegradation characteristics of Sphingopyxis sp. YF1 against crude microcystin-LR using response surface methodology. Toxins 2022, 14, 240. [Google Scholar] [CrossRef]
- Zhang, J.; Lu, Q.; Qin, D.; Pu, Y. A novel and native microcystin-degrading bacterium of Sphingopyxis sp. isolated from Lake Taihu. Int. J. Environ. Res. Public Health 2017, 14, 1187. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Eleuterio, L.; Batista, J.R. Biodegradation studies and sequencing of microcystin-LR degrading bacteria isolated from a drinking water biofilter and a fresh water lake. Toxicon 2010, 55, 1434–1442. [Google Scholar] [CrossRef] [PubMed]
- Wu, P.; Li, G.; He, Y.; Luo, D.; Yang, F. High-efficient and sustainable biodegradation of microcystin-LR using Sphingopyxis sp. YF1 immobilized Fe3O4@chitosan. Colloids Surf. B Biointerfaces 2019, 185, 110633. [Google Scholar] [CrossRef]
- Li, J.; Li, R.; Li, J. Current research scenario for microcystins biodegradation—A review on fundamental knowledge, application prospects and challenges. Sci. Total Environ. 2017, 595, 615. [Google Scholar] [CrossRef]
- Chen, L.; Chen, J.; Zhang, X.; Xie, P. A review of reproductive toxicity of microcystins. J. Hazard. Mater. 2016, 301, 381–399. [Google Scholar] [CrossRef] [PubMed]
- Cruz, A.; Antoniou, M.G.; Hiskia, A.; Pelaez, M.; Dionysiou, D.D. Can we effectively degrade microcystins?—Implications on human health. Anti-Cancer Agents Med. Chem. 2011, 11, 19–37. [Google Scholar] [CrossRef] [PubMed]
- Chintalapati, P.; Mohseni, M. Degradation of cyanotoxin microcystin-LR in synthetic and natural waters by chemical-free UV/VUV radiation. J. Hazard. Mater. 2020, 381, 120921. [Google Scholar] [CrossRef]
- Guo, Q.; Li, H.; Zhang, Q.; Zhang, Y. Fabrication, characterization and mechanism of a novel Z-scheme Ag3PO4/NG/polyimide composite photocatalyst for microcystin-LR degradation. Appl. Catal. B-Environ. 2018, 229, 192–203. [Google Scholar] [CrossRef]
- Şengül, A.B.; Ersan, G.; Tüfekçi, N. Removal of intra- and extracellular microcystin by submerged ultrafiltration (UF) membrane combined with coagulation/flocculation and powdered activated carbon (PAC) adsorption. J. Hazard. Mater. 2018, 343, 29–35. [Google Scholar] [CrossRef]
- Yang, F.; Huang, F.; Feng, H.; Wei, J.; Pu, Y. A complete route for biodegradation of potentially carcinogenic cyanotoxin microcystin-LR in a novel indigenous bacterium. Water Res. 2020, 174, 115638. [Google Scholar] [CrossRef]
- Liang, B.H.; Jing, D.Y.; Wei, Z.; Yu, F.Y.; Jian, C.; Zhi, Q.S. Isolation of a Methylobacillus sp. that degrades microcystin toxins associated with cyanobacteria. New Biotechnol. 2009, 26, 205–211. [Google Scholar]
- Kumar, P.; Hegde, K.; Brar, S.K.; Cledon, M.; Kermanshahi-pour, A.; Roy-Lachapelle, A.; Galvez-Cloutier, R. Biodegradation of microcystin-LR using acclimatized bacteria isolated from different units of the drinking water treatment plant. Environ. Pollut. 2018, 242, 407–416. [Google Scholar] [CrossRef] [Green Version]
- Jian, C.; Liang, B.H.; Wei, Z.; Shao, H.Y.; Zhi, Q.S. Degradation of microcystin-LR and RR by a Stenotrophomonas sp. strain EMS Isolated from Lake Taihu, China. Int. J. Mol. Sci. 2010, 11, 896–911. [Google Scholar]
- Bourne, D.G.; Jones, G.J.; Blakeley, R.L.; Jones, A.; Negri, A.P.; Riddles, P. Enzymatic pathway for the bacterial degradation of the cyanobacterial cyclic peptide toxin microcystin LR. Appl. Environ. Microbiol. 1996, 62, 4086–4094. [Google Scholar] [CrossRef] [Green Version]
- Zhang, L.; Gu, L.; Wei, Q.; Zhu, X.; Wang, J.; Wang, X.; Yang, Z. High temperature favors elimination of toxin-producing Microcystis and degradation of microcystins by mixotrophic Ochromonas. Chemosphere 2017, 172, 96–102. [Google Scholar] [CrossRef]
- Jing, W.; Sui, G.; Liu, S. Characteristics of a microcystin-LR biodegrading bacterial isolate: Ochrobactrum sp. FDT5. Bull. Environ. Contam. Toxicol. 2014, 92, 119–122. [Google Scholar] [CrossRef] [PubMed]
- Zhou, Y.; Yang, F.; Liang, G.; Yin, L.; Pu, Y. Biodegradation of microcystin-RR and -LR by an indigenous bacterial strain MC-LTH11 isolated from Lake Taihu. J. Southeast. Univ. 2014, 30, 68–71. [Google Scholar]
- Chen, Y.M.; Lin, T.F.; Huang, C.; Lin, J.C.; Hsieh, F.M. Degradation of phenol and TCE using suspended and chitosan-bead immobilized Pseudomonas Putida. J. Hazard. Mater. 2007, 148, 660–670. [Google Scholar] [CrossRef] [PubMed]
- Fukui, S.; Tanaka, A. Immobilized microbial cells. Annu. Rev. Microbiol 1982, 36, 145–172. [Google Scholar] [CrossRef]
- El-Naas, M.H.; Alhaija, M.A.; Al-Zuhair, S. Evaluation of an activated carbon packed bed for the adsorption of phenols from petroleum refinery wastewater. Environ. Sci. Pollut. Res. 2017, 24, 7511–7520. [Google Scholar] [CrossRef]
- Nie, H.; Nie, M.; Diwu, Z.; Wang, L.; Yan, H.; Bai, X. Immobilization of Rhodococcus qingshengii strain FF on the surface of polyethylene and its adsorption and biodegradation of mimic produced water. J. Hazard. Mater. 2021, 403, 124075. [Google Scholar] [CrossRef] [PubMed]
- Al-Zuhair, S.; El-Naas, M. Immobilization of Pseudomonas putida in PVA gel particles for the biodegradation of phenol at high concentrations. Biochem. Eng. J. 2011, 56, 46–50. [Google Scholar] [CrossRef]
- Krishnan, A.; Zhang, Y.-Q.; Mou, X. Isolation and characterization of microcystin-degrading bacteria from Lake Erie. Bull. Environ. Contam. Toxicol. 2018, 101, 617–623. [Google Scholar] [CrossRef] [PubMed]
- Wu, J.; Zhong, S.; Wang, G.Y.; Li, Y.C. Identification and activity of a bacterial strain for the biodegradation of microcystins. China Environ. Sci. 2011, 31, 116–122. [Google Scholar]
- Huang, F.; Feng, H.; Li, X.; Yi, X.; Yang, F. Anaerobic degradation of microcystin-LR by an indigenous bacterial Enterobacter sp. YF3. J. Toxicol. Environ. Health A 2019, 82, 1–9. [Google Scholar] [CrossRef]
- Park, H.D. Degradation of the cyanobacterial hepatotoxin microcystin by a new bacterium isolated from a hypertrophic lake. Environ. Toxicol. 2010, 16, 337–343. [Google Scholar] [CrossRef] [PubMed]
- Ho, L.; Meyn, T.; Keegan, A.; Hoefel, D.; Brookes, J.; Saint, C.P.; Newcombe, G. Bacterial degradation of microcystin toxins within a biologically active sand filter. Water Res. 2006, 40, 768–774. [Google Scholar] [CrossRef] [PubMed]
- Kierek-Pearson, K.; Karatan, E. Biofilm development in bacteria. Adv. Appl. Microbiol. 2005, 57, 79–111. [Google Scholar] [PubMed]
- Morón-López, J.; Molina, S. Optimization of Recycled-Membrane Biofilm Reactor (R-MBfR) as a sustainable biological treatment for microcystins removal. Biochem. Eng. J. 2019, 153, 107422. [Google Scholar] [CrossRef]
- Kumar, P.; Hegde, K.; Brar, S.K.; Cledon, M.; Kermanshahi-pour, A.; Roy-Lachapelle, A.; Galvez-Cloutier, R. Novel fluidized-bed biofilm reactor for concomitant removal of microcystin-LR and organics. Chem. Eng. J. 2019, 359, 99–111. [Google Scholar] [CrossRef]
- Karel, S.F.; Libicki, S.B.; Robertson, C.R. The immobilization of whole cells: Engineering principles. Chem. Eng. Sci. 1985, 40, 1321–1354. [Google Scholar] [CrossRef]
- Kourkoutas, Y.; Bekatorou, A.; Banat, I.M.; Marchant, R.; Koutinas, A.A. Immobilization technologies and support materials suitable in alcohol beverages production: A review. Food Microbiol. 2004, 21, 377–397. [Google Scholar] [CrossRef]
- Xiaofan, Z.; Shaohong, Y.; Lili, M.; Chunquan, C.; Chao, L. The application of immobilized microorganism technology in wastewater treatment. In Proceedings of Proceedings of the 2015 2nd International Conference on Machinery, Materials Engineering, Chemical Engineering and Biotechnology, Chongqing, China, 28–29 December 2015; pp. 103–106. [Google Scholar]
- Kadimpati, K.K.; Mondithoka, K.P.; Bheemaraju, S.; Challa, V.R.M. Entrapment of marine microalga, Isochrysis galbana, for biosorption of Cr(III) from aqueous solution: Isotherms and spectroscopic characterization. Appl. Water Sci. 2013, 3, 85–92. [Google Scholar] [CrossRef] [Green Version]
- Podder, M.S.; Majumder, C.B. Bacteria immobilization on neem leaves/MnFe2O4 composite surface for removal of As(III) and As(V) from wastewater. Arab. J. Chem. 2019, 12, 3263–3288. [Google Scholar] [CrossRef]
- Ouyang, X.; Yin, H.; Yu, X.; Guo, Z.; Zhu, M.; Lu, G.; Dang, Z. Enhanced bioremediation of 2,3′,4,4′,5-pentachlorodiphenyl by consortium GYB1 immobilized on sodium alginate-biochar. Sci. Total Environ. 2021, 788, 147774. [Google Scholar] [CrossRef] [PubMed]
- Rodríguez Couto, S. Dye removal by immobilised fungi. Biotechnol. Adv. 2009, 27, 227–235. [Google Scholar] [CrossRef]
- El-Naas, M.H.; Al-Muhtaseb, S.A.; Makhlouf, S. Biodegradation of phenol by Pseudomonas putida immobilized in polyvinyl alcohol (PVA) gel. J. Hazard. Mater. 2009, 164, 720–725. [Google Scholar] [CrossRef] [PubMed]
- Bayat, Z.; Hassanshahian, M.; Cappello, S. Immobilization of microbes for bioremediation of crude oil polluted environments: A mini review. Open Microbiol. J. 2015, 9, 48–54. [Google Scholar] [PubMed]
- Bouabidi, Z.B.; El-Naas, M.H.; Zhang, Z. Immobilization of microbial cells for the biotreatment of wastewater: A review. Environ. Chem. Lett. 2019, 17, 241–257. [Google Scholar] [CrossRef]
- Winnicki, T.; Szetela, R.; Wisniewski, J. Immobilized microorganisms in wastewater treatment. In Studies in Environmental Science; Pawlowski, L., Ed.; Elsevier: Amsterdam, The Netherlands, 1982; Volume 19, pp. 341–352. [Google Scholar]
- Zheng, Y.; Chen, D.; Li, N.; Xu, Q.; Li, H.; He, J.; Lu, J. Highly efficient simultaneous adsorption and biodegradation of a highly-concentrated anionic dye by a high-surface-area carbon-based biocomposite. Chemosphere 2017, 179, 139–147. [Google Scholar] [CrossRef]
- Dong, Z.; Lu, M.; Huang, W.; Xu, X. Treatment of oilfield wastewater in moving bed biofilm reactors using a novel suspended ceramic biocarrier. J. Hazard. Mater. 2011, 196, 123–130. [Google Scholar] [CrossRef] [PubMed]
- Bai, X.; Ye, Z.F.; Li, Y.F.; Zhou, L.C.; Yang, L.Q. Preparation of crosslinked macroporous PVA foam carrier for immobilization of microorganisms. Process Biochem. 2010, 45, 60–66. [Google Scholar] [CrossRef]
- Thornhill, S.G.; Kumar, M. Biological filters and their use in potable water filtration systems in spaceflight conditions. Life Sci. Space Res. 2018, 17, 40–43. [Google Scholar] [CrossRef] [PubMed]
- Smidsrød, O.; Skjåk-Braek, G. Alginate as immobilization matrix for cells. Trends Biotechnol. 1990, 8, 71–78. [Google Scholar] [CrossRef]
- Banerjee, A.; Ghoshal, A.K. Biodegradation of real petroleum wastewater by immobilized hyper phenol-tolerant strains of Bacillus cereus in a fluidized bed bioreactor. 3 Biotech 2016, 6, 137. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Shaharuddin, S.; Muhamad, I.I. Microencapsulation of alginate-immobilized bagasse with Lactobacillus rhamnosus NRRL 442: Enhancement of survivability and thermotolerance. Carbohydr. Polym. 2015, 119, 173–181. [Google Scholar] [CrossRef] [PubMed]
- Ren, G.; He, X.; Wu, P.; He, Y.; Yang, F. Biodegradation of microcystin-RR and nutrient pollutants using Sphingopyxis sp. YF1 immobilized activated carbon fibers-sodium alginate. Environ. Sci. Pollut. Res. 2020, 27, 1–11. [Google Scholar] [CrossRef]
- Chhipa, H. Applications of nanotechnology in agriculture. In Methods in Microbiology; Gurtler, V., Ball, A.S., Soni, S., Eds.; Academic Press: Cambridge, MA, USA, 2019; Volume 46, pp. 115–142. [Google Scholar]
- Dasgupta, N.; Ranjan, S.; Ramalingam, C. Applications of nanotechnology in agriculture and water quality management. Environ. Chem. Lett. 2017, 15, 591–605. [Google Scholar] [CrossRef]
- Madhura, L.; Singh, S.; Kanchi, S.; Sabela, M.; Bisetty, K.; Inamuddin. Nanotechnology-based water quality management for wastewater treatment. Environ. Chem. Lett. 2019, 17, 65–121. [Google Scholar] [CrossRef]
- Paterson, E. The iron oxides. Structure, properties, reactions, occurrences and uses. Clay Miner. 1999, 34, 209–210. [Google Scholar] [CrossRef]
- Chan, H.B.S.; Ellis, B.L.; Sharma, H.L.; Frost, W.; Caps, V.; Shields, R.A.; Tsang, S.C.J.A.M. Carbon-encapsulated radioactive 99mTc nanoparticles. Adv. Mater. 2004, 16, 144–149. [Google Scholar] [CrossRef]
- Bestawy, E.E.; El-Shatby, B.F.; Eltaweil, A.S. Integration between bacterial consortium and magnetite (Fe3O4) nanoparticles for the treatment of oily industrial wastewater. World J. Microbiol. Biotechnol. 2020, 36, 1–16. [Google Scholar] [CrossRef]
- Zamani, H.; Rakhshaee, R.; Garakoui, S.R. Two contrary roles of Fe3O4 nanoparticles on kinetic and thermodynamic of Paclitaxel degradation by Citrobacter amalonaticus Rashtia immobilized on sodium alginate gel beads. J. Hazard. Mater. 2017, 344, 566–575. [Google Scholar] [CrossRef] [PubMed]
- Sun, P.; Hui, C.; Wang, S.; Khan, R.A.; Zhang, Q.; Zhao, Y.H. Enhancement of algicidal properties of immobilized Bacillus methylotrophicus ZJU by coating with magnetic FeO nanoparticles and wheat bran. J. Hazard. Mater. 2016, 301, 65–73. [Google Scholar] [CrossRef] [PubMed]
- Kuo, C.H.; Liu, Y.C.; Chang, C.; Chen, J.H.; Chang, C.; Shieh, C.J. Optimum conditions for lipase immobilization on chitosan-coated Fe3O4 nanoparticles. Carbohydr. Polym 2012, 87, 2538–2545. [Google Scholar] [CrossRef]
- Li, Q.; Wang, P.; Zhou, Y.-l.; Nie, Z.; Wei, Q. A magnetic mesoporous SiO2/Fe3O4 hollow microsphere with a novel network-like composite shell: Synthesis and application on laccase immobilization. J. Sol.-Gel. Sci. Techn. 2016, 78, 523–530. [Google Scholar] [CrossRef]
- Bahrami, A.; Hejazi, P. Electrostatic immobilization of pectinase on negatively charged AOT-Fe3O4 nanoparticles. J. Mol. Catal. B Enzym. 2013, 93, 1–7. [Google Scholar] [CrossRef]
- Moron-Lopez, J.; Nieto-Reyes, L.; Senan-Salinas, J.; Molina, S.; El-Shehawy, R. Recycled desalination membranes as a support material for biofilm development: A new approach for microcystin removal during water treatment. Sci. Total Environ. 2018, 647, 785–793. [Google Scholar] [CrossRef] [PubMed]
- Sun, J.; Liu, J.; Liu, Y.; Li, Z.; Nan, J. Optimization of entrapping conditions of nitrifying bacteria and selection of entrapping agent. Procedia Environ. Sci. 2011, 8, 166–172. [Google Scholar] [CrossRef] [Green Version]
- Yoetz-Kopelman, T.; Dror, Y.; Shacham-Diamand, Y.; Freeman, A. “Cells-on-Beads”: A novel immobilization approach for the construction of whole-cell amperometric biosensors. Sensors Actuators B Chem. 2016, 232, 758–764. [Google Scholar] [CrossRef]
- Trevan, M.D. Enzyme immobilization by adsorption: A review. Adsorption 2014, 20, 801–821. [Google Scholar]
- Zhu, Y.; Ren, X.; Liu, Y.; Wei, Y.; Qing, L.; Liao, X. Covalent immobilization of porcine pancreatic lipase on carboxyl-activated magnetic nanoparticles: Characterization and application for enzymatic inhibition assays. Mat. Sci. Eng. C 2014, 38, 278–285. [Google Scholar] [CrossRef]
- Singh, R.; Paul, D.; Jain, R.K. Biofilms: Implications in bioremediation. Trends Microbiol. 2006, 14, 389–397. [Google Scholar] [CrossRef] [PubMed]
- Wu, Y.; He, J.; Yang, L. Evaluating adsorption and biodegradation mechanisms during the removal of microcystin-RR by periphyton. Environ. Sci. Technol. 2010, 44, 6319–6324. [Google Scholar] [CrossRef]
- Wu, Y.; Li, T.; Yang, L. Mechanisms of removing pollutants from aqueous solutions by microorganisms and their aggregates: A review. Bioresour. Technol. 2012, 107, 10–18. [Google Scholar] [CrossRef]
- Ho, L.; Hoefel, D.; Saint, C.P.; Newcombe, G. Isolation and identification of a novel microcystin-degrading bacterium from a biological sand filter. Water Res. 2007, 41, 4685–4695. [Google Scholar] [CrossRef] [PubMed]
- Li, J.; Shimizu, K.; Sakharkar, M.K.; Utsumi, M.; Zhang, Z.; Sugiura, N. Comparative study for the effects of variable nutrient conditions on the biodegradation of microcystin-LR and concurrent dynamics in microcystin-degrading gene abundance. Bioresour. Technol. 2011, 102, 9509–9517. [Google Scholar] [CrossRef] [PubMed]
- Bourne, D.G.; Blakeley, R.L.; Riddles, P.; Jones, G.J. Biodegradation of the cyanobacterial toxin microcystin LR in natural water and biologically active slow sand filters. Water Res. 2006, 40, 1294–1302. [Google Scholar] [CrossRef]
- Cousins, I.T.; Bealing, D.J.; James, H.A.; Sutton, A. Biodegradation of microcystin-LR by indigenous mixed bacterial populations. Water Res. 1996, 30, 481–485. [Google Scholar] [CrossRef]
- Yan, H.; Pan, G.; Zou, H.; Li, X.; Chen, H. Effective removal of microcystins using carbon nanotubes embedded with bacteria. Chinese Sci. Bull. 2004, 49, 1694–1698. [Google Scholar] [CrossRef]
- Tsuji, K.; Asakawa, M.; Anzai, Y.; Sumino, T.; Harada, K.I. Degradation of microcystins using immobilized microorganism isolated in an eutrophic lake. Chemosphere 2006, 65, 117–124. [Google Scholar] [CrossRef]
- Dziga, D. Genetically engineered bacteria immobilized in alginate as an option of cyanotoxins removal. Inter. J. Environ. Sci. Dev. 2013, 4, 360–364. [Google Scholar] [CrossRef] [Green Version]
- Phujomjai, Y.; Somdee, A.; Somdee, T. Biodegradation of microcystin [Dha7]MC-LR by a novel microcystin-degrading bacterium in an internal airlift loop bioreactor. Water Sci. Technol. 2016, 73, 267. [Google Scholar] [CrossRef] [PubMed]
- Kumar, P.; Rubio, H.D.P.; Hegde, K.; Brar, S.K.; Cledon, M.; Kermanshahi-pour, A.; Sauvé, S.; Roy-Lachapelle, A.; Galvez-Cloutier, R. Agro-industrial residues as a unique support in a sand filter to enhance the bioactivity to remove microcystin-Leucine aRginine and organics. Sci. Total Environ. 2019, 670, 971–981. [Google Scholar] [CrossRef]
- Kumar, P.; Pérez, J.A.E.; Cledon, M.; Brar, S.K.; Duy, S.V.; Sauvé, S.; Knystautas, É. Removal of microcystin-LR and other water pollutants using sand coated with bio-optimized carbon submicron particles: Graphene oxide and reduced graphene oxide. Chem Eng J 2020, 397, 125398. [Google Scholar] [CrossRef]
- Somdee, T.; Wibuloutai, J.; Somdee, T.; Somdee, A. Biodegradation of the cyanobacterial hepatotoxin [Dha7] microcystin-LR within a biologically active sand filter. Water Supply 2014, 14, 672–680. [Google Scholar] [CrossRef]
- Somdee, T.; Somdee, A.; Ruck, J.; Allison, M.; Page, R. Removal of microcystin from lake water using an internal airlift loop ceramic honeycomb support bioreactor. Chiang Mai J. Sci. 2016, 43, 1048–1058. [Google Scholar]
- Elhadi, S.L.N.; Huck, P.M.; Slawson, R.M. Factors affecting the removal of geosmin and MIB in drinking water biofilters. J. AWWA 2006, 98, 108–119. [Google Scholar] [CrossRef]
- Wang, H.; Ho, L.; Lewis, D.M.; Brookes, J.D.; Newcombe, G. Discriminating and assessing adsorption and biodegradation removal mechanisms during granular activated carbon filtration of microcystin toxins. Water Res. 2007, 41, 4262–4270. [Google Scholar] [CrossRef]
- Ho, L.; Schwarz, C.; Hoefel, D.; Steele David, A. Comparison of growth and biofilm attachment of two microcystin-degrading bacteria to plasma polymerized surface coatings. J. Environ. Eng. 2015, 141, 06015003. [Google Scholar] [CrossRef]

| Microorganism | Carrier | Microcystins | Initial Concentration (μg/mL) | Degradation Period | Degradation % | Degradation Rate (µg/L/h) | Container | Condition | Reference |
|---|---|---|---|---|---|---|---|---|---|
| Sphingopyxis sp. YF1 | ACF-SA | MC-RR | 12 | 8 h | 100 | 7.6 × 102 | - | 30 °C, pH 7.0 | [76] |
| Sphingopyxis sp. YF1 | Fe3O4@CTS | MC-LR | 10 | 12 h | 100 | 6.5 × 105 | - | 30 °C, pH 7.2 (Deionized water) | [30] |
| Ralstonia solanacearum | carbon nanotubes | MC-RR | 52.5 | within 24 h | 100 | 2.18 × 103 | flasks | 30 °C, pH 7.0 (PBS) | [100] |
| MC-LR | 29.5 | ||||||||
| B-9 | polyester (Fabios) | MC-RR | 0.2 | 24 h | 100 | 1.25 × 104 | Aeration bioreactor | 25 °C, pH 7.4 (PBS) | [101] |
| E. coli BL21_MlrA | alginate | MC-LR | 0.035 | - | 100 | 100.3 | Column | (freshwater) | [102] |
| Sphingomonas isolate NV-3 | ceramic | [Dha7]MC-LR | 25 | 30 h | 100 | 8.3 × 102 | IAL-CHS bioreactor | 30 °C, pH 7.2 (synthetic wastewater) | [103] |
| Arthrobacter ramosus + HBC | K1 Kaldness media | MC-LR | 50 | 6 days | 93.75 | 3.5 × 102 | Fluidized bed biofilm reactor | 15–19 °C, pH 7.2 (PBS) | [57] |
| Bacillus sp. + HBC | K1 Kaldness media | MC-LR | 50 | 6 days | 90.24 | 34.72 | |||
| Arthrobacter ramosus (NRRL B-3159) + native bacterial | deinking sludge + sand | MC-LR | 0.05 | 7 cycle study (49 days) | 87 ± 14 | - | sand filter | pH 6.4 (Lake water) | [104] |
| hemp fiber + sand | 82 ± 7 | pH 6.5 (Lake water) | |||||||
| paper-pulp dry sludge + sand | 78 ± 4 | pH 6.6 (Lake water) | |||||||
| Arthrobacter ramosus + native bacterial species | rGO-coated sand | MC-LR | 50 | stage (3): 6 cycles | 91.4 ± 5.6 | - | sand filter | (Lake water) | [105] |
| Novosphingobium sp. KKU15 | sand | [Dha7]MC-LR | 5 | 7 d EBCT: not mentioned | 100 | - | slow sand filter | 30 °C, pH 7.2 (Mineral salt medium) | [106] |
| Novosphingobium sp. KKU25s | plastic medium | [Dha7]MC-LR | 0.025 | within 24 h | 100 | 1.04 | sterile bioreactor | 30 °C, pH 7.2 (fresh synthetic wastewater) | [103] |
| Sphingopyxis sp. strain IM-1 | RO membranes | MCs | 2 | 24 h | 100 | 83.33 | flasks | 27 °C, pH 7.2 (Mineral salt medium) | [88] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zhang, J.; Wei, J.; Massey, I.Y.; Peng, T.; Yang, F. Immobilization of Microbes for Biodegradation of Microcystins: A Mini Review. Toxins 2022, 14, 573. https://doi.org/10.3390/toxins14080573
Zhang J, Wei J, Massey IY, Peng T, Yang F. Immobilization of Microbes for Biodegradation of Microcystins: A Mini Review. Toxins. 2022; 14(8):573. https://doi.org/10.3390/toxins14080573
Chicago/Turabian StyleZhang, Jiajia, Jia Wei, Isaac Yaw Massey, Tangjian Peng, and Fei Yang. 2022. "Immobilization of Microbes for Biodegradation of Microcystins: A Mini Review" Toxins 14, no. 8: 573. https://doi.org/10.3390/toxins14080573






