AgTx2-GFP, Fluorescent Blocker Targeting Pharmacologically Important Kv1.x (x = 1, 3, 6) Channels
Abstract
:1. Introduction
2. Results and Discussion
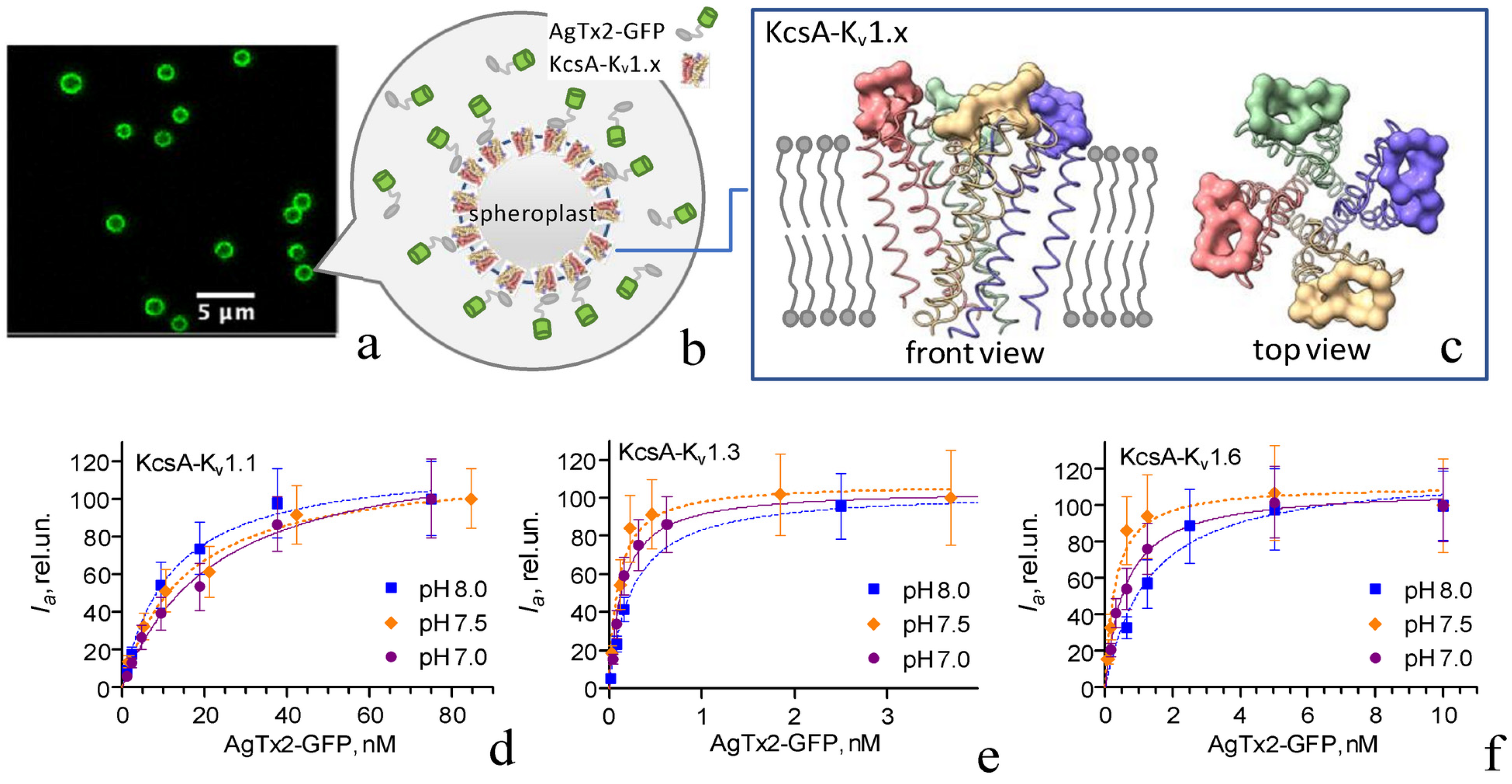
2.1. Interactions of AgTx2-GFP with KcsA-Kv1.x (x = 1, 3, 6) Channels
2.2. Electrophysiological Studies of AgTx2-GFP
2.3. Interaction of AgTx2-GFP with Kv1.3 on Mammalian Cell Membranes
3. Conclusions
4. Materials and Methods
4.1. Reagents
4.2. Experiments with KcsA and KcsA-Kv1.x (x = 1, 3, 6) Channels
4.3. Electrophysiological Experiments
4.4. Experiments with Neuro2a Cells Expressing Kv1.3 Channels
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Kuzmenkov, A.I.; Grishin, E.V.; Vassilevski, A.A. Diversity of Potassium Channel Ligands: Focus on Scorpion Toxins. Biochemistry 2015, 80, 1764–1799. [Google Scholar] [CrossRef] [PubMed]
- Rodríguez de la Vega, R.C.; Possani, L.D. Current Views on Scorpion Toxins Specific for K+-Channels. Toxicon 2004, 43, 865–875. [Google Scholar] [CrossRef] [PubMed]
- Kuang, Q.; Purhonen, P.; Hebert, H. Structure of Potassium Channels. Cell. Mol. Life Sci. 2015, 72, 3677–3693. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Fan, Y.; Zong-Yun, C.; Ying-Liang, W. Unique Interactions between Scorpion Toxins and Small Conductance Ca2+-Activated Potassium Channels. Acta Physiol. Sin. 2015, 67, 255–260. [Google Scholar] [CrossRef]
- Gutman, G.A.; Chandy, K.G.; Grissmer, S.; Lazdunski, M.; McKinnon, D.; Pardo, L.A.; Robertson, G.A.; Rudy, B.; Sanguinetti, M.C.; Stühmer, W.; et al. International Union of Pharmacology. LIII. Nomenclature and Molecular Relationships of Voltage-Gated Potassium Channels. Pharmacol. Rev. 2005, 57, 473–508. [Google Scholar] [CrossRef] [PubMed]
- Mouhat, S.; Visan, V.; Ananthakrishnan, S.; Wulff, H.; Andreotti, N.; Grissmer, S.; Darbon, H.; De Waard, M.; Sabatier, J.-M. K+ Channel Types Targeted by Synthetic OSK1, a Toxin from Orthochirus Scrobiculosus Scorpion Venom. Biochem. J. 2005, 385, 95–104. [Google Scholar] [CrossRef] [Green Version]
- Kuzmenkov, A.I.; Nekrasova, O.V.; Kudryashova, K.S.; Peigneur, S.; Tytgat, J.; Stepanov, A.V.; Kirpichnikov, M.P.; Grishin, E.V.; Feofanov, A.V.; Vassilevski, A.A. Fluorescent Protein-Scorpion Toxin Chimera Is a Convenient Molecular Tool for Studies of Potassium Channels. Sci. Rep. 2016, 6, 33314. [Google Scholar] [CrossRef]
- Mouhat, S.; Teodorescu, G.; Homerick, D.; Visan, V.; Wulff, H.; Wu, Y.; Grissmer, S.; Darbon, H.; De Waard, M.; Sabatier, J.-M. Pharmacological Profiling of Orthochirus Scrobiculosus Toxin 1 Analogs with a Trimmed N-Terminal Domain. Mol. Pharmacol. 2006, 69, 354–362. [Google Scholar] [CrossRef] [Green Version]
- Restano-Cassulini, R.; Korolkova, Y.V.; Diochot, S.; Gurrola, G.; Guasti, L.; Possani, L.D.; Lazdunski, M.; Grishin, E.V.; Arcangeli, A.; Wanke, E. Species Diversity and Peptide Toxins Blocking Selectivity of Ether-a-Go-Go-Related Gene Subfamily K+ Channels in the Central Nervous System. Mol. Pharmacol. 2006, 69, 1673–1683. [Google Scholar] [CrossRef]
- Garcia, M.L.; Garcia-Calvo, M.; Hidalgo, P.; Lee, A.; MacKinnon, R. Purification and Characterization of Three Inhibitors of Voltage-Dependent K+ Channels from Leiurus Quinquestriatus Var. Hebraeus Venom. Biochemistry 1994, 33, 6834–6839. [Google Scholar] [CrossRef]
- Varga, Z.; Gurrola-Briones, G.; Papp, F.; Rodriguez De La Vega, R.C.; Pedraza-Alva, G.; Tajhya, R.B.; Gaspar, R.; Cardenas, L.; Rosenstein, Y.; Beeton, C.; et al. Vm24, a Natural Immunosuppressive Peptide, Potently and Selectively Blocks Kv1.3 Potassium Channels of Human T Cells. Mol. Pharmacol. 2012, 82, 372–382. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kuzmenkov, A.I.; Nekrasova, O.V.; Peigneur, S.; Tabakmakher, V.M.; Gigolaev, A.M.; Fradkov, A.F.; Kudryashova, K.S.; Chugunov, A.O.; Efremov, R.G.; Tytgat, J.; et al. Kv1.2 Channel-Specific Blocker from Mesobuthus Eupeus Scorpion Venom: Structural Basis of Selectivity. Neuropharmacology 2018, 143, 228–238. [Google Scholar] [CrossRef]
- Zhao, Y.; Chen, Z.; Cao, Z.; Li, W.; Wu, Y. Diverse Structural Features of Potassium Channels Characterized by Scorpion Toxins as Molecular Probes. Molecules 2019, 24, 2045. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ortiz, E.; Possani, L.D. Scorpion Toxins to Unravel the Conundrum of Ion Channel Structure and Functioning. Toxicon 2018, 150, 17–27. [Google Scholar] [CrossRef]
- Zundler, S.; Caioni, M.; Müller, M.; Strauch, U.; Kunst, C.; Woelfel, G. K+ Channel Inhibition Differentially Regulates Migration of Intestinal Epithelial Cells in Inflamed vs. Non-Inflamed Conditions in a PI3K/Akt-Mediated Manner. PLoS ONE 2016, 11, e0147736. [Google Scholar] [CrossRef] [Green Version]
- Gossen, D.; Gesquière, J.C.; Tastenoy, M.; De Neef, P.; Waelbroeck, M.; Christophe, J. Characterization and Regulation of the Expression of Scyllatoxin (Leiurotoxin I) Receptors in the Human Neuroblastoma Cell Line NB-OK 1. FEBS Lett. 1991, 285, 271–274. [Google Scholar] [CrossRef]
- Veytia-Bucheli, J.I.; Jiménez-Vargas, J.M.; Melchy-Pérez, E.I.; Sandoval-Hernández, M.A.; Possani, L.D.; Rosenstein, Y. Kv1.3 Channel Blockade with the Vm24 Scorpion Toxin Attenuates the CD4+ Effector Memory T Cell Response to TCR Stimulation. Cell Commun. Signal. 2018, 16, 45. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Nicolazzo, J.A.; Pan, Y.; Di Stefano, I.; Choy, K.H.C.; Reddiar, S.B.; Low, Y.L.; Wai, D.C.C.; Norton, R.S.; Jin, L. Blockade of Microglial Kv1.3 Potassium Channels by the Peptide HsTX1[R14A] Attenuates Lipopolysaccharide-Mediated Neuroinflammation. J. Pharm. Sci. 2022, 111, 638–647. [Google Scholar] [CrossRef]
- Banerjee, A.; Lee, A.; Campbell, E.; MacKinnon, R. Structure of a Pore-Blocking Toxin in Complex with a Eukaryotic Voltage-Dependent K+ Channel. Elife 2013, 2, e00594. [Google Scholar] [CrossRef]
- Qiu, S.; Yi, H.; Liu, H.; Cao, Z.; Wu, Y.; Li, W. Molecular Information of Charybdotoxin Blockade in the Large Conductance Calcium-Activated Potassium Channel. J. Chem. Inf. Model. 2009, 49, 1831–1838. [Google Scholar] [CrossRef]
- Koeberle, P.D.; Schlichter, L.C. Targeting KV Channels Rescues Retinal Ganglion Cells in Vivo Directly and by Reducing Inflammation. Channels 2010, 4, 337. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Menteyne, A.; Levavasseur, F.; Audinat, E.; Avignone, E. Predominant Functional Expression of Kv1.3 by Activated Microglia of the Hippocampus after Status Epilepticus. PLoS ONE 2009, 4, e6770. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Cañas, C.A.; Castaño-Valencia, S.; Castro-Herrera, F. Pharmacological Blockade of KV1.3 Channel as a Promising Treatment in Autoimmune Diseases. J. Transl. Autoimmun. 2022, 5, 100146. [Google Scholar] [CrossRef] [PubMed]
- Comes, N.; Bielanska, J.; Vallejo-Gracia, A.; Serrano-Albarrás, A.; Marruecos, L.; Gómez, D.; Soler, C.; Condom, E.; Ramón y Cajal, S.; Hernández-Losa, J.; et al. The Voltage-Dependent K(+) Channels Kv1.3 and Kv1.5 in Human Cancer. Front. Physiol. 2013, 4, 283. [Google Scholar] [CrossRef] [Green Version]
- Feske, S.; Wulff, H.; Skolnik, E.Y. Ion Channels in Innate and Adaptive Immunity. Annu. Rev. Immunol. 2015, 33, 291–353. [Google Scholar] [CrossRef] [Green Version]
- Kazama, I. Targeting Lymphocyte Kv1.3-Channels to Suppress Cytokine Storm in Severe COVID-19: Can It Be a Novel Therapeutic Strategy? Drug Discov. Ther. 2020, 14, 143–144. [Google Scholar] [CrossRef]
- Jen, J.C.; Wan, J. Episodic Ataxias. Handb. Clin. Neurol. 2018, 155, 205–215. [Google Scholar] [CrossRef]
- D’adamo, M.C.; Liantonio, A.; Rolland, J.F.; Pessia, M.; Imbrici, P. Kv1.1 Channelopathies: Pathophysiological Mechanisms and Therapeutic Approaches. Int. J. Mol. Sci. 2020, 21, 2935. [Google Scholar] [CrossRef]
- Serrano-Albarrás, A.; Cirera-Rocosa, S.; Sastre, D.; Estadella, I.; Felipe, A. Fighting Rheumatoid Arthritis: Kv1.3 as a Therapeutic Target. Biochem. Pharmacol. 2019, 165, 214–220. [Google Scholar] [CrossRef] [Green Version]
- Peter Muiruri, K.; Zhong, J.; Yao, B.; Lai, R.; Luo, L. Bioactive Peptides from Scorpion Venoms: Therapeutic Scaffolds and Pharmacological Tools. Chin. J. Nat. Med. 2023, 21, 19–35. [Google Scholar] [CrossRef]
- Bartok, A.; Fehér, K.; Bodor, A.; Rákosi, K.; Tóth, G.K.; Kövér, K.E.; Panyi, G.; Varga, Z. An Engineered Scorpion Toxin Analogue with Improved Kv1.3 Selectivity Displays Reduced Conformational Flexibility. Sci. Rep. 2015, 5, 18397. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Rashid, M.H.; Heinzelmann, G.; Huq, R.; Tajhya, R.B.; Chang, S.C.; Chhabra, S.; Pennington, M.W.; Beeton, C.; Norton, R.S.; Kuyucak, S. A Potent and Selective Peptide Blocker of the Kv1.3 Channel: Prediction from Free-Energy Simulations and Experimental Confirmation. PLoS ONE 2013, 8, e78712. [Google Scholar] [CrossRef] [PubMed]
- Varga, Z.; Tajti, G.; Panyi, G. The Kv1.3 K + Channel in the Immune System and Its “Precision Pharmacology” Using Peptide Toxins. Biol. Futur. 2021, 72, 75–83. [Google Scholar] [CrossRef] [PubMed]
- Takacs, Z.; Toups, M.; Kollewe, A.; Johnson, E.; Cuello, L.G.; Driessens, G.; Biancalana, M.; Koide, A.; Ponte, C.G.; Perozo, E.; et al. A Designer Ligand Specific for Kv1.3 Channels from a Scorpion Neurotoxin-Based Library. Proc. Natl. Acad. Sci. USA 2009, 106, 22211–22216. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kuzmenkov, A.I.; Vassilevski, A.A. Labelled Animal Toxins as Selective Molecular Markers of Ion Channels: Applications in Neurobiology and Beyond. Neurosci. Lett. 2018, 679, 15–23. [Google Scholar] [CrossRef] [PubMed]
- Koschak, A.; Koch, R.O.; Liu, J.; Kaczorowski, G.J.; Reinhart, P.H.; Garcia, M.L.; Knaus, H.G. [125I]Iberiotoxin-D19Y/Y36F, the First Selective, High Specific Activity Radioligand for High-Conductance Calcium-Activated Potassium Channels. Biochemistry 1997, 36, 1943–1952. [Google Scholar] [CrossRef]
- Koschak, A.; Bugianesi, R.M.; Mitterdorfer, J.; Kaczorowski, G.J.; Garcia, M.L.; Knaus, H.G. Subunit Composition of Brain Voltage-Gated Potassium Channels Determined by Hongotoxin-1, a Novel Peptide Derived from Centruroides Limbatus Venom. J. Biol. Chem. 1998, 273, 2639–2644. [Google Scholar] [CrossRef] [Green Version]
- Knaus, H.G.; Schwarzer, C.; Koch, R.O.A.; Eberhart, A.; Kaczorowski, G.J.; Glossmann, H.; Wunder, F.; Pongs, O.; Garcia, M.L.; Sperk, G. Distribution of High-Conductance Ca(2+)-Activated K+ Channels in Rat Brain: Targeting to Axons and Nerve Terminals. J. Neurosci. 1996, 16, 955–963. [Google Scholar] [CrossRef] [Green Version]
- Freudenthaler, G.; Axmann, M.; Schindler, H.; Pragl, B.; Knaus, H.G.; Schütz, G.J. Ultrasensitive Pharmacological Characterisation of the Voltage-Gated Potassium Channel K(V)1.3 Studied by Single-Molecule Fluorescence Microscopy. Histochem. Cell Biol. 2002, 117, 197–202. [Google Scholar] [CrossRef]
- Coyote-Maestas, W.; Nedrud, D.; Suma, A.; He, Y.; Matreyek, K.A.; Fowler, D.M.; Carnevale, V.; Myers, C.L.; Schmidt, D. Probing Ion Channel Functional Architecture and Domain Recombination Compatibility by Massively Parallel Domain Insertion Profiling. Nat. Commun. 2021, 12, 7114. [Google Scholar] [CrossRef]
- Wai, D.C.C.; Naseem, M.U.; Mocsár, G.; Babu Reddiar, S.; Pan, Y.; Csoti, A.; Hajdu, P.; Nowell, C.; Nicolazzo, J.A.; Panyi, G.; et al. A Fluorescent Peptide Toxin for Selective Visualization of the Voltage-Gated Potassium Channel Kv1.3. Bioconjug. Chem. 2022, 33, 2197–2212. [Google Scholar] [CrossRef]
- Orlov, N.A.; Ignatova, A.A.; Kryukova, E.V.; Yakimov, S.A.; Kirpichnikov, M.P.; Nekrasova, O.V.; Feofanov, A.V. Combining MKate2-Kv1.3 Channel and Atto488-Hongotoxin for the Studies of Peptide Pore Blockers on Living Eukaryotic Cells. Toxins 2022, 14, 858. [Google Scholar] [CrossRef]
- Denisova, K.R.; Orlov, N.A.; Yakimov, S.A.; Kirpichnikov, M.P.; Feofanov, A.V.; Nekrasova, O.V. Atto488-Agitoxin 2-A Fluorescent Ligand with Increased Selectivity for Kv1.3 Channel Binding Site. Bioengineering 2022, 9, 295. [Google Scholar] [CrossRef] [PubMed]
- Kudryashova, K.S.; Nekrasova, O.V.; Kuzmenkov, A.I.; Vassilevski, A.A.; Ignatova, A.A.; Korolkova, Y.V.; Grishin, E.V.; Kirpichnikov, M.P.; Feofanov, A.V. Fluorescent System Based on Bacterial Expression of Hybrid KcsA Channels Designed for Kv1.3 Ligand Screening and Study. Anal. Bioanal. Chem. 2013, 405, 2379–2389. [Google Scholar] [CrossRef] [PubMed]
- Denisova, K.R.; Orlov, N.A.; Yakimov, S.A.; Kryukova, E.A.; Dolgikh, D.A.; Kirpichnikov, M.P.; Feofanov, A.V.; Nekrasova, O.V. GFP-Margatoxin, a Genetically Encoded Fluorescent Ligand to Probe Affinity of Kv1.3 Channel Blockers. Int. J. Mol. Sci. 2022, 23, 1724. [Google Scholar] [CrossRef] [PubMed]
- Nekrasova, O.V.; Primak, A.L.; Ignatova, A.A.; Novoseletsky, V.N.; Geras’kina, O.V.; Kudryashova, K.S.; Yakimov, S.A.; Kirpichnikov, M.P.; Arseniev, A.S.; Feofanov, A.V. N-Terminal Tagging with GFP Enhances Selectivity of Agitoxin 2 to Kv1.3-Channel Binding Site. Toxins 2020, 12, 802. [Google Scholar] [CrossRef]
- Primak, A.L.; Skutel, M.A.; Nekrasova, O.V.; Arseniev, A.S.; Kirpichnikov, M.P.; Feofanov, A.V. Kv1 Potassium Channel Ligands Based on Hongotoxin 1 and Red Fluorescent Protein. Russ. J. Bioorg. Chem. 2020, 46, 1011–1017. [Google Scholar] [CrossRef]
- Merzlyak, E.M.; Goedhart, J.; Shcherbo, D.; Bulina, M.E.; Shcheglov, A.S.; Fradkov, A.F.; Gaintzeva, A.; Lukyanov, K.A.; Lukyanov, S.; Gadella, T.W.J.; et al. Bright Monomeric Red Fluorescent Protein with an Extended Fluorescence Lifetime. Nat. Methods 2007, 4, 555–557. [Google Scholar] [CrossRef]
- Zhang, G.; Gurtu, V.; Kain, S.R. An Enhanced Green Fluorescent Protein Allows Sensitive Detection of Gene Transfer in Mammalian Cells. Biochem. Biophys. Res. Commun. 1996, 227, 707–711. [Google Scholar] [CrossRef]
- Nekrasova, O.V.; Volyntseva, A.D.; Kudryashova, K.S.; Novoseletsky, V.N.; Lyapina, E.A.; Illarionova, A.V.; Yakimov, S.A.; Korolkova, Y.V.; Shaitan, K.V.; Kirpichnikov, M.P.; et al. Complexes of Peptide Blockers with Kv1.6 Pore Domain: Molecular Modeling and Studies with KcsA-Kv1.6 Channel. J. Neuroimmune Pharmacol. 2017, 12, 260–276. [Google Scholar] [CrossRef]
- Kuzmenkov, A.I.; Vassilevski, A.A.; Kudryashova, K.S.; Nekrasova, O.V.; Peigneur, S.; Tytgat, J.; Feofanov, A.V.; Kirpichnikov, M.P.; Grishin, E.V. Variability of Potassium Channel Blockers in Mesobuthus Eupeus Scorpion Venom with Focus on Kv1.1: An Integrated Transcriptomic and Proteomic Study. J. Biol. Chem. 2015, 290, 12195–12209. [Google Scholar] [CrossRef] [Green Version]
- Kudryashova, K.S.; Nekrasova, O.V.; Kirpichnikov, M.P.; Feofanov, A.V. Chimeras of KcsA and Kv1 as a Bioengineering Tool to Study Voltage-Gated Potassium Channels and Their Ligands. Biochem. Pharmacol. 2021, 190, 114646. [Google Scholar] [CrossRef] [PubMed]
- Anangi, R.; Koshy, S.; Huq, R.; Beeton, C.; Chuang, W.J.; King, G.F. Recombinant Expression of Margatoxin and Agitoxin-2 in Pichia Pastoris: An Efficient Method for Production of KV1.3 Channel Blockers. PLoS ONE 2012, 7, e52965. [Google Scholar] [CrossRef] [Green Version]
- Hao, J.; Padilla, F.; Dandonneau, M.; Lavebratt, C.; Lesage, F.; Noël, J.; Delmas, P. Kv1.1 Channels Act as Mechanical Brake in the Senses of Touch and Pain. Neuron 2013, 77, 899–914. [Google Scholar] [CrossRef] [Green Version]
- Pimentel, C.; M’Barek, S.; Visan, V.; Grissmer, S.; Sampieri, F.; Sabatier, J.-M.; Darbon, H.; Fajloun, Z. Chemical Synthesis and 1H-NMR 3D Structure Determination of AgTx2-MTX Chimera, a New Potential Blocker for Kv1.2 Channel, Derived from MTX and AgTx2 Scorpion Toxins. Protein Sci. 2007, 17, 107–118. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Attali, B.; Romey, G.; Honore, E.; Schmid-Alliana, A.; Mattei, M.G.; Lesage, F.; Ricard, P.; Barhanin, J.; Lazdunski, M. Cloning, Functional Expression, and Regulation of Two K+ Channels in Human T Lymphocytes. J. Biol. Chem. 1992, 267, 8650–8657. [Google Scholar] [CrossRef]
- Hu, L.; Pennington, M.; Jiang, Q.; Whartenby, K.A.; Calabresi, P.A. Characterization of the Functional Properties of the Voltage-Gated Potassium Channel Kv1.3 in Human CD4+ T Lymphocytes. J. Immunol. 2007, 179, 4563–4570. [Google Scholar] [CrossRef] [Green Version]
- Fomina, A.F.; Nguyen, H.M.; Wulff, H. Kv1.3 Inhibition Attenuates Neuroinflammation through Disruption of Microglial Calcium Signaling. Channels 2021, 15, 67–78. [Google Scholar] [CrossRef] [PubMed]
- Teisseyre, A.; Palko-Labuz, A.; Sroda-Pomianek, K.; Michalak, K. Voltage-Gated Potassium Channel Kv1.3 as a Target in Therapy of Cancer. Front. Oncol. 2019, 9, 933. [Google Scholar] [CrossRef] [PubMed]
- Pérez-Verdaguer, M.; Capera, J.; Serrano-Novillo, C.; Estadella, I.; Sastre, D.; Felipe, A. The Voltage-Gated Potassium Channel Kv1.3 Is a Promising Multitherapeutic Target against Human Pathologies. Expert. Opin. Ther. Targets 2016, 20, 577–591. [Google Scholar] [CrossRef]
- Paulhus, K.; Ammerman, L.; Glasscock, E. Clinical Spectrum of KCNA1 Mutations: New Insights into Episodic Ataxia and Epilepsy Comorbidity. Int. J. Mol. Sci. 2020, 21, 2802. [Google Scholar] [CrossRef] [PubMed]
- Robbins, C.A.; Tempel, B.L. Kv1.1 and Kv1.2: Similar Channels, Different Seizure Models. Epilepsia 2012, 53 (Suppl. S1), 134–141. [Google Scholar] [CrossRef] [PubMed]
- Al-Sabi, A.; Daly, D.; Hoefer, P.; Kinsella, G.K.; Metais, C.; Pickering, M.; Herron, C.; Kaza, S.K.; Nolan, K.; Dolly, J.O. A Rational Design of a Selective Inhibitor for Kv1.1 Channels Prevalent in Demyelinated Nerves That Improves Their Impaired Axonal Conduction. J. Med. Chem. 2017, 60, 2245–2256. [Google Scholar] [CrossRef] [PubMed]
- Otuyemi, B.; Jackson, T.; Ma, R.; Monteiro, A.R.; Seifi, M.; Swinny, J.D. Domain and Cell Type-Specific Immunolocalisation of Voltage-Gated Potassium Channels in the Mouse Striatum. J. Chem. Neuroanat. 2023, 128, 102233. [Google Scholar] [CrossRef]
- Bindels, D.S.; Haarbosch, L.; van Weeren, L.; Postma, M.; Wiese, K.E.; Mastop, M.; Aumonier, S.; Gotthard, G.; Royant, A.; Hink, M.A.; et al. MScarlet: A Bright Monomeric Red Fluorescent Protein for Cellular Imaging. Nat. Methods 2017, 14, 53–56. [Google Scholar] [CrossRef] [PubMed]
- Zarowny, L.; Clavel, D.; Johannson, R.; Duarte, K.; Depernet, H.; Dupuy, J.; Baker, H.; Brown, A.; Royant, A.; Campbell, R.E. Cyan Fluorescent Proteins Derived from MNeonGreen. Protein Eng. Des. Sel. 2022, 35, gzac004. [Google Scholar] [CrossRef]
- Hirano, M.; Ando, R.; Shimozono, S.; Sugiyama, M.; Takeda, N.; Kurokawa, H.; Deguchi, R.; Endo, K.; Haga, K.; Takai-Todaka, R.; et al. A Highly Photostable and Bright Green Fluorescent Protein. Nat. Biotechnol. 2022, 40, 1132–1142. [Google Scholar] [CrossRef]
- Nekrasova, O.; Kudryashova, K.; Fradkov, A.; Yakimov, S.; Savelieva, M.; Kirpichnikov, M.; Feofanov, A. Straightforward Approach to Produce Recombinant Scorpion Toxins—Pore Blockers of Potassium Channels. J. Biotechnol. 2017, 241, 127–135. [Google Scholar] [CrossRef]
- Koren, G.; Liman, E.R.; Logothetis, D.E.; Nadal-Ginard, B.; Hess, P. Gating Mechanism of a Cloned Potassium Channel Expressed in Frog Oocytes and Mammalian Cells. Neuron 1990, 4, 39–51. [Google Scholar] [CrossRef]
- Peigneur, S.; Cheneval, O.; Maiti, M.; Leipold, E.; Heinemann, S.H.; Lescrinier, E.; Herdewijn, P.; de Lima, M.E.; Craik, D.J.; Schroeder, C.I.; et al. Where Cone Snails and Spiders Meet: Design of Small Cyclic Sodium-Channel Inhibitors. FASEB J. 2019, 33, 3693–3703. [Google Scholar] [CrossRef]
- Vriens, K.; Peigneur, S.; de Coninck, B.; Tytgat, J.; Cammue, B.P.A.; Thevissen, K. The Antifungal Plant Defensin AtPDF2.3 from Arabidopsis Thaliana Blocks Potassium Channels. Sci. Rep. 2016, 6, 32121. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Peigneur, S.; Billen, B.; Derua, R.; Waelkens, E.; Debaveye, S.; Béress, L.; Tytgat, J. A Bifunctional Sea Anemone Peptide with Kunitz Type Protease and Potassium Channel Inhibiting Properties. Biochem. Pharmacol. 2011, 82, 81–90. [Google Scholar] [CrossRef] [PubMed] [Green Version]




| pH | KcsA-Kv1.1 | KcsA-Kv1.3 | KcsA-Kv1.6 |
|---|---|---|---|
| 7.0 | 23 ± 4 | 0.14 ± 0.02 | 0.60 ± 0.06 |
| 7.5 | 15 ± 3 * | 0.10 ± 0.01 * | 0.30 ± 0.08 * |
| 8.0 | 12 ± 2 * | 0.25 ± 0.02 * | 1.3 ± 0.4 * |
| Toxin | KcsA-Kv1.1 | KcsA-Kv1.3 | KcsA-Kv1.6 |
|---|---|---|---|
| AgTx2 | 3.7 ± 0.8 | 0.039 ± 0.007 | 0.027 ± 0.004 |
| KTx1 | 3.8 ± 0.8 | 0.032 ± 0.006 | 0.016 ± 0.002 |
| Ligand | Kv1.1 | Kv1.2 | Kv1.3 | Kv1.6 |
|---|---|---|---|---|
| AgTx2-GFP | 3.5 ± 0.5 1 | - 2 | 2.31 ± 0.22 | 1.30 ± 0.15 |
| AgTx2 | 4.2 ± 0.3 | 34 ± 6 | 0.17 ± 0.03 | 0.60 ± 0.11 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Primak, A.L.; Orlov, N.A.; Peigneur, S.; Tytgat, J.; Ignatova, A.A.; Denisova, K.R.; Yakimov, S.A.; Kirpichnikov, M.P.; Nekrasova, O.V.; Feofanov, A.V. AgTx2-GFP, Fluorescent Blocker Targeting Pharmacologically Important Kv1.x (x = 1, 3, 6) Channels. Toxins 2023, 15, 229. https://doi.org/10.3390/toxins15030229
Primak AL, Orlov NA, Peigneur S, Tytgat J, Ignatova AA, Denisova KR, Yakimov SA, Kirpichnikov MP, Nekrasova OV, Feofanov AV. AgTx2-GFP, Fluorescent Blocker Targeting Pharmacologically Important Kv1.x (x = 1, 3, 6) Channels. Toxins. 2023; 15(3):229. https://doi.org/10.3390/toxins15030229
Chicago/Turabian StylePrimak, Alexandra L., Nikita A. Orlov, Steve Peigneur, Jan Tytgat, Anastasia A. Ignatova, Kristina R. Denisova, Sergey A. Yakimov, Mikhail P. Kirpichnikov, Oksana V. Nekrasova, and Alexey V. Feofanov. 2023. "AgTx2-GFP, Fluorescent Blocker Targeting Pharmacologically Important Kv1.x (x = 1, 3, 6) Channels" Toxins 15, no. 3: 229. https://doi.org/10.3390/toxins15030229







