Molecular Phylogeny, Morphology, Growth and Toxicity of Three Benthic Dinoflagellates Ostreopsis sp. 9, Prorocentrum lima and Coolia monotis Developing in Strait of Gibraltar, Southwestern Mediterranean
Abstract
:1. Introduction
2. Results
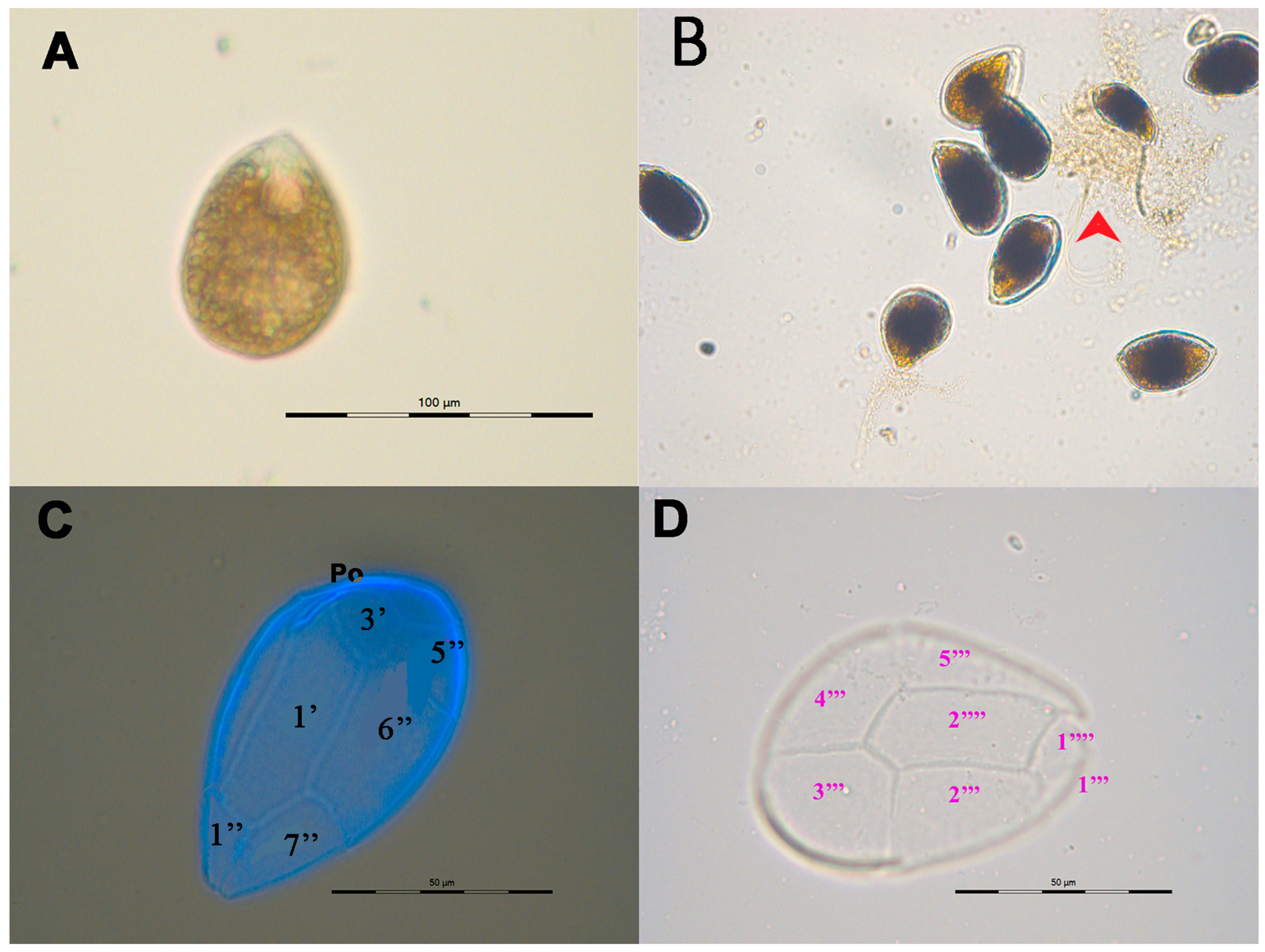
2.1. Culture Characteristics and Morphology
2.2. Molecular Analysis and Phylogeny
2.3. Toxins
2.4. Growth Characteristics
2.5. Effect of Temperature on Toxin Content of Prorocentrum lima
2.6. Effect of Nitrogen Source on Toxin Content of Prorocentrum lima
3. Discussion
3.1. Ostreopsis sp. 9
3.2. Prorocentrum lima
3.2.1. Distribution and Growth Characteristics
3.2.2. Toxin Content
3.2.3. Effect of Temperature on Toxin Content of P. lima
3.2.4. Effect of Nitrogen Source on LST Content of P. lima
3.3. Coolia monotis
4. Conclusions
5. Material and Methods
5.1. Sampling Sites
5.2. Isolation and Culture Conditions
5.3. Morphology and Identification
5.4. Molecular Analysis and Phylogeny
5.4.1. DNA Extraction and PCR
5.4.2. Amplification and Sequencing
5.4.3. Phylogeny
5.5. Toxin Analysis
5.5.1. LC–MS/MS Analysis of Lipophilic Toxins Produced by Strain PLCM17
5.5.2. LC–MS/MS Analysis of Toxins Produced by Strain OSCM17
5.5.3. LC–MS/MS Analysis of Compounds Produced by Strain CMON15
5.6. Growth Characteristics
5.7. Effect of Temperature and Nitrogen Sources (Ammonia and Nitrate) on Toxin Content of Prorocentrum lima
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Anderson, D.M.; Hoagland, P.; Kaoru, Y.; White, A.W. Estimated Annual Economic Impacts from Harmful Algal Blooms (HABs) in the United States; National Oceanic and Atmospheric Administration: Washington, DC, USA, 2000. [Google Scholar]
- Hoagland, P.; Anderson, D.M.; Kaoru, Y.; White, A.W. The Economic Effects of Harmful Algal Blooms in the United States: Estimates, Assessment Issues, and Information Needs. Estuaries 2002, 25, 819–837. [Google Scholar] [CrossRef]
- Kim, H. An Overview on the Occurrences of Harmful Algal Blooms (HABs) and Migration Strategies in Korean Coastal Waters; Terrapub and Nagasaki University: Busan, Republic of Korea, 2010. [Google Scholar]
- Gorbi, S.; Bocchetti, R.; Binelli, A.; Bacchiocchi, S.; Orletti, R.; Nanetti, L.; Raffaelli, F.; Vignini, A.; Accoroni, S.; Totti, C.; et al. Biological Effects of Palytoxin-like Compounds from Ostreopsis cf. ovata: A Multibiomarkers Approach with Mussels Mytilus galloprovincialis. Chemosphere 2012, 89, 623–632. [Google Scholar] [CrossRef]
- Park, T.G.; Lim, W.A.; Park, Y.T.; Lee, C.K.; Jeong, H.J. Economic Impact, Management and Mitigation of Red Tides in Korea. Harmful Algae 2013, 30, S131–S143. [Google Scholar] [CrossRef]
- Momigliano, P.; Sparrow, L.; Blair, D.; Heimann, K. The Diversity of Coolia spp. (Dinophyceae Ostreopsidaceae) in the Central Great Barrier Reef Region. PLoS One 2013, 8, 1–11. [Google Scholar] [CrossRef]
- Berdalet, E.; Fleming, L.E.; Gowen, R.; Davidson, K.; Hess, P.; Backer, L.C.; Moore, S.K.; Hoagland, P.; Enevoldsen, H. Marine Harmful Algal Blooms, Human Health and Wellbeing: Challenges and Opportunities in the 21st Century. J. Mar. Biol. Assoc. United Kingd. 2016, 96, 61–91. [Google Scholar] [CrossRef] [PubMed]
- Ben-Gharbia, H.; Yahia, O.K.D.; Amzil, Z.; Chomérat, N.; Abadie, E.; Masseret, E.; Sibat, M.; Triki, H.Z.; Nouri, H.; Laabir, M. Toxicity and Growth Assessments of Three Thermophilic Benthic Dinoflagellates (Ostreopsis cf. ovata, Prorocentrum lima and Coolia monotis) Developing in the Southern Mediterranean Basin. Toxins 2016, 8, 297. [Google Scholar] [CrossRef] [PubMed]
- Berdalet, E.; Pavaux, A.S.; Abós-Herràndiz, R.; Travers, M.; Appéré, G.; Vila, M.; Thomas, J.; de Haro, L.; Estrada, M.; Medina-Pérez, N.; et al. Environmental, Human Health and Socioeconomic Impacts of Ostreopsis spp. Blooms in the NW Mediterranean. Harmful Algae 2022, 119, 102320. [Google Scholar] [CrossRef] [PubMed]
- Accoroni, S.; Romagnoli, T.; Colombo, F.; Pennesi, C.; di Camillo, C.G.; Marini, M.; Battocchi, C.; Ciminiello, P.; Dell’Aversano, C.; Dello Iacovo, E.; et al. Ostreopsis cf. ovata Bloom in the Northern Adriatic Sea during Summer 2009: Ecology, Molecular Characterization and Toxin Profile. Mar. Pollut. Bull. 2011, 62, 2512–2519. [Google Scholar] [CrossRef]
- Hallegaef, G.M. Phycological Reviews 13 A Review of Harmful Algal Blooms and Their Apparent Global Increase. Phycologia 1993, 32, 79–99. [Google Scholar] [CrossRef]
- Masó, M.; Garcés, E. Harmful Microalgae Blooms (HAB); Problematic and Conditions That Induce Them. Mar. Pollut. Bull. 2006, 53, 620–630. [Google Scholar] [CrossRef]
- Aligizaki, K.; Nikolaidis, G. Morphological Identification of Two Tropical Dinoflagellates of the Genera Gambierdiscus and Sinophysis in the Mediterranean Sea. J. Biol. Res. 2008, 9, 75–82. [Google Scholar]
- Shears, N.T.; Ross, P.M. Blooms of Benthic Dinoflagellates of the Genus Ostreopsis; an Increasing and Ecologically Important Phenomenon on Temperate Reefs in New Zealand and Worldwide. Harmful Algae 2009, 8, 916–925. [Google Scholar] [CrossRef]
- Aligizaki, K.; Nikolaidis, G.; Katikou, P.; Baxevanis, A.D.; Abatzopoulos, T.J. Potentially Toxic Epiphytic Prorocentrum (Dinophyceae) Species in Greek Coastal Waters. Harmful Algae 2009, 8, 299–311. [Google Scholar] [CrossRef]
- Rhodes, L. World-Wide Occurrence of the Toxic Dinoflagellate Genus Ostreopsis Schmidt. Toxicon 2011, 57, 400–407. [Google Scholar] [CrossRef] [PubMed]
- Monserrat, M.; Catania, D.; Asnaghi, V.; Chiantore, M.; Lemée, R.; Mangialajo, L. The Role of Habitat in the Facilitation of Ostreopsis spp. Blooms. Harmful Algae 2022, 113, 102199. [Google Scholar] [CrossRef] [PubMed]
- Granéli, E.; Vidyarathna, N.K.; Funari, E.; Cumaranatunga, P.R.T.; Scenati, R. Can Increases in Temperature Stimulate Blooms of the Toxic Benthic Dinoflagellate Ostreopsis ovata? Harmful Algae 2011, 10, 165–172. [Google Scholar] [CrossRef]
- Zingone, A.; Escalera, L.; Aligizaki, K.; Fernández-Tejedor, M.; Ismael, A.; Montresor, M.; Mozetič, P.; Taş, S.; Totti, C. Toxic Marine Microalgae and Noxious Blooms in the Mediterranean Sea: A Contribution to the Global HAB Status Report. Harmful Algae 2021, 102, 101843. [Google Scholar] [CrossRef] [PubMed]
- Vila, M.; Camp, J.; Garcés, E.; Masó, M.; Delgado, M. High Resolution Spatio-Temporal Detection of Potentially Harmful Dinoflagellates in Confined Waters of the NW Mediterranean. J. Plankton Res. 2001, 23, 497–514. [Google Scholar] [CrossRef]
- Turki, S. Distribution of Toxic Dinoflagellates along the Leaves of Seagrass Posidonia oceanica and Cymodocea nodosa from the Gulf of Tunis. Cah. Biol. Mar 2005, 46, 29–34. [Google Scholar]
- Turki, S.; Harzallah, A.; Sammari, C. Occurrence of Harmful Dinoflagellates in Two Different Tunisian Ecosystems: The Lake of Bizerte and the Gulf of Gabès. Cah. Biol. Mar 2006, 47, 253–259. [Google Scholar]
- Mangialajo, L.; Ganzin, N.; Accoroni, S.; Asnaghi, V.; Blanfuné, A.; Cabrini, M.; Cattaneo-Vietti, R.; Chavanon, F.; Chiantore, M.; Cohu, S.; et al. Trends in Ostreopsis Proliferation along the Northern Mediterranean Coasts. Toxicon 2011, 57, 408–420. [Google Scholar] [CrossRef] [PubMed]
- Cohu, S.; Thibaut, T.; Mangialajo, L.; Labat, J.P.; Passafiume, O.; Blanfuné, A.; Simon, N.; Cottalorda, J.M.; Lemée, R. Occurrence of the Toxic Dinoflagellate Ostreopsis cf. ovata in Relation with Environmental Factors in Monaco (NW Mediterranean). Mar. Pollut. Bull. 2011, 62, 2681–2691. [Google Scholar] [CrossRef] [PubMed]
- Cohu, S.; Mangialajo, L.; Thibaut, T.; Blanfuné, A.; Marro, S.; Lemée, R. Proliferation of the Toxic Dinoflagellate Ostreopsis cf. ovata in Relation to Depth, Biotic Substrate and Environmental Factors in the North West Mediterranean Sea. Harmful Algae 2013, 24, 32–44. [Google Scholar] [CrossRef]
- Illoul, H.; Rodríguez, F.; Vila, M.; Adjas, N.; Younes, A.A.; Bournissa, M.; Koroghli, A.; Marouf, N.; Rabia, S.; Ameur, F.L.K. The Genus Ostreopsis along the Algerian Coastal Waters (SW Mediterranean Sea) Associated with a Human Respiratory Intoxication Episode. Cryptogam. Algol. 2012, 33, 209–216. [Google Scholar] [CrossRef]
- Amzil, Z.; Sibat, M.; Chomerat, N.; Grossel, H.; Marco-Miralles, F.; Lemee, R.; Nezan, E.; Sechet, V. Ovatoxin-a and Palytoxin Accumulation in Seafood in Relation to Ostreopsis cf. ovata Blooms on the French Mediterranean Coast. Mar. Drugs 2012, 10, 477–496. [Google Scholar] [CrossRef] [PubMed]
- Ismael, A.A. First Record of Coolia monotis Meunier along Alexandria Coast - Egypt. Egypt. J. Aquat. Res. 2014, 40, 19–25. [Google Scholar] [CrossRef]
- Blanfuné, A.; Boudouresque, C.F.; Grossel, H.; Thibaut, T. Distribution and Abundance of Ostreopsis spp. and Associated Species (Dinophyceae) in the Northwestern Mediterranean: The Region and the Macroalgal Substrate Matter. Environ. Sci. Pollut. Res. 2015, 22, 12332–12346. [Google Scholar] [CrossRef]
- Ben-Gharbia, H.; Laabir, M.; Ben Mhamed, A.; Gueroun, S.K.M.; Daly Yahia, M.N.; Nouri, H.; M’Rabet, C.; Shili, A.; Kéfi-Daly Yahia, O. Occurrence of Epibenthic Dinoflagellates in Relation to Biotic Substrates and to Environmental Factors in Southern Mediterranean (Bizerte Bay and Lagoon, Tunisia): An Emphasis on the Harmful Ostreopsis spp., Prorocentrum lima and Coolia monotis. Harmful Algae 2019, 90, 101704. [Google Scholar] [CrossRef]
- Moncer, M.; Hamza, A.; Feki-Sahnoun, W.; Mabrouk, L.; Hassen, M.B. Variability Patterns of Epibenthic Microalgae in Eastern Tunisian Coasts; Patrones de Variabilidad de Las Microalgas Epibentónicas En La Costa Este Tunecina. Sci. Mar. Sci. Mar. Barc. SCI. MAR 2017, 81, 487–498. [Google Scholar]
- Hachani, M.A.; Dhib, A.; Fathalli, A.; Ziadi, B.; Turki, S.; Aleya, L. Harmful Epiphytic Dinoflagellate Assemblages on Macrophytes in the Gulf of Tunis. Harmful Algae 2018, 77, 29–42. [Google Scholar] [CrossRef]
- de Virgilio, M.; Cifarelli, S.; Garofoli, G.; Lamberti, G.; Massari, V.; Degryse, B. Citizen Science in the Monitoring of Ostreopsis ovata Blooms in Southern Italy: A Five-Year Study. Mar. Pollut. Bull. 2021, 173, 112981. [Google Scholar] [CrossRef] [PubMed]
- Ibghi, M.; Elkbiach, M.L.; Rijal Leblad, B.; Aboualaalaa, H.; Hervé, F.; Sibat, M.; Masseret, E.; Chair, A.; Maamour, N.; Daoudi, M.; et al. Occurrence of Three Dominant Epibenthic Dinoflagellates (Ostreopsis spp., Coolia monotis and Prorocentrum lima ) in Relation to Biotic Substrates and Environmental Factors in a Highly Dynamic Ecosystem, the Strait of Gibraltar (Southwestern Mediterran). Environ. Monit. Assess. 2022, 194, 810. [Google Scholar] [CrossRef] [PubMed]
- Penna, A.; Vila, M.; Fraga, S.; Giacobbe, M.G.; Francesco, A.; Riobó, P.; Vernesi, C. Characterization of Ostreopsis and Coolia (Dinophyceae) Isolates in the Western Mediterranean Sea Based on Morphology, Toxicity and Internal Transcribed Spacer 5.8s RDNA Sequences. J. Phycol. 2005, 41, 212–225. [Google Scholar] [CrossRef]
- Pfannkuchen, M.; Godrijan, J.; Marić Pfannkuchen, D.; Iveša, L.; Kružić, P.; Ciminiello, P.; Dell’Aversano, C.; Dello Iacovo, E.; Fattorusso, E.; Forino, M.; et al. Toxin-Producing Ostreopsis cf. ovata Are Likely to Bloom Undetected along Coastal Areas. Environ. Sci. Technol. 2012, 46, 5574–5582. [Google Scholar] [CrossRef]
- Accoroni, S.; Totti, C. The Toxic Benthic Dinoflagellates of the Genus Ostreopsis in Temperate Areas: A Review. Adv. Oceanogr. Limnol. 2016, 7, 1–15. [Google Scholar] [CrossRef]
- Açaf, L.; Abboud-Abi Saab, M.; Khoury-Hanna, M.; Lemée, R. Bloom Dynamics of the Newly Described Toxic Benthic Dinoflagellate Ostreopsis fattorussoi along the Lebanese Coast (Eastern Mediterranean). Reg. Stud. Mar. Sci. 2020, 38, 101338. [Google Scholar] [CrossRef]
- Riobó, P.; Paz, B.; Franco, J.M. Analysis of Palytoxin-like in Ostreopsis Cultures by Liquid Chromatography with Precolumn Derivatization and Fluorescence Detection. Anal. Chim. Acta 2006, 566, 217–223. [Google Scholar] [CrossRef]
- Durando, P.; Ansaldi, F.; Oreste, P.; Moscatelli, P.; Marensi, L.; Grillo, C.; Gasparini, R.; Icardi, G.; Collaborative Group for the Ligurian Syndromic Algal Surveillance. Ostreopsis ovata and Human Health: Epidemiological and Clinical Features of Respiratory Syndrome Outbreaks from a Two-Year Syndromic Surveillance, 2005-06, in North-West Italy. Euro Surveill. 2007, 12, 3212. [Google Scholar] [CrossRef]
- Ciminiello, P.; Dell’Aversano, C.; Fattorusso, E.; Forino, M.; Tartaglione, L.; Grillo, C.; Melchiorre, N. Putative Palytoxin and Its New Analogue, Ovatoxin-a, in Ostreopsis ovata Collected Along the Ligurian Coasts During the 2006 Toxic Outbreak. J. Am. Soc. Mass Spectrom. 2008, 19, 111–120. [Google Scholar] [CrossRef]
- Tichadou, L.; Glaizal, M.; Armengaud, A.; Grossel, H.; Lemée, R.; Kantin, R.; Lasalle, J.L.; Drouet, G.; Rambaud, L.; Malfait, P.; et al. Health Impact of Unicellular Algae of the Ostreopsis Genus Blooms in the Mediterranean Sea: Experience of the French Mediterranean Coast Surveillance Network from 2006 to 2009. Clin. Toxicol. 2010, 48, 839–844. [Google Scholar] [CrossRef]
- Chomérat, N.; Antajan, E.; Auby, I.; Bilien, G.; Carpentier, L.; de Casamajor, M.N.; Ganthy, F.; Hervé, F.; Labadie, M.; Méteigner, C.; et al. First Characterization of Ostreopsis cf. ovata (Dinophyceae) and Detection of Ovatoxins during a Multispecific and Toxic Ostreopsis Bloom on French Atlantic Coast. Mar. Drugs 2022, 20, 461. [Google Scholar] [CrossRef] [PubMed]
- Fernández-Zabala, J.; Amorim, A.; Tuya, F.; Herrera, R.; Soler-Onís, E. Playing Hide and Seek: Distribution with Depth of Potentially Harmful Epibenthic Dinoflagellates of Southern EL HIERRO Island, Canary Islands (Ne Atlantic). Harmful Algae 2022, 117, 102271. [Google Scholar] [CrossRef] [PubMed]
- Nguyen-Ngoc, L.; Doan-Nhu, H.; Phan-Tan, L.; Huynh-Thi, D.N.; Nguyen-Tam, V.; Pham, A.H.; Tran-Thi, H.M.; Le Tran-Thi, V.; Tester, P. Seasonal Occurrence of the Potentially Toxic Benthic Armoured Dinoflagellates in Nha Trang Bay, South Central Coast of Viet Nam. Reg. Stud. Mar. Sci. 2022, 55, 102627. [Google Scholar] [CrossRef]
- Chomérat, N.; Bilien, G.; Viallon, J.; Hervé, F.; Réveillon, D.; Henry, K.; Zubia, M.; Vieira, C.; Ung, A.; Gatti, C.M.I.; et al. Taxonomy and Toxicity of a Bloom-Forming Ostreopsis Species (Dinophyceae, Gonyaulacales) in Tahiti Island (South Pacific Ocean): One Step Further towards Resolving the Identity of O. siamensis. Harmful Algae 2020, 98, 101888. [Google Scholar] [CrossRef] [PubMed]
- Fukuyo, Y. Taxonomical Study on Benthic Dinoflagellates Collected in Coral Reefs. Bull. Jap. Soc. Sci. Ficheries 1981, 47, 967–978. [Google Scholar] [CrossRef]
- Faust, M.A.; Morton, S.L.; Quod, J.P. Further Sem Study of Marine Dinoflagellates: The Genus Ostreopsis (Dinophyceae). J. Phycol. 1996, 32, 1053–1065. [Google Scholar] [CrossRef]
- Boisnoir, A.; Bilien, G.; Lemée, R.; Chomérat, N. First Insights on the Diversity of the Genus Ostreopsis (Dinophyceae, Gonyaulacales) in Guadeloupe Island, with Emphasis on the Phylogenetic Position of O. heptagona. Eur. J. Protistol. 2022, 83, 125875. [Google Scholar] [CrossRef] [PubMed]
- Amorim, A.; Veloso, V.; Penna, A. First Detection of Ostreopsis cf. siamensis in Portuguese Coastal Waters. Harmful Algae News 2010, 42, 6–7. [Google Scholar]
- David, H.; Laza-Martínez, A.; Miguel, I.; Orive, E. Ostreopsis cf. siamensis and Ostreopsis cf. ovata from the Atlantic Iberian Peninsula: Morphological and Phylogenetic Characterization. Harmful Algae 2013, 30, 44–55. [Google Scholar] [CrossRef]
- Aligizaki, K.; Nikolaidis, G. The Presence of the Potentially Toxic Genera Ostreopsis and Coolia (Dinophyceae) in the North Aegean Sea, Greece. Harmful Algae 2006, 5, 717–730. [Google Scholar] [CrossRef]
- Alkhatib, H.; Bennouna, A.; Abouabdellah, R.; Amzil, Z.; Herve, F.; Moussavou-Mouity, C.A.; Elayoubi, S.; Bekkali, M.; Fahde, A. Ostreopsis spp.: Morphology, Proliferation and Toxic Profile in the North-West of Agadir (North Atlantic Ocean). Reg. Stud. Mar. Sci. 2022, 56, 102713. [Google Scholar] [CrossRef]
- Bennouna, A.; EL Attar, J.; Abouabdellah, R.; Chafik, A.; Penna, A.; Oliveira, P.B.; Palma, S.; Moita, M.T. Ostreopsis cf. siamensis Blooms in Moroccan Atlantic Upwelling Waters (2004–2009). In Proceedings of the 14th International Conference on Harmful Algae, Hersonissos-Crete, Greece, 1–5 November 2013; International Society for the Study of Harmful Algae and Intergovernmental Oceanographic Commission of UNESCO: Athens, Greece, 2012; pp. 19–22. [Google Scholar]
- Chang, F.H.; Shimizu, Y.; Hay, B.; Stewart, R.; Mackay, G.; Tasker, R. Three Recently Recorded Ostreopsis spp. (Dinophyceae) in New Zealand: Temporal and Regional Distribution in the Upper North Island from 1995 to 1997. N. Z. J. Mar. Freshw. Res. 2000, 34, 29–39. [Google Scholar] [CrossRef]
- Rhodes, L.; Adamson, J.; Suzuki, T.; Briggs, L.; Garthwaite, I. Toxic Marine Epiphytic Dinoflagellates, Ostreopsis siamensis and Coolia monotis (Dinophyceae), in New Zealand. N. Z. J. Mar. Freshw. Res. 2000, 34, 371–383. [Google Scholar] [CrossRef]
- Laza-Martinez, A.; Orive, E.; Miguel, I. Morphological and Genetic Characterization of Benthic Dinoflagellates of the Genera Coolia, Ostreopsis and Prorocentrum from the South-Eastern Bay of Biscay. Eur. J. Phycol. 2011, 46, 45–65. [Google Scholar] [CrossRef]
- Saab, M.A.A.; Fakhri, M.; Kassab, M.T.; Matar, N. Seasonal and Spatial Variations of the Dinoflagellate Ostreopsis siamensis in the Lebanese Coastal Waters (Eastern Mediterranean). Cryptogam. Algol. 2013, 34, 57–67. [Google Scholar] [CrossRef]
- Mabrouk, L.; Hamza, A.; Mahfoudi, M.; Bradai, M.N.N. Spatial and Temporal Variations of Epiphytic Ostreopsis siamensis on Posidonia oceanica (L.) Delile Leaves in Mahdia (Tunisia). CBM-Cah. Biol. Mar. 2012, 53, 419–427. [Google Scholar]
- Ukena, T.; Satake, M.; Usami, M.; Oshima, Y.; Naoki, H.; Fujita, T.; Kan, Y.; Yasumoto, T. Structure Elucidation of Ostreocin D, a Palytoxin Analog Isolated from the Dinoflagellate Ostreopsis siamensis. Biosci. Biotechnol. Biochem. 2001, 65, 2585–2588. [Google Scholar] [CrossRef] [PubMed]
- Verma, A.; Hoppenrath, M.; Harwood, T.; Brett, S.; Rhodes, L.; Murray, S. Molecular Phylogeny, Morphology and Toxigenicity of Ostreopsis cf. siamensis (Dinophyceae) from Temperate South-East Australia. Phycol. Res. 2016, 64, 146–159. [Google Scholar] [CrossRef]
- Ciminiello, P.; Dell’Aversano, C.; Dello Iacovo, E.; Fattorusso, E.; Forino, M.; Tartaglione, L.; Yasumoto, T.; Battocchi, C.; Giacobbe, M.; Amorim, A.; et al. Investigation of Toxin Profile of Mediterranean and Atlantic Strains of Ostreopsis cf. siamensis (Dinophyceae) by Liquid Chromatography-High Resolution Mass Spectrometry. Harmful Algae 2013, 23, 19–27. [Google Scholar] [CrossRef]
- Guerrini, F.; Pezzolesi, L.; Feller, A.; Riccardi, M.; Ciminiello, P.; Dell’Aversano, C.; Tartaglione, L.; Dello Iacovo, E.; Fattorusso, E.; Forino, M.; et al. Comparative Growth and Toxin Profile of Cultured Ostreopsis ovata from the Tyrrhenian and Adriatic Seas. Toxicon 2010, 55, 211–220. [Google Scholar] [CrossRef]
- Paz, B.; Riobó, P.; Franco, J.M. Preliminary Study for Rapid Determination of Phycotoxins in Microalgae Whole Cells Using Matrix-Assisted Laser Desorption/Ionization Time-of-Flight Mass Spectrometry. Rapid Commun. Mass Spectrom. 2011, 25, 3627–3639. [Google Scholar] [CrossRef] [PubMed]
- Accoroni, S.; Glibert, P.M.; Pichierri, S.; Romagnoli, T.; Marini, M.; Totti, C. A Conceptual Model of Annual Ostreopsis cf. ovata Blooms in the Northern Adriatic Sea Based on the Synergic Effects of Hydrodynamics, Temperature, and the N:P Ratio of Water Column Nutrients. Harmful Algae 2015, 45, 14–25. [Google Scholar] [CrossRef]
- Murakami, Y.; Oshima, Y.; Yasumoto, T. Identification of Okadaic Acid As a Toxic Component of a Marine Dinoflagellate Prorocentrum lima. Nippon. Suisan Gakkaishi Jpn. Ed. 1982, 48, 69–72. [Google Scholar] [CrossRef]
- Bouacha, N.; Chézeau, A.; Turquet, J.; Quod, J.P.; Puiseux-Dao, S. Morphological and Toxicological Variability of Prorocentrum lima Clones Isolated from Four Locations in the South-West Indian Ocean. Toxicon 2001, 39, 1195–1202. [Google Scholar] [CrossRef] [PubMed]
- Maranda, L.; Corwin, S.; Hargraves, P.E. Prorocentrum lima (Dinophyceae) in Northeastern USA Coastal Waters. I. Abundance and Distribution. Harmful Algae 2007, 6, 623–631. [Google Scholar] [CrossRef]
- Nascimento, S.M.; Purdie, D.A.; Morris, S. Morphology, Toxin Composition and Pigment Content of Prorocentrum lima Strains Isolated from a Coastal Lagoon in Southern UK. Toxicon 2005, 45, 633–649. [Google Scholar] [CrossRef]
- Hou, D.-y.; Mao, X.-t.; Gu, S.; Li, H.-y.; Liu, J.-s.; Yang, W. dong Systems-Level Analysis of Metabolic Mechanism Following Nitrogen Limitation in Benthic Dinoflagellate Prorocentrum lima. Algal Res. 2018, 33, 389–398. [Google Scholar] [CrossRef]
- Toyofuku, H. Joint FAO/WHO/IOC Activities to Provide Scientific Advice on Marine Biotoxins (Research Report). Mar. Pollut. Bull. 2006, 52, 1735–1745. [Google Scholar] [CrossRef]
- Prassopoulou, E.; Katikou, P.; Georgantelis, D.; Kyritsakis, A. Detection of Okadaic Acid and Related Esters in Mussels during Diarrhetic Shellfish Poisoning (DSP) Episodes in Greece Using the Mouse Bioassay, the PP2A Inhibition Assay and HPLC with Fluorimetric Detection. Toxicon 2009, 53, 214–227. [Google Scholar] [CrossRef]
- Louppis, A.P.; Badeka, A.V.; Katikou, P.; Paleologos, E.K.; Kontominas, M.G. Determination of Okadaic Acid, Dinophysistoxin-1 and Related Esters in Greek Mussels Using HPLC with Fluorometric Detection, LC-MS/MS and Mouse Bioassay. Toxicon 2010, 55, 724–733. [Google Scholar] [CrossRef]
- Mountfort, D.O.; Suzuki, T.; Truman, P. Protein Phosphatase Inhibition Assay Adapted for Determination of Total DSP in Contaminated Mussels. Toxicon 2001, 39, 383–390. [Google Scholar] [CrossRef] [PubMed]
- Draisci, R.; Lucentini, L.; Giannetti, L.; Boria, P.; Poletti, R. First Report of Pectenotoxin-2 (PTX-2) in Algae (Dinophysis Fortii) Related to Seafood Poisoning in Europe. Toxicon 1996, 34, 923–935. [Google Scholar] [CrossRef] [PubMed]
- Nishimura, T.; Uchida, H.; Noguchi, R.; Oikawa, H.; Suzuki, T.; Funaki, H.; Ihara, C.; Hagino, K.; Arimitsu, S.; Tanii, Y.; et al. Abundance of the Benthic Dinoflagellate Prorocentrum and the Diversity, Distribution, and Diarrhetic Shellfish Toxin Production of Prorocentrum lima Complex and P. Caipirignum in Japan. Harmful Algae 2020, 96, 101687. [Google Scholar] [CrossRef]
- Marr, J.C.; Jackson, A.E.; McLachlan, J.L. Occurrence of Prorocentrum lima, a DSP Toxin-Producing Species from the Atlantic Coast of Canada. J. Appl. Phycol. 1992, 4, 17–24. [Google Scholar] [CrossRef]
- Morton, S.L.; Norris, D.R.; Bomber, J.W. Effect of Temperature, Salinity and Light Intensity on the Growth and Seasonality of Toxic Dinoflagellates Associated with Ciguatera. J. Exp. Mar. Bio. Ecol. 1992, 157, 79–90. [Google Scholar] [CrossRef]
- Nascimento, S.M.; Salgueiro, F.; Menezes, M.; Oliveira, F.d.A.; Magalhães, V.C.P.; De Paula, J.C.; Morris, S. Prorocentrum lima from the South Atlantic: Morphological, Molecular and Toxicological Characterization. Harmful Algae 2016, 57, 39–48. [Google Scholar] [CrossRef]
- Pires, E.; de Melo Tarouco, F.; Alves, T.P.; da Rosa, C.E.; da Cunha Lana, P.; Mafra, L.L. Antioxidant Responses and Okadaic Acid Accumulation in Laeonereis acuta (Annelida) Exposed to the Harmful Dinoflagellate Prorocentrum cf. lima. Toxicon 2021, 203, 104–114. [Google Scholar] [CrossRef] [PubMed]
- Gamboa-Marquez, J.F.; Sanchez-Suarez, I.G.; Barbera-Sanchez, A. Dinoflagelados (Pyrrophyta) Del Archipielago Los Roques (Venezuela): Familias Prorocentraceae y Ostreopsidaceae. Biol. Mar. 1994, 45, 140–152. [Google Scholar]
- Gayoso, A.M.; Dover, S.; Morton, S.; Busman, M.; Moeller, P.; Fulco, V.K.; Maranda, L. Diarrhetic Shellfish Poisoning Associated with Prorocentrum lima (Dinophyceae) in Patagonian Gulfs (Argentina). J. Shellfish Res. 2002, 21, 461–463. [Google Scholar]
- Delgado, G.; Popowski, G.; García, C.; Lagos, N.; Lechuga, D.; Lechuga-Devéze, C.H. Presence of DSP-Toxins in Prorocentrum lima (Ehrenberg) Dodge in Cuba. Rev. Investig. Mar. 2005, 26, 229–234. [Google Scholar]
- Foden, J.; Purdie, D.A.; Morris, S.; Nascimento, S. Epiphytic Abundance and Toxicity of Prorocentrum lima Populations in the Fleet Lagoon, UK. Harmful Algae 2005, 4, 1063–1074. [Google Scholar] [CrossRef]
- Vale, P.; Veloso, V.; Amorim, A. Toxin Composition of a Prorocentrum lima Strain Isolated from the Portuguese Coast. Toxicon 2009, 54, 145–152. [Google Scholar] [CrossRef] [PubMed]
- Álvarez, E.A.; Klemm, K.; Hoppenrath, M.; Cembella, A.; John, U.; Karlson, B. Temporal and Spatial Distribution of Epibenthic Dinoflagellates in the Kattegat-Skagerrak, NE Atlantic–Focus on Prorocentrum lima and Coolia monotis. Harmful Algae 2022, 118, 102318. [Google Scholar] [CrossRef] [PubMed]
- Barbier, M.; Amzil, Z.; Mondeguer, F.; Bhaud, Y.; Soyer-Gobillard, M.O.; Lassus, P. Okadaic Acid and PP2A Cellular Immunolocalization in Prorocentrum lima (Dinophyceae). Phycologia 1999, 38, 41–46. [Google Scholar] [CrossRef]
- Sahraoui, I.; Bouchouicha, D.; Mabrouk, H.H.; Hlaili, A.S. Driving Factors of the Potentially Toxic and Harmful Species of Prorocentrum Ehrenberg in a Semi-Enclosed Mediterranean Lagoon (Tunisia, SW Mediterranean). Mediterr. Mar. Sci. 2013, 14, 353–362. [Google Scholar] [CrossRef]
- Penna, A.; Ingarao, C.; Ercolessi, M.; Rocchi, M.; Penna, N. Potentially Harmful Microalgal Distribution in an Area of the NW Adriatic Coastline: Sampling Procedure and Correlations with Environmental Factors. Estuar. Coast. Shelf Sci. 2006, 70, 307–316. [Google Scholar] [CrossRef]
- Nakajima, I.; Oshima, Y.; Yasumoto, T. Toxicity of Benthic Dinoflagellates Found in Coral Reef-II. Toxicity of Benthic Dinoflagellates in Okinawa. Nippon Suisan Gakkaishi 1981, 47, 1029–1033. [Google Scholar] [CrossRef]
- Lee, J.S.; Igarashi, T.; Fraga, S.; Dahl, E.; Hovgaard, P.; Yasumoto, T. Determination of Diarrhetic Shellfish Toxins in Various Dinoflagellate Species. J. Appl. Phycol. 1989, 1, 147–152. [Google Scholar] [CrossRef]
- Holmes, M.J.; Lee, F.C.; Khoo, H.W.; Teo, S.L.M. Production of 7-Deoxy-Okadaic Acid by a New Caledonian Strain of Prorocentrum lima (Dinophyceae). J. Phycol. 2001, 37, 280–288. [Google Scholar] [CrossRef]
- Lu, C.K.; Lee, G.H.; Huang, R.; Chou, H.N. Spiro-Prorocentrimine, a Novel Macrocyclic Lactone from a Benthic Prorocentrum sp. of Taiwan. Tetrahedron Lett. 2001, 42, 1713–1716. [Google Scholar] [CrossRef]
- Luo, Z.; Zhang, H.; Krock, B.; Lu, S.; Yang, W.; Gu, H. Morphology, Molecular Phylogeny and Okadaic Acid Production of Epibenthic Prorocentrum (Dinophyceae) Species from the Northern South China Sea. Algal Res. 2017, 22, 14–30. [Google Scholar] [CrossRef]
- Yan, M.; Leung, P.T.Y.; Gu, J.; Lam, V.T.T.; Murray, J.S.; Harwood, D.T.; Wai, T.C.; Lam, P.K.S. Hemolysis Associated Toxicities of Benthic Dinoflagellates from Hong Kong Waters. Mar. Pollut. Bull. 2020, 155, 111114. [Google Scholar] [CrossRef]
- Meunier, A. Microplankton de La Mer Flamande. III Les Péridiniens; Mem. Mus.: Belgium, 1919; Available online: https://www.biodiversitylibrary.org/bibliography/8577 (accessed on 11 January 2024).
- Dolapsakis, N.P.; Kilpatrick, M.W.; Economou-Amilli, A.; Tafas, T. Morphology and RDNA Phylogeny of a Mediterranean Coolia monotis (Dinophyceae) Strain from Greece. Sci. Mar. 2006, 70, 67–76. [Google Scholar] [CrossRef]
- Holmes, M.J.; Lewis, R.J.; Jones, A.; Hoy, A.W.W. Cooliatoxin, the First Toxin from Coolia monotis (Dinophyceae). Nat. Toxins 1995, 3, 355–362. [Google Scholar] [CrossRef] [PubMed]
- Griffith, A.W.; Gobler, C.J. Harmful Algal Blooms: A Climate Change Co-Stressor in Marine and Freshwater Ecosystems. Harmful Algae 2020, 91, 101590. [Google Scholar] [CrossRef] [PubMed]
- Ralston, D.K.; Moore, S.K. Modeling Harmful Algal Blooms in a Changing Climate. Harmful Algae 2020, 91, 101729. [Google Scholar] [CrossRef] [PubMed]
- Tester, P.A.; Litaker, R.W.; Berdalet, E. Climate Change and Harmful Benthic Microalgae. Harmful Algae 2020, 91, 101655. [Google Scholar] [CrossRef]
- Wells, M.L.; Karlson, B.; Wulff, A.; Kudela, R.; Trick, C.; Asnaghi, V.; Berdalet, E.; Cochlan, W.; Davidson, K.; De Rijcke, M.; et al. Future HAB Science: Directions and Challenges in a Changing Climate. Harmful Algae 2020, 91, 101655. [Google Scholar] [CrossRef]
- Taleb, H.; Vale, P.; Jaime, E.; Blaghen, M. Study of Paralytic Shellfish Poisoning Toxin Profile in Shellfish from the Mediterranean Shore of Morocco. Toxicon 2001, 39, 1855–1861. [Google Scholar] [CrossRef]
- Aboualaalaa, H.; Rijal Leblad, B.; Hormat-Allah, A.; Savar, V.; Ennaskhi, I.; Hammi, I.; Elkbiach, M.L.B.; Ibghi, M.; Maamour, N.; Medhioub, W.; et al. New Insights into the Dynamics of Causative Dinoflagellates and the Related Contamination of Molluscs by Paralytic Toxins in the Southwestern Mediterranean Coastal Waters of Morocco. Mar. Pollut. Bull. 2022, 185, 114349. [Google Scholar] [CrossRef]
- Rijal Leblad, B.; Amnhir, R.; Reqia, S.; Sitel, F.; Daoudi, M.; Marhraoui, M.; Ouelad Abdellah, M.K.; Veron, B.; Er-Raioui, H.; Laabir, M. Seasonal Variations of Phytoplankton Assemblages in Relation to Environmental Factors in Mediterranean Coastal Waters of Morocco, a Focus on HABs Species. Harmful Algae 2020, 96, 101819. [Google Scholar] [CrossRef] [PubMed]
- Rijal Leblad, B.; Lundholm, N.; Goux, D.; Veron, B.; Sagou, R.; Taleb, H.; Nhhala, H.; Er-raioui, H. Pseudo-nitzschia Peragallo (Bacillariophyceae) Diversity and Domoic Acid Accumulation in Tuberculate cockles and Sweet clams in M’diq Bay, Morocco. Acta Bot. Croat 2013, 72, 35–47. [Google Scholar] [CrossRef]
- Rijal Leblad, B.; Nhhala, H.; Daoudi, M.; Marhraoui, M.; Ouelad Abdellah, M.K.O.; Veron, B.; Er-Raioui, H. Contamination and Depuration of Paralytic Shellfish Poisoning by Acanthocardia tuberculata Cockles and Callista chione Clams in Moroccan Waters. J. Mater. Environ. Sci 2017, 8, 4634–4641. [Google Scholar]
- Katoh, K.; Standley, D.M. MAFFT Multiple Sequence Alignment Software Version 7: Improvements in Performance and Usability. Mol. Biol. Evol. 2013, 30, 772–780. [Google Scholar] [CrossRef]
- Penna, A.; Fraga, S.; Battocchi, C.; Casabianca, S.; Perini, F.; Capellacci, S.; Casabianca, A.; Riobo, P.; Giacobbe, M.G.; Totti, C.; et al. Genetic Diversity of the Genus Ostreopsis Schmidt: Phylogeographical Considerations and Molecular Methodology Applications for Field Detection in the Mediterranean Sea. Cryptogam. Algol. 2012, 33, 153–163. [Google Scholar] [CrossRef]
- Toldrà, A.; Alcaraz, C.; Andree, K.B.; Fernández-Tejedor, M.; Diogène, J.; Katakis, I.; O’Sullivan, C.K.; Campàs, M. Colorimetric DNA-Based Assay for the Specific Detection and Quantification of Ostreopsis cf. ovata and Ostreopsis cf. siamensis in the Marine Environment. Harmful Algae 2019, 84, 27–35. [Google Scholar] [CrossRef]
- Vanucci, S.; Guerrini, F.; Milandri, A.; Pistocchi, R. Effects of Different Levels of N- and P-Deficiency on Cell Yield, Okadaic Acid, DTX-1, Protein and Carbohydrate Dynamics in the Benthic Dinoflagellate Prorocentrum lima. Harmful Algae 2010, 9, 590–599. [Google Scholar] [CrossRef]
- Santos, M.; Oliveira, P.B.; Moita, M.T.; David, H.; Caeiro, M.F.; Zingone, A.; Amorim, A.; Silva, A. Ocurrence of Ostreopsis in Two Temperate Coastal Bays (SW Iberia): Insights from the Plankton. Harmful Algae 2019, 86, 20–36. [Google Scholar] [CrossRef]
- Drouet, K.; Jauzein, C.; Herviot-Heath, D.; Hariri, S.; Laza-Martinez, A.; Lecadet, C.; Plus, M.; Seoane, S.; Sourisseau, M.; Lemée, R.; et al. Current Distribution and Potential Expansion of the Harmful Benthic Dinoflagellate Ostreopsis cf. siamensis towards the Warming Waters of the Bay of Biscay, North-East Atlantic. Environ. Microbiol. 2021, 23, 4956–4979. [Google Scholar] [CrossRef]
- Verma, A.; Hoppenrath, M.; Dorantes-Aranda, J.J.; Harwood, D.T.; Murray, S.A. Molecular and Phylogenetic Characterization of Ostreopsis (Dinophyceae) and the Description of a New Species, Ostreopsis rhodesae sp. nov., from a Subtropical Australian Lagoon. Harmful Algae 2016, 60, 116–130. [Google Scholar] [CrossRef] [PubMed]
- Parsons, M.L.; Aligizaki, K.; Bottein, M.Y.D.; Fraga, S.; Morton, S.L.; Penna, A.; Rhodes, L. Gambierdiscus and Ostreopsis: Reassessment of the State of Knowledge of Their Taxonomy, Geography, Ecophysiology, and Toxicology. Harmful Algae 2012, 14, 107–129. [Google Scholar] [CrossRef]
- Nascimento, S.M.; Neves, R.A.F.; De’Carli, G.; Borsato, G.T.; Silva, R.A.F.; Melo, G.A.; De Morais, A.M.; Cockell, T.C.; Fraga, S.; Menezes-salgueiro, A.; et al. Ostreopsis cf. ovata (Dinophceae) Molecular Phylogeny, Morphology, and Detection of Ovatoxins in Strains and Field Samples from Brazil. Toxins 2020, 12, 70. [Google Scholar] [CrossRef] [PubMed]
- Schmidt, J. Flora of Koh Chang. Contributions to the Knowledge of the Vegetationin the Gulf of Siam. Bot. Tidsskr. 1901, 24, 241–327. [Google Scholar]
- Tawong, W.; Nishimura, T.; Sakanari, H.; Sato, S.; Yamaguchi, H.; Adachi, M. Distribution and Molecular Phylogeny of the Dinoflagellate Genus Ostreopsis in Thailand. Harmful Algae 2014, 37, 160–171. [Google Scholar] [CrossRef]
- Gu, H.; Wang, Y.; Derrien, A.; Hervé, F.; Wang, N.; Pransilpa, M.; Lim, P.T.; Leaw, C.P. Two Toxigenic Ostreopsis species, O. cf. ovata and O. siamensis (Dinophyceae), from the South China Sea, Tropical Western Pacific. Harmful Algae 2022, 113, 102206. [Google Scholar] [CrossRef] [PubMed]
- Holmes, M.J.; Gillespie, N.J.; Lewis, R.J. Toxicity and Morphology of Ostreopsis cf. siamensis Cultured from a Ciguatera Endemic Region of Queensland, Australia. In Proceedings of the 6th International Coral Reef Symposium, Townsville, Australia, 8–12 August 1988. [Google Scholar]
- Silva, A.; Brotas, V.; Orive, E.; Neto, A. First Records of Ostreopsis heptagona, O. cf. siamensis and O. cf. ovata in the Azores Archipelago, Portugal. Harmful Algae News 2010, 42, 1–2. [Google Scholar]
- Pereira, C.J. Ocurrencia de Floraciones Nocivas de Microalgas En Las Costas Del Estado Miranda, Venezuela. Cienc. Ambient. Y Clima 2020, 3, 55–71. [Google Scholar] [CrossRef]
- Selina, M.S.; Morozova, T.V.; Vyshkvartsev, D.I.; Orlova, T.Y. Seasonal Dynamics and Spatial Distribution of Epiphytic Dinoflagellates in Peter the Great Bay (Sea of Japan) with Special Emphasis on Ostreopsis species. Harmful Algae 2014, 32, 1–10. [Google Scholar] [CrossRef]
- Tanimoto, Y.; Yamaguchi, H.; Yoshimatsu, T.; Sato, S.; Adachi, M. Effects of Temperature, Salinity and Their Interaction on Growth of Toxic Ostreopsis sp. 1 and Ostreopsis sp. 6 (Dinophyceae) Isolated from Japanese Coastal Waters. Fish. Sci. 2013, 79, 285–291. [Google Scholar] [CrossRef]
- Brissard, C.; Herrenknecht, C.; Séchet, V.; Hervé, F.; Pisapia, F.; Harcouet, J.; Lémée, R.; Chomérat, N.; Hess, P.; Amzil, Z. Complex Toxin Profile of French Mediterranean Ostreopsis cf. ovata Strains, Seafood Accumulation and Ovatoxins Prepurification. Mar. Drugs 2014, 12, 2851–2876. [Google Scholar] [CrossRef]
- Nascimento, S.M.; Corrêa, E.V.; Menezes, M.; Varela, D.; Paredes, J.; Morris, S. Growth and Toxin Profile of Ostreopsis cf. ovata (Dinophyta) from Rio de Janeiro, Brazil. Harmful Algae 2012, 13, 1–9. [Google Scholar] [CrossRef]
- Shah, M.M.R.; Samarakoon, K.W.; Ko, J.-Y.; Lakmal, H.H.C.; Lee, J.-Y.; An, S.-J.; Jeon, Y.-J.; Lee, J.-B. Potentiality of Benthic Dinoflagellate Cultures and Screening of Their Bioactivities in Jeju Island, Korea. African J. Biotechnol. 2014, 13, 792–805. [Google Scholar] [CrossRef]
- Yamaguchi, H.; Tanimoto, Y.; Yoshimatsu, T.; Sato, S.; Nishimura, T.; Uehara, K.; Adachi, M. Culture Method and Growth Characteristics of Marine Benthic Dinoflagellate Ostreopsis spp. Isolated from Japanese Coastal Waters. Fish. Sci. 2012, 78, 993–1000. [Google Scholar] [CrossRef]
- Nagahama, Y.; Murray, S.; Tomaru, A.; Fukuyo, Y. Species Boundaries in the Toxic Dinoflagellate Prorocentrum lima (Dinophyceae, Prorocentrales), Based on Morphological and Phylogenetic Characters. J. Phycol. 2011, 47, 178–189. [Google Scholar] [CrossRef] [PubMed]
- Zhang, H.; Li, Y.; Cen, J.; Wang, H.; Cui, L.; Dong, Y.; Lu, S. Morphotypes of Prorocentrum lima (Dinophyceae) from Hainan Island, South China Sea: Morphological and Molecular Characterization. Phycologia 2015, 54, 503–516. [Google Scholar] [CrossRef]
- Arteaga-Sogamoso, E.; Rodríguez, F.; Amato, A.; Ben-Gigirey, B.; Fraga, S.; Mafra, L.L.; Fernandes, L.F.; de Azevedo Tibiriçá, C.E.J.; Chomérat, N.; Nishimura, T.; et al. Morphology and Phylogeny of Prorocentrum porosum sp. nov. (Dinophyceae): A New Benthic Toxic Dinoflagellate from the Atlantic and Pacific Oceans. Harmful Algae 2023, 121, 102356. [Google Scholar] [CrossRef] [PubMed]
- Giussani, V.; Asnaghi, V.; Pedroncini, A.; Chiantore, M. Management of Harmful Benthic Dinoflagellates Requires Targeted Sampling Methods and Alarm Thresholds. Harmful Algae 2017, 68, 97–104. [Google Scholar] [CrossRef]
- Aissaoui, A.; Armi, Z.; Akrout, F.; Ben Hassine, O.K. Environmental Factors and Seasonal Dynamics of Prorocentrum lima Population in Coastal Waters of the Gulf of Tunis, South Mediterranean. Water Environ. Res. 2014, 86, 2256–2270. [Google Scholar] [CrossRef]
- Pan, Y.; Cembella, A.D.; Quilliam, M.A. Cell Cycle and Toxin Production in the Benthic Dinoflagellate Prorocentrum lima. Mar. Biol. 1999, 134, 541–549. [Google Scholar] [CrossRef]
- Morton, S.L.; Tindall, D.R. Morphological and Biochemical Variability of the Toxic Dinoflagellate Prorocentrum lima Isolated From Three Locations At Heron Island, Australia. J. Phycol. 1995, 31, 914–921. [Google Scholar] [CrossRef]
- Wang, S.; Chen, J.; Li, Z.; Wang, Y.; Fu, B.; Han, X.; Zheng, L. Cultivation of the Benthic Microalga Prorocentrum lima for the Production of Diarrhetic Shellfish Poisoning Toxins in a Vertical Flat Photobioreactor. Bioresour. Technol. 2015, 179, 243–248. [Google Scholar] [CrossRef] [PubMed]
- Bravo, I.; Fernández, M.L.; Ramilo, I.; Martínez, A. Toxin Composition of the Toxic Dinoflagellate Prorocentrum lima Isolated from Different Locations along the Galician Coast (NW Spain). Toxicon 2001, 39, 1537–1545. [Google Scholar] [CrossRef] [PubMed]
- Hashimoto, K.; Uchida, H.; Nishimura, T.; Oikawa, H.; Funaki, H.; Honma, C.; Yamaguchi, H.; Suzuki, T.; Adachi, M. Determination of Optimal Culture Conditions for Toxin Production by a Prorocentrum lima Complex Strain with High Diarrhetic Shellfish Toxins Yield. Harmful Algae 2021, 103, 102025. [Google Scholar] [CrossRef]
- Glibert, P.M.; Burkholder, J.A.M.; Kana, T.M. Recent Insights about Relationships between Nutrient Availability, Forms, and Stoichiometry, and the Distribution, Ecophysiology, and Food Web Effects of Pelagic and Benthic Prorocentrum Species. Harmful Algae 2012, 14, 231–259. [Google Scholar] [CrossRef]
- Lee, T.C.H.; Fong, F.L.Y.; Ho, K.C.; Lee, F.W.F. The Mechanism of Diarrhetic Shellfish Poisoning Toxin Production in Prorocentrum spp.: Physiological and Molecular Perspectives. Toxins 2016, 8, 272. [Google Scholar] [CrossRef]
- López-Rosales, L.; Gallardo-Rodríguez, J.J.; Sánchez-Mirón, A.; Cerón-García, M.D.C.; Belarbi, E.H.; García-Camacho, F.; Molina-Grima, E. Simultaneous Effect of Temperature and Irradiance on Growth and Okadaic Acid Production from the Marine Dinoflagellate Prorocentrum belizeanum. Toxins 2013, 6, 229–253. [Google Scholar] [CrossRef]
- Aquino-Cruz, A.; Purdie, D.A.; Morris, S. Effect of Increasing Sea Water Temperature on the Growth and Toxin Production of the Benthic Dinoflagellate Prorocentrum lima. Hydrobiologia 2018, 813, 103–122. [Google Scholar] [CrossRef]
- McLachlan, J.L.; Marr, J.C.; Conlon-Keily, A.; Adamson, A. Effects of Nitrogen Concentration and Cold Temperature on DSP-toxin Concentrations in the Dinoflagellate Prorocentrum lima (Prorocentrales, Dinophyceae). Nat. Toxins 1994, 2, 263–270. [Google Scholar] [CrossRef]
- Almada, E.V.C.; De Carvalho, W.F.; Nascimento, S.M. Investigation of Phagotrophy in Natural Assemblages of the Benthic Dinoflagellates Ostreopsis, Prorocentrum and Coolia. Braz. J. Oceanogr. 2017, 65, 392–399. [Google Scholar] [CrossRef]
- Lee, B.; Park, M.G. Genetic Analyses of the RbcL and PsaA Genes from Single Cells Demonstrate a Rhodophyte Origin of the Prey in the Toxic Benthic Dinoflagellate Ostreopsis. Front. Mar. Sci. 2018, 5, 1–11. [Google Scholar] [CrossRef]
- Stoecker, D.K.; Hansen, P.J.; Caron, D.A.; Mitra, A. Mixotrophy in the Marine Plankton. Ann. Rev. Mar. Sci. 2017, 9, 311–335. [Google Scholar] [CrossRef] [PubMed]
- Varkitzi, I.; Pagou, K.; Granéli, E.; Hatzianestis, I.; Pyrgaki, C.; Pavlidou, A.; Montesanto, B.; Economou-Amilli, A. Unbalanced N:P Ratios and Nutrient Stress Controlling Growth and Toxin Production of the Harmful Dinoflagellate Prorocentrum lima (Ehrenberg) Dodge. Harmful Algae 2010, 9, 304–311. [Google Scholar] [CrossRef]
- Aquino-Cruz, A.; Okolodkov, Y.B. Impact of Increasing Water Temperature on Growth, Photosynthetic Efficiency, Nutrient Consumption, and Potential Toxicity of Amphidinium cf. carterae and Coolia monotis (Dinoflagellata). Rev. Biol. Mar. Oceanogr. 2016, 51, 565–580. [Google Scholar] [CrossRef]
- Hou, D.-y.; Liang, J.-j.; Zou, C.; Li, H.-y.; Liu, J.-s.; Yang, W. dong MRP Functional Activity and Character in the Dinoflagellate Prorocentrum lima. J. Appl. Phycol. 2016, 28, 1667–1676. [Google Scholar] [CrossRef]
- Collos, Y.; Harrison, P.J. Acclimation and Toxicity of High Ammonia Concentrations to Unicellular Algae. Mar. Pollut. Bull. 2014, 80, 8–23. [Google Scholar] [CrossRef] [PubMed]
- Leaw, C.P.; Tan, T.H.; Lim, H.C.; Teng, S.T.; Yong, H.L.; Smith, K.F.; Rhodes, L.; Wolf, M.; Holland, W.C.; Vandersea, M.W.; et al. New Scenario for Speciation in the Benthic Dinoflagellate Genus Coolia (Dinophyceae). Harmful Algae 2016, 55, 137–149. [Google Scholar] [CrossRef]
- Abdennadher, M.; Zouari, A.B.; Sahnoun, W.F.; Walha, L.D.; Mahfoudi, M.; Hamza, A. A Long-Term Study on Coolia monotis Distribution from the South-East Mediterranean Sea. Cont. Shelf Res. 2020, 211, 104267. [Google Scholar] [CrossRef]
- Tanaka, I.; Matsuoka, S.; Murata, M.; Tachibana, K. A New Ceramide with a Novel Branched-Chain Fatty Acid Isolated from the Epiphytic Dinoflagellate Coolia monotis. J. Nat. Prod. 1998, 61, 685–688. [Google Scholar] [CrossRef]
- Liang, J.L.; Long, L.J.; Zhang, S.; Wu, J. Cooliatin, an Unprecedented Natural Dioxocyclononane from Dinoflagellate Coolia monotis from South China Sea. Chem. Pharm. Bull. 2009, 57, 888–889. [Google Scholar] [CrossRef]
- Rhodes, L.L.; Smith, K.F.; Munday, R.; Selwood, A.I.; McNabb, P.S.; Holland, P.T.; Bottein, M.Y. Toxic Dinoflagellates (Dinophyceae) from Rarotonga, Cook Islands. Toxicon 2010, 56, 751–758. [Google Scholar] [CrossRef]
- Ho, T.V.; Nguyen, L.N. Morphology and Distribu- Tion of the Three Epiphytic Dinoflagellate Species Coolia monotis, C. tropicalis, and C. canariensis (Ostreopsidaceae, Gonyaulacales, Dinophyceae) from Vietnamese Coastal Waters. Ocean Sci. J. 2014, 49, 211–221. [Google Scholar] [CrossRef]
- Tan, T.-H.; Lim, P.-T.; Mujahid, A.; Usup, G.; Leaw, C.-P. Benthic Harmful Dinoflagellate Assemblages in a Fringing Reef of Sampadi Island, Sarawak, Malaysia. Mar. Res. Indones. 2013, 38, 11–21. [Google Scholar] [CrossRef]
- Yong, H.L.; Mustapa, N.I.; Lee, L.K.; Lim, Z.F.; Tan, T.H.; Usup, G.; Gu, H.; Litaker, R.W.; Tester, P.A.; Lim, P.T.; et al. Habitat Complexity Affects Benthic Harmful Dinoflagellate Assemblages in the Fringing Reef of Rawa Island, Malaysia. Harmful Algae 2018, 78, 56–68. [Google Scholar] [CrossRef]
- Parke, M.; Dixon, P.S. Check-List of British Marine Algae-Third Revision. J. Mar. Biol. Assoc. United Kingd. 1976, 56, 527–594. [Google Scholar] [CrossRef]
- Fraga, S.; Penna, A.; Bianconi, I.; Paz, B.; Zapata, M. Coolia canariensis sp. nov. (Dinophyceae), a New Nontoxic Epiphytic Benthic Dinoflagellate from the Canary Islands. J. Phycol. 2008, 44, 1060–1070. [Google Scholar] [CrossRef]
- David, H.; Kromkamp, J.C.; Orive, E. Journal of Experimental Marine Biology and Ecology Relationship between Strains of Coolia monotis (Dinophyceae) from the Atlantic Iberian Peninsula and Their Sampling Sites. J. Exp. Mar. Bio. Ecol. 2017, 487, 59–67. [Google Scholar] [CrossRef]
- Lewis, N.I.; Wolny, J.L.; Achenbach, J.C.; Ellis, L.; Pitula, J.S.; Rafuse, C.; Rosales, D.S.; McCarron, P. Identification, Growth and Toxicity Assessment of Coolia Meunier (Dinophyceae) from Nova Scotia, Canada. Harmful Algae 2018, 75, 45–56. [Google Scholar] [CrossRef]
- Mabrouk, L.; Hamza, A.; Brahim, M.B.; Bradai, M.N. Temporal and Depth Distribution of Microepiphytes on Posidonia oceanica (L.) Delile Leaves in a Meadow off Tunisia. Mar. Ecol. 2011, 32, 148–161. [Google Scholar] [CrossRef]
- Abdennadher, M.; Zouari, A.B.; Medhioub, W.; Penna, A. Characterization of Coolia spp. ( Gonyaucales, Dinophyceae ) from Southern Tunisia: First Record of Coolia malayensis in the Mediter- Ranean Sea. Algae 2021, 36, 175–193. [Google Scholar] [CrossRef]
- Faust, M.A. Ciguatera-Causing Dinoflagellates in a Coral-Reef Mangrove Ecosystem, Belize. Atoll Res. Bull. 2009, 1–32. [Google Scholar] [CrossRef]
- Armi, Z.; Turki, S.; Trabelsi, E.; Ben Maiz, N. First Recorded Proliferation of Coolia monotis (Meunier, 1919) in the North Lake of Tunis (Tunisia) Correlation with Environmental Factors. Environ. Monit. Assess. 2010, 164, 423–433. [Google Scholar] [CrossRef] [PubMed]
- Mohammad-Noor, N.; Moestrup, Ø.; Lundholm, N.; Fraga, S.; Adam, A.; Holmes, M.J.; Saleh, E. Autecology and Phylogeny of Coolia tropicalis and Coolia malayensis (Dinophyceae), with Emphasis on Taxonomy of C. tropicalis Based on Light Microscopy, Scanning Electron Microscopy and LSU RDNA1. J. Phycol. 2013, 49, 536–545. [Google Scholar] [CrossRef] [PubMed]
- Tibiriçá, C.E.J.D.A.; Sibat, M.; Fernandes, L.F.; Bilien, G.; Chomérat, N.; Hess, P.; Mafra, L.L. Diversity and Toxicity of the Genus Coolia Meunier in Brazil, and Detection of 44-Methyl Gambierone in Coolia tropicalis. Toxins 2020, 12, 327. [Google Scholar] [CrossRef]
- Karafas, S.; York, R.; Tomas, C. Morphological and Genetic Analysis of the Coolia monotis Species Complex with the Introduction of Two New Species, Coolia santacroce sp. nov. and Coolia palmyrensis sp. nov. (Dinophyceae). Harmful Algae 2015, 46, 18–33. [Google Scholar] [CrossRef]
- Aboualaalaa, H.; El Kbiach, M.L.B.; Rijal Leblad, B.; Hervé, F.; Hormat-Allah, A.; Baudy, L.; Ennaskhi, I.; Hammi, I.; Ibghi, M.; Elmortaji, H.; et al. Development of Harmful Algal Blooms Species Responsible for Lipophilic and Amnesic Shellfish Poisoning Intoxications in Southwestern Mediterranean Coastal Waters. Toxicon 2022, 219, 106916. [Google Scholar] [CrossRef] [PubMed]
- Harrison, P.J.; Waters, R.E.; Taylor, F.J.R. A Broad Spectrum Artificial Sea Water Medium for Coastal and Open Ocean Phytoplankton. J. Phycol. 1980, 16, 28–35. [Google Scholar] [CrossRef]
- Guillard, R.R.L.; Hargraves, P.E. Stichochrysis Immobilis Is a Diatom, Not a Chrysophyte. Phycologia 1993, 32, 234–236. [Google Scholar] [CrossRef]
- Scholin, C.A.; Herzog, M.; Sogin, M.; Anderson, D.M. Identification of Group and Strain Specific Genetic Markers for Globally Distributed Alexandrium (Dinophyceae). II. Sequence Analysis of a Fragment of the LSU RRNA Gene. J. Phycol. 1994, 30, 999–1011. [Google Scholar] [CrossRef]
- Nunn, G.B.; Theisen, B.F.; Christensen, B.; Arctander, P. Simplicity-Correlated Size Growth of the Nuclear 28S Ribosomal RNA D3 Expansion Segment in the Crustacean Order Isopoda. J. Mol. Evol. 1996, 42, 211–223. [Google Scholar] [CrossRef]
- Chinain, M.; Germain, M.; Deparis, X.; Pauillac, S.; Legrand, A.M. Seasonal Abundance and Toxicity of the Dinoflagellate Gambierdiscus spp. (Dinophyceae), the Causative Agent of Ciguatera in Tahiti, French Polynesia. Mar. Biol. 1999, 135, 259–267. [Google Scholar] [CrossRef]
- Tillmann, U.; Bantle, A.; Krock, B.; Elbrächter, M.; Gottschling, M. Recommendations for Epitypification of Dinophytes Exemplified by Lingulodinium polyedra and Molecular Phylogenetics of the Gonyaulacales Based on Curated RRNA Sequence Data. Harmful Algae 2021, 104. [Google Scholar] [CrossRef] [PubMed]
- Gouy, M.; Guindon, S.; Gascuel, O. Sea View Version 4: A Multiplatform Graphical User Interface for Sequence Alignment and Phylogenetic Tree Building. Mol. Biol. Evol. 2010, 27, 221–224. [Google Scholar] [CrossRef] [PubMed]
- Edgar, R.C. MUSCLE: Multiple Sequence Alignment with High Accuracy and High Throughput. Nucleic Acids Res. 2004, 32, 1792–1797. [Google Scholar] [CrossRef] [PubMed]
- Guindon, S.; Gascuel, O. A Simple, Fast, and Accurate Algorithm to Estimate Large Phylogenies by Maximum Likelihood. Syst. Biol. 2003, 52, 696–704. [Google Scholar] [CrossRef]
- Ronquist, F.; Huelsenbeck, J.P. MrBayes 3: Bayesian Phylogenetic Inference under Mixed Models. Bioinformatics 2003, 19, 1572–1574. [Google Scholar] [CrossRef]
- Posada, D. JModelTest: Phylogenetic Model Averaging. Mol. Biol. Evol. 2008, 25, 1253–1256. [Google Scholar] [CrossRef]
- Guillard, R.R.L. Handbook of Phycological Methods: Culture Methods and Growth Measurements; Stein, J.R., Ed.; Cambridge University Press: Cambridge, UK, 1973; ISBN 9780521297479. [Google Scholar]















| Ostreopsis sp. 9 | Prorocentrum lima | Coolia monotis | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Mean ± SD | Min | Max | Mean ± SD | Min | Max | Mean ± SD | Min | Max | ||
| Exponential phase | Length (µm) | 48.81 ± 3.43 | 38.07 | 58.98 | 42.94 ± 1.79 | 37 | 47.83 | 34.92 ± 2.02 | 30.24 | 38.06 |
| Width (µm) | 35.79 ± 3.56 | 28.67 | 43.67 | 31.3 ± 1.23 | 28.15 | 35.12 | 32.53 ± 2.09 | 28.21 | 36.64 | |
| Stationary phase | Length (µm) | 53.24 ± 3.01 | 45.96 | 60.81 | 44.83 ± 1.70 | 40.43 | 48.29 | 34.99 ± 2.58 | 30.19 | 39.47 |
| Width (µm) | 36.20 ± 2.39 | 30.02 | 44.57 | 32.46 ± 1.43 | 28.91 | 35.38 | 32.28 ± 2.36 | 27.94 | 38 | |
| Okadaic Acid | DTX-1 | Length (µm) | Width (µm) | Size Ratio (L/W) | Maximum Growth Rate (d−1) | |||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Exp | Stat | Exp | Stat | Exp | Stat | Exp | Stat | Exp | Stat | |||
| Temperature | 15 °C | 11.63 | 9.51 | 15.27 | 10.64 | 45.26 | 45.42 | 34.32 | 34.61 | 1.32 | 1.31 | 0.08 |
| 20 °C | 4.21 | 3.90 | 3.47 | 3.58 | 44.62 | 43.31 | 33.17 | 32.96 | 1.35 | 1.31 | 0.22 | |
| 24 °C | 6.04 | 26.97 | 5.19 | 25.27 | 42.94 | 44.83 | 31.31 | 32.49 | 1.37 | 1.38 | 0.21 | |
| 29 °C | 6.05 | 5.98 | 6.53 | 5.75 | 44.7 | 45.37 | 33.43 | 34.44 | 1.34 | 1.32 | 0.06 | |
| Nitrate | 441 µM | 1.07 | 0.14 | 1.15 | 0.25 | 41.42 | 42.67 | 32.81 | 35.03 | 1.26 | 1.22 | 0.19 |
| 882 µM | 1.48 | 0.73 | 1.66 | 1.95 | 42.91 | 42.53 | 35.33 | 32.94 | 1.21 | 1.29 | 0.21 | |
| 1764 µM | 2.25 | 0.47 | 2.63 | 1.25 | 42.12 | 43.05 | 32.9 | 33.6 | 1.28 | 1.28 | 0.22 | |
| Ammonia | 441 µM | 0.86 | 1.83 | 1.31 | 3.10 | 40.5 | 40.46 | 32.73 | 32.2 | 1.24 | 1.26 | 0.18 |
| 882 µM | 0.53 | 6.11 | 1.80 | 4.40 | 39.84 | 43.59 | 32.85 | 32.69 | 1.21 | 1.33 | 0.17 | |
| Latitude | Longitude | Year of Observation/Isolation | In Situ or Lab | Temp (°C) | Sal | Light µmol Photons m−2 s−1 | Toxicity | Growth Rate (µ, d−1) | Maximum Cell Density FWM: Fresh Weight of Macrophyte | Ref. | |
|---|---|---|---|---|---|---|---|---|---|---|---|
| Mediterranean water | |||||||||||
| Greece | |||||||||||
| Ostreopsis cf. siamensis | 39.76141° | 22.50722° | 2003–2004 | In situ | 13.9–29.7 | 16.7–37.1 | – | - | - | 4.05 × 105 cells g−1 FWM | [52] |
| 39.76141° | 22.50722° | 2003–2005 | f/2 or K | 19.0 ± 1 | - | 70 | - | - | |||
| Italy | |||||||||||
| Ostreopsis cf. siamensis | 37.85011° | 15.30071° | 2005–2009 | f/2 | 23.1 | - | 100 | Not toxic | - | - | [62] |
| 38.22121° | 15.60987° | 1999–2000 | K, f/20, f/2 | 17 ± 1 | - | 100 | - | - | - | [35] | |
| France | |||||||||||
| Ostreopsis cf. siamensis | 43.40271° | −2.36951° | 2018 | In situ | 14.0–25.9 | 24.8–37.0 | - | - | - | 105 cells g−1 FWM | [113] |
| 43.39860° | −1.66280° | 2010 | f/4 | 20 | - | 80 | - | - | - | [51] | |
| Spain | |||||||||||
| Ostreopsis cf. siamensis | 41.85562° | 3.14320° | 2016 | f/2 | 24 ± 2 | 36 | 110 | - | - | - | [110] |
| 36.82037° | −2.44678° | ||||||||||
| 36.81699° | −2.46840° | 1999–2000 | K, f/20, f/2 | 17 ± 1 | - | 100 | - | - | - | [35] | |
| 41.84566° | 3.146161° | ||||||||||
| Morocco | |||||||||||
| Ostreopsis sp. 9 | 35.81203° | −5.75029° | 2017 | ENSW | 24 | 36 | 90 | Not toxic | 0.18 | 8.75 × 103 cells mL−1 | This study |
| Tunisia | |||||||||||
| Ostreopsis siamensis | 35.50249° | 11.09569° | 2008–2009 | In situ | - | - | - | - | - | ≥5 × 102 cells.100 g−1 FWM | [59] |
| Ostreopsis cf. siamensis | 37.32533° | 9.92102° | 2008 | In situ | - | - | - | - | - | 3.75 × 104 cells L−1 | [59] |
| Atlantic Water | |||||||||||
| France | |||||||||||
| Ostreopsis sp. 9 | 43.44881° | −1.60637° | 2021 | L1 | 17 | 35 | 160 | Not toxic | - | 9.63 × 104 cells mL−1 | [43] |
| Guadeloupe France | |||||||||||
| Ostreopsis siamensis | 16.17203° | −61.36256° | 2017–2018 | In situ | - | - | - | - | - | - | [49] |
| Spain | |||||||||||
| Ostreopsis cf. siamensis | 43.41660° | −3.39960° | 2010 | f/4 | 20 | - | 80 | - | - | - | [51] |
| 43.35250° | −3.07800° | 2010 | f/4 | 20 | - | 80 | - | - | - | ||
| 43.32150° | −1.9870° | 2010–2011 | f/4 | 20 | - | 80 | - | - | - | ||
| 43.44332° | −2.981080° | 2007–2009 | f/2 | 17–22 | 30–35 | 60 | Toxic for Artemia nauplii | - | - | [57] | |
| Ostreopsis siamensis | 27.63750° | −17.98833° | 2017 | In situ | - | - | - | - | - | - | [44] |
| Portugal | |||||||||||
| Ostreopsis cf. siamensis | 38.00000° | −25.00000° | 2009 | In situ | 20–24 | - | - | - | - | 105 cells L−1 | [121] |
| 37.95333° | −8.85883° | 2008–2009 | f/20-Si | 19 | 35 | 40 | - | - | - | [50] | |
| 37.08150° | −8.66820° | 2011 | f/4 | 20 | - | 80 | - | - | - | [51] | |
| 38.70180° | −9.40840° | 2010 | f/4 | 20 | - | 80 | - | - | - | ||
| 37.07990° | −8.31560° | 2010–2011 | f/4 | 20 | - | 80 | - | - | - | ||
| 38.69356° | −9.41470° | 2010–2011 | f/20 | 20 | 33–34 | - | - | - | - | [112] | |
| 37.09126° | −8.66902° | 2015–2016 | f/20 | 20 | 33–34 | - | - | - | - | ||
| 37.95055° | −8.86471° | 2005–2009 | f/2 | 23.1 | - | 100 | Not toxic | - | - | [62] | |
| 38.69333° | −9.40927° | 2005–2009 | f/2 | 23.1 | - | 100 | Not toxic | - | - | ||
| Morocco | |||||||||||
| Ostreopsis cf. siamensis | 30.62479° | −9.95794° | 2009 | In situ | 20–24 | - | - | Toxic (Mouse Bioassay) | - | 105 cells L−1 | [53] |
| 29.95772° | −9.79462° | 2009 | In situ | 20–24 | - | - | - | - | |||
| Venezuela | |||||||||||
| Ostreopsis cf. siamensis | 10.59090° | −66.07386° | 2010–2015 | In situ | - | - | - | - | - | 3596 cells L−1 | [122] |
| Pacific and Indo-Pacific waters | |||||||||||
| Vietnam | |||||||||||
| Ostreopsis siamensis | 12.18200° | 109.29200° | 2020 | In situ | 24–30 | 31–34 | - | - | - | 14,790 cells 100 cm−2 | [45] |
| New Zealand | |||||||||||
| Ostreopsis siamensis | −36.58878° | 175.89378° | 2004 | In situ | 21 | - | - | - | - | 1.4 × 106 cells g−1 FWM | [14] |
| −34.63077° | 173.55539° | 1995–1997 | In situ | 15.3–22.5 | - | - | - | - | 3.6 × 102 cells g−1 FWM | [55] | |
| −35.29552° | 174.53340° | 1997–1999 | GP Medium | 18 | - | 100 | Toxic for Artemia salina | 0.3 | - | [56] | |
| Malaysia | |||||||||||
| Ostreopsis siamensis | 5.28278° | 103.22669° | 2015–2018 | f/2 Si | 25.0 | 30 | 90 | Toxic | - | - | [119] |
| French Polynesia | |||||||||||
| Ostreopsis siamensis | −17.64046° | −149.61025° | 2019 | f10k | 26 | 36 | 60 | Toxic | - | - | [46] |
| Japan | |||||||||||
| Ostreopsis siamensis | 24.43447° | 124.28901° | - | - | - | - | - | - | - | - | [47] |
| Ostreopsis cf. siamensis | 43.066736° | 131.94946° | 2010 | In situ | 18 | 30–34 | - | - | - | 52 × 103 cells g−1 FWM | [123] |
| Australia | |||||||||||
| Ostreopsis cf. siamensis | −23.44213° | 151.91069° | - | - | - | - | - | - | - | - | [120] |
| −24.11266° | 152.71095° | ||||||||||
| Ostreopsis sp. 9 | −36.88330° | 149.91671° | 2013 | f/2, f/10 | 18.0 | 35.0 | 60–100 | Toxic | 0.39 | 2.37 × 104 cell mL−1 | [114] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ibghi, M.; Rijal Leblad, B.; L’Bachir El Kbiach, M.; Aboualaalaa, H.; Daoudi, M.; Masseret, E.; Le Floc’h, E.; Hervé, F.; Bilien, G.; Chomerat, N.; et al. Molecular Phylogeny, Morphology, Growth and Toxicity of Three Benthic Dinoflagellates Ostreopsis sp. 9, Prorocentrum lima and Coolia monotis Developing in Strait of Gibraltar, Southwestern Mediterranean. Toxins 2024, 16, 49. https://doi.org/10.3390/toxins16010049
Ibghi M, Rijal Leblad B, L’Bachir El Kbiach M, Aboualaalaa H, Daoudi M, Masseret E, Le Floc’h E, Hervé F, Bilien G, Chomerat N, et al. Molecular Phylogeny, Morphology, Growth and Toxicity of Three Benthic Dinoflagellates Ostreopsis sp. 9, Prorocentrum lima and Coolia monotis Developing in Strait of Gibraltar, Southwestern Mediterranean. Toxins. 2024; 16(1):49. https://doi.org/10.3390/toxins16010049
Chicago/Turabian StyleIbghi, Mustapha, Benlahcen Rijal Leblad, Mohammed L’Bachir El Kbiach, Hicham Aboualaalaa, Mouna Daoudi, Estelle Masseret, Emilie Le Floc’h, Fabienne Hervé, Gwenael Bilien, Nicolas Chomerat, and et al. 2024. "Molecular Phylogeny, Morphology, Growth and Toxicity of Three Benthic Dinoflagellates Ostreopsis sp. 9, Prorocentrum lima and Coolia monotis Developing in Strait of Gibraltar, Southwestern Mediterranean" Toxins 16, no. 1: 49. https://doi.org/10.3390/toxins16010049





