Bee Venom Stimulates Growth Factor Release from Adipose-Derived Stem Cells to Promote Hair Growth
Abstract
1. Introduction
2. Results
2.1. Cell Viability
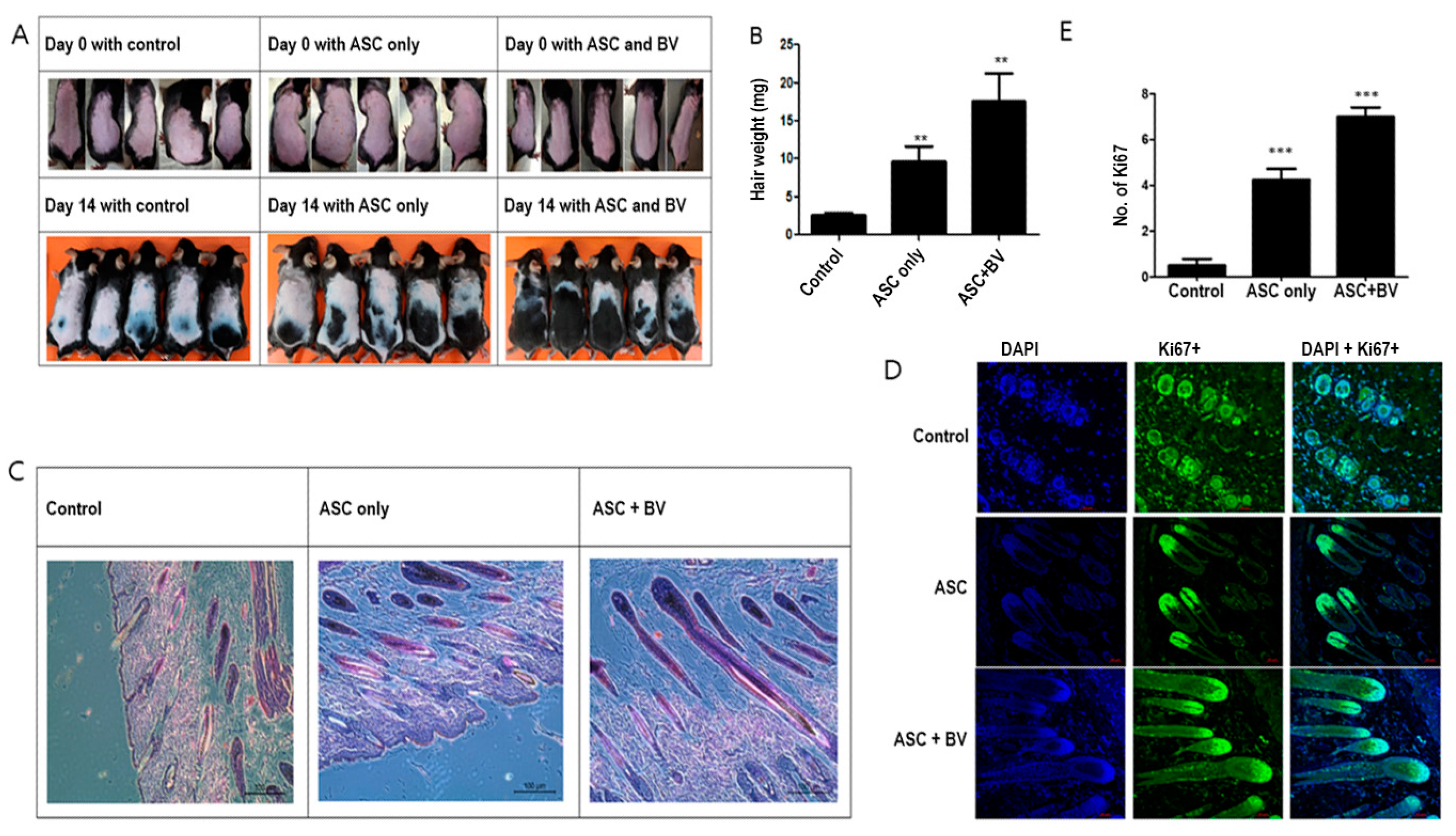
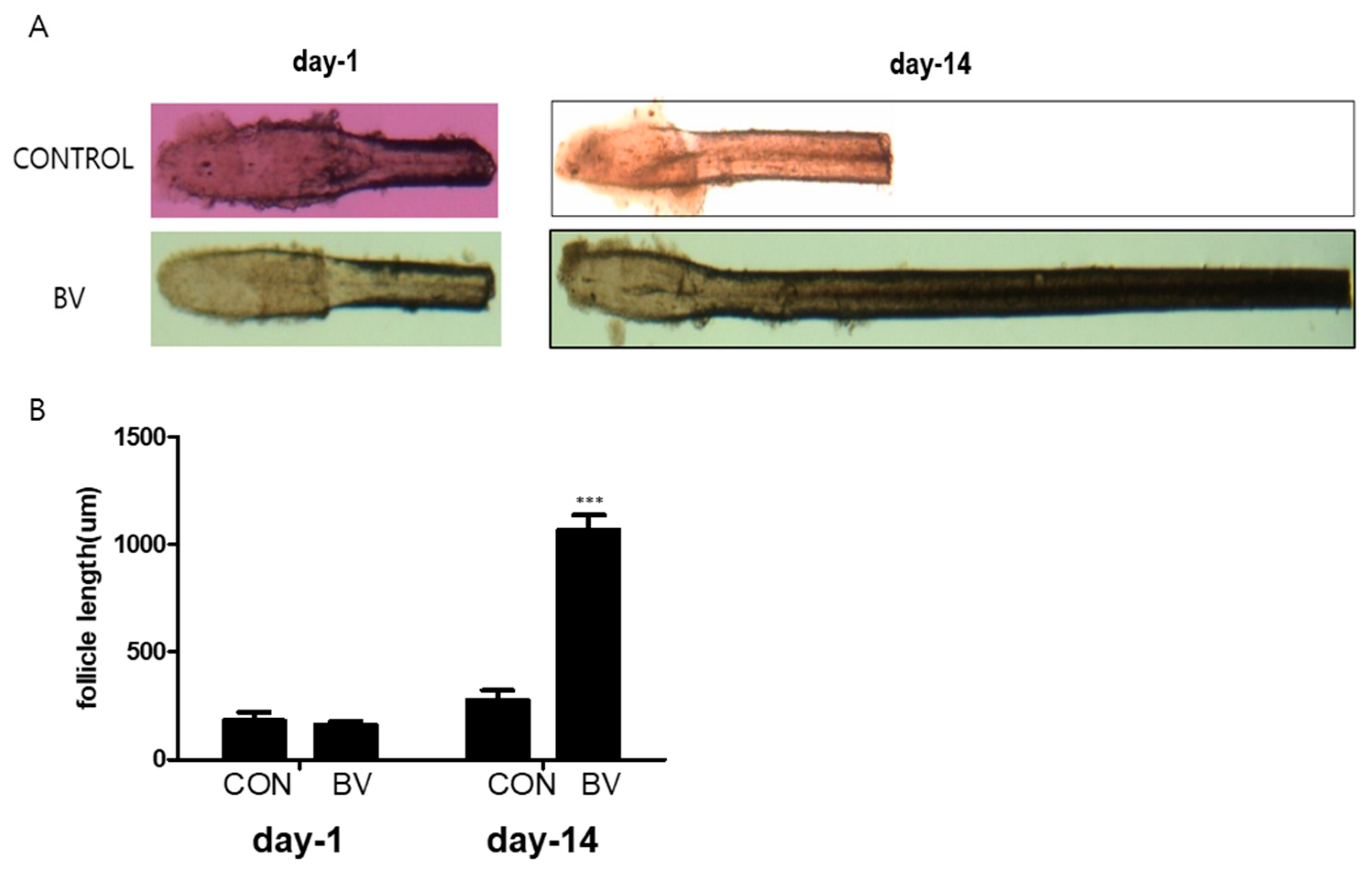
2.2. BV-Pretreated ASCs Promote Hair Growth In Vivo
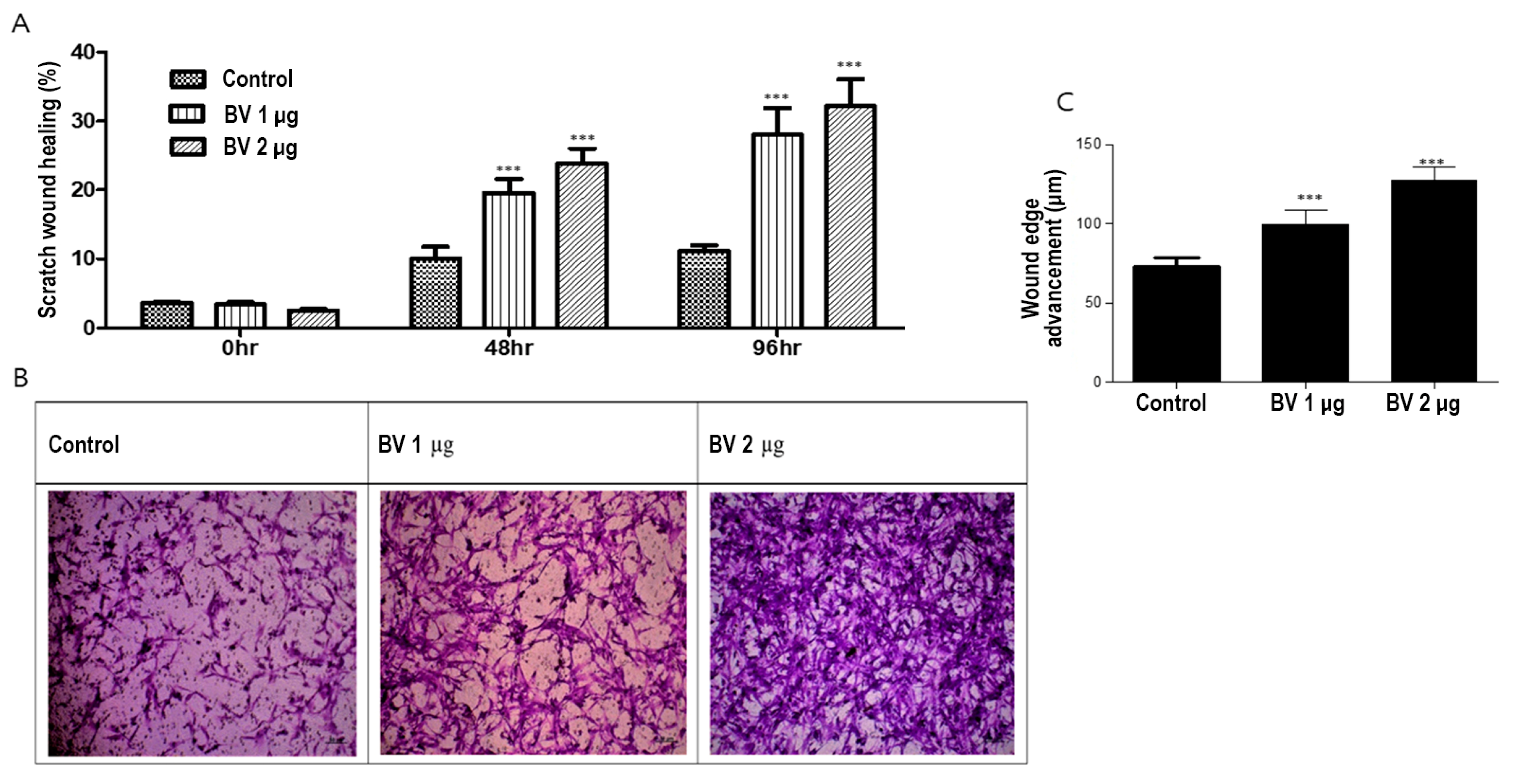
2.3. BV Can Induce Cell Migration in ASCs
2.4. PDGF-C, ECGF, FGF-1, FGF-6, and ALP Induce Hair Growth
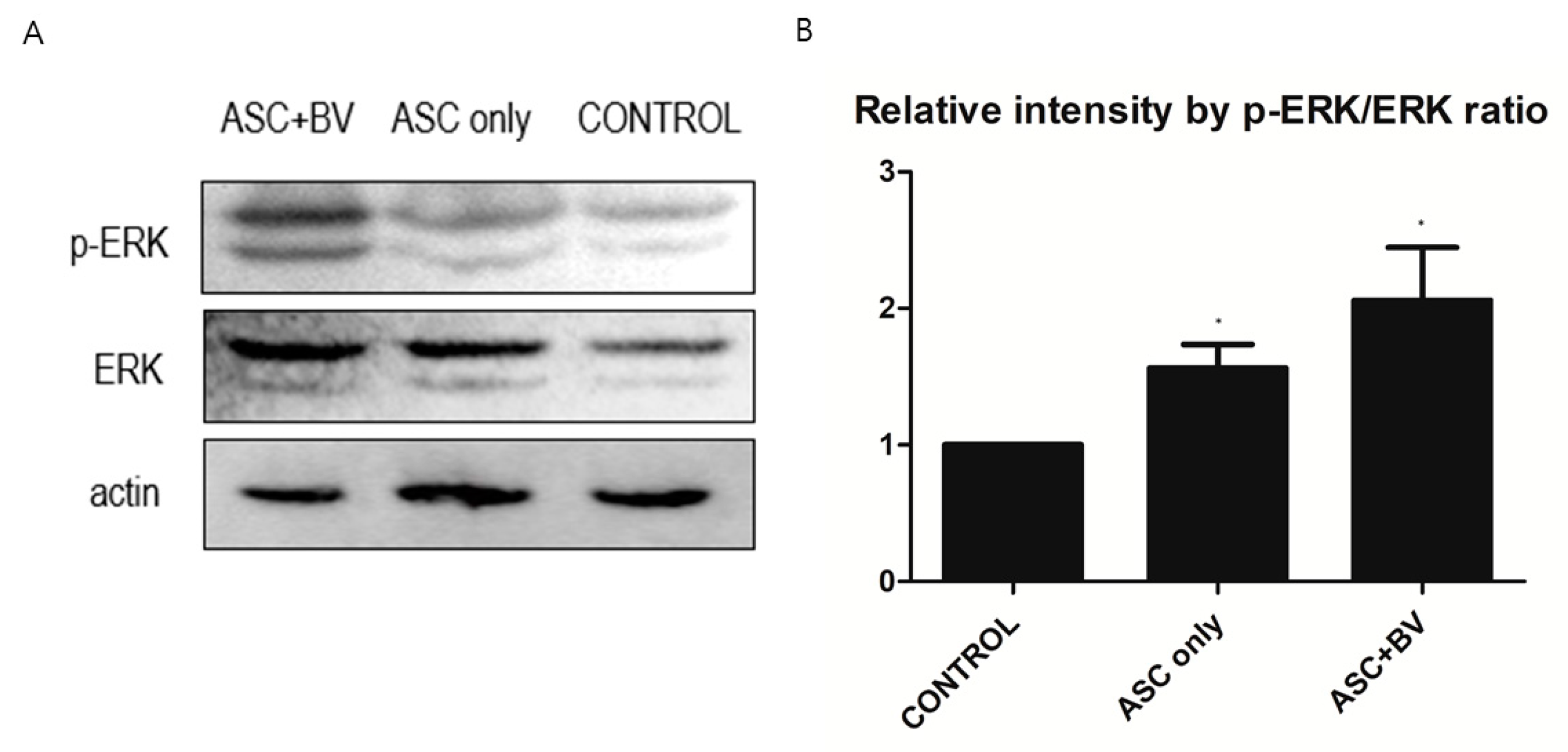
2.5. BV Can Activate ERK Pathway in ASCs
3. Discussion
4. Conclusions
5. Materials and Methods
5.1. Adiopose-Derived Stem Cells
5.2. Cell Culture
5.3. Cytotoxicity Assessment
5.4. Scratch Migration Assay
5.5. Boyden Chamber Assay
5.6. Cell Growth Assay
5.7. Animal Experiment
5.8. Hematoxylin/Eosin and Immunofluorescence Staining
5.9. Ki+ and DAPI Staining
5.10. RNA Extraction and Quantitative RT-PCR
5.11. Western Blot
5.12. Statistical Analysis
5.13. AI Language Model Usage
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Appendix A. Primer Sequence In Vivo Study
| Primer | Forward | Reverse |
|---|---|---|
| mouse GAPDH | GTG CTG AGT ATG TCG TGG AG | ATT TCT CGT GGT TCA CAC CC |
| mouse FGF1 | TTA TAC GGC TCG CAG ACA CC | GGC CCA CAA ACC AGT TCT TC |
| mouse FGF2 | GGC TCT ACT GCA AGA ACG G | GGC ACA CAC TCC CTT GAT AG |
| mouse FGF6 | GCC ATG AAC AGT AAA GGA AGA G | CTG ACT CGT AGG CGT TGT AG |
| mouse FGF10 | CGG GAC CAA GAA TGA AGA CTG | TAG TTG CTG TTG ATG GCT TTG |
| mouse TRPV1 | CAC CAC GGC TGC TTA CTA TC | AGA CTC CTC CTG ACA CAG AC |
| mouse TRPV4 | GTG GTC TCC TAT CTG TGT GC | CTC CTG TGA AGA GCG TGA TG |
| mouse TRPV6 | GAG AGT ATG GTC TTG GCG AC | CCA TCT TGT TGC TGG AAG GC |
| mouse CXCL1 | GCT GGG ATT CAC CTC AAG AAC | TGT GGC TAT GAC TTC GGT TTG |
| mouse CXCL9 | AGG CAC GAT CCA CTA CAA ATC | GGC AGG TTT GAT CTC CGT TC |
| mouse CXCL10 | GAT CCC TCT CGC AAG GAC G | TCG TGG CAA TGA TCT CAA CAC |
| mouse CXCL12 | CAG AGC CAA CGT CAA GCA TC | TTC TTG TTC TTC AGC CGT GC |
| mouse SOX2 | CTC TGC ACA TGA AGG AGC AC | CGG GAA GCG TGT ACT TAT CC |
| mouse ALP | AGC TCA ACA CCA ATG TAG CC | AAT TTG TCC ATC TCC AGC CG |
| mouse PDGFA | GCT CGA AGT CAG ATC CAC AG | TCT CGG GCA CAT GGT TAA TG |
| mouse PDGFB | GAC TCC GTA GAT GAA GAT GGG | AGA TGA GCT TTC CAA CTC GAC |
| mouse PDGFC | GAC AGC CTC TAC AAG CCA AC | CCC GTA TGG ACA CTG AGA AG |
| mouse PDGFD | AAG ATT TCC AAC CGG AAG CAG | ATG GAG AGT GAT AGG ACA CCC |
| Mouse PDECGF | GGG CAT CTG AGA GAA GCA G | TGT CTC TTC TAG GTT CAT TCC C |
References
- Ji, S.; Zhu, Z.; Sun, X.; Fu, X. Functional hair follicle regeneration: An updated review. Signal Transduct. Target. Ther. 2021, 6, 66. [Google Scholar] [CrossRef]
- Wu, P.; Jiang, T.-X.; Lei, M.; Chen, C.-K.; Li, S.-M.H.; Widelitz, R.B.; Chuong, C.-M. Cyclic growth of dermal papilla and regeneration of follicular mesenchymal components during feather cycling. Development 2021, 148, dev198671. [Google Scholar] [CrossRef]
- Choi, N.; Shin, S.; Song, S.U.; Sung, J.-H. Minoxidil promotes hair growth through stimulation of growth factor release from adipose-derived stem cells. Int. J. Mol. Sci. 2018, 19, 691. [Google Scholar] [CrossRef]
- Korta, D.Z.; Christiano, A.M.; Bergfeld, W.; Duvic, M.; Ellison, A.; Fu, J.; Harris, J.E.; Hordinsky, M.K.; King, B.; Kranz, D. Alopecia areata is a medical disease. J. Am. Acad. Dermatol. 2018, 78, 832–834. [Google Scholar] [CrossRef] [PubMed]
- Hosking, A.-M.; Juhasz, M.; Atanaskova Mesinkovska, N. Complementary and alternative treatments for alopecia: A comprehensive review. Ski. Appendage Disord. 2019, 5, 72–89. [Google Scholar] [CrossRef] [PubMed]
- Suchonwanit, P.; Iamsumang, W.; Rojhirunsakool, S. Efficacy of topical combination of 0.25% finasteride and 3% minoxidil versus 3% minoxidil solution in female pattern hair loss: A randomized, double-blind, controlled study. Am. J. Clin. Dermatol. 2019, 20, 147–153. [Google Scholar] [CrossRef]
- Sung, S.-H.; Kim, J.-W.; Han, J.-E.; Shin, B.-C.; Park, J.-K.; Lee, G. Animal venom for medical usage in pharmacopuncture in Korean medicine: Current status and clinical implication. Toxins 2021, 13, 105. [Google Scholar] [CrossRef] [PubMed]
- Bava, R.; Castagna, F.; Musella, V.; Lupia, C.; Palma, E.; Britti, D. Therapeutic use of bee venom and potential applications in veterinary medicine. Vet. Sci. 2023, 10, 119. [Google Scholar] [CrossRef]
- Gu, H.; Han, S.M.; Park, K.-K. Therapeutic effects of apamin as a bee venom component for non-neoplastic disease. Toxins 2020, 12, 195. [Google Scholar] [CrossRef]
- Nilforoushzadeh, M.A.; Aghdami, N.; Taghiabadi, E. Effects of adipose-derived stem cells and platelet-rich plasma exosomes on the inductivity of hair dermal papilla cells. Cell J. 2021, 23, 576. [Google Scholar]
- Wu, J.; Yang, Q.; Wu, S.; Yuan, R.; Zhao, X.; Li, Y.; Wu, W.; Zhu, N. Adipose-derived stem cell exosomes promoted hair regeneration. Tissue Eng. Regen. Med. 2021, 18, 685–691. [Google Scholar] [CrossRef] [PubMed]
- Kazi, T.; Nagata, A.; Nakagawa, T.; Matsuzaki, T.; Inui, S. Dermal papilla cell-derived extracellular vesicles increase hair inductive gene expression in adipose stem cells via β-catenin activation. Cells 2022, 11, 202. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.H.; Kim, W.K.; Sung, Y.K.; Kwack, M.H.; Song, S.Y.; Choi, J.S.; Sung, J.H. The molecular mechanism underlying the proliferating and preconditioning effect of vitamin C on adipose-derived stem cells. Stem Cells Dev. 2014, 23, 1364–1376. [Google Scholar] [CrossRef] [PubMed]
- Won, C.H.; Yoo, H.G.; Kwon, O.S.; Sung, M.Y.; Kang, Y.J.; Chung, J.H.; Park, B.S.; Sung, J.-H.; Kim, W.S.; Kim, K.H. Hair growth promoting effects of adipose tissue-derived stem cells. J. Dermatol. Sci. 2010, 57, 134–137. [Google Scholar] [CrossRef] [PubMed]
- González, R.; Moffatt, G.; Hagner, A.; Sinha, S.; Shin, W.; Rahmani, W.; Chojnacki, A.; Biernaskie, J. Platelet-derived growth factor signaling modulates adult hair follicle dermal stem cell maintenance and self-renewal. NPJ Regen. Med. 2017, 2, 11. [Google Scholar] [CrossRef] [PubMed]
- Lachgar, S.; Moukadiri, H.; Jonca, F.; Charveron, M.; Bouhaddioui, N.; Gall, Y.; Bonafe, J.L.; Plouët, J. Vascular endothelial growth factor is an autocrine growth factor for hair dermal papilla cells. J. Investig. Dermatol. 1996, 106, 17–23. [Google Scholar] [CrossRef] [PubMed]
- Lin, W.H.; Xiang, L.J.; Shi, H.X.; Zhang, J.; Jiang, L.P.; Cai, P.T.; Lin, Z.L.; Lin, B.B.; Huang, Y.; Zhang, H.L.; et al. Fibroblast growth factors stimulate hair growth through β-catenin and Shh expression in C57BL/6 mice. BioMed Res. Int. 2015, 2015, 730139. [Google Scholar]
- Lee, S.-H.; Yoon, J.; Shin, S.H.; Zahoor, M.; Kim, H.J.; Park, P.J.; Park, W.-S.; Min, D.S.; Kim, H.-Y.; Choi, K.-Y. Valproic acid induces hair regeneration in murine model and activates alkaline phosphatase activity in human dermal papilla cells. PLoS ONE 2012, 7, e34152. [Google Scholar] [CrossRef]
- Park, S.; Erdogan, S.; Hwang, D.; Hwang, S.; Han, E.H.; Lim, Y.-H. Bee venom promotes hair growth in association with inhibiting 5α-reductase expression. Biol. Pharm. Bull. 2016, 39, 1060–1068. [Google Scholar] [CrossRef]
- Tu, W.-C.; Wu, C.-C.; Hsieh, H.-L.; Chen, C.-Y.; Hsu, S.-L. Honeybee venom induces calcium-dependent but caspase-independent apoptotic cell death in human melanoma A2058 cells. Toxicon 2008, 52, 318–329. [Google Scholar] [CrossRef]
- Festa, E.; Fretz, J.; Berry, R.; Schmidt, B.; Rodeheffer, M.; Horowitz, M.; Horsley, V. Adipocyte lineage cells contribute to the skin stem cell niche to drive hair cycling. Cell 2011, 146, 761–771. [Google Scholar] [CrossRef]
- Li, L.; Clevers, H. Coexistence of quiescent and active adult stem cells in mammals. Science 2010, 327, 542–545. [Google Scholar] [CrossRef]
- Xu, Z.C.; Zhang, Q.; Li, H. Differentiation of human hair follicle stem cells into endothelial cells induced by vascular endothelial and basic fibroblast growth factors. Mol. Med. Rep. 2014, 9, 204–210. [Google Scholar] [CrossRef][Green Version]
- Madaan, A.; Verma, R.; Singh, A.T.; Jaggi, M. Review of hair follicle dermal papilla cells as in vitro screening model for hair growth. Int. J. Cosmet. Sci. 2018, 40, 429–450. [Google Scholar] [CrossRef]
- Gomez-Salinero, J.M.; Rafii, S. Endothelial cell adaptation in regeneration. Science 2018, 362, 1116–1117. [Google Scholar] [CrossRef]
- Hsu, Y.-C.; Pasolli, H.A.; Fuchs, E. Dynamics between stem cells, niche, and progeny in the hair follicle. Cell 2011, 144, 92–105. [Google Scholar] [CrossRef]
- Danilenko, D.M.; Ring, B.D.; Pierce, G.F. Growth factors and cytokines in hair follicle development and cycling: Recent insights from animal models and the potentials for clinical therapy. Mol. Med. Today 1996, 2, 460–467. [Google Scholar] [CrossRef]
- Kawano, M.; Komi-Kuramochi, A.; Asada, M.; Suzuki, M.; Oki, J.; Jiang, J.; Imamura, T. Comprehensive analysis of FGF and FGFR expression in skin: FGF18 is highly expressed in hair follicles and capable of inducing anagen from telogen stage hair follicles. J. Investig. Dermatol. 2005, 124, 877–885. [Google Scholar] [CrossRef] [PubMed]
- Rendl, M.; Lewis, L.; Fuchs, E. Molecular dissection of mesenchymal–epithelial interactions in the hair follicle. PLoS Biol. 2005, 3, e331. [Google Scholar] [CrossRef] [PubMed]
- Hagiwara, H.; Hiruma, Y.; Inoue, A.; Yamaguchi, A.; Hirose, S. Deceleration by angiotensin II of the differentiation and bone formation of rat calvarial osteoblastic cells. J. Endocrinol. 1998, 156, 543–550. [Google Scholar] [CrossRef] [PubMed]
- Kwack, M.H.; Kang, B.M.; Kim, M.K.; Kim, J.C.; Sung, Y.K. Minoxidil activates β-catenin pathway in human dermal papilla cells: A possible explanation for its anagen prolongation effect. J. Dermatol. Sci. 2011, 62, 154–159. [Google Scholar] [CrossRef] [PubMed]
- Almalki, S.G.; Agrawal, D.K. ERK signaling is required for VEGF-A/VEGFR2-induced differentiation of porcine adipose-derived mesenchymal stem cells into endothelial cells. Stem Cell Res. Ther. 2017, 8, 113. [Google Scholar] [CrossRef] [PubMed]
- Zhang, X.; Shen, R.; Shu, Z.; Zhang, Q.; Chen, Z. S100A12 promotes inflammation and apoptosis in ischemia/reperfusion injury via ERK signaling in vitro study using PC12 cells. Pathol. Int. 2020, 70, 403–412. [Google Scholar] [CrossRef] [PubMed]


Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kim, J.H.; Kim, T.Y.; Goo, B.; Park, Y. Bee Venom Stimulates Growth Factor Release from Adipose-Derived Stem Cells to Promote Hair Growth. Toxins 2024, 16, 84. https://doi.org/10.3390/toxins16020084
Kim JH, Kim TY, Goo B, Park Y. Bee Venom Stimulates Growth Factor Release from Adipose-Derived Stem Cells to Promote Hair Growth. Toxins. 2024; 16(2):84. https://doi.org/10.3390/toxins16020084
Chicago/Turabian StyleKim, Jung Hyun, Tae Yoon Kim, Bonhyuk Goo, and Yeoncheol Park. 2024. "Bee Venom Stimulates Growth Factor Release from Adipose-Derived Stem Cells to Promote Hair Growth" Toxins 16, no. 2: 84. https://doi.org/10.3390/toxins16020084
APA StyleKim, J. H., Kim, T. Y., Goo, B., & Park, Y. (2024). Bee Venom Stimulates Growth Factor Release from Adipose-Derived Stem Cells to Promote Hair Growth. Toxins, 16(2), 84. https://doi.org/10.3390/toxins16020084




