2.1. Site-Directed Mutagenesis, Expression, and Purification of Mutant EF Proteins
The pQE30-EF(H351A) and pQE30-EF(H351R) expression plasmids containing mutations of His351 to alanine (A) or arginine (R), respectively, within the catalytic site of EF [
8] were confirmed by sequencing. According to previous reports, circular dichroism profiles indicate that the mutant proteins do not exhibit any gross structural differences compared to the wild-type EF [
8] and remain competent in binding to PA [
13]. The wild-type and mutant proteins were expressed as
N-terminal (His)
6-tagged fusion proteins in
E. coli M15 strain. Following induction with isopropyl-1-thio-β-
d-galactopyranoside (IPTG), the cells were harvested and the periplasmic fraction was isolated. After Ni-NTA affinity chromatography and SP-sepharose cation-exchange column, wild-type EF or mutant proteins were isolated from other
E. coli cell constituents. Purified wild-type and mutant proteins displayed identical purification characteristics (>90%) with respect to their molecular mass (89 kDa) and final yields (5–6 mg/L of culture) and sodium dodecyl sulfate-polyacrylamide gel electrophoresis (SDS–PAGE) analysis confirmed that homogenous preparations of structurally stable mutants were obtained (
Figure 1).
Figure 1.
Purification of wild-type EF, EF(H351A) and EF(H351R) proteins expressed in E. coli M15 strain. (a) SDS-PAGE analysis of Ni-NTA affinity purified wild-type EF: M, molecular weight marker; lane 1, total soluble fraction; lane 2, elution fraction; (b) SDS-PAGE analysis of SP-sepharose cation-exchange chromatography purified wild-type EF: M, molecular weight marker; lane 1, total soluble fraction; lane 2, elution fraction. Similar results were obtained following purification of EF(H351A) and EF(H351R) proteins using the same procedures; (c) SDS-PAGE (10% gel) analysis of 1 μg of purified EF, EF(H351A), and EF(H351R): M, molecular weight marker; Lane 1, EF; lane 2, EF(H351A); lane 3, EF(H351R).
Figure 1.
Purification of wild-type EF, EF(H351A) and EF(H351R) proteins expressed in E. coli M15 strain. (a) SDS-PAGE analysis of Ni-NTA affinity purified wild-type EF: M, molecular weight marker; lane 1, total soluble fraction; lane 2, elution fraction; (b) SDS-PAGE analysis of SP-sepharose cation-exchange chromatography purified wild-type EF: M, molecular weight marker; lane 1, total soluble fraction; lane 2, elution fraction. Similar results were obtained following purification of EF(H351A) and EF(H351R) proteins using the same procedures; (c) SDS-PAGE (10% gel) analysis of 1 μg of purified EF, EF(H351A), and EF(H351R): M, molecular weight marker; Lane 1, EF; lane 2, EF(H351A); lane 3, EF(H351R).
2.2. Adenylate Cyclase Activity of Wild-Type and Mutant Proteins
EF is a calmodulin-dependent AC, which elevates intracellular cAMP levels in the presence of PA. The cAMP level in CHO cells was measured within 2 h exposed to the expressed proteins accompanied with PA, and to forskolin or SQ22536. As expected, forskolin, an AC activator, could improve the intracellular cAMP level, while SQ22536, an AC inhibitor, decreased the cAMP level (
Figure 2a). To verify the activity of the expressed proteins, CHO cells were treated with varying concentrations (0.5–500 ng/mL) of EF, EF(H351R), and EF(H351A) in the presence of 1 μg/mL PA for 2 h. A sharp increase in cAMP was observed in a dose-dependent manner when CHO cells were exposed to EF combined with PA, while a relatively mild but not significant increase when exposed to EF(H351R) or EF(H351A) (
Figure 2b). Non-cellular reaction systems containing MgCl
2, calmodulin, CaCl
2, BSA, and ATP are commonly used to analyze the activity of EF or its mutants, but the detection sensitivity can be variable due to the difference in formulas. Previous evaluation of the EF(H351A) and EF(H351R) mutants in an in vitro AC assay showed a 200-fold reduction in the catalytic activity of these two mutant proteins compared to that of the native EF [
5]. However, in another study, the AC activity of EF(H351A) was below the detectable limits of a similar, but not identical, analysis system [
7]. So whether the substitution of His351 by alanine or arginine of EF remains AC activity of physiological and pathological impact may need further evaluation.
Figure 2.
In vitro adenylate cyclase activity assay of EF, EF(H351A), or EF(H351R). (a) CHO cells were incubated for 2 h with forskolin (50 μg/mL and 5 μg/mL), SQ22536(50 μg/mL and 5 μg/mL) or DMSO control. Intracellular cAMP was measured using an EIA kit; (b) CHO cells were incubated for 2 h with PA (1 μg/mL) and EF, EF(H351A), or EF(H351R) at the indicated concentrations. Intracellular cAMP was measured using an EIA kit. An average of 13 pmol of cAMP/107 CHO cells was detected in control cells (untreated cells or cells treated with only one toxin component). Results are expressed as mean ± standard error. Statistical significance: (** p < 0.01, * p < 0.05).
Figure 2.
In vitro adenylate cyclase activity assay of EF, EF(H351A), or EF(H351R). (a) CHO cells were incubated for 2 h with forskolin (50 μg/mL and 5 μg/mL), SQ22536(50 μg/mL and 5 μg/mL) or DMSO control. Intracellular cAMP was measured using an EIA kit; (b) CHO cells were incubated for 2 h with PA (1 μg/mL) and EF, EF(H351A), or EF(H351R) at the indicated concentrations. Intracellular cAMP was measured using an EIA kit. An average of 13 pmol of cAMP/107 CHO cells was detected in control cells (untreated cells or cells treated with only one toxin component). Results are expressed as mean ± standard error. Statistical significance: (** p < 0.01, * p < 0.05).
2.3. Footpad Edema Induced by Wild-Type and Mutant Toxins
Although ET is an important toxin relevant to anthrax pathogenesis, it plays a more comprehensive role in the aspects of bacterial infection that are not related to lethality. ET causes edema through targeting of an unknown cellular target (not endothelial cells) in the early stages of infection. Subcutaneous injection of ET induces edema in experimental animals [
8,
14] and a mouse model of footpad edema induction has been used to evaluate the effect of antibodies against ET [
12]; therefore, we used this model to further investigate the activity of EF(H351A) and EF(H351R).
Forskolin is an AC activator which elevates the intracellular cAMP level, and SQ22536 is an AC inhibitor which decreases the intracellular cAMP level. In this study, 25 μg of each activator or inhibitor was injected subcutaneously into the footpads of mice. Local edema was induced by forskolin with an increase of cAMP in the tissue, but not by SQ22536 (
Figure 3a,b), demonstrating that edema was induced by the elevation of intracellular cAMP.
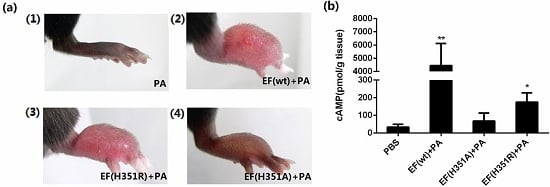
The progression of lesions induced in the mouse footpad by subcutaneous ET injection was then observed. When 500 ng EF was administered in combination with 1000 ng PA, rapid footpad swelling was observed in experimental animals (
Figure 3c), with peak swelling (footpad thickness) and ulceration detected after approximately 53 h passed (
Figure 3e). Modest edema was observed following application of identical amounts of mutant toxins, EF(H351A) or EF(H351R) in combination with PA (
Figure 3c–e). Meanwhile, the cAMP levels in footpads were increased about 100 times by native ET, four times by ET (H351R), and two times by ET (H351A) in 2 h of treatment (
Figure 3d). No edema was observed following administration of EF, EF(H351A), EF(H351R), or PA (data not shown). The lesion caused by native or mutant ET challenge gradually healed within two weeks without any intervention (data not shown). The edema induction effects of varying amounts of wild-type or mutant toxins were also studied, and the peak edema values observed at 24 h and 53 h after injection were analyzed (
Figure 3f,g). Although neither the wild-type nor mutant EFs induced edema at very low doses (0.5 ng and 5 ng), toxic effects emerged when the challenge dose was increased to 50 ng. Significant differences (
p < 0.05) observed in the degree of edema caused by wild-type EF and the mutants, while there were no significant differences in the effects caused by EF(H351R) and EF(H351A). When the toxin challenge doses were elevated from 500 ng to 2500 ng, the degree of edema caused by the wild-type EF did not increase, while that caused by EF(H351R) increased such there was no significant difference between the effects of the wild-type EF and EF(H351R) (
Figure 3d,e). These results indicate that the sensitivity of this mouse footpad edema model is sufficient to distinguish not only the weak toxicity of EF(H351R) and EF(H351A), but also increased toxicity of EF(H351R) compared with EF(H351A).
Figure 3.
ET-induced footpad edema in mice. 25 μg of forskolin or SQ22536 dissolved in 25 μL DMSO were injected subcutaneously into the footpad; 25 μL of DMSO was administered as the solvent control. (a) Footpad thickness was measured at 0, 5, 17, and 49 h after injection; (b) cAMP levels in the treated footpads tissue. Mice were injected subcutaneously with 500 ng EF and 1000 ng PA (in 25 μL PBS), 500 ng EF(H351R) and 1000 ng PA or 500 ng EF(H351A) and 1000 ng PA; (c) Footpad thickness was measured at 0, 12, 24, 53, and 76 h after injection; (d) cAMP levels in the treated footpads tissue; (e) Comparison of right footpads treated with 1000 ng PA only (1), 500 ng EF and 1000 ng PA (2), 500 ng EF(H351R) and 1000 ng PA (3), or 500 ng EF(H351A) and 1000 ng PA (4). Mutant EF(H351A) induced modest edema, while wild-type EF caused much higher levels of edema; (f) and (g) Serial doses (0.5, 5, 50, 500, and 2500 ng) of EF, EF(H351R) or EF(H351A) in combination with double concentrations of PA were subcutaneously injected into the footpad; the degree edema at 24 h and 53 h after injection are shown. Significant differences between the groups are indicated by p-values. The p-values of the indicated groups versus the solvent control group are shown. Data represent the mean ± standard error of the mean based on n = 3 mice per treatment. At least two biological replicates were performed for each experiment. (** p < 0.01, * p < 0.05).
Figure 3.
ET-induced footpad edema in mice. 25 μg of forskolin or SQ22536 dissolved in 25 μL DMSO were injected subcutaneously into the footpad; 25 μL of DMSO was administered as the solvent control. (a) Footpad thickness was measured at 0, 5, 17, and 49 h after injection; (b) cAMP levels in the treated footpads tissue. Mice were injected subcutaneously with 500 ng EF and 1000 ng PA (in 25 μL PBS), 500 ng EF(H351R) and 1000 ng PA or 500 ng EF(H351A) and 1000 ng PA; (c) Footpad thickness was measured at 0, 12, 24, 53, and 76 h after injection; (d) cAMP levels in the treated footpads tissue; (e) Comparison of right footpads treated with 1000 ng PA only (1), 500 ng EF and 1000 ng PA (2), 500 ng EF(H351R) and 1000 ng PA (3), or 500 ng EF(H351A) and 1000 ng PA (4). Mutant EF(H351A) induced modest edema, while wild-type EF caused much higher levels of edema; (f) and (g) Serial doses (0.5, 5, 50, 500, and 2500 ng) of EF, EF(H351R) or EF(H351A) in combination with double concentrations of PA were subcutaneously injected into the footpad; the degree edema at 24 h and 53 h after injection are shown. Significant differences between the groups are indicated by p-values. The p-values of the indicated groups versus the solvent control group are shown. Data represent the mean ± standard error of the mean based on n = 3 mice per treatment. At least two biological replicates were performed for each experiment. (** p < 0.01, * p < 0.05).
![Toxins 08 00035 g003a]()
![Toxins 08 00035 g003b]()
2.4. Systemic Effects of Wild-type and His351 EF Mutants
Systemic administration of ET
in vivo results in a wide variety of organ lesions caused by the AC activity of EF, while PA alone is not toxic [
15]. Subcutaneous injection of ET into the mouse footpad causes dramatic local edema, while systemic administration induces liver edema, fluid accumulation in the intestinal lumen, diverse tissue damage, and death with a minimum lethal dose for mice between 20 and 30 μg [
14]. EF(H351A) is considered to be a potential prophylactic agent for anthrax based on its lack of toxicity in sensitive cell lines and its non-lethal effects when administered to mice in combination with an equal dose of PA [
13]. However, the present study indicates that His531 mutation does not result in complete abolition of the toxicity of EF and therefore, caution should be exercised in using such mutants to prevent anthrax infection.
In this study, we challenged each mouse with 50 μg of the wild-type or mutant EF in combination with 100 μg PA, and mice challenged with the individual proteins served as controls. The survival curves shown in
Figure 4a show that EF(H351A) or EF(H351R) in combination with PA were not lethal and did not cause significant signs of illness, while 100% mortality was observed in mice within 12 h of challenge with an equal dose of wild-type EF combined with PA. When the doses of toxins was doubled (100 μg wild-type or mutant EF in combination with 200 μg PA), all animals in the EF and PA treated group died with 3 h of administration, while none died in the groups treated with EF(H351A) or EF(H351R) in combination with PA. However, histological analysis revealed some unexpected lesions induced by EF(H351A) and EF(H351R). The liver is considered to be the key target of ET-induced lethality and deletion of anthrax toxin receptor CMG2 from hepatocytes in mice led to remarkable resistance to ET [
14]. In this study, necrotic areas in the liver were observed both in mice treated with the wild-type and mutant ET, while the lesions induced by EF(H351A) were relatively mild (
Figure 4b). Furthermore, serious damage was found in the lungs of wild-type EF treated mice but not in those treated with the mutant toxins. cAMP levels within the liver and lung tissues in 2 h after the injection of the wild-type and mutant ET were also determined (
Figure 4c,d). The cAMP amount in liver but not in lung was elevated by ET (H351A), while that in both of the organs were greatly increased by wild-type ET.
Figure 4.
Systemic effects of wild-type and His351 EF mutants. (a) Survival rate of groups of three C57BL/6 mice challenged with wild-type or mutant EF (50 μg) and PA (100 μg). Mice challenged with EF (wild-type) + PA died within 12 h, while none died in the groups challenged with EF(H351R) + PA or EF(H351A) + PA, or in the control groups (EF or PA only). Mice were injected intravenously with PBS, EF (100 μg) and PA (200 μg), or EF(H351A) (100 μg) and PA (200 μg); (b) Histology of liver and lung tissues of mice treated with ET or ET(H351A). Mice treated with EF in combination with PA died within 3 h, and were immediately necropsied. Mice treated EF(H351A) in combination with PA were euthanized 6 h later by CO2 inhalation and immediately necropsied. In the liver, sinusoid stenosis (left arrow), scattered necrotic lesions, and inflammatory cell infiltration (right arrow) were found in mice treated with EF(H351A) in combination with PA, while more marked necrotic lesions and inflammatory cell infiltration (right arrow) accompanied by hemorrhaging (left arrow) were identified in mice treated with EF in combination with and PA. In the lungs, hemorrhaging, edema, and inflammatory cell infiltration (arrows) were found around the trachea and bronchus in mice treated with EF in combination with PA but not in those treated with EF(H351A) in combination with PA; (c) and (d) cAMP levels in liver and lung tissues of mice treated with ET or ET (H351A). Mice were euthanized by CO2 inhalation 2 h after injection and immediately necropsied. Significant differences between the groups are indicated by p-values. The p-values of the indicated groups versus the solvent control group are shown. Data represent the mean ± standard error of the mean based on n = 3 mice per treatment. At least two biological replicates were performed for each experiment. (** p < 0.01, * p < 0.05).
Figure 4.
Systemic effects of wild-type and His351 EF mutants. (a) Survival rate of groups of three C57BL/6 mice challenged with wild-type or mutant EF (50 μg) and PA (100 μg). Mice challenged with EF (wild-type) + PA died within 12 h, while none died in the groups challenged with EF(H351R) + PA or EF(H351A) + PA, or in the control groups (EF or PA only). Mice were injected intravenously with PBS, EF (100 μg) and PA (200 μg), or EF(H351A) (100 μg) and PA (200 μg); (b) Histology of liver and lung tissues of mice treated with ET or ET(H351A). Mice treated with EF in combination with PA died within 3 h, and were immediately necropsied. Mice treated EF(H351A) in combination with PA were euthanized 6 h later by CO2 inhalation and immediately necropsied. In the liver, sinusoid stenosis (left arrow), scattered necrotic lesions, and inflammatory cell infiltration (right arrow) were found in mice treated with EF(H351A) in combination with PA, while more marked necrotic lesions and inflammatory cell infiltration (right arrow) accompanied by hemorrhaging (left arrow) were identified in mice treated with EF in combination with and PA. In the lungs, hemorrhaging, edema, and inflammatory cell infiltration (arrows) were found around the trachea and bronchus in mice treated with EF in combination with PA but not in those treated with EF(H351A) in combination with PA; (c) and (d) cAMP levels in liver and lung tissues of mice treated with ET or ET (H351A). Mice were euthanized by CO2 inhalation 2 h after injection and immediately necropsied. Significant differences between the groups are indicated by p-values. The p-values of the indicated groups versus the solvent control group are shown. Data represent the mean ± standard error of the mean based on n = 3 mice per treatment. At least two biological replicates were performed for each experiment. (** p < 0.01, * p < 0.05).
![Toxins 08 00035 g004]()
Together, these results clearly demonstrate that the mutated EF(H351A), which is generally used as a “loss” of AC activity control, retains limited AC activity and is associated with systemic toxicity, although the effects are significantly weaker than those of the wild-type EF. As reported in a previous study [
13], mutants such as EF(H351A) which retain PA-binding ability represent potential toxin decoys, but our findings demonstrate that the liver toxicity of these mutants should be carefully considered for such applications. Furthermore, our results indicate that the mice footpad edema model is a highly sensitive method to determine the remaining AC activity of these mutants.