Fundamental Study of Decellularization Method Using Cyclic Application of High Hydrostatic Pressure
Abstract
:1. Introduction
2. Materials and Methods
2.1. NB1RGB Cell Culture
2.2. High Hydrostatic Pressure Application to NB1RGB Cell Suspension
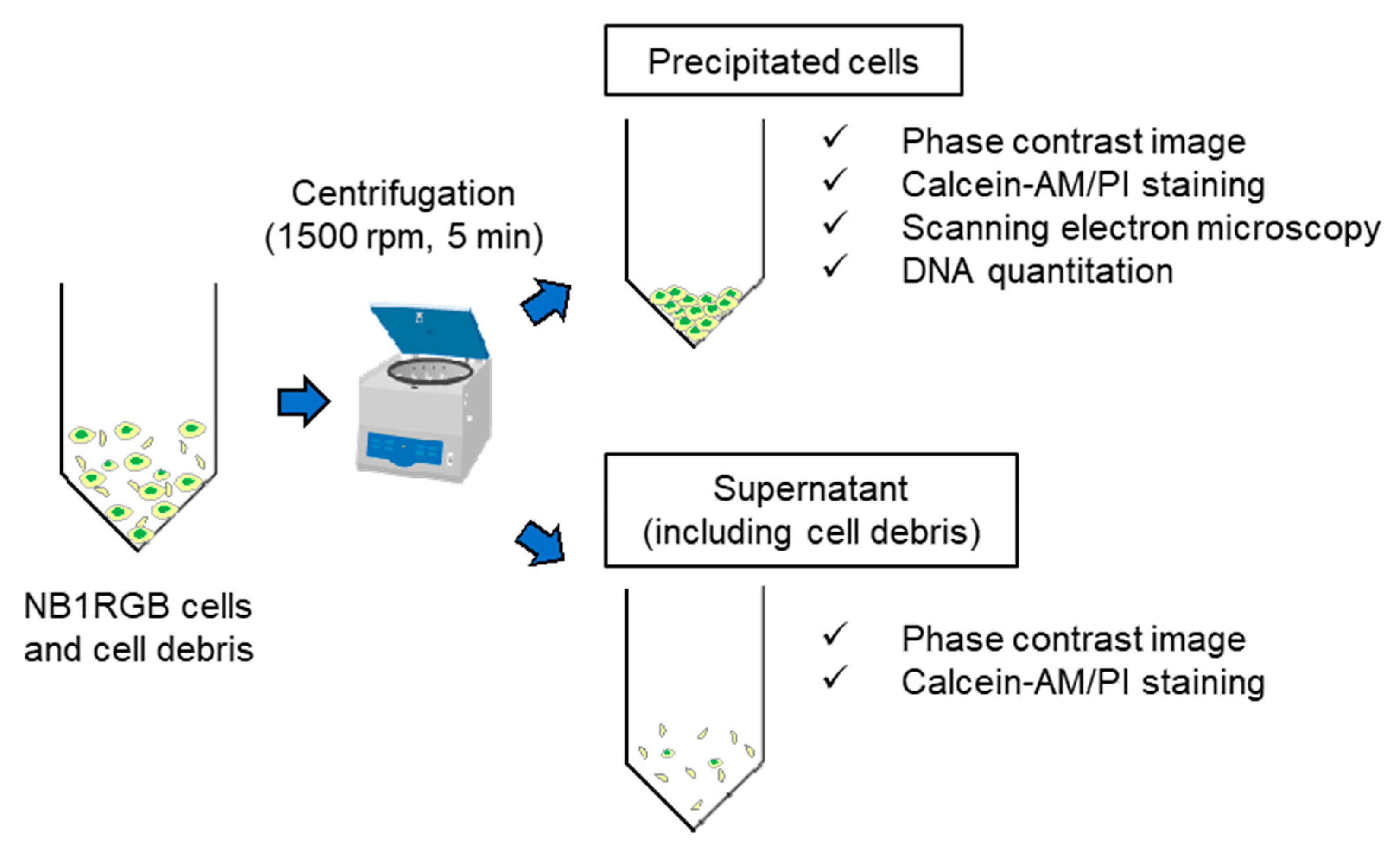
2.3. Biochemical Characterization
2.4. Statistical Analysis
3. Results
3.1. Effect of Cyclic Application of HHP on Cell Morphology
3.2. Effect of Cyclic Application of HHP on Cell Viability
3.3. Effect of Cyclic Application of HHP on Cell Proliferation
4. Discussion
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Ikada, Y. Challenges in tissue engineering. J. R. Soc. Interface 2006, 3, 589–601. [Google Scholar] [CrossRef]
- Yusen, Q.; Xingzhi, L.; Xichao, Z.; Hongbo, Z.; Wen, Z.; Wei, X.; Guoqing, P.; Wenguo, C.; Hélder, A.S.; Qin, S. Gelatin Templated Polypeptide Co-Cross-Linked Hydrogel for Bone Regeneration. Adv. Healthc. Mater. 2020, 9, e1901239. [Google Scholar]
- Dulnik, J.; Kołbuk, D.; Denis, P.; Sajkiewicz, P. The effect of a solvent on cellular response to PCL/gelatin and PCL/collagen electrospun nano-fibers. Eur. Polym. J. 2018, 104, 147–156. [Google Scholar] [CrossRef]
- Shaghayegh, H.J.; Jamal, A.; Anousheh, Z.K.; Elham, V. In vitro hemocompatibility and cytocompatibility of a three-layered vascular scaffold fabricated by sequential electrospinning of PCL, collagen, and PLLA nanofibers. J. Biomater. Appl. 2016, 31, 438–449. [Google Scholar]
- Wang, L.; Dormer, N.H.; Bonewald, L.F.; Detamore, M.S. Osteogenic differentiation of human umbilical cord mesenchymal stromal cells in polyglycolic acid scaffolds. Tissue Eng. Part A 2010, 16, 1937–1948. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bahcecioglu, G.; Hasirci, N.; Hasirci, V. Cell behavior on the alginate-coated PLLA/PLGA scaffolds. Int. J. Biol. Macromol. 2019, 124, 444–450. [Google Scholar] [CrossRef] [PubMed]
- Zhang, K.; Huang, D.; Yan, Z.; Wang, C. Heparin/collagen encapsulating nerve growth factor multilayers coated aligned PLLA nanofibrous scaffolds for nerve tissue engineering. J. Biomed. Mater. Res. A 2017, 105, 1900–1910. [Google Scholar] [CrossRef]
- Zha, F.; Chen, W.; Zhang, L.; Yu, D. Electrospun natural polymer and its composite nanofibrous scaffolds for nerve tissue engineering. J. Biomater. Sci. Polym. Ed. 2020, 31, 519–548. [Google Scholar] [CrossRef]
- Boni, R.; Ali, A.; Shavandi, A.; Clarkson, A.N. Current and novel polymeric biomaterials for neural tissue engineering. J. Biomed. Sci. 2018, 25, 90. [Google Scholar] [CrossRef] [Green Version]
- Yu, L.; Dean, K.; Li, L. Polymer blends and composites from renewable resources. Prog. Polym. Sci. 2006, 31, 576–602. [Google Scholar] [CrossRef]
- Park, K.S.; Kim, B.J.; Lih, E.; Park, W.; Lee, S.H.; Joung, Y.K.; Han, D.K. Versatile effects of magnesium hydroxide nanoparticles in PLGA scaffold-mediated chondrogenesis. Acta Biomater. 2018, 73, 204–216. [Google Scholar] [CrossRef] [PubMed]
- Mohammadi Nasr, S.; Rabiee, N.; Hajebi, S.; Ahmadi, S.; Fatahi, Y.; Hosseini, M.; Bagherzadeh, M.; Ghadiri, A.M.; Rabiee, M.; Jajarmi, V.; et al. Biodegradable Nanopolymers in Cardiac Tissue Engineering: From Concept Towards Nanomedicine. Int. J. Nanomed. 2020, 15, 4205–4224. [Google Scholar] [CrossRef] [PubMed]
- Andrea, P.; Elena, S.; Silvia, B.; Francesca, G.; Veronica, M.; Raffaele, D.C. Tissue-Engineered Grafts from Human Decellularized Extracellular Matrices: A Systematic Review and Future Perspectives. Int. J. Mol. Sci. 2018, 19, 4117. [Google Scholar]
- Pu, W.; Ren, J.; Chen, Y.; Shu, J.; Cui, L.; Han, Y.; Xi, J.; Pei, X.; Yue, W.; Han, Y. Injectable human decellularized adipose tissue hydrogel containing stem cells enhances wound healing in mouse. Colloids Surf. A Phys. Eng. Asp. 2020, 604, 125268. [Google Scholar] [CrossRef]
- David, M.H.; Elisabeth, M.O.; Gwen, E.O.; Dina, O.H.; Scott, M.G.; Chris, M.B.; Craig, M.N.; Russell, T.K.; Joseph, P.V. The retention of extracellular matrix proteins and angiogenic and mitogenic cytokines in a decellularized porcine dermis. Biomaterials 2010, 31, 6730–6737. [Google Scholar]
- Luciana, C.W.; Paula, H.S.; Leticia, K.; Victoria, S.R.; Lúcia, N.; Francisco, D.A.C.; Felipe, F.T. Histological and Biomechanical Characteristics of Human Decellularized Allograft Heart Valves After Eighteen Months of Storage in Saline Solution. Biopreserv. Biobank. 2020, 18, 90–101. [Google Scholar]
- Stefanie, I.; Karl, H.S.; Janos, M.K.; Heinz, R.; Richard, O.C.O. Harnessing Human Decellularized Blood Vessel Matrices and Cellular Construct Implants to Promote Bone Healing in an Ex Vivo Organotypic Bone Defect Model. Adv. Healthc. Mater. 2019, 8, e1800088. [Google Scholar]
- Peter, M.C.; Thomas, W.G.; Stephen, F.B. An overview of tissue and whole organ decellularization processes. Biomaterials 2011, 32, 3233–3243. [Google Scholar]
- Wei, S.; Karsten, B.; Sze, W.T.; Yun, W.L. Rapid and Detergent-Free Decellularization of Cartilage. Tissue Eng. Part C Methods 2020, 26, 201–206. [Google Scholar]
- Erik, A.R.; Luiz, F.Q.; Carolina, K.V.N.; Bruno, S.F.S. Advances in Hepatic Tissue Bioengineering with Decellularized Liver Bioscaffold. Stem. Cells Int. 2019, 2019, 2693189. [Google Scholar]
- Yu, S.K.; Marjan, M.; Anthony, J.M.; Antonios, G.M. Applications of decellularized extracellular matrix in bone and cartilage tissue engineering. Bioeng. Transl. Med. 2018, 4, 83–95. [Google Scholar]
- Wonil, H.; Narendra, K.S.; Joeng, J.K.; Hyeonji, K.; Byoung, S.K.; Ju, Y.P.; Jinah, J.; Dong, W.C. Directed differential behaviors of multipotent adult stem cells from decellularized tissue/organ extracellular matrix bioinks. Biomaterials 2019, 224, 119496. [Google Scholar]
- Liao, J.; Joyce, E.M.; Sacks, M.S. Effects of decellularization on the mechanical and structural properties of the porcine aortic valve leaflet. Biomaterials 2008, 29, 1065–1074. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Anna, G.; Yong, Y. Decellularization Strategies for Regenerative Medicine: From Processing Techniques to Applications. Biomed. Res. Int. 2017, 2017, 9831534. [Google Scholar]
- Susanne, P.R.; Sina, M.G.; Amelie, P.; Ina, E.; Sandra, H.; Janina, B. Automated freeze-thaw cycles for decellularization of tendon tissue—A pilot study. BMC Biotechnol. 2017, 17, 13. [Google Scholar]
- Janina, B.; Ina, E.; Dagmar, B.; Johannes, K.; Cornelia, K.; Bastian, P.; Karsten, W.; Walter, B. Freeze-thaw cycles enhance decellularization of large tendons. Tissue Eng. Part C Methods 2014, 20, 276–284. [Google Scholar]
- Yaniv, Z.; David, K.; Natalie, L.; Jonathan, L.; Elad, M. Optimization of Irreversible Electroporation Protocols for In-vivo Myocardial Decellularization. PLoS ONE 2016, 11, e0165475. [Google Scholar]
- Mary, P.; Elad, M.; Boris, R. Nonthermal irreversible electroporation for tissue decellularization. J. Biomech. Eng. 2010, 132, 091003. [Google Scholar]
- Jun, N.; Seiichi, F.; Tsuyoshi, K.; Kwangoo, N.; Tetsuya, H.; Akio, K. Porcine radial artery decellularization by high hydrostatic pressure. J. Tissue Eng. Regen. Med. 2015, 9, E144–E151. [Google Scholar]
- Erna, G.S.; Keita, Y.; Yasushi, H.; Masanori, A.; Osamu, Y.; Akio, K.; Yutaka, O.; Shigeru, S.; Takashi, U.; Katsuko, S.F. Application of detergents or high hydrostatic pressure as decellularization processes in uterine tissues and their subsequent effects on in vivo uterine regeneration in murine models. PLoS ONE 2014, 9, e103201. [Google Scholar]
- Potekhin, S.A.; Senin, A.A.; Abdurakhmanov, N.N.; Tiktopulo, E.I. High pressure stabilization of collagen structure. Biochim. Biophys. Acta 2009, 1794, 1151–1158. [Google Scholar] [CrossRef] [PubMed]
- Shoji, S.; Masayasu, A.; Nobuko, N.; Naoko, Y.; Masako, O.; Rikimaru, H. Effects of hydrostatic pressure on the ultrastructure and leakage of internal substances in the yeast. Appl. Microbiol. Biotechnol. 1993, 40, 123–131. [Google Scholar]
- Ulmer, H.M.; Gänzle, M.G.; Vogel, R.F. Effects of high pressure on survival and metabolic activity of Lactobacillus plantarum TMW1.460. Appl. Envrion. Microbiol. 2000, 66, 3966–3973. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Dooley, C.; Cafferky, D.; Lee, T.C.; Taylor, D. Fatigue failure of osteocyte cellular processes: Implications for the repair of bone. Eur. Cell Mater. 2014, 27, 39–48. [Google Scholar] [CrossRef] [PubMed]
- Kim, Y.J.; Sah, R.L.; Doong, J.Y.; Grodzinsky, A.J. Fluorometric assay of DNA in cartilage explants using Hoechst 33258. Anal. Biochem. 1988, 174, 168–176. [Google Scholar] [CrossRef]
- Forsey, R.W.; Chaudhuri, J.B. Validity of DNA analysis to determine cell numbers in tissue engineering scaffolds. Biotechnol. Lett. 2009, 31, 819–823. [Google Scholar] [CrossRef] [PubMed]
- Mahara, A.; Morimoto, N.; Sakuma, T.; Fujisato, T.; Yamaoka, T. Complete cell killing by applying high hydrostatic pressure for acellular vascular graft preparation. Biomed. Res. Int. 2014, 2014, 379607. [Google Scholar] [CrossRef]








| Maximum Pressure (MPa) | Application Condition | |
|---|---|---|
| 150-static | 150 | static |
| 200-static | 200 | static |
| 250-static | 250 | static |
| 150-cyclic | 150 | cyclic |
| 200-cyclic | 200 | cyclic |
| 250-cyclic | 250 | cyclic |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zemmyo, D.; Yamamoto, M.; Miyata, S. Fundamental Study of Decellularization Method Using Cyclic Application of High Hydrostatic Pressure. Micromachines 2020, 11, 1008. https://doi.org/10.3390/mi11111008
Zemmyo D, Yamamoto M, Miyata S. Fundamental Study of Decellularization Method Using Cyclic Application of High Hydrostatic Pressure. Micromachines. 2020; 11(11):1008. https://doi.org/10.3390/mi11111008
Chicago/Turabian StyleZemmyo, Daiki, Masashi Yamamoto, and Shogo Miyata. 2020. "Fundamental Study of Decellularization Method Using Cyclic Application of High Hydrostatic Pressure" Micromachines 11, no. 11: 1008. https://doi.org/10.3390/mi11111008





