Simple Fabrication of Solid-State Nanopores on a Carbon Film
Abstract
:1. Introduction
2. Materials and Methods
2.1. Experimental Materials
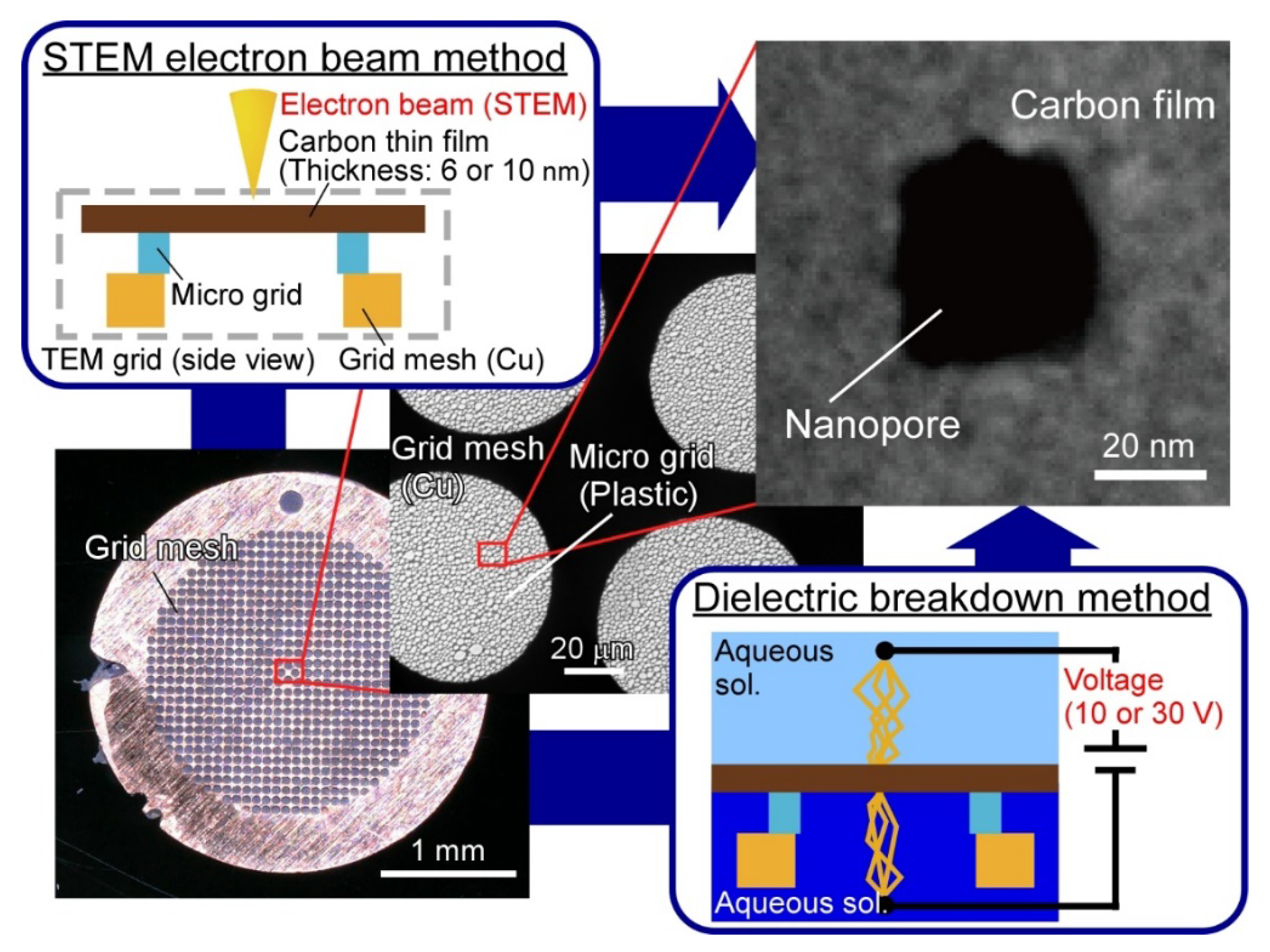
2.2. Fabrication of Nanopores by STEM EB Method
2.3. Fabrication of Nanopores by DB Method
2.4. Channel Current Measurements and Data Analysis
3. Results
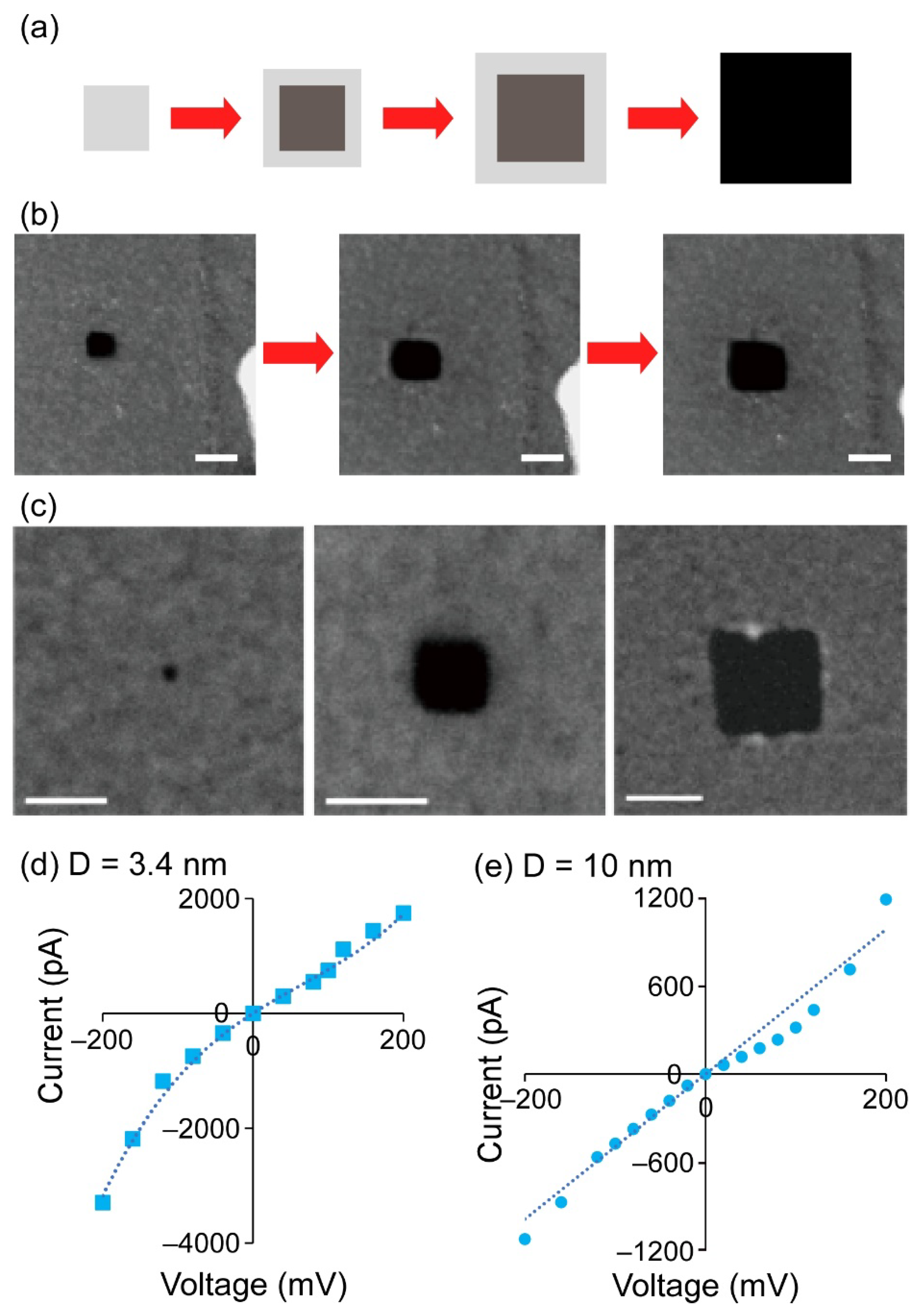
3.1. Nanopores Fabricated by the STEM EB Method
3.2. Nanopores Fabricated by the DB Method
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Gu, L.-Q.; Braha, O.; Conlan, S.; Cheley, S.; Bayley, H. Stochastic Sensing of Organic Analytes by a Pore-Forming Protein Containing a Molecular Adapter. Nature 1999, 398, 686–690. [Google Scholar] [CrossRef] [PubMed]
- Shi, W.; Friedman, A.K.; Baker, L.A. Nanopore Sensing. Anal. Chem. 2017, 89, 157–188. [Google Scholar] [CrossRef] [Green Version]
- Liu, P.; Kawano, R. Recognition of Single-Point Mutation Using a Biological Nanopore. Small Methods 2020, 4, 2000101. [Google Scholar] [CrossRef]
- Miyagi, M.; Takiguchi, S.; Hakamada, K.; Yohda, M.; Kawano, R. Single Polypeptide Detection Using a Translocon EXP2 Nanopore. Proteomics 2021, 2100070. [Google Scholar] [CrossRef]
- Kasianowicz, J.J.; Brandin, E.; Branton, D.; Deamer, D.W. Characterization of Individual Polynucleotide Molecules Using a Membrane Channel. Proc. Natl. Acad. Sci. USA 1996, 93, 13770–13773. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ameur, A.; Kloosterman, W.P.; Hestand, M.S. Single-Molecule Sequencing: Towards Clinical Applications. Trends Biotechnol. 2019, 37, 72–85. [Google Scholar] [CrossRef]
- Vlassiouk, I.; Smirnov, S.; Siwy, Z. Ionic Selectivity of Single Nanochannels. Nano Lett. 2008, 8, 1978–1985. [Google Scholar] [CrossRef]
- Macha, M.; Marion, S.; Nandigana, V.V.R.; Radenovic, A. 2D Materials as an Emerging Platform for Nanopore-Based Power Generation. Nat. Rev. Mater. 2019, 4, 588–605. [Google Scholar] [CrossRef]
- Akeson, M.; Branton, D.; Kasianowicz, J.J.; Brandin, E.; Deamer, D.W. Microsecond Time-Scale Discrimination among Polycytidylic Acid, Polyadenylic Acid, and Polyuridylic Acid as Homopolymers or as Segments Within Single RNA Molecules. Biophys. J. 1999, 77, 3227–3233. [Google Scholar] [CrossRef] [Green Version]
- Derrington, I.M.; Butler, T.Z.; Collins, M.D.; Manrao, E.; Pavlenok, M.; Niederweis, M.; Gundlach, J.H. Nanopore DNA Sequencing with MspA. Proc. Natl. Acad. Sci. USA 2010, 107, 16060–16065. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Watanabe, H.; Gubbiotti, A.; Chinappi, M.; Takai, N.; Tanaka, K.; Tsumoto, K.; Kawano, R. Analysis of Pore Formation and Protein Translocation Using Large Biological Nanopores. Anal. Chem. 2017, 89, 11269–11277. [Google Scholar] [CrossRef] [PubMed]
- Kawano, R. Synthetic Ion Channels and DNA Logic Gates as Components of Molecular Robots. ChemPhysChem 2018, 19, 359–366. [Google Scholar] [CrossRef] [PubMed]
- Ohara, M.; Takinoue, M.; Kawano, R. Nanopore Logic Operation with DNA to RNA Transcription in a Droplet System. ACS Synth. Biol. 2017, 6, 1427–1432. [Google Scholar] [CrossRef] [PubMed]
- Takiguchi, S.; Kawano, R. Nanopore Decoding for a Hamiltonian Path Problem. Nanoscale 2021, 13, 6192–6200. [Google Scholar] [CrossRef]
- Matsushita, M.; Shoji, K.; Takai, N.; Kawano, R. Biological Nanopore Probe: Probing of Viscous Solutions in a Confined Nanospace. J. Phys. Chem. B 2020, 124, 2410–2416. [Google Scholar] [CrossRef]
- Steinbock, L.J.; Otto, O.; Chimerel, C.; Gornall, J.; Keyser, U.F. Detecting DNA Folding with Nanocapillaries. Nano Lett. 2010, 10, 2493–2497. [Google Scholar] [CrossRef] [PubMed]
- Ma, T.; Janot, J.-M.; Balme, S. Track-Etched Nanopore/Membrane: From Fundamental to Applications. Small Methods 2020, 4, 2000366. [Google Scholar] [CrossRef]
- Dekker, C. Solid-State Nanopores. Nat. Nanotechnol. 2007, 2, 209–215. [Google Scholar] [CrossRef]
- Lee, K.; Park, K.-B.; Kim, H.-J.; Yu, J.-S.; Chae, H.; Kim, H.-M.; Kim, K.-B. Recent Progress in Solid-State Nanopores. Adv. Mater. 2018, 30, 1704680. [Google Scholar] [CrossRef] [PubMed]
- Li, J.; Stein, D.; McMullan, C.; Branton, D.; Aziz, M.J.; Golovchenko, J.A. Ion-Beam Sculpting at Nanometre Length Scales. Nature 2001, 412, 166–169. [Google Scholar] [CrossRef] [Green Version]
- Emmrich, D.; Beyer, A.; Nadzeyka, A.; Bauerdick, S.; Meyer, J.C.; Kotakoski, J.; Gölzhäuser, A. Nanopore Fabrication and Characterization by Helium Ion Microscopy. Appl. Phys. Lett. 2016, 108, 163103. [Google Scholar] [CrossRef] [Green Version]
- Storm, A.J.; Chen, J.H.; Ling, X.S.; Zandbergen, H.W.; Dekker, C. Fabrication of Solid-State Nanopores with Single-Nanometre Precision. Nat. Mater. 2003, 2, 537–540. [Google Scholar] [CrossRef]
- Kim, M.J.; Wanunu, M.; Bell, D.C.; Meller, A. Rapid Fabrication of Uniformly Sized Nanopores and Nanopore Arrays for Parallel DNA Analysis. Adv. Mater. 2006, 18, 3149–3153. [Google Scholar] [CrossRef]
- Briggs, K.; Kwok, H.; Tabard-Cossa, V. Automated Fabrication of 2-Nm Solid-State Nanopores for Nucleic Acid Analysis. Small 2014, 10, 2077–2086. [Google Scholar] [CrossRef] [PubMed]
- Kwok, H.; Briggs, K.; Tabard-Cossa, V. Nanopore Fabrication by Controlled Dielectric Breakdown. PLoS ONE 2014, 9, e92880. [Google Scholar] [CrossRef] [Green Version]
- Goto, Y.; Yanagi, I.; Matsui, K.; Yokoi, T.; Takeda, K. Integrated Solid-State Nanopore Platform for Nanopore Fabrication via Dielectric Breakdown, DNA-Speed Deceleration and Noise Reduction. Sci. Rep. 2016, 6, 31324. [Google Scholar] [CrossRef]
- Kim, M.J.; McNally, B.; Murata, K.; Meller, A. Characteristics of Solid-State Nanometre Pores Fabricated Using a Transmission Electron Microscope. Nanotechnology 2007, 18, 205302. [Google Scholar] [CrossRef] [Green Version]
- Xie, P.; Xiong, Q.; Fang, Y.; Qing, Q.; Lieber, C.M. Local Electrical Potential Detection of DNA by Nanowire–Nanopore Sensors. Nat. Nanotechnol. 2012, 7, 119–125. [Google Scholar] [CrossRef] [Green Version]
- Storm, A.J.; Storm, C.; Chen, J.; Zandbergen, H.; Joanny, J.-F.; Dekker, C. Fast DNA Translocation through a Solid-State Nanopore. Nano Lett. 2005, 5, 1193–1197. [Google Scholar] [CrossRef] [Green Version]
- Venkatesan, B.M.; Dorvel, B.; Yemenicioglu, S.; Watkins, N.; Petrov, I.; Bashir, R. Highly Sensitive, Mechanically Stable Nanopore Sensors for DNA Analysis. Adv. Mater. 2009, 21, 2771–2776. [Google Scholar] [CrossRef] [Green Version]
- Venkatesan, B.M.; Shah, A.B.; Zuo, J.M.; Bashir, R. DNA Sensing Using Nano-Crystalline Surface Enhanced Al2O3 Nanopore Sensors. Adv. Funct. Mater. 2010, 20, 1266–1275. [Google Scholar] [CrossRef] [Green Version]
- Garaj, S.; Hubbard, W.; Reina, A.; Kong, J.; Branton, D.; Golovchenko, J.A. Graphene as a Subnanometre Trans-Electrode Membrane. Nature 2010, 467, 190–193. [Google Scholar] [CrossRef]
- Merchant, C.A.; Healy, K.; Wanunu, M.; Ray, V.; Peterman, N.; Bartel, J.; Fischbein, M.D.; Venta, K.; Luo, Z.; Johnson, A.T.C.; et al. DNA Translocation through Graphene Nanopores. Nano Lett. 2010, 10, 2915–2921. [Google Scholar] [CrossRef]
- Schneider, G.F.; Kowalczyk, S.W.; Calado, V.E.; Pandraud, G.; Zandbergen, H.W.; Vandersypen, L.M.K.; Dekker, C. DNA Translocation through Graphene Nanopores. Nano Lett. 2010, 10, 3163–3167. [Google Scholar] [CrossRef] [Green Version]
- Liu, S.; Lu, B.; Zhao, Q.; Li, J.; Gao, T.; Chen, Y.; Zhang, Y.; Liu, Z.; Fan, Z.; Yang, F.; et al. Boron Nitride Nanopores: Highly Sensitive DNA Single-Molecule Detectors. Adv. Mater. 2013, 25, 4549–4554. [Google Scholar] [CrossRef]
- Liu, K.; Feng, J.; Kis, A.; Radenovic, A. Atomically Thin Molybdenum Disulfide Nanopores with High Sensitivity for DNA Translocation. ACS Nano 2014, 8, 2504–2511. [Google Scholar] [CrossRef]
- Nayak, L.; Rahaman, M.; Giri, R. Surface Modification/Functionalization of Carbon Materials by Different Techniques: An Overview. In Carbon-Containing Polymer Composites; Rahaman, M., Khastgir, D., Aldalbahi, A.K., Eds.; Springer Series on Polymer and Composite Materials; Springer: Singapore, 2019; pp. 65–98. ISBN 9789811326882. [Google Scholar]
- Wanunu, M.; Meller, A. Chemically Modified Solid-State Nanopores. Nano Lett. 2007, 7, 1580–1585. [Google Scholar] [CrossRef] [PubMed]
- Gyurcsányi, R.E. Chemically-Modified Nanopores for Sensing. TrAC Trends Anal. Chem. 2008, 27, 627–639. [Google Scholar] [CrossRef]
- Ying, Y.-L.; Hu, Y.-X.; Gao, R.; Yu, R.-J.; Gu, Z.; Lee, L.P.; Long, Y.-T. Asymmetric Nanopore Electrode-Based Amplification for Electron Transfer Imaging in Live Cells. J. Am. Chem. Soc. 2018, 140, 5385–5392. [Google Scholar] [CrossRef] [PubMed]
- Hille, B. Ion Channels of Excitable Membranes, 3rd ed.; Sinauer: Sunderland, MA, USA, 2001; ISBN 978-0-87893-321-1. [Google Scholar]
- Siwy, Z.S. Ion-Current Rectification in Nanopores and Nanotubes with Broken Symmetry. Adv. Funct. Mater. 2006, 16, 735–746. [Google Scholar] [CrossRef]
- White, H.S.; Bund, A. Ion Current Rectification at Nanopores in Glass Membranes. Langmuir 2008, 24, 2212–2218. [Google Scholar] [CrossRef] [PubMed]
- Fragasso, A.; Schmid, S.; Dekker, C. Comparing Current Noise in Biological and Solid-State Nanopores. ACS Nano 2020, 14, 1338–1349. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wang, Y.; Ying, C.; Zhou, W.; de Vreede, L.; Liu, Z.; Tian, J. Fabrication of Multiple Nanopores in a SiN x Membrane via Controlled Breakdown. Sci. Rep. 2018, 8, 1234. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Yanagi, I.; Hamamura, H.; Akahori, R.; Takeda, K. Two-Step Breakdown of a SiN Membrane for Nanopore Fabrication: Formation of Thin Portion and Penetration. Sci. Rep. 2018, 8, 10129. [Google Scholar] [CrossRef]
- Briggs, K.; Charron, M.; Kwok, H.; Le, T.; Chahal, S.; Bustamante, J.; Waugh, M.; Tabard-Cossa, V. Kinetics of Nanopore Fabrication during Controlled Breakdown of Dielectric Membranes in Solution. Nanotechnology 2015, 26, 084004. [Google Scholar] [CrossRef]
- Zhang, Y.; Miyahara, Y.; Derriche, N.; Yang, W.; Yazda, K.; Capaldi, X.; Liu, Z.; Grutter, P.; Reisner, W. Nanopore Formation via Tip-Controlled Local Breakdown Using an Atomic Force Microscope. Small Methods 2019, 3, 1900147. [Google Scholar] [CrossRef]
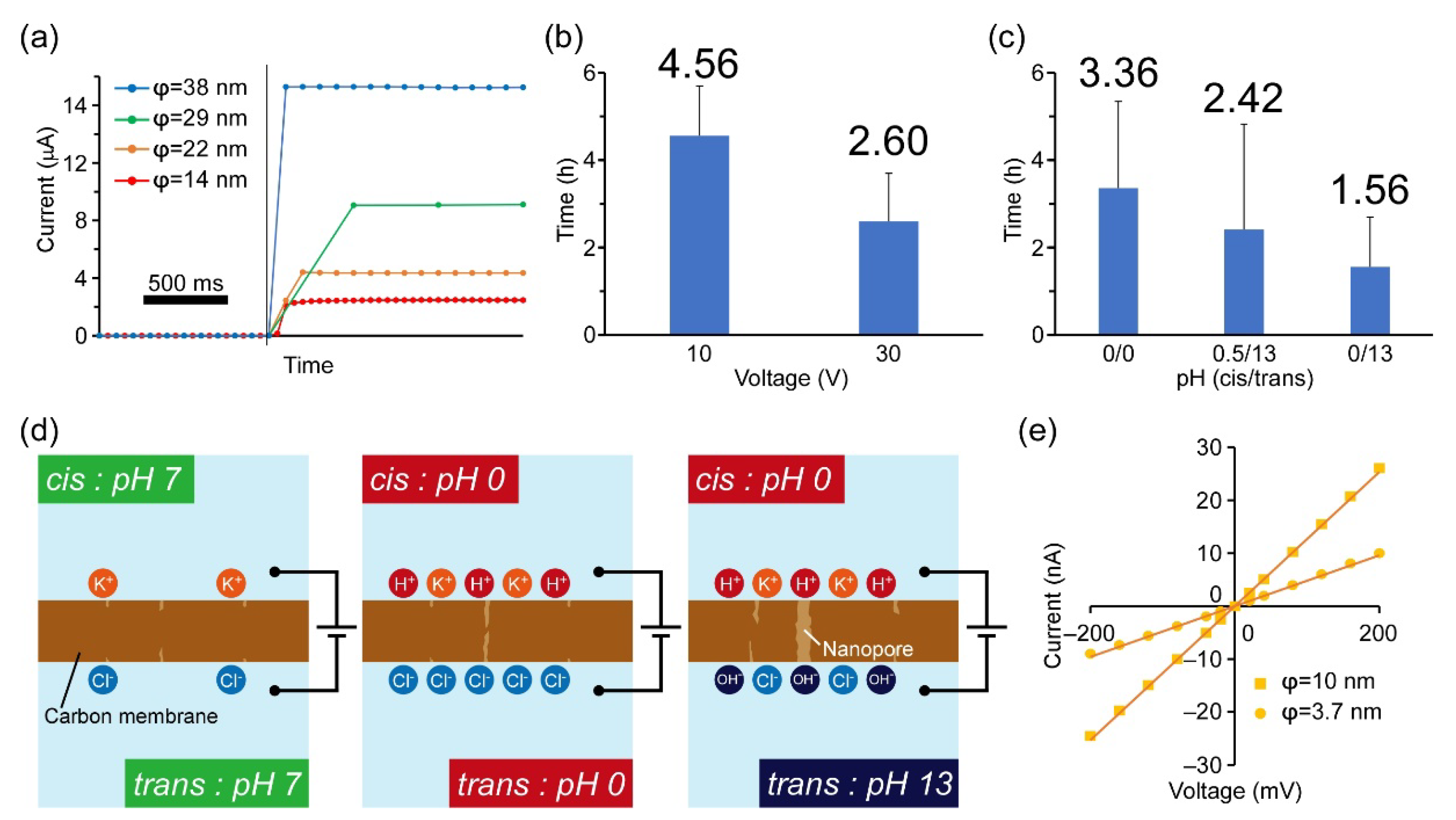
| Applied Voltage (V) | pH (Cis/Trans) | Thickness of the Membrane (nm) | Fabrication Time (h) |
|---|---|---|---|
| 10 | 0.5/0.5 | 6 | 4.56 ± 1.14 |
| 30 | 0.5/0.5 | 6 | 2.60 ± 1.10 |
| 10 | 0/0 | 10 | 3.36 ± 1.99 |
| 10 | 0.5/13 | 10 | 2.42 ± 2.41 |
| 10 | 0/13 | 10 | 1.56 ± 1.14 |
| Methods | Materials | Pore Size (nm) | Size Controllability | Thickness (nm) | Pre-Micro and Nano Fabrication | Fabrication Time (min) | Membrane Stability | REF. |
|---|---|---|---|---|---|---|---|---|
| TEM EB | Carbon film (Micro-grid) | 2~135 | Excellent | −10 | Not required | 2~90 | low | This work |
| DB | Carbon film (Micro-grid) | 3.7~1345 (75% of nanopores are less than 40 nm) | Poor | −10 | Not required | 90~300 | low | This work |
| TEM EB | Si3N4 | 2~20 | Excellent | 10- | Required | 0.5~ | high | [23] |
| DB | SiNx | 1~25 | Good | 10- | Required | −10 | high | [25] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Takai, N.; Shoji, K.; Maki, T.; Kawano, R. Simple Fabrication of Solid-State Nanopores on a Carbon Film. Micromachines 2021, 12, 1135. https://doi.org/10.3390/mi12091135
Takai N, Shoji K, Maki T, Kawano R. Simple Fabrication of Solid-State Nanopores on a Carbon Film. Micromachines. 2021; 12(9):1135. https://doi.org/10.3390/mi12091135
Chicago/Turabian StyleTakai, Natsumi, Kan Shoji, Tei Maki, and Ryuji Kawano. 2021. "Simple Fabrication of Solid-State Nanopores on a Carbon Film" Micromachines 12, no. 9: 1135. https://doi.org/10.3390/mi12091135
APA StyleTakai, N., Shoji, K., Maki, T., & Kawano, R. (2021). Simple Fabrication of Solid-State Nanopores on a Carbon Film. Micromachines, 12(9), 1135. https://doi.org/10.3390/mi12091135






