2.1. Kinetic Model of TEA Droplet Self-Ignition in Air at NPT Conditions
For a kinetic analysis of the self-ignition stage, it is necessary to draw up a scheme of elementary reactions. By definition, the primary reaction in the scheme should be a heterogeneous reaction that occurs when oxygen molecules available in air collide with TEA droplets. Such a reaction, leading to the self-ignition of a mixture occupying a limited volume, should be characterized by a sufficiently low activation energy. Presumably, this may be a heterogeneous reaction of the intrusion of an O
2 molecule to TEA with the formation of the (C
2H
5)
2Al–O–O–(C
2H
5) molecule directly in the collision of TEA and O
2 molecules
or by forming an intermediate complex Al(C
2H
5)
3O
2 according to the scheme [
17]
where
= 113 kcal/mol. Reaction (3) or (4) can be followed by the monomolecular decomposition reaction through two channels:
where
= −77.9 kcal/mol and
= −90.5 kcal/mol [
17]. Reactions (3 or 4) + (5) and (3 or 4) + (6) can be considered as exothermic bimolecular reactions. These reactions occur during collisions of gas-phase molecules with the surfaces of TEA droplets (heterogeneous reactions). In this case, volatile active radicals C
2H
5 and C
2H
5O enter the gas phase (air) and interact with oxygen, releasing heat and giving rise to other sequential and parallel reactions, the same as in the gas-phase kinetics of the oxidation, self-ignition, and combustion of light alkanes (methane, ethane, and butane) and their derivatives. Kinetic schemes, corresponding equations of chemical kinetics, algorithms, and codes that describe similar gas-phase processes exist, and they can be readily used as subroutines for the numerical solution of the problem under consideration. It is worth emphasizing that we consider only the initial stage of the self-ignition process, rather than the entire process of TEA droplet combustion. At this stage, the size and chemical composition of droplets, as well as the oxygen concentration in the gas, change only a little, and can be considered constant.
Based on this prerequisite, the following kinetic model of TEA self-ignition in air at NPT conditions is proposed. The rate constant of reactions (3 or 4) + (5) and (3 or 4) + (6) is approximated as
where
is the preexponential factor;
is the temperature; and
is the activation energy. The consumption rate of TEA molecules per unit volume of the mixture can be expressed by the formula
where
is the number of TEA molecules per unit volume;
is the number of oxygen molecules per unit volume;
is the thermal velocity of oxygen molecules;
is the number of collisions of oxygen molecules with a unit surface of a droplet per unit time;
is the surface area of a TEA droplet;
is the TEA droplet radius;
is the number of TEA droplets per unit volume;
is the reaction probability in one collision; and λ is the steric factor. This latter factor is unknown. Its value is probably in the range of 0.1–0.01. According to the kinetic theory of gases, the thermal velocity
at temperature
is
By definition, the derivative
can be also expressed as
where
is the thickness of the outer molecular monolayer in a TEA droplet;
is the volume of the outer molecular monolayer in a TEA droplet;
is the number of TEA molecules per unit droplet volume;
is the number of TEA molecules in the volume
. Substituting Equation (7) into Equation (8) and comparing Equation (8) with Equation (10), one obtains
Equation (11) has a simple physical meaning. The values of
and
are expressed in terms of the effective radius,
, of the TEA molecule:
,
. From here and from Equation (11), one obtains
where
is the effective collision cross-section. Formula (12) coincides with the definition of the preexponential factor in the thermal theory of the rate constants of bimolecular reactions in gases [
24] up to a factor of 2/3. It follows from Equations (7), (10), and (11) that
It follows from Equation (13) that the rate of reactions (3 or 4) + (5) and (3 or 4) + (6) depends on the TEA droplet size,
, the oxygen concentration in the environment,
, and the local instantaneous air temperature,
:
Therefore, it could be expected that the self-ignition delay of TEA spray in ambient air could be a function of the spray injection pressure, as the droplet diameter generally depends on the injection pressure: the higher the injection pressure, the smaller the droplet size and the shorter the self-ignition delay. According to Equation (14), the self-ignition delay is shorter if the environment contains more oxygen, and if the local instantaneous air temperature is higher.
In addition to the uncertainty in the value of
, the rate constant (7) contains an unknown activation energy
. There exist empirical formulae establishing the relationship between
and the heat of the exothermic reaction,
, in a linear approximation:
with positive parameters
and
. The values of
and
vary depending on the type (set) of reactions. These formulae include the well-known Polanyi–Semenov rule [
24]:
This rule, when applied to bimolecular reactions (3 or 4) + (5) and (3 or 4) + (6), gives, respectively,
As noted in [
21], the formulae such as (16) must be used with great caution. The same is true for estimates (17). They can only be considered as a rough approximation, which must be verified and refined experimentally. In experiments, the parameters of the reaction rate in Equation (13) are not measured directly. However, the induction period before the self-ignition of TEA droplets and some other kinetic and thermodynamic parameters can be measured, which depend on the rate constant (7). In this case, the activation energy
can be found by solving the inverse problem. To do this, one must first solve the direct problem, which consists in calculating the self-ignition delay with a variation in activation energy
.
2.2. Self-Ignition Delays for C2H5–Air and C2H5O–Air Mixtures
The mathematical statement of the problem is the statement of the standard problem of the self-ignition of a gas mixture [
25] with a given detailed kinetics [
26], which is supplemented by a heterogeneous mechanism for the formation of C
2H
5 or C
2H
5O radicals. The equations for the conservation of the energy and mass of the components have the form
where
is time;
is the number of components in the gas mixture;
is the mass fraction of the
th component;
is the heat capacity of the gas mixture at constant pressure;
is the density of the mixture;
is the heat release in chemical reactions;
is the component consumption in chemical reactions; and
is the formation of C
2H
5 or C
2H
5O in reaction (6) or (5), respectively. The system of Equations (18) and (19) is supplemented by the ideal gas equation of state, expressions for
and
[
27], and by the polynomial relationship for the heat capacity, while the polynomial coefficients are taken from [
28].
Compared to the standard problem formulation for gas mixture self-ignition, the expression for and all other considerations associated with this circumstance are new. It is assumed that the mixture is initially represented by pure air, and radicals C2H5 or C2H5O appear in the gas due to the heterogeneous reaction (6) or (5), respectively. Self-ignition delays depend on the rates of formation of C2H5 and C2H5O radicals in heterogeneous reactions (6) and (5) and on the rates of their interaction with oxygen in the gas phase. As both reactions, (6) and (5), are possible, for determining the effect of these radicals on the self-ignition delay, the problem must be solved for two options: (i) for a C2H5–air mixture and (ii) for a C2H5O–air mixture.
2.2.1. Option i: C2H5–Air Mixture
Here,
is the molecular mass of C
2H
5;
is the rate of change in the concentration (mol/cm
3/s) of C
2H
5 radicals in the heterogeneous reaction (6), which, in accordance with reaction (6) and Equation (13), satisfies the equation
2.2.2. Option ii: C2H5O–Air Mixture
For this option, the same system of equations is solved as for option i, but in Equations (20) and (21) everywhere in the rows and in the indices, C2H5 must be replaced by C2H5O. To solve the problem for these two options, a special computational code has been developed.
2.3. Experimental Setup
Before designing the experimental setup, it was implied that the self-ignition delay, , of the TEA/TEB spray in ambient air could be estimated experimentally either by measuring the time delay between the start of spray injection and the appearance of self-ignition luminosity, if TEA/TEB is sprayed in a short pulse mode, or by measuring the width of the dark zone between the injector nozzle face and the luminous combustion plume, if the TEA/TEB is sprayed continuously. It could be assumed that ignition occurs much later than the pulsed injection of TEA/TEB. During this time, TEA/TEB droplets slow down, causing the ignition to occur in a virtually quiescent and spatially homogeneous mixture. With an average droplet path of around 10 cm and a speed of escape from the injector nozzle of the order of 104 cm/s, the deceleration time is around 1 ms. This time is much shorter than the expected self-ignition delay time, even at the lowest estimated value of activation energy in Equation (17), kcal/mol. With the continuous spraying of TEA/TEB into the ambient air, one could expect the appearance of a quasi-stationary luminous combustion plume. In this case, TEA/TEB droplets ignite due to the air flow around them, and the self-ignition delay, , of the spray is determined by the time given to a droplet to enter the zone of the luminous combustion plume. The value of can be estimated experimentally based on the width, , of the dark zone between the injector nozzle face and luminous combustion plume measured along the spray axis. The value is related to and the speed of the TEA/TEB spray at the nozzle exit, , as .
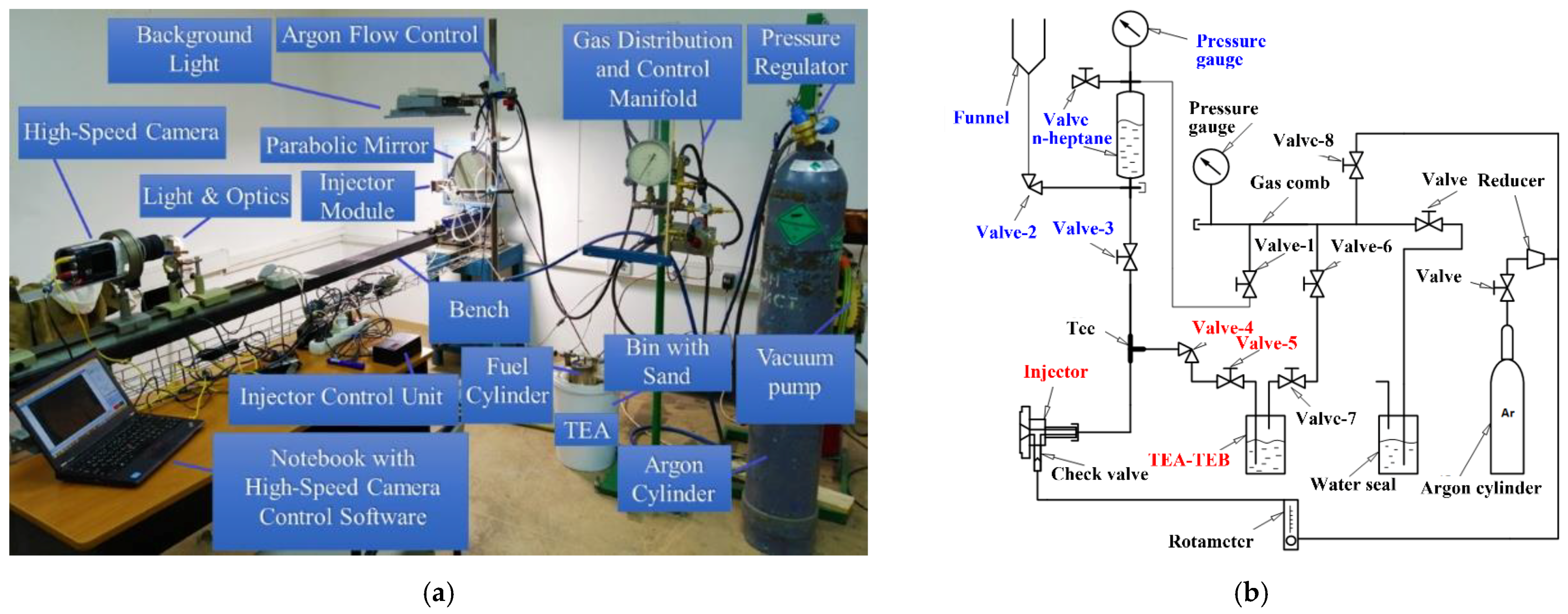
Based on these implications, the experimental setup for fuel spraying in air was designed and manufactured. The experimental setup consisted of an electromagnetic fuel injector (BOSCH 0 280 158 017) and a system for ensuring its operation (hydraulic system and microprocessor control unit), an optical system, a high-speed camera (Phantom Miro LC310), and a safety system. The elements of the setup are shown in
Figure 1a. The injector nozzle had 4 holes with a diameter of 0.2 mm. The microprocessor control unit monitored the current through the injector and the voltage applied, and issued synchronization and trigger signals for the high-speed video camera. In the preliminary experiments, the standard 13%TEA–87%TEB mixture provided by the production company was used. The density of the TEA–TEB mixture was 0.703 g/cm
3. To prevent clogging of the setup communications by the condensed reaction products of the TEA–TEB mixture with air, the injector was sprayed with n-heptane before and after each experiment. n-heptane (density 0.684 g/cm
3) was also used for estimating the flow rate and characteristic droplet size in the spray at different injection pressures. The nominal flow rate of n-heptane at an overpressure of 3 and 6 atm was 2.55 ± 0.08 mL/s (1.74 ± 0.05 g/s) and 5.1 ± 0.2 mL/s (3.5 ± 0.1 g/s), respectively. The droplet diameter in n-heptane sprays at the injection overpressure of 3 and 6 atm measured by the slide sampling method [
29,
30] was ~80–120 and ~30–50 μm, respectively. The operation frequency of the injector in the pulsed mode as well as the injection duration time were varied (see below).
Figure 1b shows the hydraulic scheme of the experimental setup. The experimental procedure was as follows. (i) Before supplying the TEA–TEB mixture to the injector, all communications were thoroughly purged with argon; (ii) liquid n-heptane was poured into the transparent measuring tank through the funnel and pressurized by argon to an overpressure of 3 atm using valve 1; (iii) with valves 2, 4, and 5 closed, valve 3 open, and the injector turned on, the communications were spilled with n-heptane; (iv) valve 3 was closed and the pressure was relieved by briefly turning on the injector; (v) valves 6 and 7 were opened and the TEA–TEB tank was pressurized by argon to the overpressure of 3 to 6 atm; valve 8 was used to control the pressure level; (vi) valves 6 to 8 were closed and valves 1, 4, and 5 opened; (vii) using a rotameter, a small flow rate of argon (around 1 L/min) was established around the injector nozzle to avoid nozzle clogging by the condensed reaction products of the TEA–TEB mixture with air available in the vicinity of the nozzle face; and (viii) the injector was turned on and operated for a preset time either in the pulsed or continuous injection mode.
Figure 2 shows two options used for fastening the injector in the housing, which differed in the means of supplying argon and the geometry of the insulating cavity. The first series of experiments was performed using the recessed injector of
Figure 2a and direct video registration of spray self-luminosity during ignition and combustion in ambient air (see
Section 3.3). As the dark zone between the flame and the nozzle mouth could be quite short, the second series of experiments was performed using the flat Injector of
Figure 2b (see
Section 3.3). In the latter case, spray self-ignition and combustion was registered both by direct video registration of self-luminosity and by the schlieren method.