Effect of Combustion Chamber Geometrical Parameters on the Decomposition and Combustion Characteristics of an ADN-Based Thruster
Abstract
:1. Introduction
2. Description of Numerical Simulations and CFD Methods
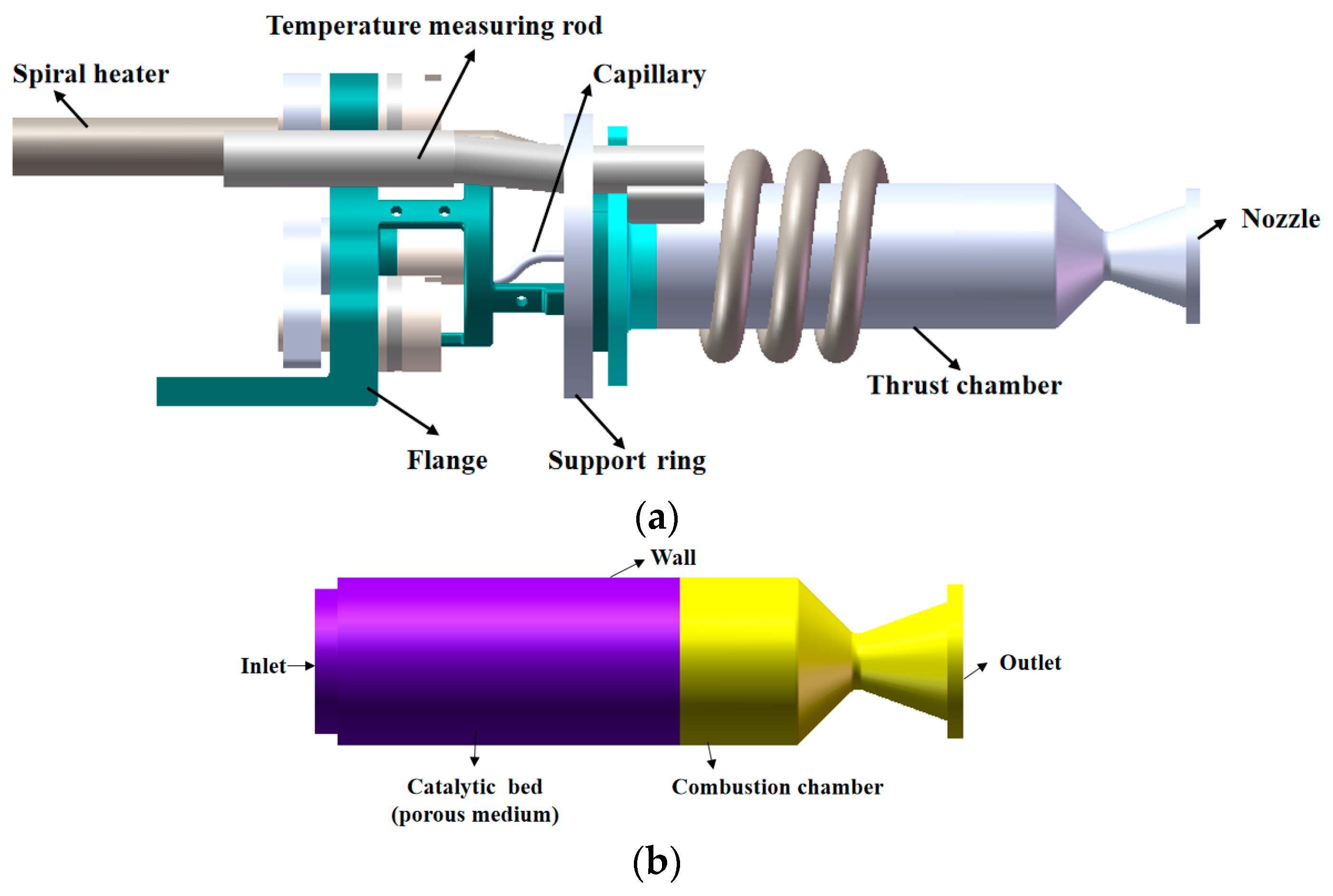
2.1. Thruster Geometry and Calculation Settings
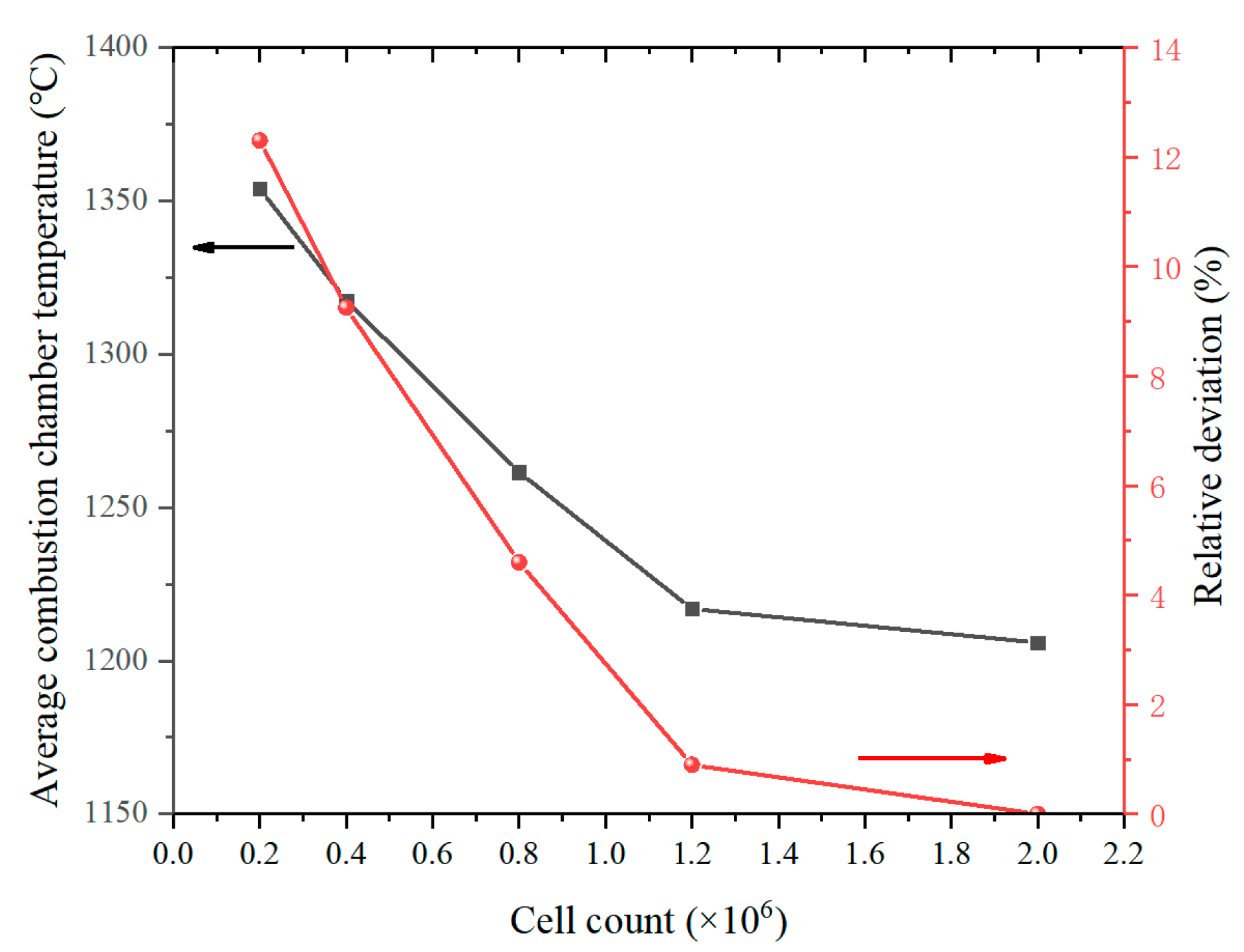
2.2. Numerical Description
2.3. 0.2 N-Class ADN-Based Thruster Vacuum Ground Test
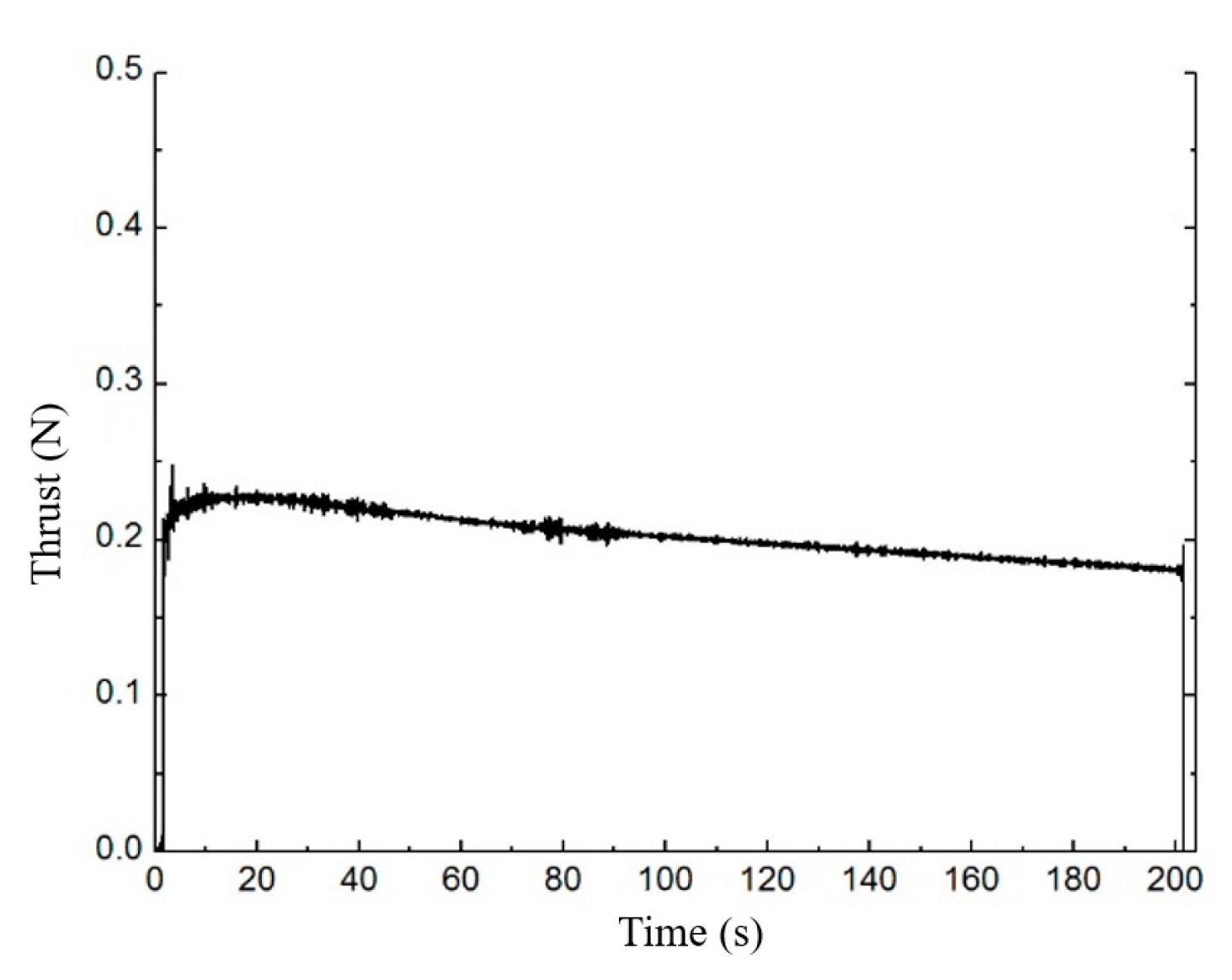
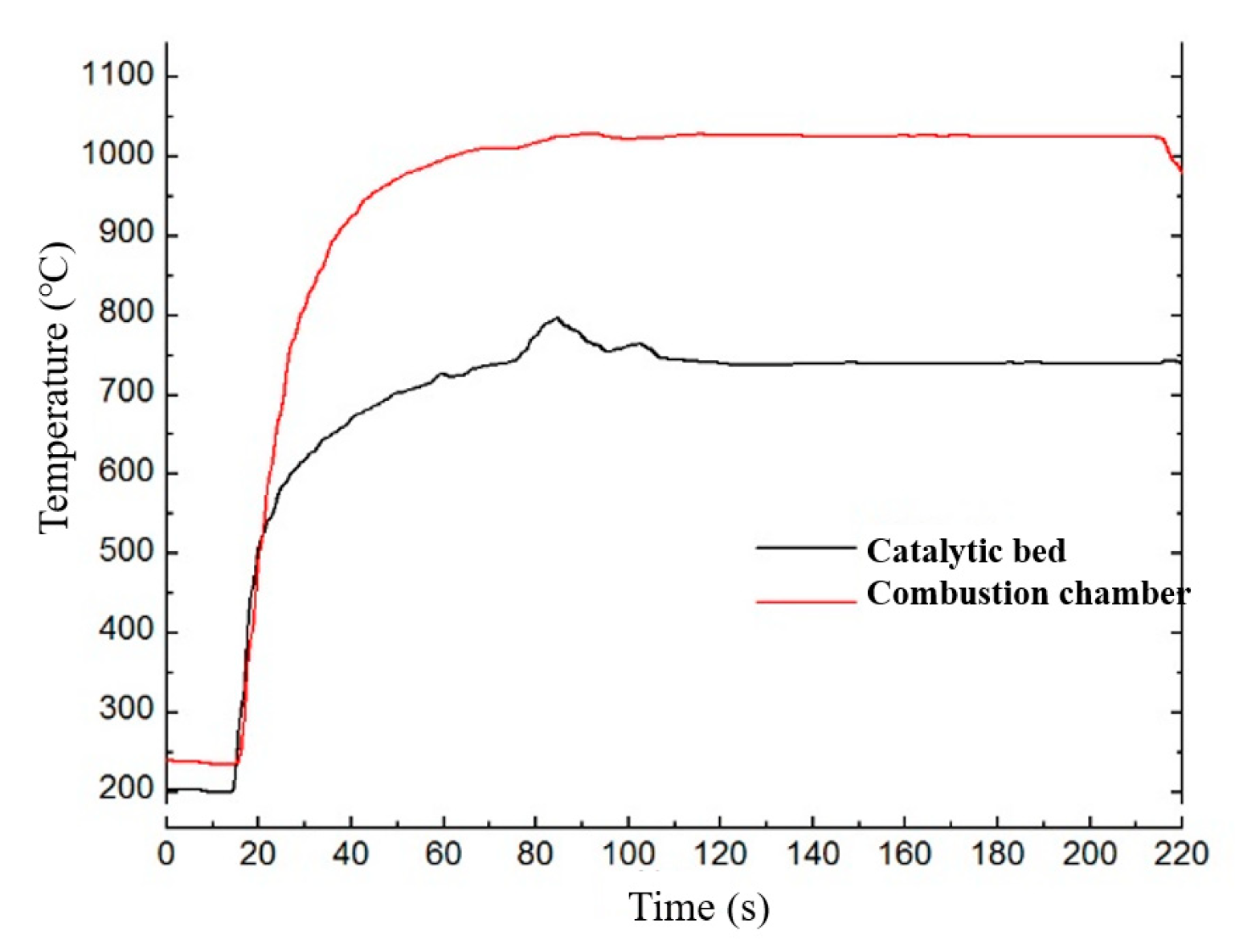
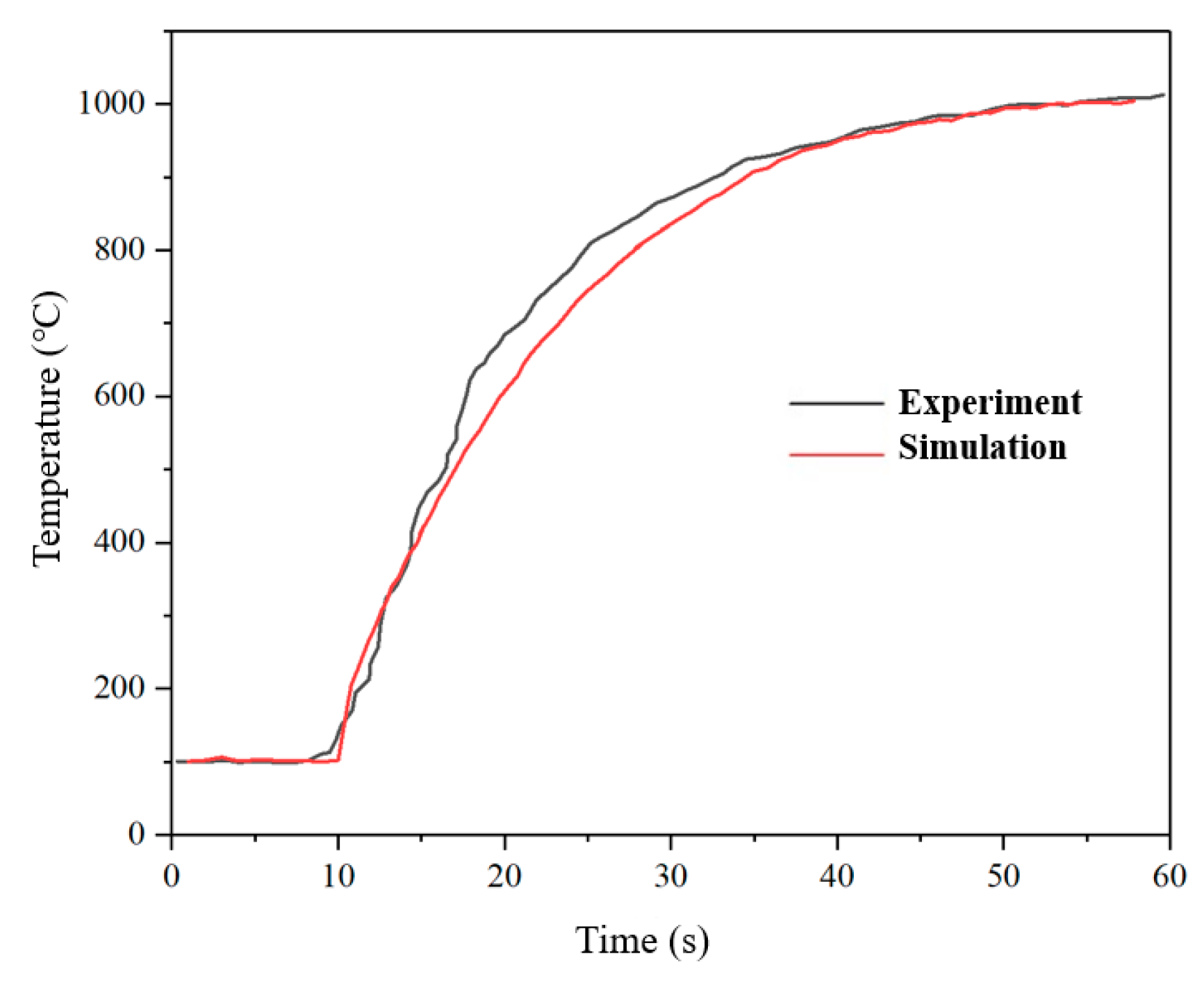
2.4. Model Validation
3. Results and Discussion
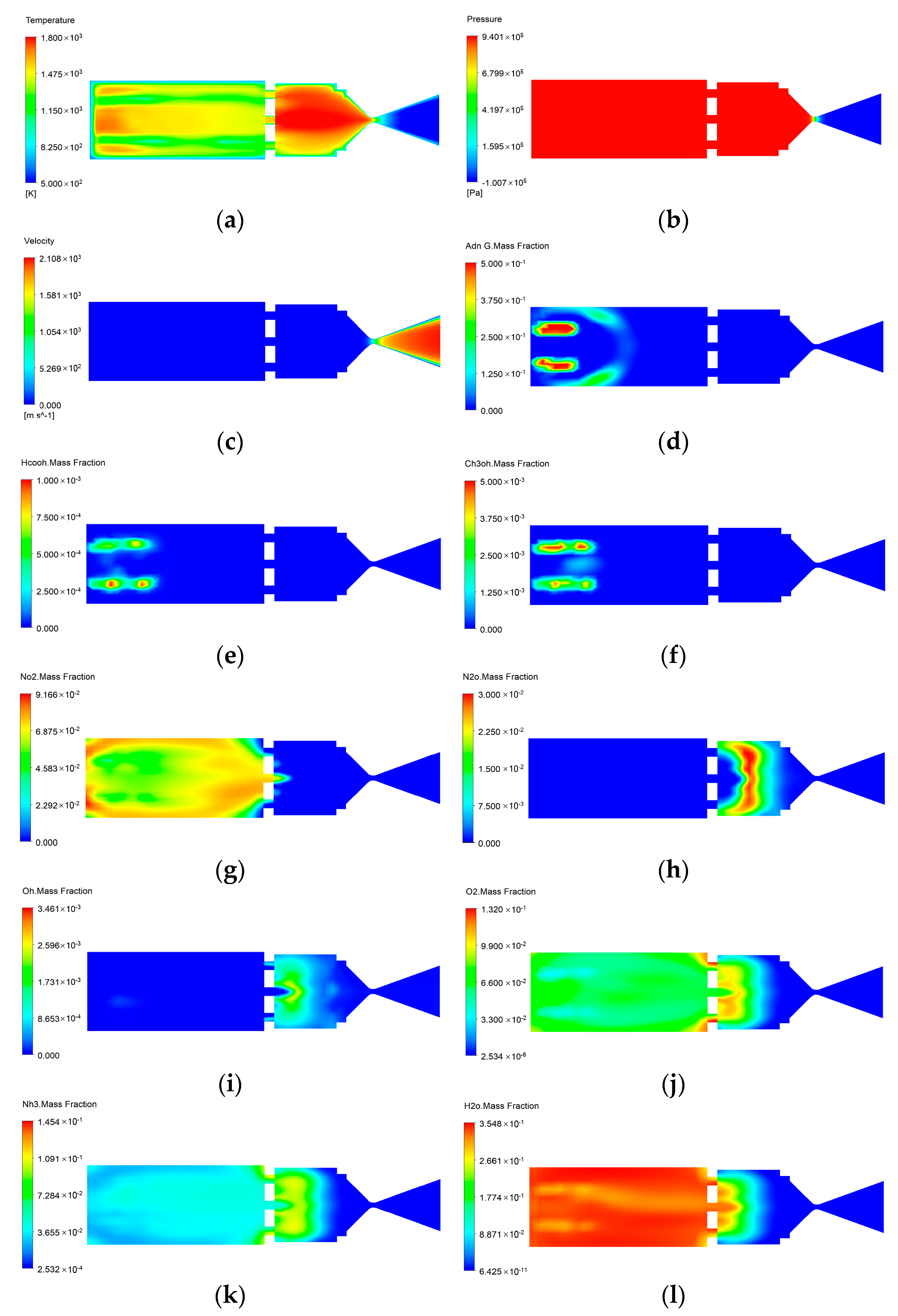
3.1. Catalytic Decomposition and Combustion Processes in ADN-Based Thrusters
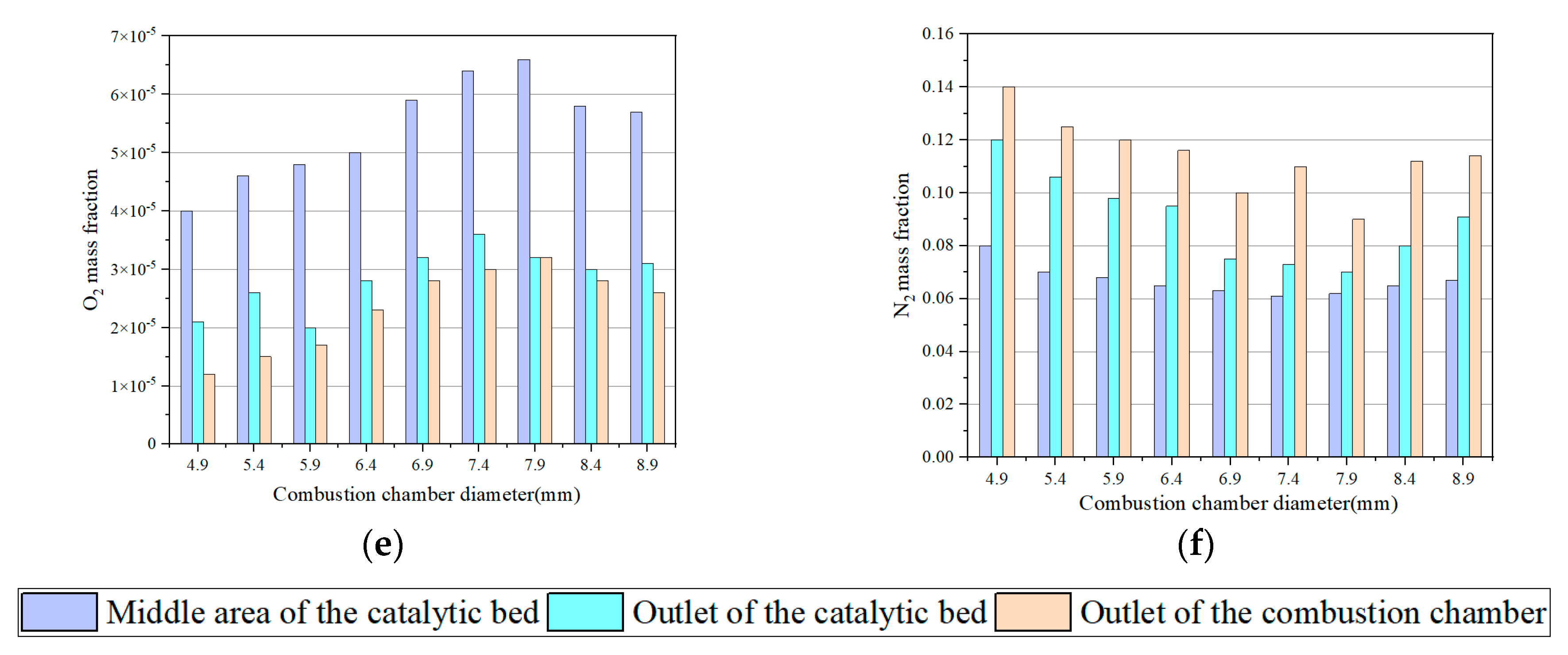
3.2. Effect of Combustion Chamber Diameter on Thruster Decomposition and Combustion Characteristics
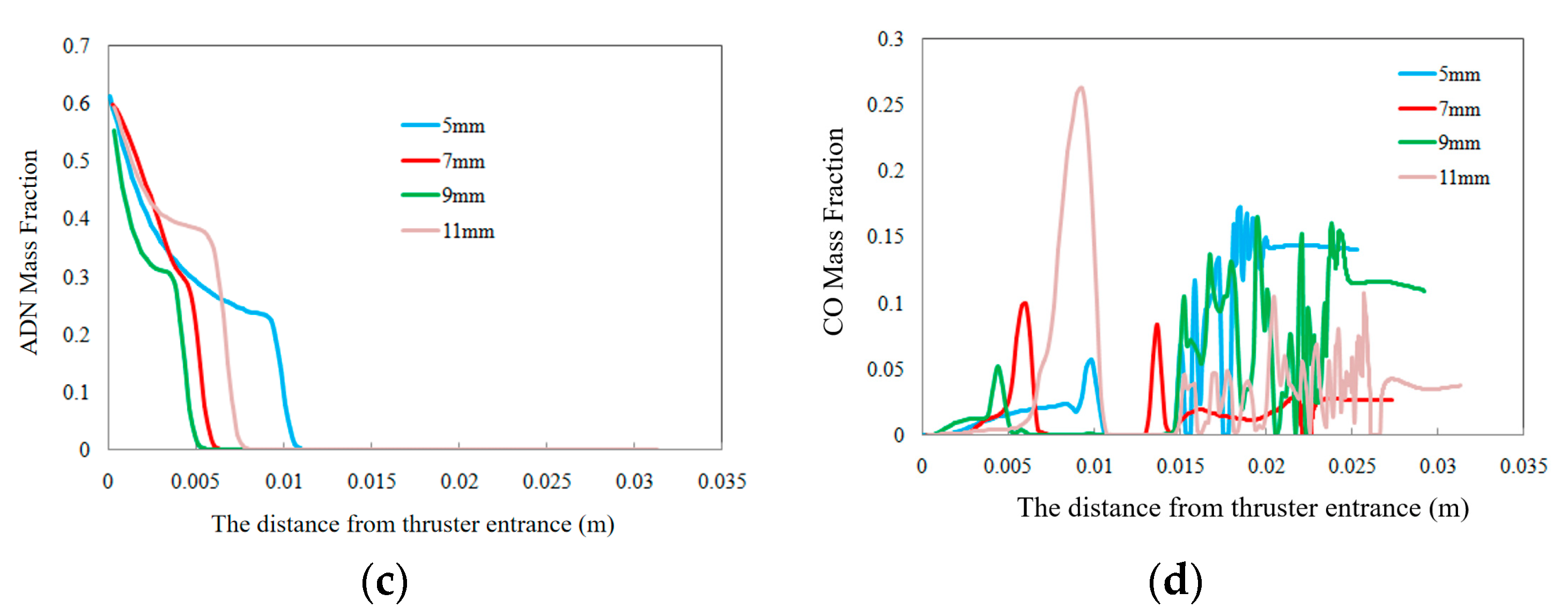
3.3. Effects of Combustion Chamber Length on Decomposition and Combustion Characteristics
4. Conclusions
- (1)
- The results show that the two geometrical parameters of combustion chamber length and diameter have significant effects on the temperature, pressure, and concentration of key product components in the thruster. By comparing the specific impulse and thrust at various geometrical parameters, it is found that the improvement of thruster performance tends to level off with the increase in combustion chamber length and diameter, and there exists a theoretical optimum design value to achieve the best performance of the thruster;
- (2)
- Because of the wall effect, the decomposition rate of an ADN near the wall is slower than that in the center of the catalytic bed, and the decomposition of an ADN is mainly in the catalytic bed area, while the oxidation reaction of methanol is concentrated in the combustion chamber.
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
Nomenclature
| AN | hydroxylamine nitrate | on the droplet surface | |
| mass fraction of the droplets | inside the droplet | ||
| mean diameter | pressure of evaporation | ||
| propagation parameter | in the droplet | ||
| in the droplet | droplet diameter | ||
| thermal conductivity of the continuous phase | |||
| surface area of the droplet | specific heat capacity of the continuous phase | ||
| fluid | |||
| solid | porosity of porous media | ||
| Fluid-phase thermal conductivity | Solid-phase thermal conductivity | ||
| thermal conductivity between fluid and solid | Fluid–solid interfacial density | ||
| solid density | sensible enthalpy | ||
| solid heat transfer coefficient | solid temperature | ||
| volume source term | heat flow | ||
| heat flow of fluid | heat flow of solid | ||
| specific impulse | DPM | discrete phase model | |
| UDF | User-defined function |
References
- Whitmore, A.; Merkley, P.; Eilers, D.; Judson, I. Development and Testing of a Green Monopropellant Ignition System. In Proceedings of the 49th AIAA/ASME/SAE/ASEE Joint Propulsion Conference and Exhibit, San Jose, CA, USA, 15–17 July 2013. [Google Scholar]
- Freudenmann, D.; Ciezki, H.K. ADN and HAN-based monopropellants-A minireview on compatibility and chemical stability in aqueous media. Explos. Pyrotech. 2019, 44, 1084–1089. [Google Scholar] [CrossRef]
- Yu, Y.-S.; Li, G.-X.; Zhang, T.; Chen, J.; Wang, M. Effects of catalyst-bed’s structure parameters on decomposition and combustion characteristics of an ammonium dinitramide (ADN)-based thruster. Energy Convers. Manag. 2015, 106, 566–575. [Google Scholar] [CrossRef]
- Yang, R.; Thakre, P.; Yang, V. Thermal Decomposition and Combustion of Ammonium Dinitramide (Review). Combust. Explos. Shock Waves 2005, 41, 657–679. [Google Scholar] [CrossRef]
- Oliveira, J. ADN-The new oxidizer around the corner for an environmentally friendly smokeless propellant. J. Aerosp. Technol. Manag. 2009, 1, 153–160. [Google Scholar]
- Handy, S. Applications of Ionic Liquid in Science and Technology; InTech Publishing: New York, NY, USA, 2011. [Google Scholar]
- Jharapla, P.K.; Vaitheeswaran, G.; Gupta, M.; Mittal, R. Comparative study of electronic structure, optical properties, lattice dynamics and thermal expansion behaviour of energetic ammonium and potassium dinitramide salts. Mater. Chem. Phys. 2021, 267, 124645. [Google Scholar] [CrossRef]
- Yao, Z.; Zhang, W.; Wang, M.; Chen, J.; Shen, Y.; Wei, Y.; Yu, X.; Li, F.; Zeng, H. Tunable diode laser absorption spectroscopy measurements of high-pressure ammonium dinitramide combustion. Aerosp. Sci. Technol. 2015, 45, 140–149. [Google Scholar] [CrossRef]
- Anflo, K.; Crowe, B. In-space demonstration of an ADN-based propulsion system. In Proceedings of the 47th AIAA/ASME/SAE/ASEE Joint Propulsion Conference & Exhibit, San Diego, CA, USA, 31 July–3 August 2011. [Google Scholar]
- Izato, Y.-I.; Miyake, A. Detailed kinetic model for ammonium dinitramide decomposition. Combust. Flame 2018, 198, 222–229. [Google Scholar] [CrossRef]
- Wingborg, N.; Eldsäter, C.; Skifs, H. Formulation and characterization of ADN-based liquid monopropellants. In Proceedings of the 41th AIAA/ASME/SAE/ASEE Joint Propulsion Conference & Exhibit, Tucson, AZ, USA, 10–13 July 2004. [Google Scholar]
- Jones, D.E.G.; Kwok, Q.S.M.; Vachon, M.; Badeen, C.; Ridley, W. Characterization of ADN and ADN-Based Propellants. Propellants Explos. Pyrotech. 2005, 30, 140–147. [Google Scholar] [CrossRef]
- Wingborg, N. Heat of formation ADN-based liquid monopropellants. Propellants Explos. Pyrotech. 2019, 44, 1090–1095. [Google Scholar] [CrossRef]
- Wingborg, N.; Johansson, M.; Bodin, L. ADN-based liquid monopropellants: Propellant selection and initial thruster development. In Proceedings of the 3rd International Conference on Green Propellants for Space Propulsion, Futuroscope, France, 17–20 September 2006. [Google Scholar]
- Jing, L.; You, X.; Huo, J.; Zhu, M.; Yao, Z. Experimental and numerical studies of ammonium dinitramide based liquid propellant combustion in space thruster. Aerosp. Sci. Technol. 2017, 69, 161–170. [Google Scholar] [CrossRef]
- Grönland, T.A.; Anflo, K.; Bergman, G. ADN-based propulsion for spacecraft-Key requirements and experimental verification. In Proceedings of the 40th AIAA/ASME/SAE/ASEE Joint Propulsion Conference & Exhibit, Fort Lauderdale, FL, USA, 11–14 July 2004. [Google Scholar]
- Farhat, K.; Kappenstein, C.; Batonneau, Y. Thermal and catalytic decomposition of AN-, ADN and HNF-based ionic monopropellants. In Proceedings of the 44th AIAA/ASME/SAE/ASEE Joint Propulsion Conference & Exhibit, Hartford, CT, USA, 21–23 July 2008. [Google Scholar]
- Amrousse, R.; Hori, K.; Fetimi, W. HAN and ADN as liquid ionic monopropellants: Thermal and catalytic decomposition processes. Appl. Catal. B Environ. 2012, 127, 121–128. [Google Scholar] [CrossRef]
- Kim, J.W.; Baek, S.; Jung, Y.; Yoon, W.; Ban, H.S.; Kwon, S. An alternative ADN based monopropellant mixed with tetraglyme. Acta Astronaut. 2021, 178, 241–249. [Google Scholar] [CrossRef]
- Zhang, T.; Li, G.; Yu, Y.; Sun, Z.; Wang, M.; Chen, J. Numerical simulation of ammonium dinitramide (ADN)-basednon-toxic aerospace propellant decomposition and combustion in a monopropellant thruster. Energy Convers. Manag. 2014, 87, 965–974. [Google Scholar] [CrossRef]
- Chen, J.; Li, G.; Zhang, T.; Liu, Y.; Yang, R.; Chen, Y. Catalytic bed slenderness ratio and ADN/methanol ratio for decomposition and combustion characteristics within ammonium dinitramide (ADN)-based green aerospace thruster. Chin. J. Chem. Eng. 2019, 27, 1159–1165. [Google Scholar] [CrossRef]
- Bingue, J.P.; Saveliev, A.V.; Fridman, A.A. Hydrogen production in ultra-rich filtration combustion of methane and hydrogen sulfide. Int. J. Hydrog. Energ. 2002, 27, 643–649. [Google Scholar] [CrossRef]
- Koopmans, R.J.; Shrimpton, J.S.; Roberts, G.T. A one-dimensional multicomponent two-fluid model of a reacting packed bed including mass, momentum and energy interphase transfer. Int. J. Multiph. Flow 2013, 57, 10–28. [Google Scholar] [CrossRef]
- Hitt, D.L.; Zhou, X. One-dimensional modeling of catalyzed H2O2 decomposition in microchannel flows. In Proceedings of the 33rd AIAA Fluid Dynamics Conference and Exhibit, Orlando, FL, USA, 23–26 June 2003. [Google Scholar]
- Kuwahara, F.; Shirota, M.; Nakayama, A. A numerical study of interfacial convective heat transfer coefficient in two-energy equation model for convection in porous media. Int. J. Heat Mass Transf. 2001, 44, 1153–1159. [Google Scholar] [CrossRef]
- Hou, B.; Wang, X.; Li, T. Steady-state behavior of liquid fuel hydrazine decomposition in packed bed. AIChE J. 2015, 61, 1064–1080. [Google Scholar] [CrossRef]
- Yu, Q.; Yang, Y.; Wang, Z.; Zhu, H. Modeling and parameter sensitivity analysis of fluidized bed solid particle/sCO2 heat exchanger for concentrated solar power plant. Appl. Therm. Eng. 2021, 197, 117429. [Google Scholar] [CrossRef]
- Sun, Y.N.; Gao, D.R.; Zhang, Z.Y. Study on flow field and temperature field characteristics of gas-liquid two-fluid snowmaking nozzle. Int. J. Hydromechatron. 2020, 1, 1. [Google Scholar] [CrossRef]
- Gunawan, R.; Zhang, D. Thermal stability and kinetics of decomposition of ammonium nitrate in the presence of pyrite. J. Hazard. Mater. 2009, 165, 751–758. [Google Scholar] [CrossRef]
- Korobeinichev, O.P.; Bolshova, T.A.; Paletsky, A.A. Modeling the chemical reactions of ammonium dinitramide (ADN) in a flame. Combust. Flame 2001, 126, 1516–1523. [Google Scholar] [CrossRef]
- Foustoukos, D.I.; Stern, J.C. Oxidation pathways for formic acid under low temperature hydrothermal conditions: Implications for the chemical and isotopic evolution of organics on Mars. Geochim. Cosmochim. Acta 2012, 76, 14–28. [Google Scholar] [CrossRef] [Green Version]
- Heo, S.; Jo, S.; Yun, Y. Effect of dual-catalytic bed using two different catalyst sizes for hydrogen peroxide thruster. Aerosp. Sci. Technol. 2018, 78, 26–32. [Google Scholar] [CrossRef]





| Parameter | Value | |
|---|---|---|
| Thruster geometry parameters | Catalyst bed length (m) | 1.5 × 10−2 |
| Catalyst bed diameter (m) | 6.9 × 10−3 | |
| Combustion chamber length (m) | 5.3 × 10−3 | |
| Combustion chamber diameter (m) | 6.9 × 10−3 | |
| Nozzle expansion ratio | 50 |
| Value | Literature | |
|---|---|---|
| Density of liquid (kg/m3) | 1290 | [3] |
| Cp (Specific Heat) (J/kg·k) | 2350 | [3] |
| Viscosity (kg/m·s) | 0.0046 | [3] |
| n | 7.726 | [20] |
| d () | 80.7 | [20] |
| 0.5 | [21] | |
| (W/m·K) | 0.8 | [22] |
| (W/m·K) | 25.1 | [22] |
| ADN(G) + M => NH3 + HN3O4 + M HN3O4 = HNNO2 + NO2 2CH3OH + 2NO2 = 2HCOOH + N2 + 2H2O HNNO2 + M = N2O + OH + M NH3 + OH = NH2 + H2O OH + OH = H2O + O HNNO2 + OH = H2O + 2NO NH2 + NO2 = H2NO + NO NO + NO = N2 + O2 CH3OH + O2 = CH2OH + HO2 | CH3OH + OH = CH2OH + H2O CH2OH + O = CH2O + OH CH2O + OH = HCO + H2O HCO + OH = H2O + CO H2NO + O = NH2 + O2 NH2 + NO = N2 + H2O N2O + M = N2 + O + M N2O + O = N2 + O2 2HCOOH + O2 = 2CO2 + 2H2O CO + O + M = CO2 + M |
| Experimental Result | Numerical Result | Error | |
|---|---|---|---|
| Mean thrust (N) | 0.21 | 0.203 | 3.33% |
| Temperature of combustion chamber (°C) | 1013 | 1004 | 0.89% |
| Temperature of catalytic bed (°C) | 706 | 721 | 2.12% |
| Middle of the Catalytic Bed | Middle of the Combustion Chamber | |
|---|---|---|
| temperature (℃) | 1126 | 1525 |
| Pressure (MPa) | 0.96 | 0.94 |
| Velocity (m/s) | 43 | 86 |
| ADN mass fraction | 0.25 | 6 × 10−3 |
| HCOOH mass fraction | 6.1 × 10−4 | 9.4 × 10−5 |
| CH3OH mass fraction | 0.03 | 0.025 |
| NO2 mass fraction | 2.4 × 10−5 | 1.3 × 10−5 |
| N2O mass fraction | 3.6 × 10−3 | 5 × 10−4 |
| OH mass fraction | 5 × 10−4 | 2.3 × 10−3 |
| O2 mass fraction | 4.3 × 10−5 | 1.9 × 10−5 |
| NH3 mass fraction | 0.04 | 0.073 |
| H2O mass fraction | 0.28 | 0.16 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hou, Y.; Yu, Y.; Liu, X.; Cao, J. Effect of Combustion Chamber Geometrical Parameters on the Decomposition and Combustion Characteristics of an ADN-Based Thruster. Micromachines 2022, 13, 605. https://doi.org/10.3390/mi13040605
Hou Y, Yu Y, Liu X, Cao J. Effect of Combustion Chamber Geometrical Parameters on the Decomposition and Combustion Characteristics of an ADN-Based Thruster. Micromachines. 2022; 13(4):605. https://doi.org/10.3390/mi13040605
Chicago/Turabian StyleHou, Yangyang, Yusong Yu, Xuhui Liu, and Jie Cao. 2022. "Effect of Combustion Chamber Geometrical Parameters on the Decomposition and Combustion Characteristics of an ADN-Based Thruster" Micromachines 13, no. 4: 605. https://doi.org/10.3390/mi13040605
APA StyleHou, Y., Yu, Y., Liu, X., & Cao, J. (2022). Effect of Combustion Chamber Geometrical Parameters on the Decomposition and Combustion Characteristics of an ADN-Based Thruster. Micromachines, 13(4), 605. https://doi.org/10.3390/mi13040605







