Review on Surface-Modified Electrodes for the Enhanced Electrochemical Detection of Selective Serotonin Reuptake Inhibitors (SSRIs)
Abstract
:1. Introduction
2. Polymer-Based Modification of Electrode Surface
2.1. Fluoxetine
2.2. Citalopram
2.3. Fluvoxamine
2.4. Sertraline
3. Carbon Nanomaterial-Based Modification of Electrode Surface
3.1. Escitalopram
3.2. Paroxetine
4. Metal/Metal Oxide Nanomaterial-Based Modification of Electrode Surface
4.1. Escitalopram
4.2. Sertraline
5. Carbon–Metal/Metal Oxide Nanocomposite-Based Modification of Electrode Surface
5.1. Citalopram
5.2. Escitalopram
5.3. Sertraline
6. Carbon–Polymer Composite-Based Modification of Electrode Surface
6.1. Fluoxetine
6.2. Citalopram
6.3. Paroxetine
6.4. Fluvoxamine
6.5. Sertraline
7. Metal–Polymer Composite-Based Modification of Electrode Surface
Fluoxetine
8. Carbon–Metal–Polymer Composite-Based Modification Electrode Surface
8.1. Fluoxetine
8.2. Citalopram
8.3. Paroxetine
8.4. Sertraline
9. Bio-Functionalised Polymer-Based Modification of Electrode Surface
Paroxetine
10. Concluding Remarks and Future Perspectives
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Xu, J.; Tao, J.; Su, L.; Wang, J.; Jiao, T. A critical review of carbon quantum dots: From synthesis toward applications in electrochemical biosensors for the determination of a depression-related neurotransmitter. Materials 2021, 14, 3987. [Google Scholar] [CrossRef] [PubMed]
- Ondicova, K.; Tillinger, A.; Pecenak, J.; Mravec, B. The vagus nerve role in antidepressants action: Efferent vagal pathways participate in peripheral anti-inflammatory effect of fluoxetine. Neurochem. Int. 2019, 125, 47–56. [Google Scholar] [CrossRef] [PubMed]
- Bishnoi, S.; Sharma, A.; Singhal, R.; Goyal, R.N. Edge plane pyrolytic graphite as a sensing surface for the determination of fluvoxamine in urine samples of obsessive-compulsive disorder patients. Biosens. Bioelectron. 2020, 168, 112489. [Google Scholar] [CrossRef]
- Olędzka, I.; Plenis, A.; Kowalski, P.; Bączek, T.; Roszkowska, A. Analytical aspects of sample handling during the quantification of selective serotonin reuptake inhibitors in clinical applications. TrAC Trends Anal. Chem. 2023, 162, 117026. [Google Scholar] [CrossRef]
- Ivrigh, Z.J.-N.; Bigdeli, A.; Jafarinejad, S.; Hormozi-Nezhad, M.R. Multiplex detection of antidepressants with a single component condition-based colorimetric sensor array. Sensors Actuators B Chem. 2022, 363, 131855. [Google Scholar] [CrossRef]
- Eshun-Wilson, I.; Akena, D.H.; Siegfried, N.; Obuku, E.; Stein, D.J.; Joska, J.A. Antidepressants for depression in adults with HIV infection. Cochrane Database Syst. Rev. 2014, 1. [Google Scholar] [CrossRef]
- Pei, S.; Liu, L.; Zhong, Z.; Wang, H.; Lin, S.; Shang, J. Risk of prenatal depression and stress treatment: Alteration on serotonin system of offspring through exposure to Fluoxetine. Sci. Rep. 2016, 6, 33822. [Google Scholar] [CrossRef] [Green Version]
- Williams, L.J.; Berk, M.; Hodge, J.M.; Kotowicz, M.A.; Stuart, A.L.; Chandrasekaran, V.; Cleminson, J.; Pasco, J.A. Selective Serotonin Reuptake Inhibitors (SSRIs) and Markers of Bone Turnover in Men. Calcif. Tissue Int. 2018, 103, 125–130. [Google Scholar] [CrossRef]
- Silva, L.J.G.; Pereira, A.M.P.T.; Rodrigues, H.; Meisel, L.M.; Lino, C.M.; Pena, A. SSRIs antidepressants in marine mussels from Atlantic coastal areas and human risk assessment. Sci. Total Environ. 2017, 603–604, 118–125. [Google Scholar] [CrossRef]
- Hudson, A.D.; Solà, R.; Ueta, J.T.; Battell, W.; Jamieson, O.; Dunbar, T.; Maciá, B.; Peeters, M. Synthesis of Optimized Molecularly Imprinted Polymers for the Isolation and Detection of Antidepressants via HPLC. Biomimetics 2019, 4, 18. [Google Scholar] [CrossRef] [Green Version]
- Coleman, J.A.; Navratna, V.; Antermite, D.; Yang, D.; Bull, J.A.; Gouaux, E. Chemical and structural investigation of the paroxetine-human serotonin transporter complex. Elife 2020, 9, e56427. [Google Scholar] [CrossRef] [PubMed]
- Edinoff, A.N.; Akuly, H.A.; Hanna, T.A.; Ochoa, C.O.; Patti, S.J.; Ghaffar, Y.A.; Kaye, A.D.; Viswanath, O.; Urits, I.; Boyer, A.G.; et al. Selective serotonin reuptake inhibitors and adverse effects: A narrative review. Neurol. Int. 2021, 13, 387–401. [Google Scholar] [CrossRef] [PubMed]
- Ran, C.; Zhou, H.; Tan, C.; Tan, J.; Zhang, Z.; Zhao, W. Detection and Evaluation of Adverse Drug Reaction Signals of Antidepressants Based on FDA Adverse Event Reporting System Database. Open J. Depress. 2020, 09, 17–25. [Google Scholar] [CrossRef]
- Das, R.; Agrawal, Y.K. Trends and Advances in Separation and Detection of SSRIs and SNRIs in Biological Matrices. Chromatogr. Res. Int. 2013, 2013, 139459. [Google Scholar] [CrossRef]
- Lotrich, F.E.; Pollock, B.G. Candidate genes for antidepressant response to selective serotonin reuptake inhibitors. Neuropsychiatr. Dis. Treat. 2005, 1, 17–35. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Porcelli, S.; Drago, A.; Fabbri, C.; Gibiino, S.; Calati, R.; Serretti, A. Pharmacogenetics of antidepressant response. J. Psychiatry Neurosci. 2011, 36, 87–113. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wu, Y.; Altuner, E.E.; El Houda Tiri, R.N.; Bekmezci, M.; Gulbagca, F.; Aygun, A.; Xia, C.; Van Le, Q.; Sen, F.; Karimi-Maleh, H. Hydrogen generation from methanolysis of sodium borohydride using waste coffee oil modified zinc oxide nanoparticles and their photocatalytic activities. Int. J. Hydrogen Energy 2022, 48, 6613–6623. [Google Scholar] [CrossRef]
- Soleymanpour, A.; Rezvani, S.A. Liquid membrane/polyaniline film coated glassy carbon sensor for highly sensitive and selective determination of fluvoxamine in pharmaceutical and biological samples. Sens. Actuators B Chem. 2017, 247, 602–608. [Google Scholar] [CrossRef]
- Magalhães, P.; Alves, G.; Llerena, A.; Falcão, A. Therapeutic drug monitoring of fluoxetine, norfluoxetine and paroxetine: A new tool based on microextraction by packed sorbent coupled to liquid chromatography. J. Anal. Toxicol. 2017, 41, 631–638. [Google Scholar] [CrossRef] [Green Version]
- Caraci, F.; Calabrese, F.; Molteni, R.; Bartova, L.; Dold, M.; Leggio, G.M.; Fabbri, C.; Mendlewicz, J.; Racagni, G.; Kasper, S.; et al. International union of basic and clinical pharmacology CIV: The neurobiology of treatment-resistant depression: From antidepressant classifications to novel pharmacological targets. Pharmacol. Rev. 2018, 70, 475–504. [Google Scholar] [CrossRef] [Green Version]
- Plöderl, M.; Hengartner, M.P. What are the chances for personalised treatment with antidepressants? Detection of patient-by-treatment interaction with a variance ratio meta-analysis. BMJ Open 2019, 9, e034816. [Google Scholar] [CrossRef] [PubMed]
- dos Santos Neto, A.G.; de Sousa, C.S.; da Silva Freires, A.; Silva, S.M.; Zanin, H.; Damos, F.S.; de Cássia Silva Luz, R. Electrochemical sensor for detection of imipramine antidepressant at low potential based on oxidized carbon nanotubes, ferrocenecarboxylic acid, and cyclodextrin: Application in psychotropic drugs and urine samples. J. Solid State Electrochem. 2018, 22, 1385–1394. [Google Scholar] [CrossRef]
- Truta, L.; Castro, A.L.; Tarelho, S.; Costa, P.; Sales, M.G.F.; Teixeira, H.M. Antidepressants detection and quantification in whole blood samples by GC–MS/MS, for forensic purposes. J. Pharm. Biomed. Anal. 2016, 128, 496–503. [Google Scholar] [CrossRef] [PubMed]
- Yagihashi, G.; Tarui, T.; Miyagi, H.; Ohnishi, H.; Watanabe, T.; Yamaguchi, Y. Diagnostic accuracy for drug detection using liquid chromatography/mass spectroscopy in overdose patients. Acute Med. Surg. 2020, 7, e487. [Google Scholar] [CrossRef] [PubMed]
- Abu-hassan, A.A.; Omar, M.A.; Derayea, S.M. Use of acetylacetone for nano-level assay of fluvoxamine maleate in pure form and pharmaceutical formulation. Luminescence 2020, 35, 1360–1365. [Google Scholar] [CrossRef]
- Martins, F.C.O.L.; Pimenta, L.C.; De Souza, D. Antidepressants determination using an electroanalytical approach: A review of methods. J. Pharm. Biomed. Anal. 2021, 206, 114365. [Google Scholar] [CrossRef]
- Zamani, M.; Wilhelm, T.; Furst, A.L. Perspective—Electrochemical Sensors for Neurotransmitters and Psychiatrics: Steps toward Physiological Mental Health Monitoring. J. Electrochem. Soc. 2022, 169, 047513. [Google Scholar] [CrossRef]
- Tajik, S.; Safaei, M.; Beitollahi, H. A sensitive voltammetric sertraline nanosensor based on ZnFe2O4 nanoparticles modified screen printed electrode. Meas. J. Int. Meas. Confed. 2019, 143, 51–57. [Google Scholar] [CrossRef]
- Sharma, S.; Singh, N.; Tomar, V.; Chandra, R. A review on electrochemical detection of serotonin based on surface modified electrodes. Biosens. Bioelectron. 2018, 107, 76–93. [Google Scholar] [CrossRef]
- Milani-Hosseini, M.R.; Karamdoust, S.; Bahman, M.; Motaharian, A.; Mohammadsadegh, S. Molecularly Imprinted Polymer (MIP) electrochemical sensor based on graphene modified platinum electrode for sertraline determination. Anal. Bioanal. Electrochem. 2020, 12, 128–140. [Google Scholar]
- Mohammadi, S.Z.I.A.; Beitollahi, H.; Rohani, T.; Allahabadi, H.; Tajik, S. La2O3/Co3O4 nanocomposite modified screen printed electrode for voltammetric determination of sertraline. J. Serbian Chem. Soc. 2020, 85, 505–515. [Google Scholar] [CrossRef] [Green Version]
- Brycht, M.; Skrzypek, S.; Karadas-Bakirhan, N.; Smarzewska, S.; Bozal-Palabiyik, B.; Ozkan, S.A.; Uslu, B. Voltammetric behavior and determination of antidepressant drug paroxetine at carbon-based electrodes. Ionics 2015, 21, 2345–2354. [Google Scholar] [CrossRef]
- Badulla, W.F.; Özcan, S.; Atkoşar, Z.; Arli, G. Study of electrochemical behavior of escitalopram oxalate using hanging mercury drop electrode and its determination in human urine and pharmaceuticals. J. Iran. Chem. Soc. 2021, 18, 739–750. [Google Scholar] [CrossRef]
- Silva, M.K.L.; Sousa, G.S.; Simoes, R.P.; Cesarino, I. Fabrication of paper-based analytical devices using a PLA 3D-printed stencil for electrochemical determination of chloroquine and escitalopram. J. Solid State Electrochem. 2022, 26, 581–586. [Google Scholar] [CrossRef]
- Qian, L.; Durairaj, S.; Prins, S.; Chen, A. Nanomaterial-based electrochemical sensors and biosensors for the detection of pharmaceutical compounds. Biosens. Bioelectron. 2021, 175, 112836. [Google Scholar] [CrossRef]
- Alqarni, S.A.; Hussein, M.A.; Ganash, A.A.; Khan, A. Composite Material–Based Conducting Polymers for Electrochemical Sensor Applications: A Mini Review. Bionanoscience 2020, 10, 351–364. [Google Scholar] [CrossRef]
- Lahcen, A.A.; Amine, A. Recent Advances in Electrochemical Sensors Based on Molecularly Imprinted Polymers and Nanomaterials. Electroanalysis 2019, 31, 188–201. [Google Scholar] [CrossRef]
- Herrera-Chacón, A.; Cetó, X.; del Valle, M. Molecularly imprinted polymers—Towards electrochemical sensors and electronic tongues. Anal. Bioanal. Chem. 2021, 413, 6117–6140. [Google Scholar] [CrossRef]
- Rebelo, P.; Costa-Rama, E.; Seguro, I.; Pacheco, J.G.; Nouws, H.P.A.; Cordeiro, M.N.D.S.; Delerue-Matos, C. Molecularly imprinted polymer-based electrochemical sensors for environmental analysis. Biosens. Bioelectron. 2021, 172, 112719. [Google Scholar] [CrossRef]
- Feroz, M.; Lopes, I.C.; Rehman, H.U.; Ata, S.; Vadgama, P. A novel molecular imprinted polymer layer electrode for enhanced sensitivity electrochemical determination of the antidepressant fluoxetine. J. Electroanal. Chem. 2020, 878, 114693. [Google Scholar] [CrossRef]
- Madani, M. Poly(Pyrrole) conducting polymer solid-state sensor for potentiometric determination of fluoxetine. Anal. Bioanal. Electrochem. 2019, 11, 647–656. [Google Scholar]
- Izadyar, A.; Arachchige, D.R.; Cornwell, H.; Hershberger, J.C. Ion transfer stripping voltammetry for the detection of nanomolar levels of fluoxetine, citalopram, and sertraline in tap and river water samples. Sens. Actuators B Chem. 2016, 223, 226–233. [Google Scholar] [CrossRef]
- Rebelo, P.; Pacheco, J.G.; Voroshylova, I.V.; Seguro, I.; Cordeiro, M.N.D.S.; Delerue-Matos, C. Computational Modelling and Sustainable Synthesis of a Highly Selective Electrochemical MIP-Based Sensor for Citalopram Detection. Molecules 2022, 27, 3315. [Google Scholar] [CrossRef]
- Lović, J.; Lađarević, J.; Trišović, N.; Andrić, F.; Mladenović, A.; Mijin, D.; Vuković, D.; Petrović, S.; Ivić, M.A. Electrochemical determination of sertraline in pharmaceutical formulation and serum using a gold electrode in a pH 8.4 bicarbonate solution. Mon. Fur Chem. 2021, 152, 185–192. [Google Scholar] [CrossRef]
- Nguyen, T.D.; Nguyen, M.T.N.; Lee, J.S. Carbon-Based Materials and Their Applications in Sensing by Electrochemical Voltammetry. Inorganics 2023, 11, 81. [Google Scholar] [CrossRef]
- Zhang, C.; Du, X. Electrochemical Sensors Based on Carbon Nanomaterial Used in Diagnosing Metabolic Disease. Front. Chem. 2020, 8, 651. [Google Scholar] [CrossRef]
- Power, A.C.; Gorey, B.; Chandra, S.; Chapman, J. Carbon nanomaterials and their application to electrochemical sensors: A review. Nanotechnol. Rev. 2018, 7, 19–41. [Google Scholar] [CrossRef]
- Fredj, Z.; Sawan, M. Advanced Nanomaterials-Based Electrochemical Biosensors for Catecholamines Detection: Challenges and Trends. Biosensors 2023, 13, 211. [Google Scholar] [CrossRef] [PubMed]
- Li, C.M.; Sun, C.Q.; Chen, W.; Pan, L. Electrochemical thin film deposition of polypyrrole on different substrates. Surf. Coat. Technol. 2005, 198, 474–477. [Google Scholar] [CrossRef]
- Medeiros, R.A.; Baccarin, M.; Fatibello-Filho, O.; Rocha-Filho, R.C.; Deslouis, C.; Debiemme-Chouvy, C. Comparative Study of Basal-Plane Pyrolytic Graphite, Boron-Doped Diamond, and Amorphous Carbon Nitride Electrodes for the Voltammetric Determination of Furosemide in Pharmaceutical and Urine Samples. Electrochim. Acta 2016, 197, 179–185. [Google Scholar] [CrossRef]
- Baccarin, M.; Cervini, P.; Cavalheiro, E.T.G. Comparative performances of a bare graphite-polyurethane composite electrode unmodified and modified with graphene and carbon nanotubes in the electrochemical determination of escitalopram. Talanta 2018, 178, 1024–1032. [Google Scholar] [CrossRef] [PubMed]
- Mendes, R.K.; Claro-Neto, S.; Cavalheiro, E.T.G. Evaluation of a new rigid carbon-castor oil polyurethane composite as an electrode material. Talanta 2002, 57, 909–917. [Google Scholar] [CrossRef] [PubMed]
- Santos, S.X.D.; Cavalheiro, É.T.G. Using of a graphite-polyurethane composite electrode modified with a schiff base as a bio-inspired sensor in the dopamine determination. J. Braz. Chem. Soc. 2014, 25, 1071–1077. [Google Scholar] [CrossRef]
- Cervini, P.; Cavalheiro, E.T.G. Possibilities of using a new graphite-castor oil polyurethane composite electrode as an amperometric flow detector. Anal. Chem. Indian J. 2006, 2, 187–194. [Google Scholar]
- Mendes, R.K.; Cervini, P.; Cavalheiro, É.T.G. The use of a graphite-castor oil polyurethane composite electrode for the determination of hydroquinone in photographic developers. Talanta 2006, 68, 708–712. [Google Scholar] [CrossRef]
- Gomes, N.O.; Mendonça, C.D.; Machado, S.A.S.; Oliveira, O.N.; Raymundo-Pereira, P.A. Flexible and integrated dual carbon sensor for multiplexed detection of nonylphenol and paroxetine in tap water samples. Microchim. Acta 2021, 188, 359. [Google Scholar] [CrossRef]
- Shahrokhian, S.; Salimian, R.; Rastgar, S. Pd-Au nanoparticle decorated carbon nanotube as a sensing layer on the surface of glassy carbon electrode for electrochemical determination of ceftazidime. Mater. Sci. Eng. C 2014, 34, 318–325. [Google Scholar] [CrossRef]
- Ehzari, H.; Gholivand, M.; Shamsipur, M. A sensitive electrochemical sensor based on multiwall carbon nanotube-ionic liquid/nickel oxide nanoparticles for simultaneous determination of the antipsychotic drugs clozapine and sertraline. Adv. Nanochem. 2021, 3, 23–33. [Google Scholar] [CrossRef]
- Fazio, E.; Spadaro, S.; Corsaro, C.; Neri, G.; Leonardi, S.G.; Neri, F.; Lavanya, N.; Sekar, C.; Donato, N.; Neri, G. Metal-oxide based nanomaterials: Synthesis, characterization and their applications in electrical and electrochemical sensors. Sensors 2021, 21, 2494. [Google Scholar] [CrossRef]
- Daneshvar, L.; Rounaghi, G.H.; Es’haghi, Z.; Chamsaz, M.; Tarahomi, S. Fabrication a new modified electrochemical sensor based on Au–Pd bimetallic nanoparticle decorated graphene for citalopram determination. Mater. Sci. Eng. C 2016, 69, 653–660. [Google Scholar] [CrossRef]
- Attia, A.K.; Mohamed, M.A.; Fekry, A.M. Electroanalytical determination of escitalopram oxalate using nickel nanoparticles modified carbon paste sensor. Acta Chim. Slov. 2017, 64, 415–421. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Tajik, S.; Beitollahi, H. Electrochemical Determination of Sertraline at Screen Printed Electrode Modified with Feather Like La3+/ZnO Nano-Flowers and Its Determination in Pharmaceutical and Biological Samples. Russ. J. Electrochem. 2020, 56, 222–229. [Google Scholar] [CrossRef]
- Zaimbashi, R.; Shamspur, T.; Mostafavi, A. Hydrothermal synthesis of ZnO nanoflowes for rapid detection of sertraline and imipramine using the modified screen-printed electrode. J. Mater. Sci. Mater. Electron. 2022, 33, 19711–19722. [Google Scholar] [CrossRef]
- Shoja, Y.; Rafati, A.A.; Ghodsi, J. Electropolymerization of Ni-LD metallopolymers on gold nanoparticles enriched multi-walled carbon nanotubes as nano-structure electrocatalyst for efficient voltammetric sertraline detection in human serum. Electrochim. Acta 2016, 203, 281–291. [Google Scholar] [CrossRef]
- Chadderdon, D.J.; Xin, L.; Qi, J.; Qiu, Y.; Krishna, P.; More, K.L.; Li, W. Electrocatalytic Oxidation of 5- Hydroxymethylfurfural to 2,5-Furandicarboxylic Acid on Supported Au and Pd Bimetallic Nanoparticles. Green Chem. 2014, 16, 3778–3786. [Google Scholar] [CrossRef]
- Madrakian, T.; Soleimani, M.; Afkhami, A. Electrochemical determination of fluvoxamine on mercury nanoparticle multi-walled carbon nanotube modified glassy carbon electrode. Sens. Actuators B Chem. 2015, 210, 259–266. [Google Scholar] [CrossRef]
- Ghaedi, H.; Afkhami, A.; Madrakian, T.; Soltani-Felehgari, F. Construction of novel sensitive electrochemical sensor for electro-oxidation and determination of citalopram based on zinc oxide nanoparticles and multi-walled carbon nanotubes. Mater. Sci. Eng. C 2016, 59, 847–854. [Google Scholar] [CrossRef]
- Karimi-Harandi, M.H.; Shabani-Nooshabadi, M.; Darabi, R. Simultaneous determination of citalopram and selegiline using an efficient electrochemical sensor based on ZIF-8 decorated with RGO and g-C3N4 in real samples. Anal. Chim. Acta 2022, 1203, 339662. [Google Scholar] [CrossRef] [PubMed]
- Keypour, H.; Saremi, S.G.; Veisi, H.; Noroozi, M. Electrochemical determination of citalopram on new Schiff base functionalized magnetic Fe3O4 nanoparticle/MWCNTs modified glassy carbon electrode. J. Electroanal. Chem. 2016, 780, 160–168. [Google Scholar] [CrossRef]
- Rasheed, T.; Rizwan, K. Metal-organic frameworks based hybrid nanocomposites as state-of–the-art analytical tools for electrochemical sensing applications. Biosens. Bioelectron. 2022, 199, 113867. [Google Scholar] [CrossRef]
- Wang, Y.; Wang, L.; Huang, W.; Zhang, T.; Hu, X.; Perman, J.A.; Ma, S. A metal-organic framework and conducting polymer based electrochemical sensor for high performance cadmium ion detection. J. Mater. Chem. A 2017, 5, 8385–8393. [Google Scholar] [CrossRef]
- Sun, S.; Zhao, R.; Hao, W.; Guo, H.; Shi, L.; Su, X. Construction of ZIF-8/AuNPs/PVP–rGO/GCE Electrochemical Sensor and Its Sensitive Determination of Salbutamol. Russ. J. Electrochem. 2019, 55, 908–919. [Google Scholar] [CrossRef]
- Liang, C.; Lin, H.; Guo, W.; Lu, X.; Yu, D.; Fan, S.; Zhang, F.; Qu, F. Amperometric sensor based on ZIF/g-C3N4/RGO heterojunction nanocomposite for hydrazine detection. Microchim. Acta 2021, 188, 48. [Google Scholar] [CrossRef]
- Alizadeh, T.; Rafiei, F. An innovative application of graphitic carbon nitride (g-C3N4) nano-sheets as silver ion carrier in a solid state potentiometric sensor. Mater. Chem. Phys. 2019, 227, 176–183. [Google Scholar] [CrossRef]
- Sravani, B.; Kiranmai, S.; Rajasekhara Reddy, G.; Park, J.P.; VeeraManohara Reddy, Y.; Madhavi, G. Highly sensitive detection of anti-cancer drug based on bimetallic reduced graphene oxide nanocomposite. Chemosphere 2022, 287, 132281. [Google Scholar] [CrossRef]
- Ghosh, P.R.; Fawcett, D.; Sharma, S.B.; Poinern, G.E.J. Production of high-value nanoparticles via biogenic processes using aquacultural and horticultural food waste. Materials 2017, 10, 852. [Google Scholar] [CrossRef] [Green Version]
- Kim, C.H.; Kim, J.H.; Lee, S.M. Transformation of Solid Waste into Nanoporous Carbon via Carbothermic Reduction. ACS Omega 2018, 3, 7904–7910. [Google Scholar] [CrossRef]
- Kah, M.; Sigmund, G.; Xiao, F.; Hofmann, T. Sorption of ionizable and ionic organic compounds to biochar, activated carbon and other carbonaceous materials. Water Res. 2017, 124, 673–692. [Google Scholar] [CrossRef] [PubMed]
- Xu, R.K.; Xiao, S.C.; Yuan, J.H.; Zhao, A.Z. Adsorption of methyl violet from aqueous solutions by the biochars derived from crop residues. Bioresour. Technol. 2011, 102, 10293–10298. [Google Scholar] [CrossRef]
- Teixidó, M.; Pignatello, J.J.; Beltrán, J.L.; Granados, M.; Peccia, J. Speciation of the ionizable antibiotic sulfamethazine on black carbon (Biochar). Environ. Sci. Technol. 2011, 45, 10020–10027. [Google Scholar] [CrossRef]
- Yang, K.; Xing, B. Adsorption of organic compounds by carbon nanomaterials in aqueous phase: Polanyi theory and its application. Chem. Rev. 2010, 110, 5989–6008. [Google Scholar] [CrossRef]
- Oliveira, P.R.; Kalinke, C.; Mangrich, A.S.; Marcolino-Junior, L.H.; Bergamini, M.F. Copper hexacyanoferrate nanoparticles supported on biochar for amperometric determination of isoniazid. Electrochim. Acta 2018, 285, 373–380. [Google Scholar] [CrossRef]
- Wang, J.; Yang, J.; Xu, P.; Liu, H.; Zhang, L.; Zhang, S.; Tian, L. Gold nanoparticles decorated biochar modified electrode for the high-performance simultaneous determination of hydroquinone and catechol. Sens. Actuators B Chem. 2020, 306, 127590. [Google Scholar] [CrossRef]
- Trindade, C.M.B.; Silva, M.K.L.; Cesarino, I. Copper nanostructures anchored on renewable carbon as electrochemical platform for the detection of dopamine, fluoxetine and escitalopram. Sens. Actuators Rep. 2022, 4, 100107. [Google Scholar] [CrossRef]
- Babaei, A.; Afrasiabi, M.; Yousefi, A. Fe3O4@MCM-48-SO3H/ Multi-Wall Carbon Nanotubes Composite Modified Glassy Carbon Electrode: An Efficient Sensor for Sensitive and Selective Simultaneous Determination of Serotonin and Sertraline in the presence of Uric Acid. Anal. Bioanal. Electrochem. 2016, 11, 1–18. [Google Scholar]
- Salimi, A.; Sharifi, E.; Noorbakhsh, A.; Soltanian, S. Direct electrochemistry and electrocatalytic activity of catalase immobilized onto electrodeposited nano-scale islands of nickel oxide. Biophys. Chem. 2007, 125, 540–548. [Google Scholar] [CrossRef]
- Yuan, C.; Xiong, S.; Zhang, X.; Shen, L.; Zhang, F.; Gao, B.; Su, L. Template-free synthesis of ordered mesoporous NiO/poly(sodium-4-styrene sulfonate) functionalized carbon nanotubes composite for electrochemical capacitors. Nano Res. 2009, 2, 722–732. [Google Scholar] [CrossRef] [Green Version]
- Atty, S.A.; Ibrahim, A.H.; Ibrahim, H.; Abdelzaher, A.M.; Abdel-Raoof, A.M.; Fouad, F.A. Simultaneous voltammetric detection of anti-depressant drug, sertraline HCl and paracetamol in biological fluid at CNT-cesium modified electrode in micellar media. Microchem. J. 2020, 159, 105524. [Google Scholar] [CrossRef]
- Alizadeh, T.; Azizi, S. Graphene/graphite paste electrode incorporated with molecularly imprinted polymer nanoparticles as a novel sensor for differential pulse voltammetry determination of fluoxetine. Biosens. Bioelectron. 2016, 81, 198–206. [Google Scholar] [CrossRef]
- Ardelean, M.; Pode, R.; Schoonman, J.; Pop, A.; Manea, F. Fast simultaneous electrochemical detection of tetracycline and fluoxetine in water. WIT Trans. Ecol. Environ. 2017, 216, 213–220. [Google Scholar] [CrossRef] [Green Version]
- Hassan, S.S.M.; Kamel, A.H.; Amr, A.E.G.E.; Hashem, H.M.; Bary, E.M.A. Imprinted polymeric beads-based screen-printed potentiometric platforms modified with multi-walled carbon nanotubes (MWCNTs) for selective recognition of fluoxetine. Nanomaterials 2020, 10, 572. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Abd-Rabboh, H.S.M.; Hashem, H.M.; Al Shagri, L.M.S.; Amr, A.E.G.E.; Almehizia, A.A.; Naglah, A.M.; Kamel, A.H. All-Solid-State Potentiometric Platforms Modified with a Multi-Walled Carbon Nanotubes for Fluoxetine Determination. Membranes 2022, 12, 446. [Google Scholar] [CrossRef] [PubMed]
- Gholivand, M.B.; Akbari, A. A novel voltammetric sensor for citalopram based on multiwall carbon nanotube/(poly(p-aminobenzene sulfonic acid)/β-cyclodextrin). Mater. Sci. Eng. C 2016, 62, 480–488. [Google Scholar] [CrossRef]
- Oghli, A.H.; Soleymanpour, A. Polyoxometalate/reduced graphene oxide modified pencil graphite sensor for the electrochemical trace determination of paroxetine in biological and pharmaceutical media. Mater. Sci. Eng. C 2020, 108, 110407. [Google Scholar] [CrossRef]
- Hassan Oghli, A.; Soleymanpour, A. Ultrasensitive electrochemical sensor for simultaneous determination of sumatriptan and paroxetine using molecular imprinted polymer/sol-gel/polyoxometalate/rGO modified pencil graphite electrode. Sens. Actuators B Chem. 2021, 344, 130215. [Google Scholar] [CrossRef]
- Hashem, M.H.; Hassan, S.M.S.; Kamel, H.A.; Amr, A.E.-G.E.; AbdelBary, E.M. Cost-Effective Potentiometric Platforms Modified with Multi-Walled Carbon Nanotubes (MWCNTs) and Based on Imprinted Receptors for Fluvoxamine Assessment. Polymers 2020, 12, 673. [Google Scholar] [CrossRef] [Green Version]
- Khosrokhavar, R.; Motaharian, A.; Milani Hosseini, M.R.; Mohammadsadegh, S. Screen-printed carbon electrode (SPCE) modified by molecularly imprinted polymer (MIP) nanoparticles and graphene nanosheets for determination of sertraline antidepressant drug. Microchem. J. 2020, 159, 105348. [Google Scholar] [CrossRef]
- Emin Çorman, M.; Cetinkaya, A.; Armutcu, C.; Uzun, L.; Ozkan, S.A. Designing of ZnO nanoparticles oriented interface imprinted electrochemical sensor for fluoxetine detection. Bioelectrochemistry 2023, 152, 108411. [Google Scholar] [CrossRef]
- Ardelean, M.; Manea, F.; Pode, R. Silver Electrodeposited Carbon Nanofiber-Epoxy Electrode for Fluoxetine Detection. Int. J. Chem. Eng. Appl. 2016, 7, 169–172. [Google Scholar] [CrossRef] [Green Version]
- Habibi, B.; Pashazadeh, S.; Saghatforoush, L.A.; Pashazadeh, A. Direct electrochemical synthesis of the copper based metal-organic framework on/in the heteroatoms doped graphene/pencil graphite electrode: Highly sensitive and selective electrochemical sensor for sertraline hydrochloride. J. Electroanal. Chem. 2021, 888, 115210. [Google Scholar] [CrossRef]
- Onorato, J.W.; Luscombe, C.K. Morphological effects on polymeric mixed ionic/electronic conductors. Mol. Syst. Des. Eng. 2019, 4, 310–324. [Google Scholar] [CrossRef]
- Madej, M.; Matoga, D.; Skaźnik, K.; Porada, R.; Baś, B.; Kochana, J. A voltammetric sensor based on mixed proton-electron conducting composite including metal-organic framework JUK-2 for determination of citalopram. Microchim. Acta 2021, 188, 184. [Google Scholar] [CrossRef] [PubMed]
- Aminikhah, M.; Babaei, A.; Taheri, A. A novel electrochemical sensor based on molecularly imprinted polymer nanocomposite platform for sensitive and ultra-selective determination of citalopram. J. Electroanal. Chem. 2022, 918, 116493. [Google Scholar] [CrossRef]
- Al-Mhyawi, S.R.; Ahmed, R.K.; El Nashar, R.M. Application of a conducting poly-methionine/gold nanoparticles-modified sensor for the electrochemical detection of paroxetine. Polymers 2021, 13, 3981. [Google Scholar] [CrossRef]
- Ajayi, R.F.; Nxusani, E.; Douman, S.F.; Jonnas, A.; Baker, P.G.L.; Iwuoha, E.I. An amperometric cytochrome P450-2D6 biosensor system for the detection of the selective serotonin reuptake inhibitors (SSRIs) paroxetine and fluvoxamine. J. Nano Res. 2016, 44, 208–228. [Google Scholar] [CrossRef]








| Analyte | Coating | Technique | Range (µM) | LOD (µM) | Interference | Sample | Ref |
|---|---|---|---|---|---|---|---|
| FLX | MIP-GCE | CV, SWV, DPV | 0.499–33.8 | 0.33 | Biological species | Blood serum | [40] |
| Poly(pyrrole) | Potentiometry | 1–1000 | 0.63 | Inorganic species and amino acids | FLX.HC:L capsules | [41] | |
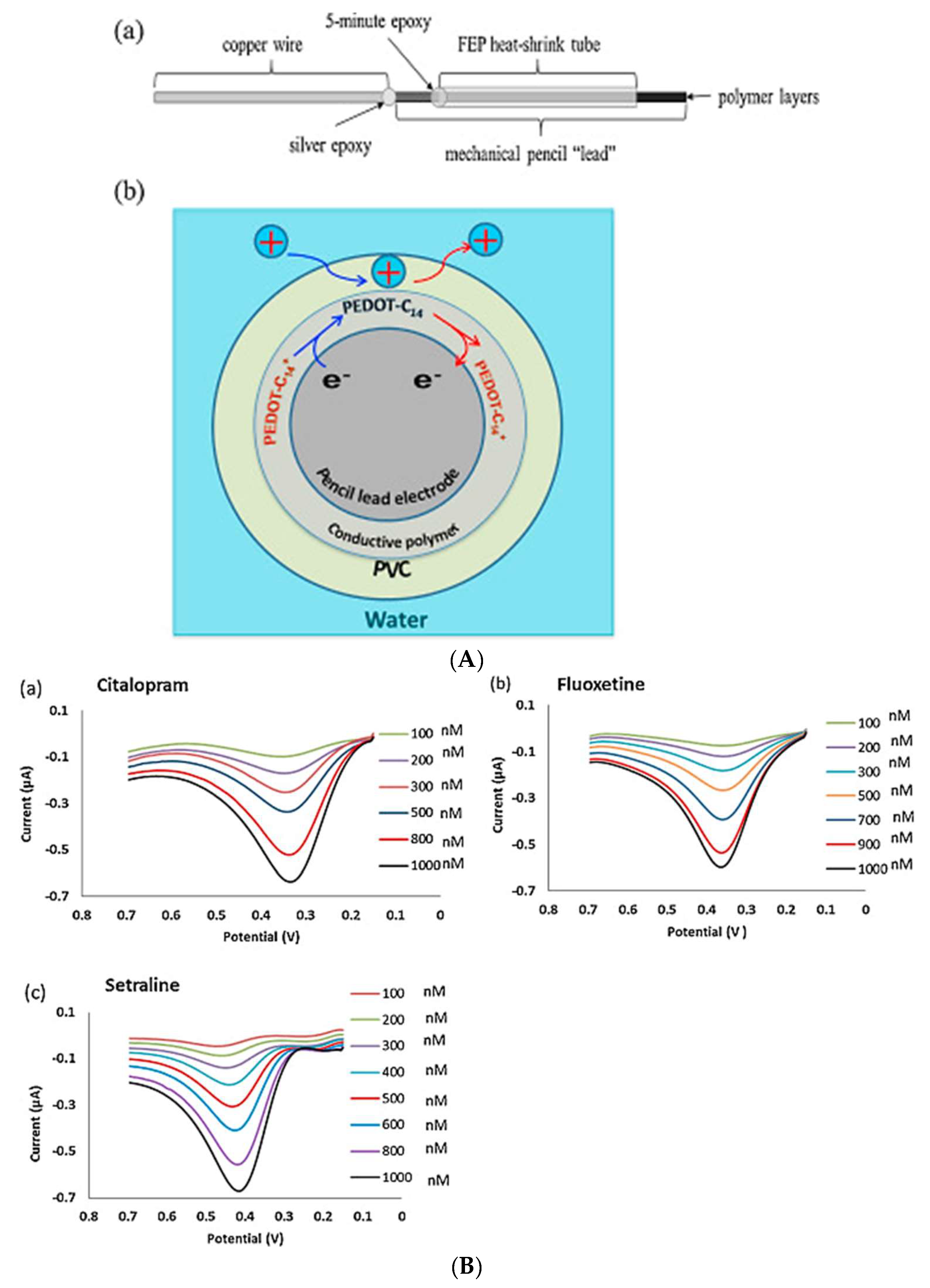
| FLX CIT STR | PVC/PEDOT-C14-PLE | ITSV | 0.1–1 | 0.035 0.025 0.045 | None | Tap and river water | [42] |
| FLV | PM-PAN-GCE | Potentiomettry | 0.18–1300 | 0.078 | Inorganic species, organic molecules, and amino acids | Human serum and urine | [18] |
| STR | HPβCD, βCD-Au electrode | CV, SWV | 0.1–0.9 | 0.026 | None | Human serum and Sidata tablet | [44] |
| Analyte | Coating | Technique | Range (µM) | LOD (µM) | Interference | Sample | Ref |
|---|---|---|---|---|---|---|---|
| ESC | EGPU-GR | DPV, SWV | 1.5–12 | 0.25 | Organic molecules and amino acids | Urine and CSF | [51] |
| PRX | CSS-SPE | SWV | 1–100 | 0.67 | Biological species | Tap water | [56] |
| Analyte | Coating | Technique | Range (µM) | LOD (µM) | Interference | Sample | Ref |
|---|---|---|---|---|---|---|---|
| ESC | NiCACP | CV, DPV, EIS | 1–70 | 0.2 | Amino acids, inorganic cations and sugars | Tablet and urine | [61] |
| STR | ZnFe2O4-SPE | DPV | 0.07–300 | 0.02 | Amino acids, sugars, inorganic-, organic molecules and biological species | Urine and tablet | [28] |
| La2O3/CO3O4-SPE | DPV | 5–400 | 1 | None | Urine and tablet | [31] | |
| La3+/ZnO-SPE | CV, DPV | 0.5–150 | 0.15 | None | Urine and tablet | [62] | |
| ZnO-NFs/GSPE | DPV | N/A | N/A | Organic and inorganic molecules | Urine and tablet | [63] |
| Analyte | Coating | Technique | Range (µM) | LOD (µM) | Interference | Sample | Ref |
|---|---|---|---|---|---|---|---|
| CIT | Au-PdNPs-GR-AuE | CV, SWV, EIS | 0.5–50 | 0.049 | Inorganic and organic molecules, amino acids, and sugars | Human plasma and tablets | [60] |
| ZnO-MWCNT-CPE | CV, ASWV | 0.012–1.54 | 0.005 | Serum, urine and tablets | [67] | ||
| Fe3O4@[(EtO)3Si-L]/MWCNTs-GCE | CV, DPV | 0.3–10,000 | 0.0532 | Blood and tablets | [69] | ||
| ZIF-8/g-C3N4/RGO-CPE | CV, DPV, EIS | 0.009–900 | 0.008 | Blood, urine and tablets | [68] | ||
| ESC | GC-RC-CuNP | CV, DPV | 0.02–5 | 0.25 | Organic and inorganic molecules, and amino acids | Tap water and urine | [84] |
| FLX | 0.1–10 | 0.05 | |||||
| STR | Fe3O4@MCM-48-SO3H/MWCNTs-GCE | CV, DPV | 0.05–100 | 0.015 | Inorganic and organic molecules, amino acids, and sugars | Blood and urine plasma | [85] |
| MWCNT-IL/NiONPs-GCE | CV, DPAdSV | 0.21–85 | 0.047 | Serum and tablets | [58] | ||
| CNT/CsM/SDS-CPE | CV, SWV | 0.06–15 | 0.0092 | Plasma and tablets | [88] |
| Analytes | Coating | Technique | Range (µM) | LOD (µM) | Interference | Sample | Ref |
|---|---|---|---|---|---|---|---|
| FLX | Nano-MIP/G2-CP | DPV | 0.006–0.1 | 0.0015 | Inorganic species, amino acids and sugars | Tablet and plasma | [89] |
| CNF | CV, DPV | 0–10 | 0.385 | None | Water | [90] | |
| MIP-MWCNTs-SPE | EIS | 0.1–10,000 | 2.1 | Organic and inorganic species, amino acids and sugars | tablet | [91] | |
| Ionophore I, Ionophore II and Ionophore III-MWCNTs-SPE | Potentiometry | 0.2–6.5 | 5.2, 4.7, 0.2 | Ionic and biological species | Blood | [92] | |
| CIT | P(pABSA)/β-CD/MWCNT-GCE | CV, EIS | 0.09–100 | 0.044 | Sugars and organic species | Tablet and serum | [93] |
| PRX | rGo/PWA/PGE | DPV | 0.008–6 | 0.9 nM | Ionic and biological species | Serum, urine and tablet | [94] |
| MIPP,S/Sol-Gel/PWA/rGO/PGE | adDPV | 0.005–2.2 | 0.7 nM | Blood, urine and tablet | [95] | ||
| FLV | MWCNTs/MIP-SPE | CP, EIS | 0.1–10,000 | 4.8 | Inorganic and organic species, amino acids and sugars | Tablet | [96] |
| STR | Graphene-MIPPtE | CV, DPV | 0.01–1 | 0.007 | Inorganic and organic species, amino acids and sugars | Serum | [30] |
| MIP/graphene-SPCE | CV, DPV | 0.005–0.075 | 0.002 | Dapoxetine, fluoxetine, citalopram, uric acid and ascorbic acid | Serum and tablet | [97] |
| Analyte | Coating | Technique | Range (µM) | LOD (µM) | Interference | Sample | Ref |
|---|---|---|---|---|---|---|---|
| FLX | FLX@ZnO-MAPA@MIP/GCE | DPV | 0.01–0.1 nM | 0.00267 nM | Antibiotics | Tap water and serum | [98] |
| Analytes | Coating | Technique | Range (µM) | LOD (µM) | Interference | Sample | Ref |
|---|---|---|---|---|---|---|---|
| FLX | AgCFE | CV, CA | 0–10 | 0.079 | None | None | [99] |
| CIT | JUK-2-MWCNTs-AuNPsGCE | CV, EIS | 0.05–115 | 0.011 | Organic and inorganic compounds | Tablets, water, urine and serum | [102] |
| MIP-hNiNS-AMWCNT@GONRs-GCE | CV, DPV, EIS | 0.5–190 | 0.043 | Tablets, urine and serum | [103] | ||
| PRX | Poly(ᴅʟ-met)/AuNPs-GCE | CV, DPV, EIS | 0.05 nM–100 µM | 0.01 nM | Sugars, organic molecules, biological species and amino acids | PRX tablets | [104] |
| STR | Ni(II)-LD/AuNPs/MWCNTs-GCE | CV | 0.05–5.5 | 0.095 | Inorganic and organic molecules, amino acids and sugars | Serum | [64] |
| CuMOF/SNDGrPGE | CV, DPV | 0.05–2.67 | 0.038 | Blood and tablet | [100] |
| Analyte | Coating | Technique | Range (µM) | LOD (µM) | Interference | Sample | Ref |
|---|---|---|---|---|---|---|---|
| PAR | Poly-2D6 | CV | 0.005–0.05 | 0.002 | N/A | N/A | [105] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Barry, S.C.L.; Franke, C.; Mulaudzi, T.; Pokpas, K.; Ajayi, R.F. Review on Surface-Modified Electrodes for the Enhanced Electrochemical Detection of Selective Serotonin Reuptake Inhibitors (SSRIs). Micromachines 2023, 14, 1334. https://doi.org/10.3390/mi14071334
Barry SCL, Franke C, Mulaudzi T, Pokpas K, Ajayi RF. Review on Surface-Modified Electrodes for the Enhanced Electrochemical Detection of Selective Serotonin Reuptake Inhibitors (SSRIs). Micromachines. 2023; 14(7):1334. https://doi.org/10.3390/mi14071334
Chicago/Turabian StyleBarry, Simone C. L., Candice Franke, Takalani Mulaudzi, Keagan Pokpas, and Rachel Fanelwa Ajayi. 2023. "Review on Surface-Modified Electrodes for the Enhanced Electrochemical Detection of Selective Serotonin Reuptake Inhibitors (SSRIs)" Micromachines 14, no. 7: 1334. https://doi.org/10.3390/mi14071334







