Evolution of Free Volumes in Polycrystalline BaGa2O4 Ceramics Doped with Eu3+ Ions
Abstract
:1. Introduction
2. Materials and Methods
3. Results and Discussion
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Noto, L.L.; Poelman, D.; Orante-Barrón, V.R.; Swart, H.C.; Mathevula, L.E.; Nyenge, R.; Chithambo, M.; Mothudi, B.M.; Dhlamini, M.S. Photoluminescence and thermoluminescence properties of BaGa2O4. Phys. B Condens. Matter 2018, 535, 268–271. [Google Scholar] [CrossRef]
- Kodu, M.; Avarmaa, T.; Mändar, H.; Jaaniso, R. Pulsed Laser Deposition of BaGa2O4. Appl. Phys. A 2008, 93, 801–805. [Google Scholar] [CrossRef]
- Gust, D.; Moore, T.A.; Moore, A.L. Solar Fuels via Artificial Photosynthesis. Acc. Chem. Res. 2009, 42, 1890–1898. [Google Scholar] [CrossRef]
- Skillen, N.; Robertson, P.K.J. Artificial photosynthesis. In Solar Energy; World Scientific Publishing Co. Pte. Ltd.: Singapore, 2016; pp. 205–241. [Google Scholar] [CrossRef]
- Acuña, W.; Tellez, J.F.; Macías, M.A.; Roussel, P.; Ricote, S.; Gauthier, G.H. Synthesis and Characterization of BaGa2O4 and Ba3Co2O6(CO3)0.6 Compounds in the Search of Alternative Materials for Proton Ceramic Fuel Cell (PCFC). Solid State Sci. 2017, 71, 61–68. [Google Scholar] [CrossRef]
- Guérineau, T.; Strutynski, C.; Skopak, T.; Morency, S.; Hanafi, A.; Calzavara, F.; Ledemi, Y.; Danto, S.; Cardinal, T.; Messaddeq, Y.; et al. Extended Germano-Gallate Fiber Drawing Domain: From Germanates to Gallates Optical Fibers. Opt. Mater. Express 2019, 9, 2437–2445. [Google Scholar] [CrossRef] [Green Version]
- Gonçalves, J.M.; Munoz, R.A.; Angnes, L. Materials for Optical, Magnetic and Electronic Devices. J. Mater. Chem. C 2021, 9, 8708–8717. [Google Scholar] [CrossRef]
- Nakauchi, D.; Okada, G.; Kawaguchi, N.; Yanagida, T. Luminescent and Scintillation Properties of Eu-Doped (Ba, Sr)Al2O4 Crystals. Opt. Mater. 2019, 87, 58–62. [Google Scholar] [CrossRef]
- Khattab, T.A.; Abd El-Aziz, M.; Abdelrahman, M.S.; El-Zawahry, M.; Kamel, S. Development of Long-persistent Photoluminescent Epoxy Resin Immobilized with Europium (II)-doped Strontium Aluminate. Luminescence 2020, 35, 478–485. [Google Scholar] [CrossRef]
- Yu, L.; den Engelsen, D.; Gorobez, J.; Fern, G.R.; Ireland, T.G.; Frampton, C.; Silver, J. Crystal Structure, Photoluminescence and Cathodoluminescence of Sr1-xCaxAl2O4 Doped with Eu2+. Opt. Mater. Express 2019, 9, 2175–2195. [Google Scholar] [CrossRef]
- Maphiri, V.M.; Mhlongo, M.R.; Hlatshwayo, T.T.; Motaung, T.E.; Koao, L.F.; Motloung, S.V. Citrate Sol-Gel Synthesis of BaAl2O4:X% Cu2+ (0 ≤ x ≤ 1) Nano-Phosphors: Structural, Morphological and Photoluminescence Properties. Opt. Mater. 2020, 109, 110244. [Google Scholar] [CrossRef]
- den Engelsen, D.; Fern, G.R.; Ireland, T.G.; Yang, F.; Silver, J. Photoluminescence and Cathodoluminescence of BaAl2O4:Eu2+ and Undoped BaAl2O4: Evidence for F-Centres. Opt. Mater. Express 2020, 10, 1962–1980. [Google Scholar] [CrossRef]
- den Engelsen, D.; Fern, G.R.; Ireland, T.G.; Silver, J. Laser-Activated Luminescence of BaAl2O4:Eu. ECS J. Solid State Sci. Technol. 2020, 9, 026001. [Google Scholar] [CrossRef]
- Shivaramu, N.J.; Coetsee, E.; Roos, W.D.; Nagabhushana, K.R.; Swart, H.C. Charge Carrier Trapping Processes in Un-Doped and BaAl2O4:Eu3+ Nanophosphor for Thermoluminescent Dosimeter Applications. J. Phys. D Appl. Phys. 2020, 53, 475305. [Google Scholar] [CrossRef]
- Xie, Q.; Li, B.; He, X.; Zhang, M.; Chen, Y.; Zeng, Q. Correlation of Structure, Tunable Colors, and Lifetimes of (Sr, Ca, Ba)Al2O4:Eu2+, Dy3+ Phosphors. Materials 2017, 10, 1198. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wang, S.; Wang, Y.; Gao, H.; Li, J.; Fang, L.; Yu, X.; Tang, S.; Zhao, X.; Sun, G. Synthesis and Characterization of BaAl2O4: Ce and Mn-Ce-Co-Doped BaAl2O4 Composite Materials by a Modified Polyacrylamide Gel Method and Prediction of Photocatalytic Activity Using Artificial Neural Network (ANN) Algorithm. Optik 2020, 221, 165363. [Google Scholar] [CrossRef]
- Malkamäki, M.; Bos, A.J.J.; Dorenbos, P.; Lastusaari, M.; Rodrigues, L.C.V.; Swart, H.C.; Hölsä, J. Persistent Luminescence Excitation Spectroscopy of BaAl2O4:Eu2+,Dy3+. Phys. B Condens. Matter 2020, 593, 411947. [Google Scholar] [CrossRef]
- Nakauchi, D.; Okada, G.; Kato, T.; Kawaguchi, N.; Yanagida, T. Crystal Growth and Scintillation Properties of Eu:BaAl2O4 Crystals. Radiat. Meas. 2020, 135, 106365. [Google Scholar] [CrossRef]
- Vrankić, M.; Šarić, A.; Bosnar, S.; Pajić, D.; Dragović, J.; Altomare, A.; Falcicchio, A.; Popović, J.; Jurić, M.; Petravić, M.; et al. Magnetic Oxygen Stored in Quasi-1D Form within BaAl2O4 Lattice. Sci. Rep. 2019, 9, 15158. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lisovskii, S.; Meganov, A.; Khrustov, V.R.; Ivanov, V. Ultraviolet Cathodoluminescence of Pure Zinc Aluminate ZnAl2O4. J. Phys. Conf. Ser. 2019, 1410, 012089. [Google Scholar] [CrossRef]
- Valiev, D.; Khasanov, O.; Dvilis, E.; Stepanov, S.; Paygin, V.; Ilela, A. Structural and Spectroscopic Characterization of Tb3+-Doped MgAl2O4 Spinel Ceramics Fabricated by Spark Plasma Sintering Technique. Phys. Status Solidi (B) 2020, 257, 1900471. [Google Scholar] [CrossRef]
- Lushchik, A.; Feldbach, E.; Kotomin, E.A.; Kudryavtseva, I.; Kuzovkov, V.N.; Popov, A.I.; Seeman, V.; Shablonin, E. Distinctive Features of Diffusion-Controlled Radiation Defect Recombination in Stoichiometric Magnesium Aluminate Spinel Single Crystals and Transparent Polycrystalline Ceramics. Sci. Rep. 2020, 10, 7810. [Google Scholar] [CrossRef] [PubMed]
- Mironova-Ulmane, N.; Popov, A.I.; Krieke, G.; Antuzevics, A.; Skvortsova, V.; Elsts, E.; Sarakovskis, A. Low-Temperature Studies of Cr3+ Ions in Natural and Neutron-Irradiated g-Al Spinel. Low Temp. Phys. 2020, 46, 1154–1159. [Google Scholar] [CrossRef]
- Platonenko, A.; Gryaznov, D.; Kotomin, E.A.; Lushchik, A.; Seeman, V.; Popov, A.I. Hybrid Density Functional Calculations of Hyperfine Coupling Tensor for Hole-Type Defects in MgAl2O4. Nucl. Instrum. Methods Phys. Res. Sect. B Beam Interact. Mater. At. 2020, 464, 60–64. [Google Scholar] [CrossRef]
- Li, Q.; Liu, T.; Xu, X.; Wang, X.; Guo, R.; Jiao, X.; Lu, Y. Study on the Optical Spectra of MgAl2O4 with Oxygen Vacancies. Mater. Technol. 2021, 36, 279–285. [Google Scholar] [CrossRef]
- Li, Q.; Liu, T.; Xu, X.; Guo, R.; Jiao, X.; Wang, X.; Lu, Y. Study of Cation Vacancies with Localized Hole States in MgAl2O4 Crystals. J. Phys. Chem. Solids 2020, 145, 109542. [Google Scholar] [CrossRef]
- Seeman, V.; Feldbach, E.; Kärner, T.; Maaroos, A.; Mironova-Ulmane, N.; Popov, A.I.; Shablonin, E.; Vasil’chenko, E.; Lushchik, A. Fast-Neutron-Induced and as-Grown Structural Defects in Magnesium Aluminate Spinel Crystals with Different Stoichiometry. Opt. Mater. 2019, 91, 42–49. [Google Scholar] [CrossRef]
- Gholami, Y.H.; Yuan, H.; Wilks, M.Q.; Josephson, L.; el Fakhri, G.; Normandin, M.D.; Kuncic, Z. Positron Annihilation Localization by Nanoscale Magnetization. Sci. Rep. 2020, 10, 20262. [Google Scholar] [CrossRef]
- Rementeria, R.; Domínguez-Reyes, R.; Capdevila, C.; Garcia-Mateo, C.; Caballero, F.G. Positron Annihilation Spectroscopy Study of Carbon-Vacancy Interaction in Low-Temperature Bainite. Sci. Rep. 2020, 10, 487. [Google Scholar] [CrossRef] [Green Version]
- Dlubek, G.; Kilburn, D.; Bondarenko, V.; Pionteck, J.; Krause-Rehberg, R.; Alam, M.A. Positron Annihilation: A Unique Method for Studying Polymers. Macromol. Symp. 2004, 210, 11–20. [Google Scholar] [CrossRef]
- Dai, H.Y.; Liu, H.Z.; Peng, K.; Ye, F.J.; Li, T.; Chen, J.; Chen, Z.P. Correlation between Vacancy Defects and Magnetic Properties of the GdMn1-xZnxO3 Multiferroic Ceramics Studied by Positron Annihilation. Mater. Res. Bull. 2019, 119, 110565. [Google Scholar] [CrossRef]
- Barad, D.; Mange, P.L.; Jani, K.K.; Mukherjee, S.; Ahmed, M.; Kumar, S.; Dolia, S.N.; Pandit, R.; Raval, P.Y.; Modi, K.B.; et al. Ca2+-Substitution Effect on the Defect Structural Changes in the Quadruple Perovskite Series Ca1+xCu3-xTi4O12 Studied by Positron Annihilation and Complementary Methods. Ceram. Int. 2021, 47, 2631–2640. [Google Scholar] [CrossRef]
- Ghanem, A.; Mohamed, K. Effect of Gamma-Ray on Producing Induced Colour Centres and on Positron Annihilation Lifetime of Bismuth-Doped Zinc Sodium Borate Glasses. Arab. J. Nucl. Sci. Appl. 2021, 54, 1–9. [Google Scholar] [CrossRef]
- Ashok, J.; Kostrzewa, M.; Ingram, A.; Reddy, M.S.; Kumar, V.R.; Gandhi, Y.; Veeraiah, N. Free Volume Estimation in Au and Ag Mixed Sodium Antimonate Glass Ceramics by Means of Positron Annihilation. Phys. B Condens. Matter 2019, 570, 266–273. [Google Scholar] [CrossRef]
- El-Gamal, S.; Elsayed, M. Positron Annihilation and Electrical Studies on the Influence of Loading Magnesia Nanoribbons on PVA-PVP Blend. Polym. Test. 2020, 89, 106681. [Google Scholar] [CrossRef]
- Zhang, H.J.; Sellaiyan, S.; Sako, K.; Uedono, A.; Taniguchi, Y.; Hayashi, K. Effect of Free-Volume Holes on Static Mechanical Properties of Epoxy Resins Studied by Positron Annihilation and PVT Experiments. Polymer 2020, 190, 122225. [Google Scholar] [CrossRef]
- Biswas, D.; Rajan, A.; Kabi, S.; Das, A.S.; Singh, L.S.; Nambissan, P.M.G. Structural Defects Characterization of Silver-Phosphate Glass Nanocomposites by Positron Annihilation and Related Experimental Studies. Mater. Charact. 2019, 158, 109928. [Google Scholar] [CrossRef]
- Fan, J.; Zhou, W.; Wang, Q.; Chu, Z.; Yang, L.; Yang, L.; Sun, J.; Zhao, L.; Xu, J.; Liang, Y.; et al. Structure Dependence of Water Vapor Permeation in Polymer Nanocomposite Membranes Investigated by Positron Annihilation Lifetime Spectroscopy. J. Membr. Sci. 2018, 549, 581–587. [Google Scholar] [CrossRef]
- Sato, K.; Tamiya, R.; Xu, Q.; Tsuchida, H.; Yoshiie, T. Detection of Deuterium Trapping Sites in Tungsten by Thermal Desorption Spectroscopy and Positron Annihilation Spectroscopy. Nucl. Mater. Energy 2016, 9, 554–559. [Google Scholar] [CrossRef] [Green Version]
- Wang, Z.F.; Wang, B.; Yang, Y.R.; Hu, C.P. Correlations between Gas Permeation and Free-Volume Hole Properties of Polyurethane Membranes. Eur. Polym. J. 2003, 39, 2345–2349. [Google Scholar] [CrossRef]
- Melikhova, O.; Kuriplach, J.; Prochazka, I.; Cizek, J.; Hou, M.; Zhurkin, E.; Pisov, S. Simulation of Positron Annihilation Response to Mechanical Deformation of Nanostructured Ni3Al. Appl. Surf. Sci. 2008, 255, 157–159. [Google Scholar] [CrossRef]
- Shpotyuk, O.; Balitska, V.; Brunner, M.; Hadzaman, I.; Klym, H. Thermally-Induced Electronic Relaxation in Structurally-Modified Cu0.1Ni0.8Co0.2Mn1.9O4 Spinel Ceramics. Phys. B Condens. Matter 2015, 459, 116–121. [Google Scholar] [CrossRef]
- Klym, H.; Ingram, A.; Shpotyuk, O.; Karbovnyk, I. Influence of CsCl Addition on the Nanostructured Voids and Optical Properties of 80GeS2-20Ga2S3 Glasses. Opt. Mater. 2016, 59, 39–42. [Google Scholar] [CrossRef]
- Kostiv, Y.; Luchechko, A.; Klym, H.; Karbovnyk, I.; Sadovyi, B.; Zaremba, O.; Kravets, O. Structural properties of polycrystalline BaGa2O4 Ceramics Doped with Eu3+ Ions. In Proceedings of the XIth International Scientific and Practical Conference on Electronics and Information Technologies, Lviv, Ukraine, 16–18 September 2019; pp. 307–311. [Google Scholar] [CrossRef]
- Klym, H.; Ingram, A.; Hadzaman, I.; Shpotyuk, O. Evolution of Porous Structure and Free-volume Entities in Magnesium Aluminate Spinel Ceramics. Ceram. Int. 2014, 40, 8561–8567. [Google Scholar] [CrossRef]
- Kansy, J.; Giebel, D. Study of Defect Structure with New Software for Numerical Analysis of PAL Spectra. J. Phys. Conf. Ser. 2011, 265, 0102030. [Google Scholar] [CrossRef] [Green Version]
- Goworek, T. Comments on the Relation: Positronium Lifetime—Free Volume Size Parameters of the Tao-Eldrup Model. Chem. Phys. Lett. 2002, 366, 184–187. [Google Scholar] [CrossRef]
- Lushchik, A.; Dolgov, S.; Feldbach, E.; Pareja, R.; Popov, A.I.; Shablonin, E.; Seeman, V. Creation and Thermal Annealing of Structural Defects in Neutron-Irradiated MgAl2O4 Single Crystals. Nucl. Instrum. Methods Phys. Res. Sect. B Beam Interact. Mater. At. 2018, 435, 31–37. [Google Scholar] [CrossRef]
- Feldbach, E.; Museur, L.; Krasnenko, V.; Zerr, A.; Kitaura, M.; Kanaev, A. Defects Induced by He+ Irradiation in γ-Si3N4. J. Lumin. 2021, 237, 118132. [Google Scholar] [CrossRef]
- Feldbach, E.; Zerr, A.; Museur, L.; Kitaura, M.; Manthilake, G.; Tessier, F.; Krasnenko, V.; Kanaev, A. Electronic Band Transitions in γ-Ge3N4. Electron. Mater. Lett. 2021, 17, 315–323. [Google Scholar] [CrossRef]
- Kozlovskiy, A.; Kenzhina, I.; Zdorovets, M.V. Optical and Structural Properties of AlN Ceramics Irradiated with Heavy Ions. Opt. Mater. 2019, 91, 130–137. [Google Scholar] [CrossRef]
- Zdorovets, M.V.; Dukenbayev, K.; Kozlovskiy, A.L. Study of Helium Swelling in Nitride Ceramics at Different Irradiation Temperatures. Materials 2019, 12, 2415. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kozlovskiy, A.; Kenzhina, I.; Dukenbayev, K.; Zdorovets, M. Influence of He-ion Irradiation of Ceramic AlN. Vacuum 2019, 163, 45–51. [Google Scholar] [CrossRef]
- Kurteeva, A.A.; Bogdanovich, N.M.; Bronin, D.I.; Porotnikova, N.M.; Vdovin, G.K.; Pankratov, A.A.; Beresnev, S.M.; Kuz’mina, L.A. Options for Adjustment of Microstructure and Conductivity of Cathodic Substrates of La(Sr)MnO3. Russ. J. Electrochem. 2010, 46, 811–819. [Google Scholar] [CrossRef]
- Porotnikova, N.M.; Eremin, V.A.; Farlenkov, A.S.; Kurumchin, E.K.; Sherstobitova, E.A.; Kochubey, D.I.; Ananyev, M.V. Effect of AO Segregation on Catalytical Activity of La0.7A0.3MnO3±δ (A = Ca, Sr, Ba) Regarding Oxygen Reduction Reaction. Catal. Lett. 2018, 148, 2839–2847. [Google Scholar] [CrossRef]
- Osinkin, D.A.; Khodimchuk, A.V.; Porotnikova, N.M.; Bogdanovich, N.M.; Fetisov, A.V.; Ananyev, M.V. Rate-Determining Steps of Oxygen Surface Exchange Kinetics on Sr2Fe1.5Mo0.5O6−δ. Energies 2020, 13, 250. [Google Scholar] [CrossRef] [Green Version]
- Suchikova, Y.O. Sulfide Passivation of Indium Phosphide Porous Surfaces. J. Nano-Electron. Phys. 2017, 9, 01006. [Google Scholar] [CrossRef]
- Suchikova, J.A. Synthesis of Indium Nitride Epitaxial Layers on a Substrate of Porous Indium Phosphide. J. Nano-Electron. Phys. 2015, 7, 03017. [Google Scholar]
- Shlimas, D.; Kenzhina, I.; Zdorovets, M. Study of the Use of Ionizing Radiation to Improve the Efficiency of Performance of Nickel Nanostructures as Anodes of Lithium-ion Batteries. Mater. Res. Express 2019, 6, 055026. [Google Scholar] [CrossRef]
- Rumiantseva, Y.; Melnichuk, I.; Garashchenko, V.; Zaporozhets, O.; Turkevich, V.; Bushlya, V. Influence of cBN Content, Al2O3 and Si3N4 Additives and Their Morphology on Microstructure, Properties, and Wear of PCBN with NbN Binder. Ceram. Int. 2020, 46, 22230–22238. [Google Scholar] [CrossRef]
- Olenych, I.B.; Aksimentyeva, O.I.; Monastyrskii, L.S.; Horbenko, Y.Y.; Partyka, M.V. Electrical and Photoelectrical Properties of Reduced Graphene Oxide—Porous Silicon Nanostructures. Nanoscale Res. Lett. 2017, 12, 272. [Google Scholar] [CrossRef]
- Luchechko, A.; Zhydachevskyy, Y.; Ubizskii, S.; Kravets, O.; Popov, A.I.; Rogulis, U.; Elsts, E.; Bulur, E.; Suchocki, A. Afterglow, TL and OSL Properties of Mn2+-doped ZnGa2O4 Phosphor. Sci. Rep. 2019, 9, 9544. [Google Scholar] [CrossRef]
- Dimza, V.; Popov, A.I.; Lāce, L.; Kundzins, M.; Kundzins, K.; Antonova, M.; Livins, M. Effects of Mn Doping on Dielectric Properties of Ferroelectric Relaxor PLZT Ceramics. Curr. Appl. Phys. 2017, 17, 169–173. [Google Scholar] [CrossRef]
- Bystrova, A.; Dekhtyar, Y.D.; Popov, A.; Coutinho, J.; Bystrov, V. Modified Hydroxyapatite Structure and Properties: Modeling and Synchrotron Data Analysis of Modified Hydroxyapatite Structure. Ferroelectrics 2015, 475, 135–147. [Google Scholar] [CrossRef]
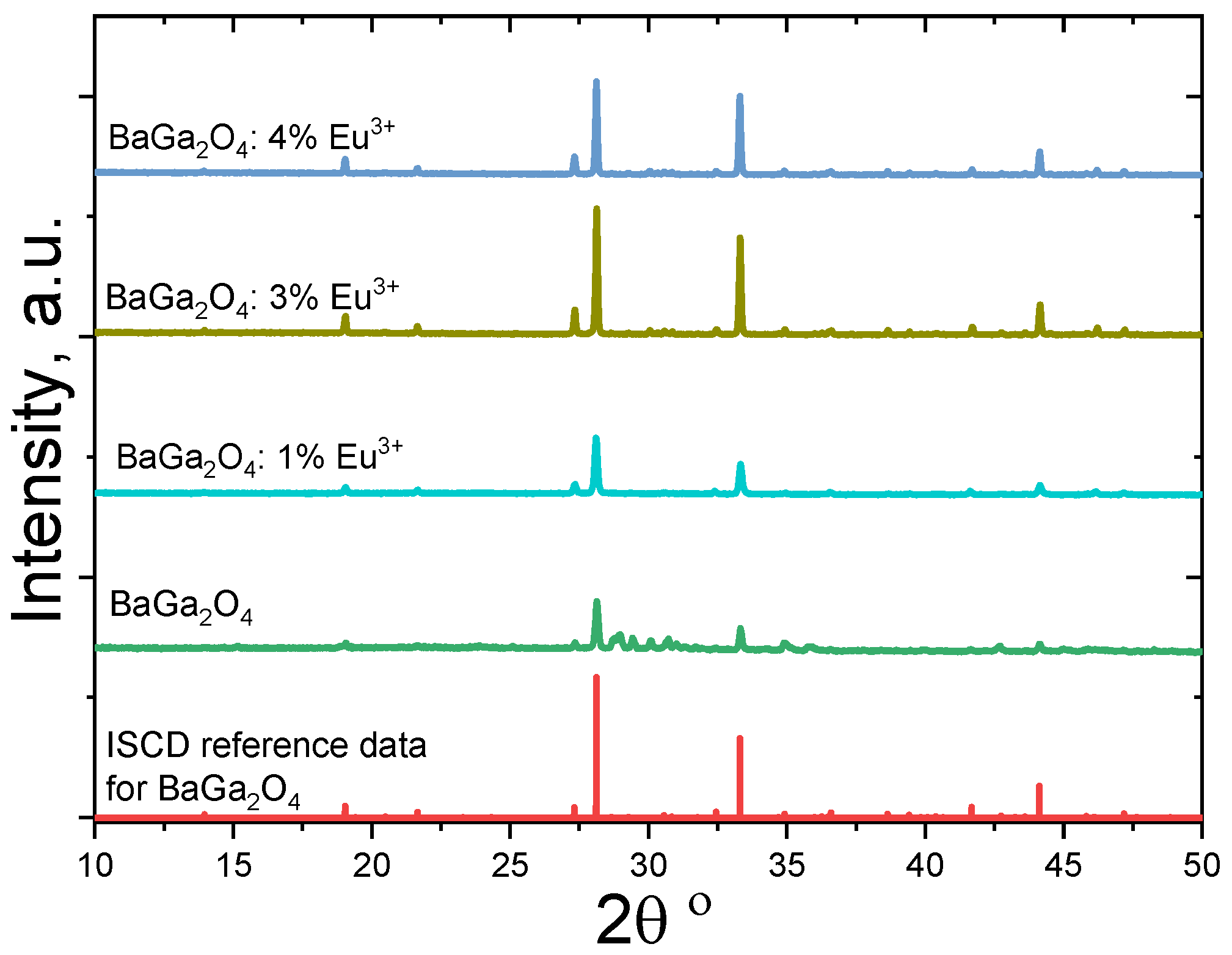

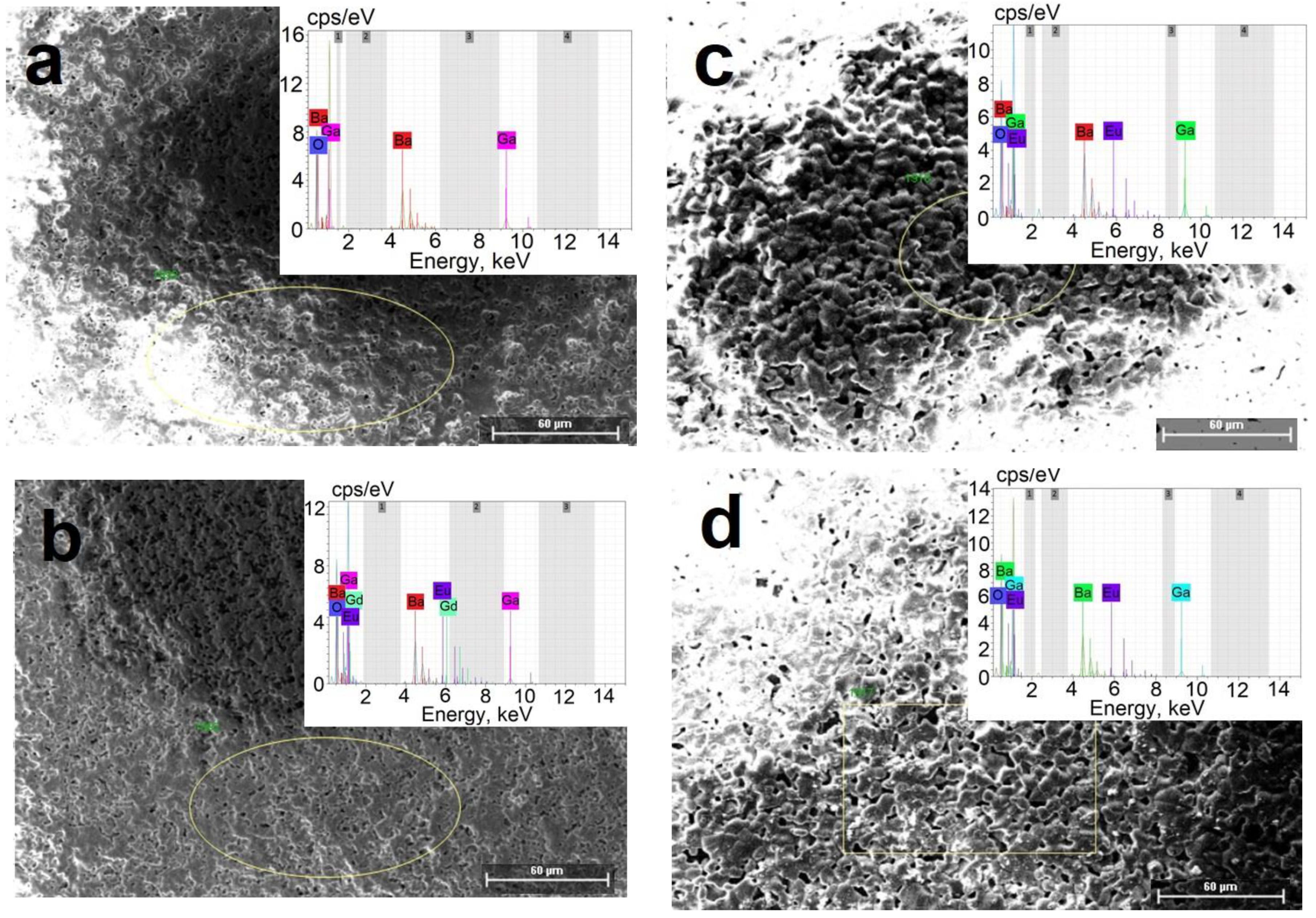
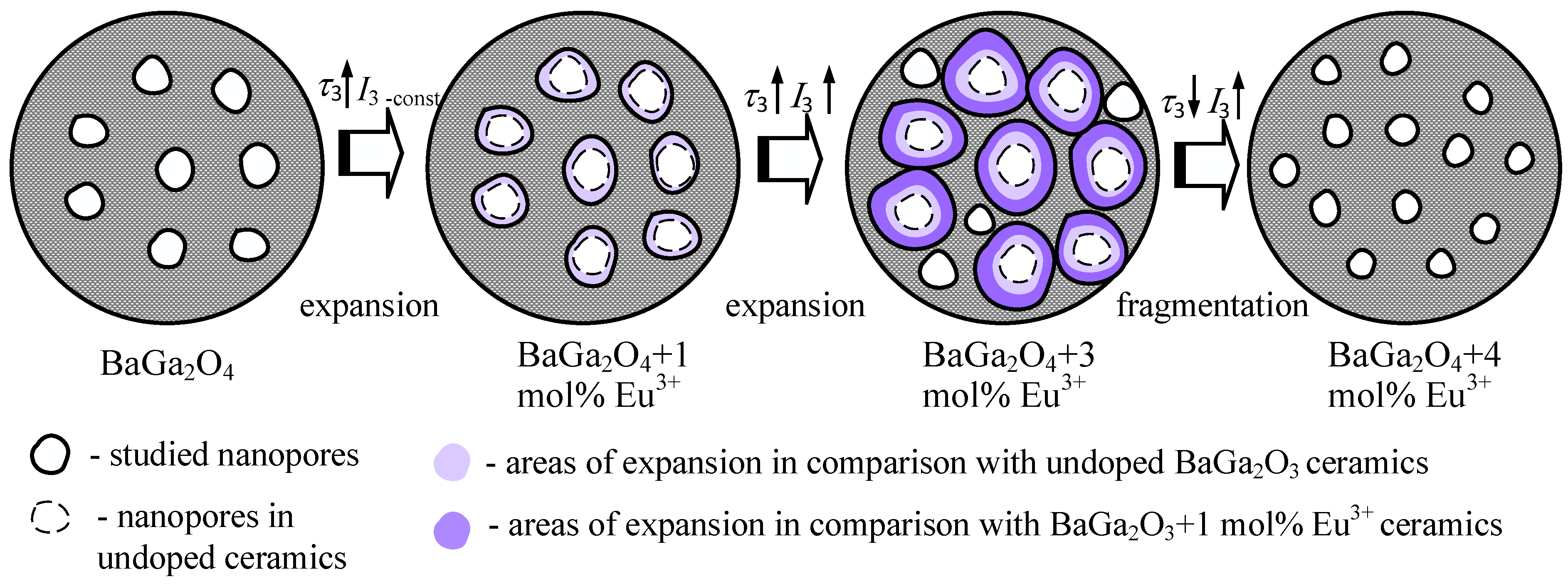
| Sample | Number of Phases | Phases, Lattice Parameters, Weight Fraction |
|---|---|---|
| BaGa2O4 | 3 phases: BaGa2O4, Ba2.84Ga11.32O19.82 and Ga2O3 | BaGa2O4: a = 18.619(1) Å, c = 8.670(1) Å fraction 34.2 wt.% Ba2.84Ga11.32O19.82: a = 15.807(1) Å, b = 11.687(1) Å, c = 5.136(1) Å, β = 107.62(1) Å, fraction 58.5 wt.% Ga2O3: a = 12.213(4) Å, b = 3.042(1) Å, c = 5.810(2) Å, β = 103.65(3) Å, fraction 7.3 wt.% |
| BaGa2O4 + 1 mol.% Eu3+ | 1 phase: (Ba,Eu)Ga2O4 | (Ba,Eu)Ga2O4: a = 18.605(1) Å, c = 8.670(1) Å, fraction 100 wt.% |
| BaGa2O4 + 3 mol.% Eu3+ | 2 phases: BaGa2O4, Eu3GaO6 | BaGa2O4: a = 18.620(1) Å, c = 8.658 (1) Å, fraction 97.3 wt.% Eu3GaO6: a = 9.026 (2) Å, b = 11.344(2) Å, c = 5.496(1) Å, fraction 2.7 wt.% |
| BaGa2O4 + 4 mol.% Eu3+ | 2 phases: BaGa2O4, Eu3GaO6 | BaGa2O4: a = 18.622(1) Å, c = 8.658(1) Å, fraction 96.7 wt.% Eu3GaO6: a = 9.031(2) Å, b = 11.343(2) Å, c = 5.497(1) Å, fraction 3.3 wt.% |
| Sample | Mass, % | Atom, % | ||||||
|---|---|---|---|---|---|---|---|---|
| O | Ga | Ba | Eu | O | Ga | Ba | Eu | |
| BaGa2O4 | 16 | 34 | 30 | - | 59 | 28 | 13 | - |
| BaGa2O4 + 1 mol.% Eu3+ | 17 | 16 | 29 | - | 71 | 15 | 14 | - |
| BaGa2O4 + 3 mol.% Eu3+ | 16 | 30 | 44 | 0.74 | 59 | 25 | 15 | 0.28 |
| BaGa2O4 + 4 mol.% Eu3+ | 16 | 25 | 32 | 4.88 | 61 | 22 | 15 | 1.98 |
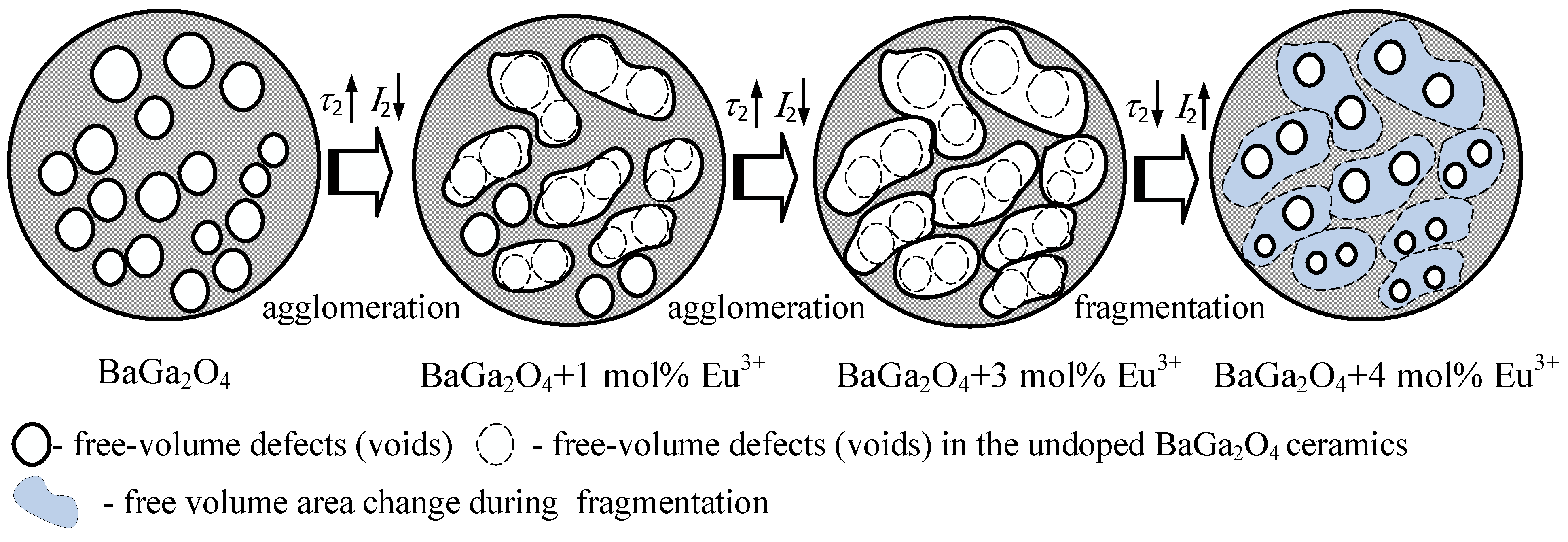
| Sample | τ1 (±0.002), ns | I1 (±0.1), % | τ2 (±0.001), ns | I2 (±0.1), % | τ3 (±0.001), ns | I3 (±0.1), % |
|---|---|---|---|---|---|---|
| BaGa2O4 | 0.200 | 83.3 | 0.424 | 14.9 | 2.196 | 1.8 |
| BaGa2O4 + 1 mol.% Eu3+ | 0.206 | 85.0 | 0.450 | 13.2 | 2.289 | 1.8 |
| BaGa2O4 + 3 mol.% Eu3+ | 0.212 | 89.9 | 0.550 | 7.9 | 2.390 | 2.2 |
| BaGa2O4 + 4 mol.% Eu3+ | 0.201 | 83.3 | 0.411 | 14.4 | 2.157 | 2.4 |
| Sample | τav, ns | τb, ns | κd, ns−1 | τ2 − τb, ns | τ2/τb | R3, nm |
|---|---|---|---|---|---|---|
| BaGa2O4 | 0.234 | 0.218 | 0.40 | 0.21 | 1.95 | 0.306 |
| BaGa2O4 + 1 mol.% Eu3+ | 0.239 | 0.222 | 0.35 | 0.23 | 2.02 | 0.314 |
| BaGa2O4 + 3 mol.% Eu3+ | 0.240 | 0.223 | 0.23 | 0.33 | 2.46 | 0.322 |
| BaGa2O4 + 4 mol.% Eu3+ | 0.232 | 0.218 | 0.37 | 0.19 | 1.89 | 0.302 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Klym, H.; Karbovnyk, I.; Luchechko, A.; Kostiv, Y.; Pankratova, V.; Popov, A.I. Evolution of Free Volumes in Polycrystalline BaGa2O4 Ceramics Doped with Eu3+ Ions. Crystals 2021, 11, 1515. https://doi.org/10.3390/cryst11121515
Klym H, Karbovnyk I, Luchechko A, Kostiv Y, Pankratova V, Popov AI. Evolution of Free Volumes in Polycrystalline BaGa2O4 Ceramics Doped with Eu3+ Ions. Crystals. 2021; 11(12):1515. https://doi.org/10.3390/cryst11121515
Chicago/Turabian StyleKlym, Halyna, Ivan Karbovnyk, Andriy Luchechko, Yuriy Kostiv, Viktorija Pankratova, and Anatoli I. Popov. 2021. "Evolution of Free Volumes in Polycrystalline BaGa2O4 Ceramics Doped with Eu3+ Ions" Crystals 11, no. 12: 1515. https://doi.org/10.3390/cryst11121515
APA StyleKlym, H., Karbovnyk, I., Luchechko, A., Kostiv, Y., Pankratova, V., & Popov, A. I. (2021). Evolution of Free Volumes in Polycrystalline BaGa2O4 Ceramics Doped with Eu3+ Ions. Crystals, 11(12), 1515. https://doi.org/10.3390/cryst11121515







