Melting points were determined using melting-point apparatus (SMP10) in open capillaries and remained uncorrected. Chemicals, reagents and solvents were purchased from Alfa Aesar and Sigma-Merck. The progress of reactions and purity of products were observed using thin-layer chromatography (TLC) on pre-coated plates with silica gel 60 F254 at a thickness of 0.25 mm (Merck). Nuclear magnetic resonance spectra (1H NMR and 13C NMR) were determined in CDCl3 and DMSO-d6 and recorded using Bruker AC 400 MHz spectrometers with TMS as an internal reference standard. δ (ppm) was used for chemical shift description and values of coupling constants were given in Hz. HREI mass spectra were recorded with a Finnigan MAT 95XP instrument. CHNS-microanalysis performed on a Flash EA-1112 instrument.
2.1. General Procedures
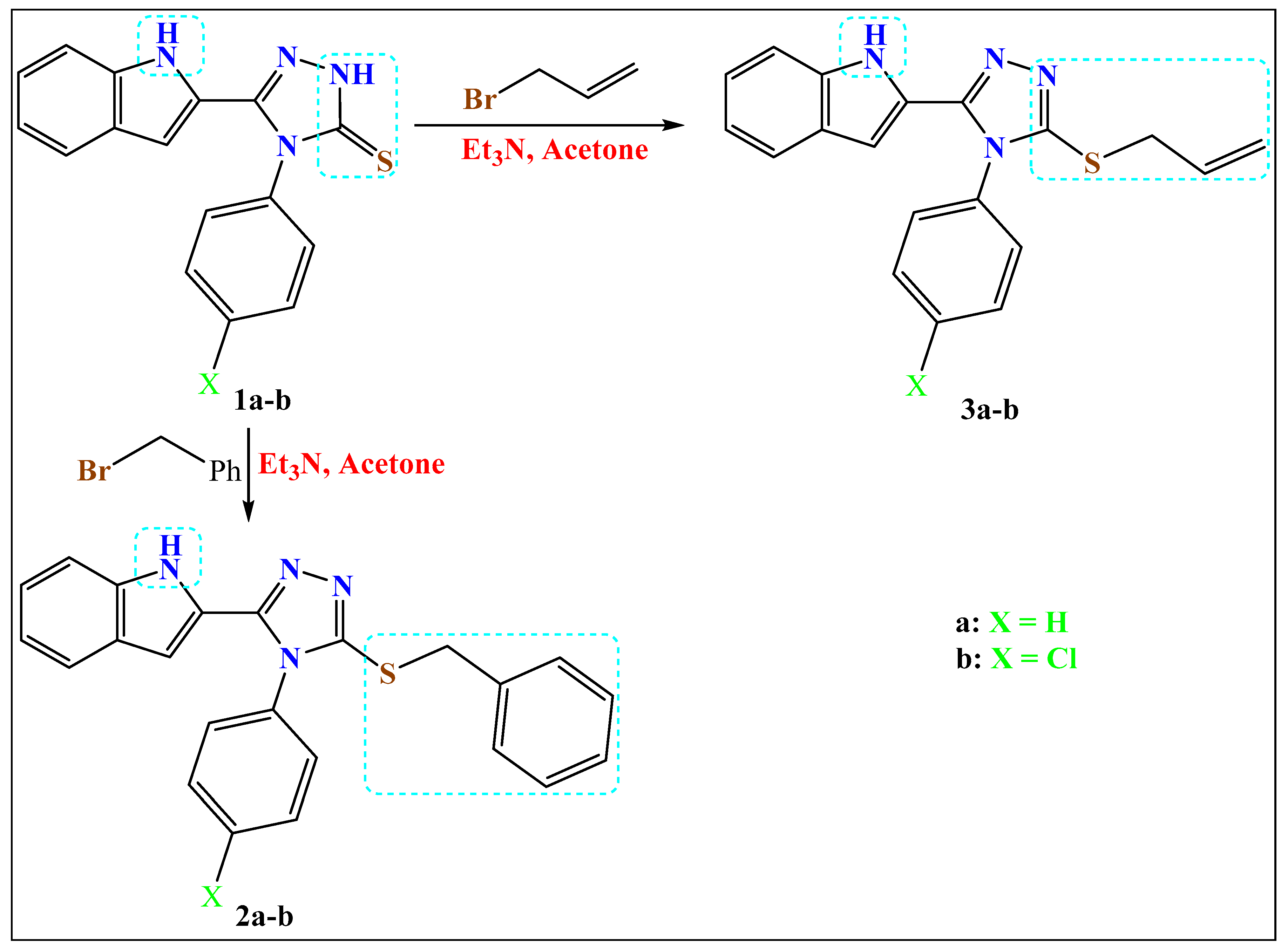
2.1.1. Method a: Synthesis of the S-Alkylated Products
The selected 4-aryl-triazole-thiones 1a–b (1.0 mmol) and Et3N (1.1 mmol) in dry acetone (10 mL) was stirred for one hour, benzyl bromide or allyl bromide (1.1 mmol) was added portion-wise, and stirring was continued overnight. The solvent was removed under vacuum, water was added, and the formed precipitates were collected, dried and recrystallized from ethanol.
Yield: 70%, m.p. 248–249 °C. 1H NMR (400 MHz, DMSO-d6): δ 4.42 (s, 2H), 5.59 (d, 1H, J = 1.3 Hz,), 6.95 (t, 1H, J = 7.4 Hz), 7.15 (t, 1H, J = 7.6 Hz), 7.27–7.45 (m, 9H), 7.61–7.70 (m, 3H), 11.97 (s, 1H); 13C NMR (100 MHz, DMSO-d6) δ 37.21, 101.89, 112.31, 120.20, 121.17, 123.60, 124.33, 127.65, 128.00, 128.39, 128.97, 129.45, 130.62, 131.10, 134.16, 137.00, 137.43, 149.67, 151.76; Elemental Analysis Calc. for [C23H18N4S]: C, 72.23; H, 4.74; N, 14.65; S, 8.38 found C, 72.35; H, 4.79; N, 14.53; S, 8.49.
Yield: 65%, m.p. 242–243 °C. 1H NMR (400 MHz, DMSO-d6): δ 4.42 (s, 2H), 5.70 (s, 1H), 6.97 (t, 1H, J = 7.1 Hz), 7.16 (t, 1H, J = 7.2 Hz), 7.21–7.56 (m, 9H), 7.62–7.81 (m, 2H), 11.97 (s, 1H); 13C NMR (CDCl3, 100 MHz) δ 37.37, 101.04, 112.31, 120.24, 121.33, 123.68, 124.13, 127.67, 128.04, 128.99, 129.46, 130.38, 130.71, 133.04, 135.75, 137.01, 137.39, 149.59, 151.69; Elemental Analysis Calc. for [C23H17ClN4S]: C, 66.26; H, 4.11; Cl, 8.50; N, 13.44; S, 7.69 found C, 66.12; H, 4.28; Cl, 8.38; N, 13.48; S, 7.83.
Yield: 81%, m.p. 218–219 °C [Lit. [
24], 214–216 °C].
1H NMR (400 MHz, CDCl
3):
δ 3.96 (d, H,
J = 6.8 Hz), 5.20 (d, 1H,
Jcis = 9.9 Hz), 5.34 (d, 1H,
Jtrans = 16.9 Hz), 5.78 (s, 1H), 5.95–6.05 (m, 1H), 7.06 (dd, 1H,
J = 7.7,
J = 8.0 Hz), 7.25 (dd, 1H,
J = 7.7,
J = 8.0 Hz), 7.36–7.96 (m, 7H,), 11.05 (br.s, 1H);
13C NMR (CDCl
3, 100 MHz)
δ 35.48, 102.74, 112.49, 119.12, 120.14, 120.99, 123.73, 127.78, 127.97, 130.29, 130.73, 132.61, 133.94, 136.93, 149.89, 152.34; HRMS (EI) calcd for C
19H
16N
4S (M
+): 332.1096. Found: 332.1090.
Yield: 79%, m.p. 229–230 °C. 1H NMR (400 MHz, CDCl3): δ 3.95 (d, 2H, J = 7.0 Hz), 5.20 (d, 1H, Jcis = 10.0 Hz), 5.34 (d, 1H, Jtrans = 16.9 Hz), 5.82 (s, 1H), 5.93–6.03 (m, 1H), 7.08 (t, 1H, J = 7.4 Hz), 7.26 (t, 1H, J = 7.6 Hz), 7.38 (d, 2H, J = 8.5 Hz), 7.47 (d, 1H, J = 8.0 Hz), 7.64 (d, 3H, J = 8.5 Hz), 10.64 (br.s, 1H); 13C NMR (CDCl3, 100 MHz) δ 35.53, 102.65, 112.08, 119.24, 120.32, 121.10, 123.38, 123.94, 127.70, 129.25, 130.58, 132.28, 132.36, 136.64, 136.87, 149.58, 152.27; HRMS (EI) calcd for [C19H15N4SCl]: 366.0706. Found: 366.0730.
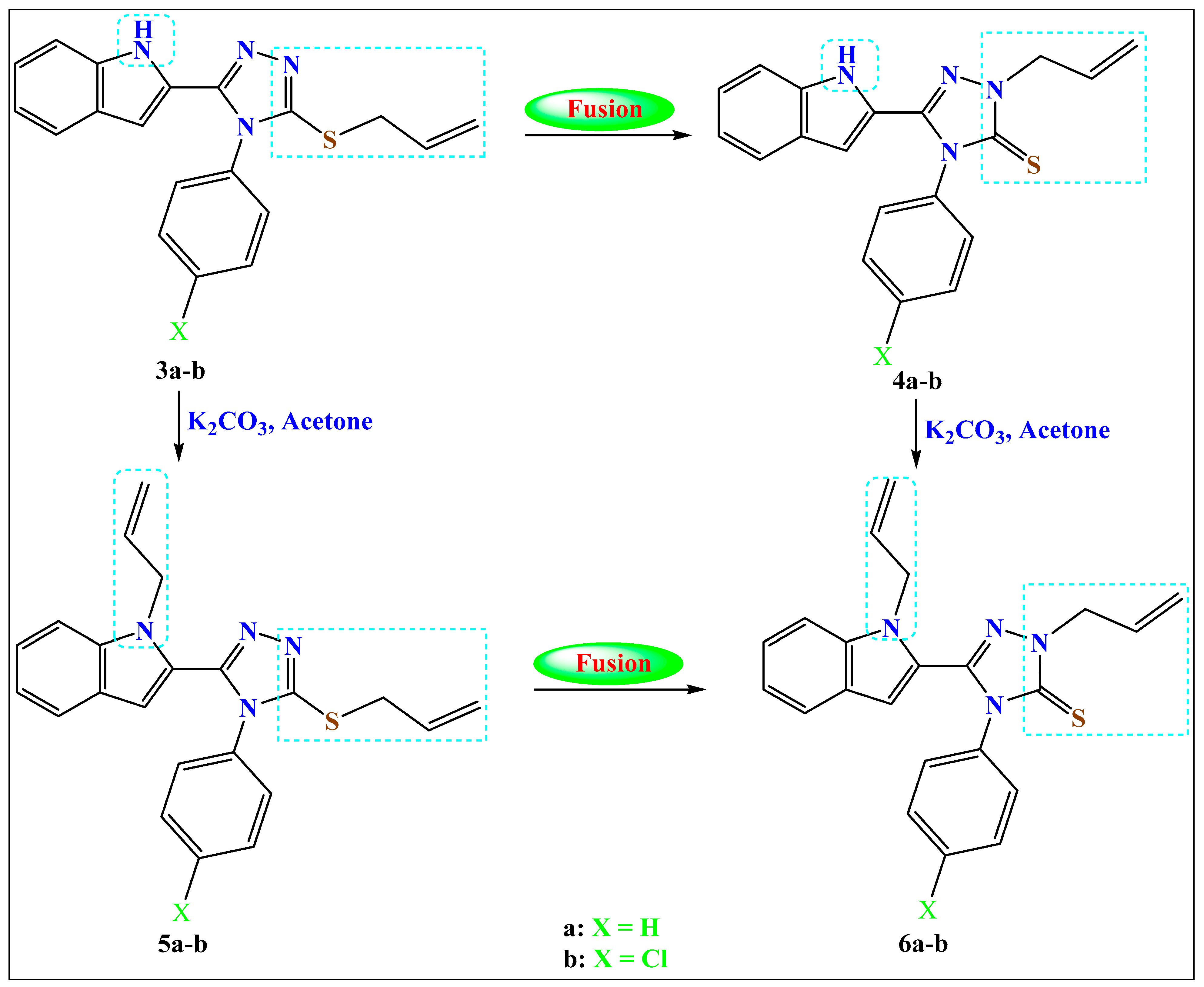
2.1.2. Method b: Fusion of the Allyl-sulfanyl Isomers (Syhthesis of the N-Allylated 4a–b and N-,N-Diallylated Compounds 6a–b)
Separately, the S-allylated compounds from 3a–b (1.0 mmol) and S,N-diallylated compounds 5a–b (1.0 mmol) were fused at temperatures just higher than the respective melting point for few minutes (about 5 min) until all S-allyl starting materials were converted to N-allyl analogues as monitored by TLC. The products were purified by recrystallization from EtOH.
Yield: 76%, m.p. 165–166 °C. 1H NMR (400 MHz, CDCl3): δ 5.00 (d, 2H, J = 6.0 Hz), 5.42 (d, 1H, Jcis = 10.2 Hz), 5.47 (d, 1H, Jtrans = 17.2 Hz), 5.77 (s, 1H), 6.05–6.24 (m, 1H), 7.09 (t, 1H, J = 7.4 Hz), 7.27 (t, 1H, J = 7.4 Hz), 7.32–7.53 (m, 4H), 7.67–7.68 (m, 3H), 8.94 (br.s, 1H); 13C NMR (CDCl3, 100 MHz) δ 51.94, 105.26, 111.23, 119.76, 120.85, 121.73, 122.05, 124.93, 127.54, 128.61, 130.24, 130.70, 134.97, 136.24, 144.04, 168.65; HRMS (EI) calcd for [C19H16N4S]: 332.1096. Found: 332.1091.
Yield: 74%, m.p. 187–188 °C. 1H NMR (400 MHz, CDCl3): δ 4.97 (d, 2H, J = 6.0 Hz), 5.41 (dd, 1H, Jcis = 10.0, Jgem = 0.8 Hz), 5.44 (dd, 1H, Jtrans = 17.2, Jgem = 0.8 Hz), 5.87 (d, 1H, J = 1.2 Hz), 6.10–6.18 (m, 1H), 7.07 (dd, 1H, J = 8.0, J = 7.2 Hz), 7.25 (dd, 1H, J = 7.2, J = 8.0 Hz), 7.33–7.38 (m, 3H), 7.45 (d, 1H, J = 8.0 Hz), 7.59 (d, 2H, J = 8.4 Hz), 8.96 (br.s, 1H); 13C NMR (CDCl3, 100 MHz) δ 51.99, 105.26, 111.25, 119.90, 121.00, 121.82, 125.11, 127.50, 130.04, 130.56, 130.67, 133.38, 136.27, 136.79, 143.80, 168.66; HRMS (EI) calcd for [C19H15N4SCl]: 366.0706. Found: 366.0716.
2.1.3. Method c: Synthesis of S,N-Diallylated Compounds 5a–b and N,N-Diallylated Compounds 6a–b
Separately, (1.0 mmol) of the selected hit compounds 3-allylsulfanyl-4-aryl-triazole-thiones 3a–b/2-(Allyl)-4-phenyl-5-indolyl-3-thioxo-1,2,4-triazoles 4a–b and anhydrous K2CO3 (1.1 mmol) in dry acetone (10 mL) were stirred for one hour, allyl bromide (1.1 mmol) was added portion-wise and all mixtures were stirred overnight (reaction progress was monitored using TLC). Acetone was removed under vacuum, water was added, and the formed precipitates were collected, dried and recrystallized from ethanol.
Yield: 51%, m.p. 116–117 °C [Lit. [
24], 113–114 °C].
1H NMR (400 MHz, CDCl
3):
δ 3.92 (d, 2H,
J = 7.2 Hz), 4.84 (d, 1H,
Jtrans = 17.2 Hz), 5.07 (d, 1H,
Jcis = 10.4 Hz), 5.14 (d, 1H,
Jcis = 10.0 Hz), 5.275–5.32 (m, 3H), 5.95–6.04 (m, 3H), 7.03 (dd, 1H,
J = 8.0,
J = 7.2 Hz), 7.19–7.24 (m, 3H), 7.33 (d, 1H,
J = 8.4 Hz), 7.38 (d, 1H,
J = 8.0 Hz), 7.47–7.52 (m, 3H);
13C NMR (CDCl
3, 100 MHz) δ 35.22, 47.10, 105.51, 110.44, 116.12, 119.10, 120.24, 121.36, 123.47, 124.59, 126.95, 127.65, 130.00, 130.20, 132.58, 134.14, 137.73, 149.05, 152.30; HRMS (EI) calcd for [C
22H
20N
4S]: 372.1409. Found: 372.1422.
Yield: 47%, m.p. 160–161 °C. 1H NMR (400 MHz, CDCl3): δ 3.92 (d, 2H, J = 7.2 Hz), 4.82 (d, 1H, Jtrans = 17.2, Jgem = 0.8 Hz), 5.06 (d, 1H, Jcis = 10.0, Jgem = 0.8 Hz), 5.15 (d, 1H, Jcis = 9.6 Hz), 5.28–5.33 (m, 3H), 5.95–6.04 (m, 3H), 7.06 (dd, 1H, J = 8.0, J = 7.2 Hz), 7.17 (d, 2H, J = 8.8 Hz), 7.23 (dd, 1H, J = 7.2, J 8.0 Hz), 7.34 (d, 1H, J = 8.0 Hz), 7.43 (d, 1H, J = 8.0 Hz), 7.47 (d, 2H, J = 8.4 Hz); 13C NMR (CDCl3, 100 MHz) δ 35.43, 47.04, 105.85, 110.46, 116.22, 119.22, 120.43, 121.45, 123.76, 124.01, 126.93, 128.95, 130.31, 132.31, 132.48, 134.06, 136.45, 137.89, 148.85, 152.25; HRMS (EI) calcd for C22H19N4SCl (M+): 406.1019. Found: 406.1022.
Yield: 73method b, 55method c%, m.p. 145–146 °C. 1H NMR (400 MHz, CDCl3): δ 4.89 (d, 1H, Jtrans = 17.2 Hz), 5.02 (d, 2H, J = 6.0 Hz), 5.08–5.12 (m, 3H), 5.36 (d, 1H, Jcis = 10.4, Jgem = 0.8 Hz), 5.42 (d, 1H, Jtrans = 17.2, Jgem = 0.8 Hz), 5.85–5.94 (m, 1H), 6.01 (s, 1H), 6.05–6.12 (m, 1H), 7.05 (dd, 1H, J = 8.0, J = 7.2 Hz), 7.23–7.32 (m, 4H), 7.40 (d, 1H, J = 0.8 Hz), 7.50–7.60 (m, 3H); 13C NMR (CDCl3, 100 MHz) δ 47.02, 51.92, 107.80, 110.34, 116.66, 119.74, 120.66, 121.78, 122.91, 124.29, 12670, 128.47, 129.85, 130.10, 130.80, 133.58, 135.28, 137.99, 143.63, 168.09; HRMS (EI) calcd for [C22H20N4S]: 372.1409. Found: 372.1390.
Yield: 69method b, 51method c%, m.p. 174–175 °C. 1H NMR (400 MHz, CDCl3): δ 4.88 (d, 1H, Jtrans = 17.1 Hz), 5.00 (d, 2H, J 5.9 Hz), 5.09–5.14 (m, 3H), 5.42 (d, 1H, Jcis = 10.4 Hz), 5.46 (d, 1H, Jtrans = 17.2 Hz), 5.90–5.97 (m, 1H), 6.11–6.18 (m, 2H), 7.13 (t, 1H, J = 7.4 Hz), 7.29–7.51 (m, 7H); 13C NMR (CDCl3, 100 MHz) δ 46.95, 51.92, 107.80, 110.31, 116.69, 119.85, 120.76, 121.81, 122.55, 124.42, 126.61, 129.76, 130.11, 130.59, 133.47, 133.64, 136.10, 137.97, 143.36, 168.03; HRMS (EI) calcd for C22H19N4SCl (M+): 406.1019. Found: 406.1023.