1. Introduction
Thyroid cancer is the most common endocrine cancer and accounts for around 2% of total cancers diagnosed globally, corresponding to 567,233 new cases in 2018 and ranking in 9th place for incidence. The incidence of thyroid cancer has increased in many countries since the early 1980s, probably due to the increase in the diagnosis of papillary thyroid cancer in relation to the improvements in diagnostic methods [
1]. Despite the fact that the overall survival rates of the main forms of thyroid cancer (papillary and follicular forms) are generally good, some forms, such as anaplastic carcinomas and radioactive iodine (RAI) refractory cancers, remain a significant therapeutic challenge [
2,
3]. Hence, study of the tumor microenvironment interactions, and understanding of drug resistance mechanisms, are crucial in the cancer field, particularly for thyroid anaplastic cancers, for which no therapy currently seems to improve the prognosis at one year.
Galectins represent a large family of lectins expressing a highly conserved sequence of approximately 130 amino acids called the “Carbohydrate Recognition Domain” (CRD). Galectins bind to a wide range of glycoproteins and glycolipids, both on the cell surface and in the extracellular matrix. By binding to these glycoconjugates, galectins transmit intracellular signals and mediate cell–cell and cell–extracellular matrix adhesion. Many studies have reported that galectins play crucial roles in cancer because they notably contribute to neoplastic transformation, tumor cell survival, angiogenesis, and tumor metastasis [
4]. They also modulate the immune and inflammatory responses and might play a key role helping tumors to evade immune surveillance [
4]. More specifically, galectin-1 (Gal-1) is reported as overexpressed in tumor cells and tumor-associated endothelial cells [
5]. Gal-1 is broadly implicated in angiogenesis, cancer cell motility and invasion, and immune system escape [
5]. An increase in its expression has been demonstrated in numerous solid cancers, including colorectal, lung, breast, pancreas, liver, and thyroid malignancies [
6]. Importantly, intracellular Gal-1 interacts with the active form of the oncogenic H-Ras (H-Ras-GTP) and, therefore, increases its anchorage to the cell membrane, a crucial step in the malignant transformation [
5]. In thyroid cancer, Gal-1 protein has been studied as a diagnostic marker and appears to be upregulated in papillary thyroid carcinoma and its lymph node metastases [
7]. The role of Gal-1 in thyroid cancer has been previously explored by our team, who reported a higher serum level of Gal-1 in patients with differentiated thyroid cancer than in healthy patients [
8]. We also demonstrated the implication of Gal-1 in thyroid cancer by using a knockdown model showing the inhibition of TPC-1 cell migration, 8505c cell proliferation, and invasion in vitro, in addition to a significant decrease in tumor and metastasis development in vivo [
9]. Overall, these data indicate that Gal-1 could represent an interesting therapeutic target in thyroid cancers.
Although antibodies and peptides may be an interesting option to inhibit Gal-1, small molecules tend to be advantageous regarding bioavailability, immunogenic activation, degradation, and ability to scale up [
10]. OTX008 is a calixarene derivative designed to bind the Gal-1 amphipathic β-sheet conformation [
11]. Nuclear magnetic resonance analysis demonstrated that OTX008 targets Gal-1 at a site distant from the lectin CRD and acts as an allosteric inhibitor of glycan/carbohydrate binding [
12]. This molecule has previously shown anti-proliferative and anti-invasive properties at micromolar concentrations in colon, breast, head and neck, prostate, ovarian, renal, and lung cancer cell lines [
11]. Results in an ovarian xenograft model confirmed the anti-proliferative effects observed in vitro, in addition to reduction in the micro-vessel diameter and the inhibition of VEGFR2 expression within the tumor [
11].
Our study aimed to evaluate the anti-tumoral effects of OTX008 in a panel of six thyroid cancer cell lines, in addition to the use of a xenograft mouse model for anaplastic cancer. Moreover, we addressed the molecular mechanisms of action of OTX008 in our anaplastic model by conducting RPPA and phosphoprotein expression assays.
2. Materials and Methods
2.1. Cell Lines
Human thyroid cancer cell lines 8505c (ATC), BCPAP (PTC), TPC-1 (PTC) were kindly provided by Prof. C. Maenhaut, (IRIBHM, Université Libre de Bruxelles, Brussels, Belgium). The FTC-133 (FTC) lines were kindly gifted by Dr. Köhrke (Institute of Experimental Endocrinology of the Charité, Humbold University, Berlin, Germany) and the TT2609C02 (FTC) and CAL-62 (ATC) cell lines were purchased from DSMZ company (Braunschweig, Germany). All cell lines except CAL-62 were maintained in RPMI-1640 with L-Glutamine (Lonza, Westburg, The Netherlands) supplemented with 10% fetal bovine serum, Brazil (Gibco™, Thermo Fisher, Bruxelles Belgium) and 1% penicillin/streptomycin (Gibco™, Thermo Fisher, Bruxelles, Belgium). The CAL-62 cell line was cultured in Dulbecco’s Modified Eagle Medium (DMEM 4.5 g/L glucose without L-Glutamine, Lonza, Westburg, The Netherlands) supplemented with 10% fetal bovine serum (Gibco™, Thermo Fisher, Bruxelles, Belgium), 2% glutamine (Gibco™, Thermo Fisher, Bruxelles, Belgium) and 1% penicillin/streptomycin (Gibco™, Thermo Fisher, Bruxelles, Belgium). All cell lines were cultured in a standard humidified incubator at 37 °C in 5% CO2 atmosphere and monthly tested for Mycoplasma by PCR test. The culture medium was changed twice a week. For routine maintenance and experimental studies, cells were detached by incubation with accutase solution (Gibco™, Thermo Fisher, Bruxelles Belgium), resuspended, and counted using an electronic cell counter (Scepter Cell Counter Sensors, Millipore, Merck, Overijse, Belgium).
2.2. Cell Proliferation Assay
Cell proliferation assays were performed in 96 well plates (Sarstedt, Berchem, Belgium) in triplicate. The 8505c, TPC-1, FTC133, and BCPAP were plated at 1500 cells/well in 100 µL of culture media, whereas the TT2609C02 were plated at 3000 cells/well and CAL-62 at 2000 cells/well. The next day, cells were supplemented with 100 µL of fresh medium containing OTX008 drug (HY-19756, MedChemExpress, Bio-Connect BV, Te Huissen, The Netherlands) at gradual concentrations or with vehicle for control. Medium with drug was removed after 72 h and cells were washed with DPBS supplemented with Mg++ and Ca++ (Lonza, Westburg, The Netherlands) and then fixed with a solution of 1% glutaraldehyde (Sigma-Aldrich, Merck, Overijse, Belgium) for 15 min. After washing, the cells were colored by a solution of 4% crystal violet (Sigma-Aldrich, Merck, Overijse, Belgium) for 30 min. The cells were washed with tap water and 96 well plates were left for a minimum of 1 h on a bench in open air to dry. Cells were finally permeabilized with a solution of Triton™ X-100 (Sigma-Aldrich, Merck, Overijse, Belgium) for 90 min. Finally, the absorbance was determined at 570 nm with a spectrophotometer (VERSA max-SoftMax Pro, VWR, Leuven, Belgium) and the results were normalized to control (untreated cells).
2.3. Clonogenic Assay
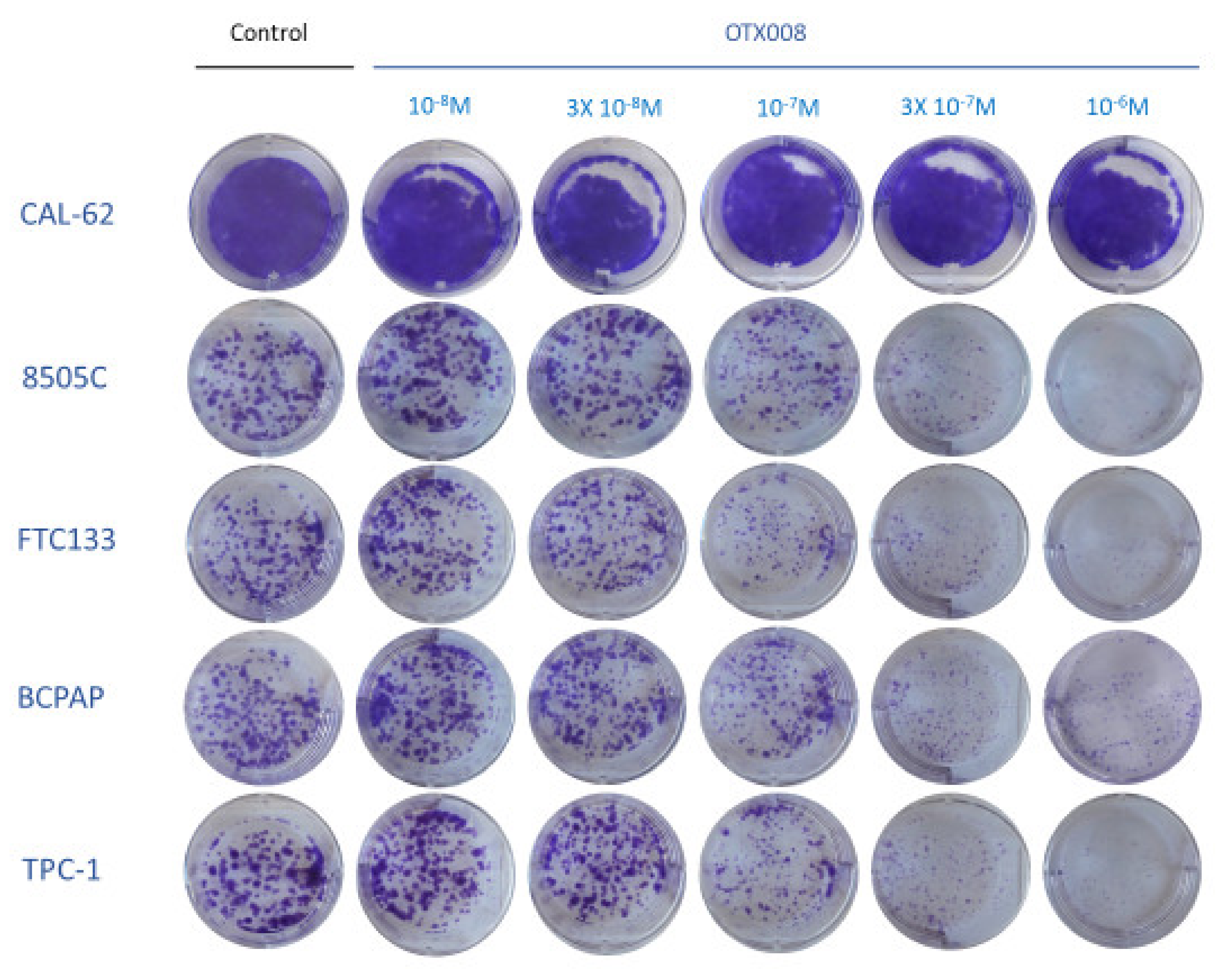
Cells (250 cells/well for each cell line) were seeded in 6 well plates (Nunc™, VWR, Leuven, Belgium) and allowed to adhere for 48 h in culture medium. The cells were then exposed to OTX008 at gradual concentrations or with vehicle for control. The medium was refreshed once during the 10 days of growth culture. At day 10, the medium was removed, and cells were washed with DPBS supplemented with Ca++ and Mg++ (Lonza, Westburg, The Netherlands). The cells were then fixed with 1% glutaraldehyde (Sigma-Aldrich, Overijse, Belgium) for 15 min and then stained with 4% crystal violet solution (Sigma-Aldrich, Overijse, Belgium) for 30 min. The excess of crystal violet was removed with tap water and the colonies were counted.
2.4. Cell Migration and Invasion Assay
The migration and invasion assay protocols were similar and briefly described below. The cell kits were purchased from Merck Company (QCM Chemotaxis Cell Migration Assay, 24 well, 8 µm and QCM ECMatrix Cell Invasion Assay, 24 well, 8 µm, Merck, Overijse, Belgium), and presented an 8 µm pore size polycarbonate membrane, alone (for migration assessment) or over which a thin layer of ECMatrix™ is dried (for invasion evaluation). Firstly, the chambers were hydrated with medium without FBS during minimum 30 min in the cell incubator. Medium supplemented with FBS was added in each well. Cells were suspended in medium without FBS at line-dependent densities. The cell suspension was placed in the center of the chamber. An equal volume of solution of medium containing OTX008 was added in cell suspension depending on the tested conditions. Plates were then incubated for 48 h at 37 °C. At the end of treatment, the cells inside the chambers were removed, and excess cells were eliminated with a cotton swab. The external bottom part of the chamber, where cells migrated or invaded, was fixed with a solution of 1% glutaraldehyde for 15 min. A solution of 4% crystal violet was added in wells, whereas chambers were deposited on it and incubated for 2 min. Finally, chambers were washed with distilled water. Cell invasion was observed under microscope. Quantification was calculated after 1 h solubilization in Triton™ X-100 solution and the relative absorbance was determined at 570 nm.
2.5. Protein Microarray
Protein microarray was performed by RPPA analysis at the Functional Proteomics Core Facility at The University of Texas MD Anderson Cancer Center. The cell extracts were sent to the University of Texas. Cells were plated in T75 and detached by incubation with accutase solution (Gibco™, Thermo Fisher, Bruxelles, Belgium). The second step was counting using an electronic cell counter (Scepter Cell Counter Sensors, Millipore, Merck, Overijse Belgium), followed by centrifugation at 4 °C, 1200 rpm for 5 min. Successive washes were performed to finally transfer 6 × 106 cells in an Eppendorf. Cells were centrifuged and stored at −80 °C.
2.6. Human Phospho-Kinase Array
A Human Phospho-Kinase Array kit (R&D Systems, Minneapolis, MN, USA) was used to evaluate variations of 37 kinase phosphorylation sites after OTX treatment. First, 8505c and CAL-62 cells were plated in dishes for 72 h. Once the cells were at confluence in dishes, they were treated with 30 µM OTX008 for 30 min. Media was then eliminated, and cells were washed with DPBS with Ca++ et Mg++ on ice and finally scratched in DPBS. Cells were centrifuged at 1200 rpm for 5 min at 4 °C and the pellets were solubilized in lysis buffer supplemented with phosphatase provided in the Human Phospho Array kit. The lysis buffer was incubated for 30 min at 4 °C. Cell suspension was finally centrifuged at 14,000 rpm for 5 min at 4 °C. The supernatant was transferred and stored at −80 °C. Quantitation of sample protein concentration was performed using BCA Protein Assay (Pierce™ BCA Protein Assay Kit, Thermo Scientific™, Waltham, MA, USA) using bovine serum albumin as standard. The nitrocellulose membranes from the kit were blocked with provided solution and the cell lysates were incubated with the membranes overnight at 4 °C on a rocking platform shaker. After 3 washings, the cocktails of antibodies were incubated with the membranes for 2 h at room temperature on a rocking platform. The next steps comprised 3 washings followed by the incubation with the streptavidin-HRP for 30 min at room temperature on a rocking platform. After a final step of washing, the chemi-reagent mix was added, and the spot revealed with a LAS-3000 CCD camera (Fujifilm, Tokyo, Japan), using software specifically designed for image acquisition (Image Reader, Raytest®, Straubenhardt, Germany). Immunoreactive spot intensities were quantified using the software AIDA® Image Analyser 3.45 (Raytest®, Straubenhardt, Germany).
2.7. Immunofluorescence Assay
Cells were plated at day 0 in 12 well plates with 1 mL of medium per well. Each well contained a coverslip. Cell densities varied according to conditions (no treatment: 3000 cells/well, 10 µM OTX008: 10,000 cells/well, 30 µM OTX008: 30 000 cells/well). At day 1, drug or vehicle was added to the medium for 72 h. The steps of fixation and antibody incubation were performed at day 4. Cells were washed with PBS and then fixed for 15 min in paraformaldehyde solution. Cells were washed again with a PBS solution containing 0.1% Triton™ X-100 for 3 successive washes of 5 min. A blocking solution of PBS-BSA 5% was added for 1 h at room temperature. Cells were again washed with PBS solution. The primary antibody (Galectin 1 Monoclonal Antibody (6C8.4-1) Thermo Fisher Scientific, Waltham, MA, USA) was added at a dilution of 1:250 in PBS containing 0.5% BSA for 3 h at room temperature (negative control lacked antibodies). Cells were washed 3 times for 5 min with 0.1% Triton™ X-100 in PBS. The secondary antibody (Alexa fluor) 1:100 in PBS containing 0.5% BSA was then added for 30 min at room temperature. Cells were finally washed 3 times with 0.1% Triton™ X-100 in PBS. A Few drops of DAPI solution (ProLong™ Gold Antifade Mountant with DAPI, Thermo Fisher Scientific, Waltham, MA, USA) were added to a glass side, which was then covered by a coverslip. Images were then acquired with NIS element viewer 5.21.
2.8. pERK Detection by HTFR Kit
The 8505c cells were plated in 96 well plates 24 h before the test. The cells were then treated with 30 µM OTX008 treatment for 10 and 30 min. A lysis buffer provided in the kit was added to the cells and incubated during 45 min on a rocking platform. The lysates were then transferred to the provided plate and the antibody added. An incubation of 4 h at room temperature was performed. The plate was finally read at 665 and 620 nm with the Spectra Max5.
2.9. Xenograft ATC Models
Four to nine week old male athymic nude mice were purchased from Charles River (490CRATHHO, Male CR ATH HO MOUSE 28–34 days, Charles River, Sait Germain Nuelles, France), which were fed ad libitum and kept in optimal conditions in a 12 h light/dark cycle. Upon arrival, animals were isolated in the animal facility for a 1 week quarantine before starting experiments. The 8505c cells (1 × 106) were resuspended in 300 μL HBSS (no calcium, no magnesium, no phenol red, Gibco™, Thermo Fisher, Bruxelles, Belgium) and injected subcutaneously on the right flank of the immunodeficient mice. A total of 9 mice were inoculated with cancer cells (3 in the control group and 6 in the drug-treated group). The treatment was administered to mice by daily intraperitoneal (IP) injections of either vehicle or OTX008 at 5 mg/kg/day for 5 days/week, 2 days off, for 3 weeks. The mice weight was measured every two days. The tumor development was monitored 3 times/week and tumor volumes were evaluated by measurements of tumor length and width in millimeters using calipers. At the end of treatment, the animals were sacrificed by lethal IP injection of Dolethal 200 mg/mL (pentobarbital sodique, Vetoquinol, Aartselaar, Belgium). Tumor tissues and lungs were collected. Tumor volumes were calculated according to the formula (length × width × thickness)/2. Tissues were fixed in 4% formaldehyde and paraffined for HE staining and histological analyses. All animal experiments were performed according to the institutional guidelines and approved by the ethics committee of the University of Mons (Mons, Belgium) (SA-04-01).
2.10. Statistical Analysis
All statistical analyses were performed using IBM SPSS Statistics 23 (IBM, Ehningen, Germany). In vitro data are expressed as means ± standard deviation, and comparison of mean values was conducted with one way ANOVA test and Tukey (multiple variables compared each other) or Dunnett (multiple variables compared to control) post hoc tests, or with the t-test (comparison of two variables). Animal data are presented in box plots (median and percentiles 5/25/75/95) and analyzed by the Mann Whitney test. Significance was indicated as * p < 0.05; ** p < 0.01; *** p < 0.001.
4. Discussion
The aim of our study was to deepen our knowledge on the importance of Gal-1 in the biology of thyroid cancer. Due to the effect of the OTX008 molecule, we observed significant inhibition of cell proliferation and invasion in vitro, and of tumor and metastasis growth in a xenografted mouse model. Experiments were also conducted to further understand the molecular mechanisms involved in the anti-cancer properties of OTX008 in thyroid cancer. OTX008, which is a small molecule acting as an allosteric inhibitor of Gal-1, appears to present therapeutic potential in cancer. OTX008 is chemically more stable and resistant to hydrolysis in comparison to other Gal-1 inhibitors such as Anginex. Its low molecular weight (937 Da), associated with the fact that it is neither a protein or a saccharide, but a phenyl-based molecule, greatly increases its bioavailability [
6].
First, our in vitro study showed that OTX008 exhibits direct anti-tumor activity in vitro due to an anti-proliferative effect on five different thyroid cancer cell lines (8505c, TPC1, BCPAP, FTC133, TT2609C02), with IC50 values ranging from 0.2 to 1.1 µM. By contrast, the anaplastic thyroid cancer cell CAL62 showed a limited response to the drug, with an IC50 value > 30 µM. The anti-proliferative properties of OTX008 were previously demonstrated in many cancer cell lines, including colon (HT29, COLO205-S, HCC2998, HCT116, COLO205-R), ovarian (A2780-1A9, OVCAR 3, IGROV1, SKOV3), prostate (PC3, DU 145), head and neck (SQ20B, SCC91, HEP2), breast (MCFS7, SK BR3, ZR-75-1, MCF7-shWISP), and renal cancer lines (CAKI 1), with IC50 values ranging from 3 to 500 µM, demonstrating the variable response which can be obtained with such a Gal-1 inhibitor [
11]. Variable results were also obtained for melanoma (WM983a, A375 M) and glioblastoma (U87MG), with IC50 values between 35 µM to 192 µM. This variability observed between all cell lines underlines the possibility of intrinsic molecular mechanisms for OTX008 action, or specific targets for OTX008 depending on cell lines, driving the response to the drug.
Then, an in vivo model was developed based on our in vitro results. This model reported a significant reduction in tumor growth, to 66% of that of the control group, and inhibition of lung metastasis development in mice treated with 5 mg/kg OTX008 for 3 weeks. In this context, Xerri’s study reported that OTX008 induced a significant reduction in tumor growth in nude mice bearing A2780-1A9 ovarian cancer xenografts after 2 weeks of treatment [
11]. OTX008 has also been shown to be efficient in two other models of carcinoma using MA148 human ovarian and B16 mouse melanoma by subcutaneous delivery of the drug with implanted osmotic mini pumps. In the MA148 model, ovarian tumor growth was reduced to 58% at 2.4 mg/kg [
13]. In B16 mouse melanoma, the inhibition of tumor growth reached 77% at 10 mg/kg in comparison to the untreated group control [
13]. Of note, at these concentrations, the authors did not observe toxicity, as assessed by animal behavior, body weight change, or hematocrit or creatinine levels (both determined by blood drawing on the last day of treatment). Moreover, after autopsy, no readily apparent abnormalities in internal organs were observed [
13]. In our study, the in vivo evaluation of OTX008 in an anaplastic thyroid carcinoma xenograft model also resulted in the absence of notable side-effects.
The aspects of invasiveness and migration studied in our work showed that OTX008 inhibits the migrative property in four thyroid cell lines (8505c, FTC133, TPC1, BCPAP), with more than 50% of inhibition observed with 3 µM treatment after 48 h. In Xerri’s study, OTX008 demonstrated its capacity to inhibit the invasion of SQ20B cells at micromolar concentrations. In addition, Zuchetti and colleagues demonstrated an inhibition, almost to the baseline level, of the endothelial cell motility and invasion after OTX008 treatment [
14]. Overall, these results confirm the anti-invasive and anti-migrative properties of OTX008 in different cell types.
A hypothesis to explain the sensitivity/resistance of thyroid cancer cells to OTX008 is the activation of cell-dependent molecular mechanisms. Based on our in vitro data, we explored the impact of OTX008 on various signaling pathways, Gal-1 expression, and subcellular localization, with regard to RAS mutation. Many studies have reported that galectins drive an intracellular signaling pathway via interaction with the small GTPase RAS [
15,
16,
17,
18]. In our study, the analysis of the RAS mutation in our cell lines revealed that only the CAL-62 line harbors a mutation in KRAS, with probably fewer interactions with Gal-1, supporting the lack of efficacy of OTX008 in this cell line.
In addition, given our results from phospho-kinase array and the literature, a cascade implicating Gal-1, integrin β, PYK2, eNOS, and HSP27 is proposed to explain the opposite response to OTX008 in CAL-62 and 8505c. Indeed, we evaluated the phosphorylation levels of many proteins and noticed some important disparities between the two lines, such as an inverse expression of HSP27 S78/S82 and eNOS S1177. Highly expressed in CAL-62, HSP27 is known as a marker of resistance and is related to the overall survival rate, notably in many cancers, such as breast cancer, prostate cancer, gastric cancer, lung cancer or liver cancer [
19,
20,
21,
22]. Therefore, our results appear to confirm the role of HSP27 in the resistance of the anaplastic thyroid cancer to OTX008. In contrast, our study demonstrated that the level of S1177 phosphorylation of eNOS was significantly lower in CAL-62 than in 8505c lines, and the final major disparity related to the Y402 phosphorylation of PYK2 (proline-rich tyrosine kinase 2), with a higher expression in CAL-62 than in 8505c cells. Overall, CAL-62 resistance is characterized by high phosphorylation of HSP27 and PYK2, and weak phosphorylation of eNOS.
Of note, PYK2 is part of the focal adhesion kinase (FAK) non-receptor tyrosine kinase family and can be activated by multiple growth factors (GFs), hormones, neuropeptides, cytokines, and chemokines [
23]. PYK2 is implicated in many signal transduction cascades and plays critical roles in controlling cell adhesion, proliferation, migration, and invasion. Most importantly, recent studies suggest a network between PYK2, eNOS, and HSP27. In breast cancer, the strong phosphorylation levels of HSP27 and PYK2 have been associated with a form of resistance to doxorubicin [
24]. In addition, the inhibition of eNOS mediated by PYK2 has been explored in cardiomyocyte survival and cardio-protection, showing a possible cascade between these molecules [
25].
As previously mentioned, OTX008 acts as an inhibitor of Gal-1, which is broadly implicated in cell–cell and cell–extracellular matrix interactions, and its implication in tumor invasion has been broadly described in the literature [
4,
5,
6]. The integrin β1 seems to exert an important connection between Gal-1 and the FAK signaling pathway. By a knockdown of Gal-1 in breast cancer, the Gal-1/integrin β1 interactions have been highlighted because the depletion of Gal-1 negatively impacts the signaling pathway downstream of FAK/SRC [
26]. Another study evaluated the impact of Gal-1 overexpression and demonstrated the activation of the FAK/PI3K/AKT/mTOR pathway, which is correlated with upper urinary urothelial carcinoma progression and survival [
27]. The characterization of integrin receptor molecules in different thyroid cancer cell lines confirmed high levels of integrin β1 in anaplastic thyroid cancers [
28]. Integrin linked kinase has also been studied, showing an elevated expression in anaplastic thyroid cancer [
28,
29,
30]. All of these arguments highlighting the relationship between Gal-1, integrin (more specifically, integrin β1), and the FAK pathway support our hypothesis of a relationship between Gal-1 and PYK2, member of the FAK family. To confirm the possible activation of PYK2 through an interaction between Gal-1 and integrin β1, it would be interesting to assess the effect of PYK2 inhibitors on the phosphorylation levels of eNOS and HSP27.