1. Introduction
Urothelial bladder cancer (BCa) accounts for approximately 3% of global cancer diagnoses and 2.1% of cancer-related deaths worldwide [
1]. Approximately 75% of BCa cases are non muscle-invasive BCa, and 25% of BCa cases are muscle-invasive BCa [
2]. Currently, muscle-invasive BCa is treated by radical cystectomy with bilateral lymphadenectomy in combination with platinum-based perioperative chemotherapy in patients with extravesical tumors and/or lymph node metastasis [
3,
4].
In addition to tumor cells (TCs), the tumor immune microenvironment is associated with prognosis [
5,
6,
7,
8,
9,
10,
11]. In particular, the presence of tumor infiltrating immune cells (ICs), such as leukocytes, including lymphocytes identified by their protein or gene expression profile, is associated with improved five-year overall survival (OS) or disease-specific survival (DSS) [
8,
10,
11].
We were interested in potential biomarkers for bladder cancer that are expressed in TCs as well as in IC cells to gain more insight into the interaction of tumor cells and the tumor microenvironment. A well-recognized marker expressed in TCs and ICs in bladder cancer is PD-L1, which has already been utilized for therapy stratification of checkpoint inhibitor therapies [
12,
13,
14]. Recently, we found that the expression of the immune cell marker chemokine CC motif ligand 2 (CCL2) was differentially associated with prognosis, depending on whether it was expressed in tumor cells (poor prognosis) or in immune cells (good prognosis) of bladder cancer patients [
15]. Another candidate could be progranulin (GP88; synonymous: granulin–epithelin precursor; proepithelin; PC cell-derived growth factor; acrogranin) as it is expressed in bladder cancer cells [
16] and is expected to be expressed in immune cells. GP88 protein expression has been shown in normal gastric mucosa in both epithelial and infiltrating immune cells (granulocytes, lymphocytes) [
17]. One of the first articles about granulins described their presence in leukocytes [
18], and another article described GP88 as a macrophage-derived factor [
19]. However, none of the studies reported its presence, particularly in immune cells, in the context of tumors.
GP88, named after its molecular weight at ~88 kDa on Western immunoblots due to glycosylation, is physiologically a growth factor that regulates cell proliferation and wound repair, but pathologically it is also a component of the tumorigenesis machinery for different cancers [
20,
21,
22,
23]. Increased GP88 expression has been reported in breast cancer, brain tumors, non-small cell lung cancer, ovarian cancer, renal carcinoma, hepatocellular carcinoma, prostate cancer, colorectal cancer and hematological cancers [
22,
24,
25,
26,
27,
28,
29,
30]. An association between increased GP88 protein expression and a poor prognosis has been reported for breast cancer, ovarian cancer, non-small cell lung carcinoma, lymphomas, esophageal cancer, prostate cancer, and colorectal cancer [
25,
26,
28,
29,
30,
31,
32]. GP88 expression is also correlated with resistance to anti-estrogen therapy in breast cancer patients [
25,
33] and platinum-based chemotherapy in non-small cell lung cancer patients [
34].
In bladder cancer, in vitro studies demonstrated that GP88 stimulated the migration and invasion of bladder cancer cells [
35]. Consistent with this finding, downregulation of GP88 in bladder cancer cells reduced their ability to proliferate in the absence of serum and inhibited migration, invasion and wound healing [
36]. GP88-depleted urothelial cancer cells exhibited markedly reduced in vivo tumor growth in orthotopic and subcutaneous xenograft tumor models [
16]. Progranulin depletion also sensitized urothelial cancer cells to cisplatin treatment [
16,
37]. Increased GP88 levels were detected in the urine of bladder cancer patients compared to healthy controls [
36,
38,
39]. GP88 RNA expression was significantly upregulated in invasive bladder cancer tissues compared with normal urothelium, and elevated RNA expression was associated with a shorter OS [
36]. Two immunohistochemical studies characterized GP88 protein expression in BCa [
16,
36]. Immunohistochemical staining in a small number of samples revealed that GP88 protein expression was significantly increased in invasive bladder tumors compared with normal bladder tissues [
36]. Another study on a tissue microarray with 69 patients with BCa confirmed that GP88 protein expression was increased in tumor tissue compared with non-tumor bladder tissue [
16]. In addition, GP88 was also overexpressed in metastatic bladder tissues, indicating that GP88 expression levels might be associated with bladder cancer metastasis. However, upregulation of GP88 was not significantly different between T1 and T2-4 urothelial carcinoma tissues [
16]. Altogether, a statistical analysis of the association of GP88 protein expression with prognosis has not yet been reported in BCa.
The aim of this study was to examine GP88 protein expression in TCs and in ICs of muscle-invasive BCa, and to analyze the association of GP88 expression with clinicopathological parameters and prognosis.
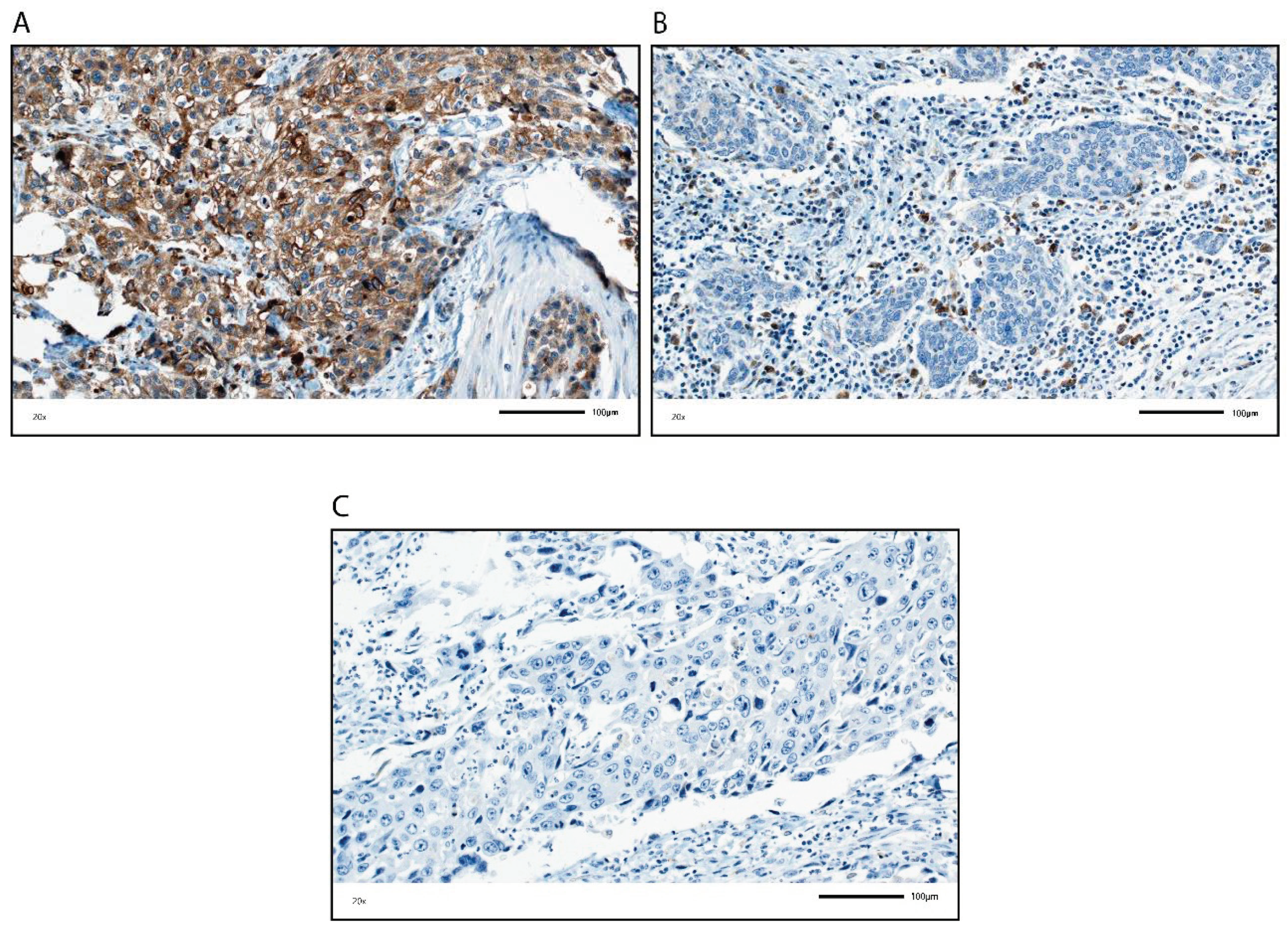
4. Discussion
We analyzed GP88 protein expression in TCs and in ICs in tumors from BCa patients by IHC and assessed its association with clinicopathological and survival data. Remarkably, GP88 staining was inversely correlated in the TCs and in the ICs. GP88 staining in TCs was positively correlated with tumor stage, lymph node stage, adjuvant chemotherapy and molecular subtype, and negatively correlated with immune cells and their markers as well as with OS, DSS and RFS.
In contrast, GP88 staining of ICs was positively correlated with the presence of immune cells and their markers, and with prognosis (OS, DSS, RFS). In addition, GP88-positive IC staining was negatively correlated with tumor grade, lymph node stage, adjuvant chemotherapy and molecular subtype. The correlation of GP88 positivity in ICs to different immune cell markers suggests that various immune cells may express GP88, but this issue awaits further clarification in future studies with staining for multiple proteins.
Previous reports of GP88 expression have exclusively concentrated on investigating GP88 expression in the tumor cells. GP88 expression has been shown to enhance proliferation and promote tumor growth in several cancer cell lines and cancers, e.g., breast, gastrointestinal, hepatic, lung, and genitourinary cancers [
22,
23,
27]. In bladder cancer, in addition to extensive in vitro and in vivo model studies [
16,
35,
36], tumor cells showed increased GP88 expression at the RNA and protein levels compared with normal tissue [
16,
36]. Furthermore, GP88 was overexpressed in metastatic bladder tissues, but Buraschi et al. could not identify significant differential upregulation of GP88 between T1 and T2-4 urothelial carcinoma tissues [
16]. In this present study, we exclusively focused on muscle-invasive BCa with tumor stages ranging from pT2 to pT4. We found a correlation between increased GP88 protein expression in TCs and higher tumor stage.
We showed for the first time that GP88 expression in TCs is an independent negative predictor for DSS and RFS in muscle-invasive BCa patients. After patient stratification based on several clinicopathological and molecular parameters, multivariate analysis revealed that GP88 expression in TCs was an independent prognostic factor, i.e., for OS, DSS and RFS in the pT3+4 group, N0 group, CT−group and luminal subgroup. Thus, a more detailed characterization can be made of, for example, the pT3+4 group, which is generally considered to have a poor prognosis; specifically, patients with a poor prognosis (TC GP88 positive) and patients with a better prognosis (TC GP88 negative) can be distinguished. Of special interest is that GP88 expression has been correlated with resistance to platinum-based chemotherapy in non-small cell lung cancer patients, and progranulin depletion sensitizes urothelial cancer cells to cisplatin treatment [
16,
34,
37]. However, until now, pretreatment gene signatures for the luminal muscle-invasive BCa subtypes that might predict chemosensitivity were not available [
42]. GP88 positivity in TCs predicts poor outcomes in the absence of chemotherapy, but not for patients treated with chemotherapy, which could indicate a chemotherapy-induced cancellation of the adverse effect of GP88 expression on TCs.
In addition, we report for the first time that GP88 expression in ICs is an independent positive predictor for OS, DSS and RFS in muscle-invasive bladder cancer patients. After stratification of the patients for several clinicopathological and molecular parameters, we found in multivariate analysis that negative GP88 expression in ICs was an independent negative prognostic factor (OS, DSS and RFS) in the pT3+4 group, in the CT− and CT+, as well as basal and luminal subgroups. However, an association was also noted between negative GP88 expression in ICs and poor RFS in the pT2 group.
Why are GP88-positive ICs associated with better prognosis? Tumors can be considered as wounds that do not heal [
45]. GP88 is a growth factor that has been described in wound-healing [
46]. In a mouse wound healing model, He et al. observed that inflammatory cells infiltrating the wound, including neutrophils and macrophages, expressed progranulin mRNA from day one [
46]. In addition, when GP88 was applied to a cutaneous wound, it increased the accumulation of immune cells (neutrophils, macrophages), blood vessels and fibroblasts in the wound.
It is known that GP88 can be produced by immune cells such as leukocytes [
17,
18,
19,
47]. The presence of different immune cells (TILs) has been associated with better prognosis and therapy response in muscle invasive bladder cancer [
10,
48,
49]. For tumor cells, GP88 might function as an autocrine growth factor [
25], while in the context of immune cells, its anti-inflammatory function might be predominant [
50]. Moreover, there are several points at which GP88 can be active in the immune system with an anti-tumorigenic activity: (i) GP88 is involved in the activation of innate immunity by serving as a soluble cofactor for TLR9 e.g., in macrophages [
51]. (ii) GP88 promotes the Th2 (T-helper 2 cells) response [
52], and can induce expression of Th2-like cytokines, IL-4 and IL-5 [
53]. Components of the Th2-mediated immune response, such as IL-4 and eosinophils, can decrease tumor growth and initiate anti-tumor activity [
54]. (iii) Binding of GP88 to TNFR2 inhibits binding of TNFα [
55]. TNFα is known for its immunosuppressive activity by intrinsic negative effect on conventional T cells activation and by boosting suppressive cells, as myeloid-derived suppressor cells (MDSC) or regulatory T cells (Tregs) [
56].
Altogether, our findings suggest that GP88 expression could mark a population of ICs that may be anti-tumorigenic in muscle-invasive BCa patients.
Finally, we focused on the combination of GP88 staining in TCs and ICs and their association with prognosis to define patient groups with worse and better prognoses. GP88 staining in TCs and ICs was negatively correlated. As expected from the single GP88 staining analyses, the group with TC-negative and IC-positive GP88 staining showed the best prognosis. In contrast, the TC-positive/IC-negative group presented the worst prognosis, whereas the other two groups were prognostically in between for OS, DSS and RFS. The combination of GP88 staining of TCs and ICs showed an additive effect for risk evaluation compared to the single analyses in either TCs or ICs. Remarkably, for the TC-positive/IC-negative group, a greater than 2.5-fold risk for death and a greater than 4-fold increased risk for either disease-specific death or recurrence could be calculated. This finding implies that GP88 evaluated in both TCs and ICs could be a relevant prognostic marker for muscle-invasive BCa cancer patients. However, targeting GP88 in tumor therapy by siRNA or GP88 monoclonal antibodies, as recently shown in a triple-negative breast cancer tumor xenograft model [
57], might be restricted to TCs and should possibly spare ICs in BCa.
Our study has some limitations. This was a retrospective study. After stratification, the patient sub-groups were rather small. Therefore, our results must be evaluated in a larger prospective study. However, altogether, the number of patients in this study allowed for the reasonable multivariate analysis of the association of one parameter, namely GP88 staining, with prognosis in muscle-invasive BCa patients. The patients were not treated with checkpoint inhibitor immunotherapy, given the retrospective nature of the study. However, GP88 expression (as PD-L1 expression) in TCs was associated with poor prognosis, whereas GP88 expression (as PD-L1 protein expression) in ICs was associated with a better prognosis. GP88 expression in TCs can be associated with chemotherapy resistance in different cancers/cancer cells. Thus, GP88 expression may have some predictive potential for future chemo- and immunotherapies, in addition to its prognostic value.
However, further characterization of the type of ICs expressing GP88 is necessary.