Enhanced Cognition and Neurogenesis in miR-146b Deficient Mice
Abstract
1. Introduction
2. Materials and Methods
2.1. Animals and Experimental Design
- 146b locus 5′ forward primer- 5′ CTCACACTCTTGTTCTTACCCAGTTCTT 3′;
- 146b locus 3′ reverse primer- 5′ CAAACAAACAAACAAAAGGTTCAGCTAAG 3′;
- 146b locus internal reverse primer-5′ACACACAGGGCATATGAGATCAGTTGGTT 3′ and same generation littermates were used in experiments. Two–three months old male mice were used for all experiments, which were undertaken in agreement with the guidelines established in the principles of laboratory animal care (Directive 2010/63/EU). All the mice were group-housed with a 12-h light/dark cycle with food and water available ad libitum. The Animal Experimentation Committee at the Estonian Ministry of Agriculture (no. 183, 2021) approved the experimental protocol. The sequence of the experiments performed is summarized in Table 1. The age of animals at the start of the experiments was approximately 2 months old. The age of animals at the time of sacrifice was approximately 3 months old.
2.2. Novel Object Recognition Test (NORT)
2.3. Contextual Fear Conditioning (CFC) and Tone Fear Recall
2.4. Open Field Test (OFT)
2.5. Elevated Plus Maze (EPM)
2.6. Tail Suspension Test (TST)
2.7. Social Dominance Test (SDT)
2.8. Flow Cytometry
2.9. Brain Volume Assessment and Immunohistochemistry
2.10. Assessment of Neurogenesis in the Adult Mouse Dentate Gyrus
2.11. Isolation of Brain Cells
2.12. RNA Extraction and RT-qPCR
2.13. Statistical Analysis
3. Results
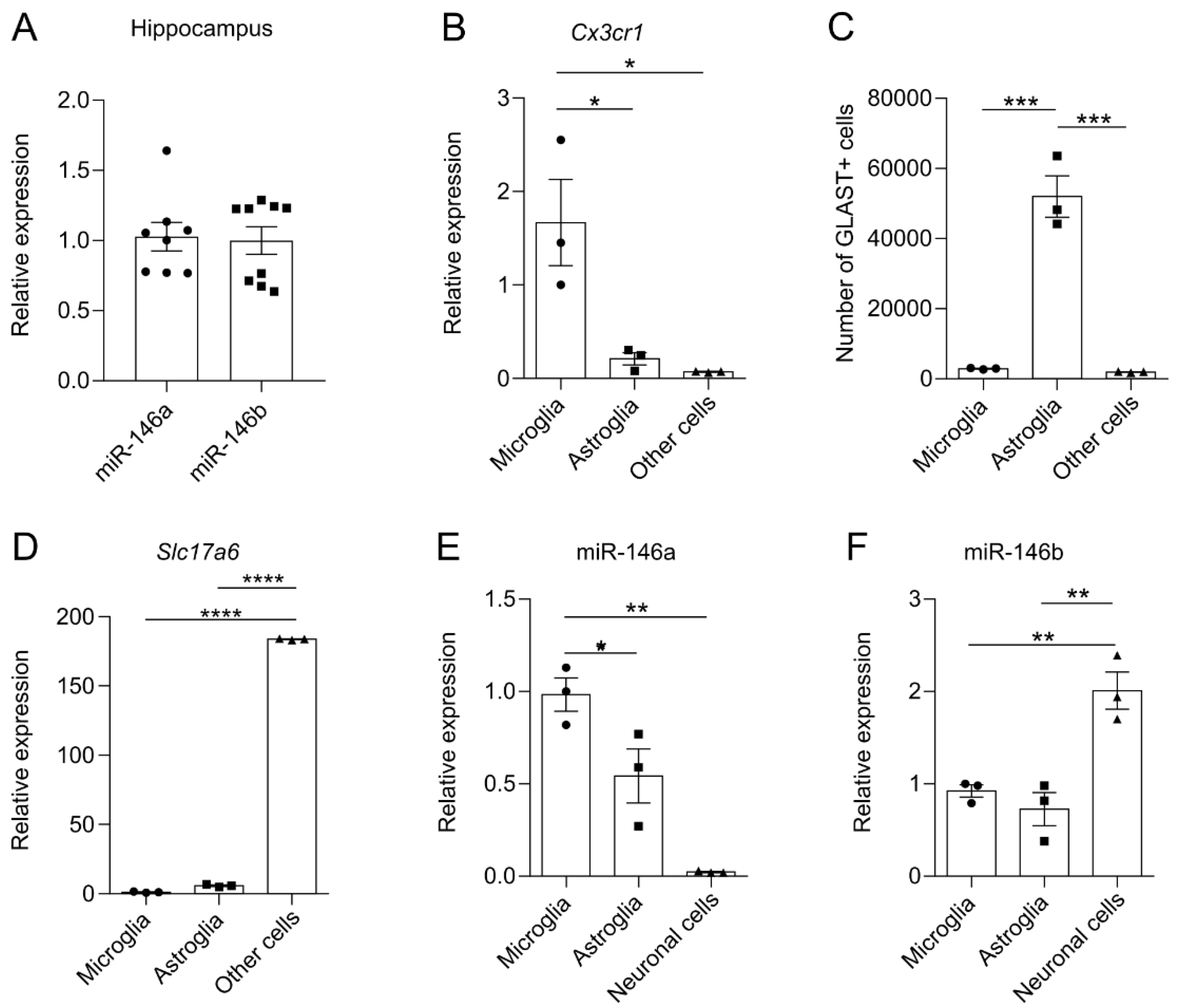
3.1. miR-146b Is Highly Expressed in Neuronal Cells in the Mouse Brain
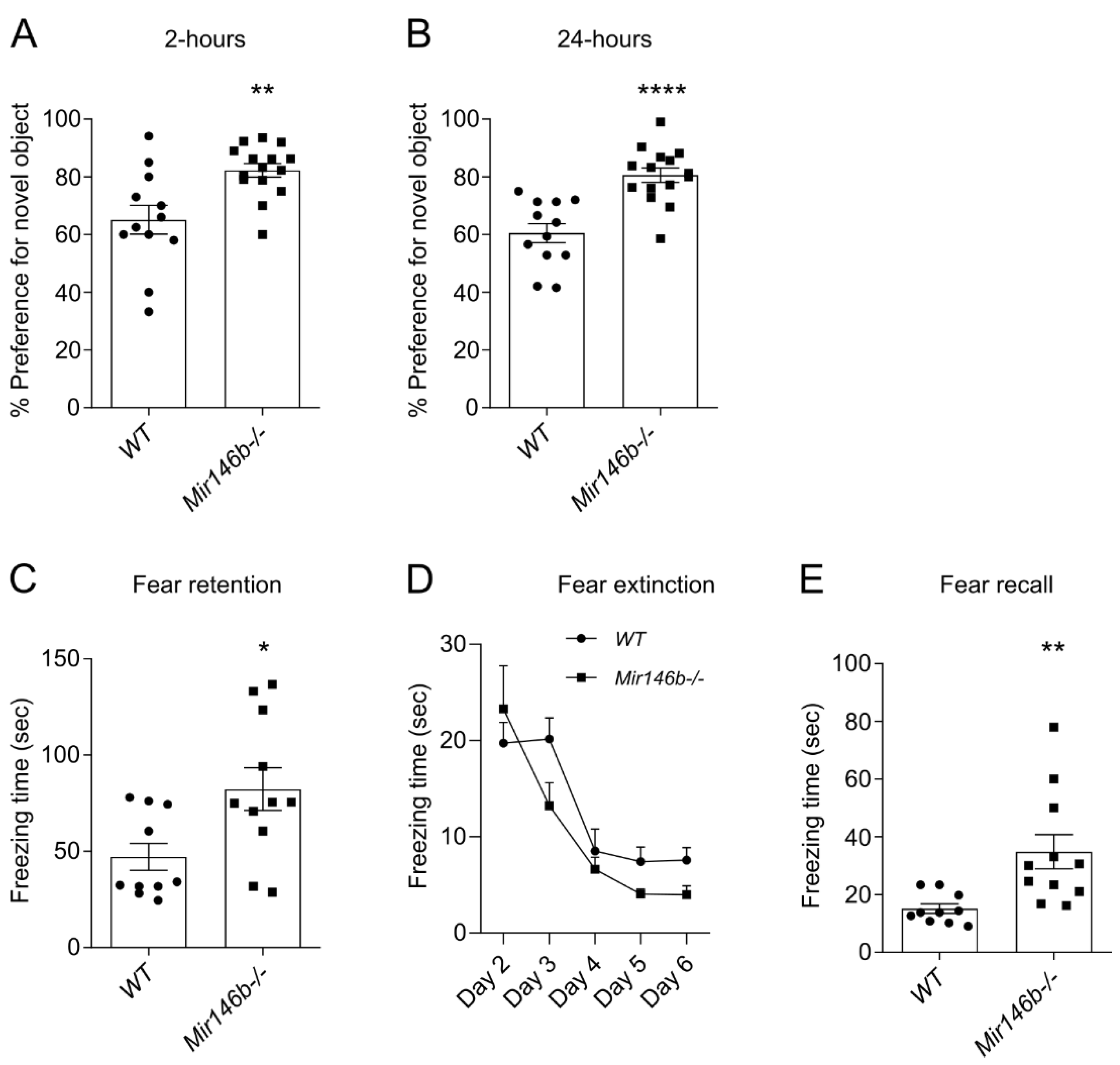
3.2. Mir146b-/- Mice Display Improved Recognition and Associative Memory
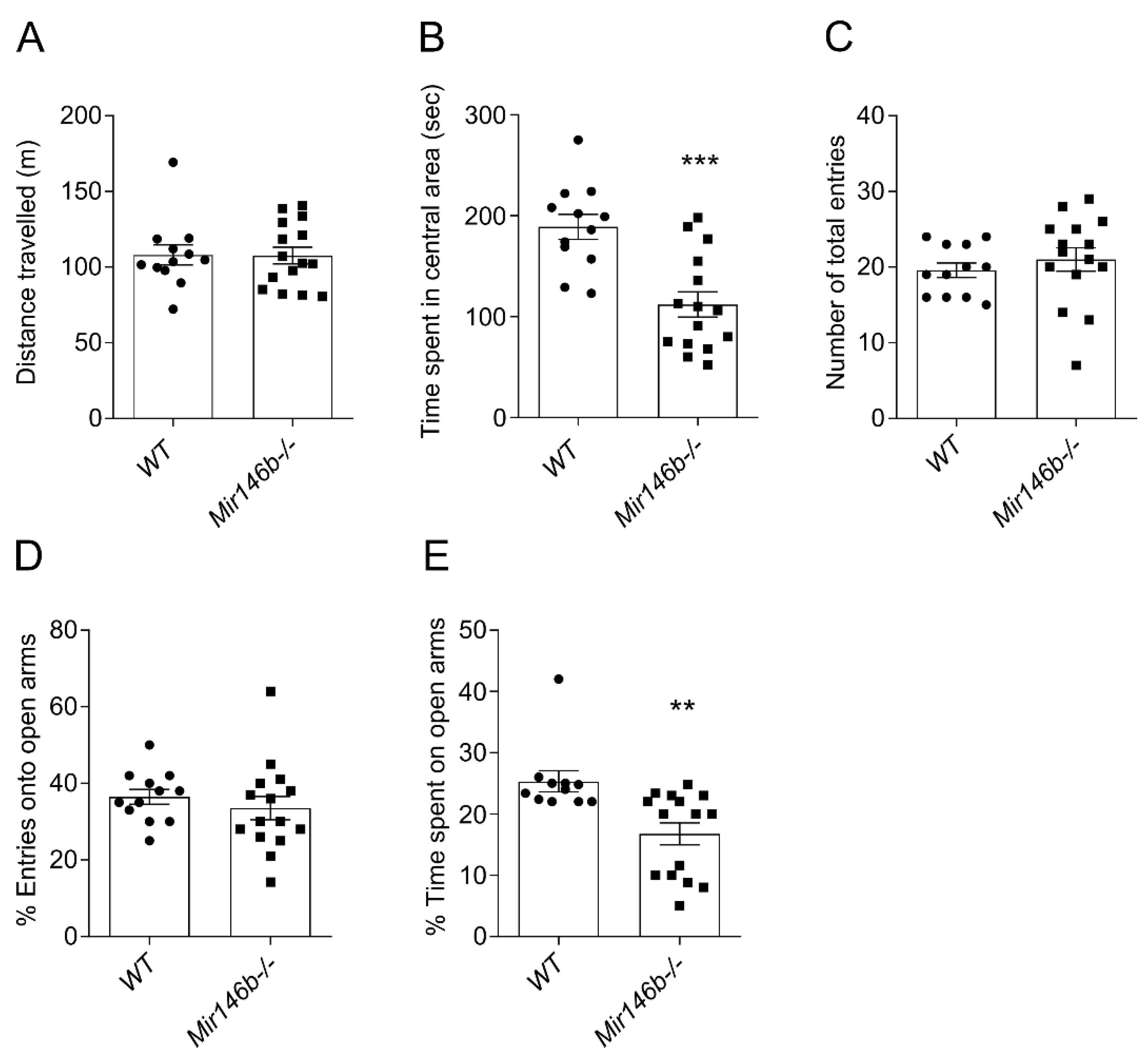
3.3. Mir146b-/- Mice Showed Anxiety-like Behaviors but Had No Differences in Depression-like and Social-Dominant Behaviors
3.4. Brain Cell Abundancy Is Altered in the Brain of Mir146b-/- Mice
3.5. Increased Hippocampal Neurogenesis in Adult Mir146b-/- Mice
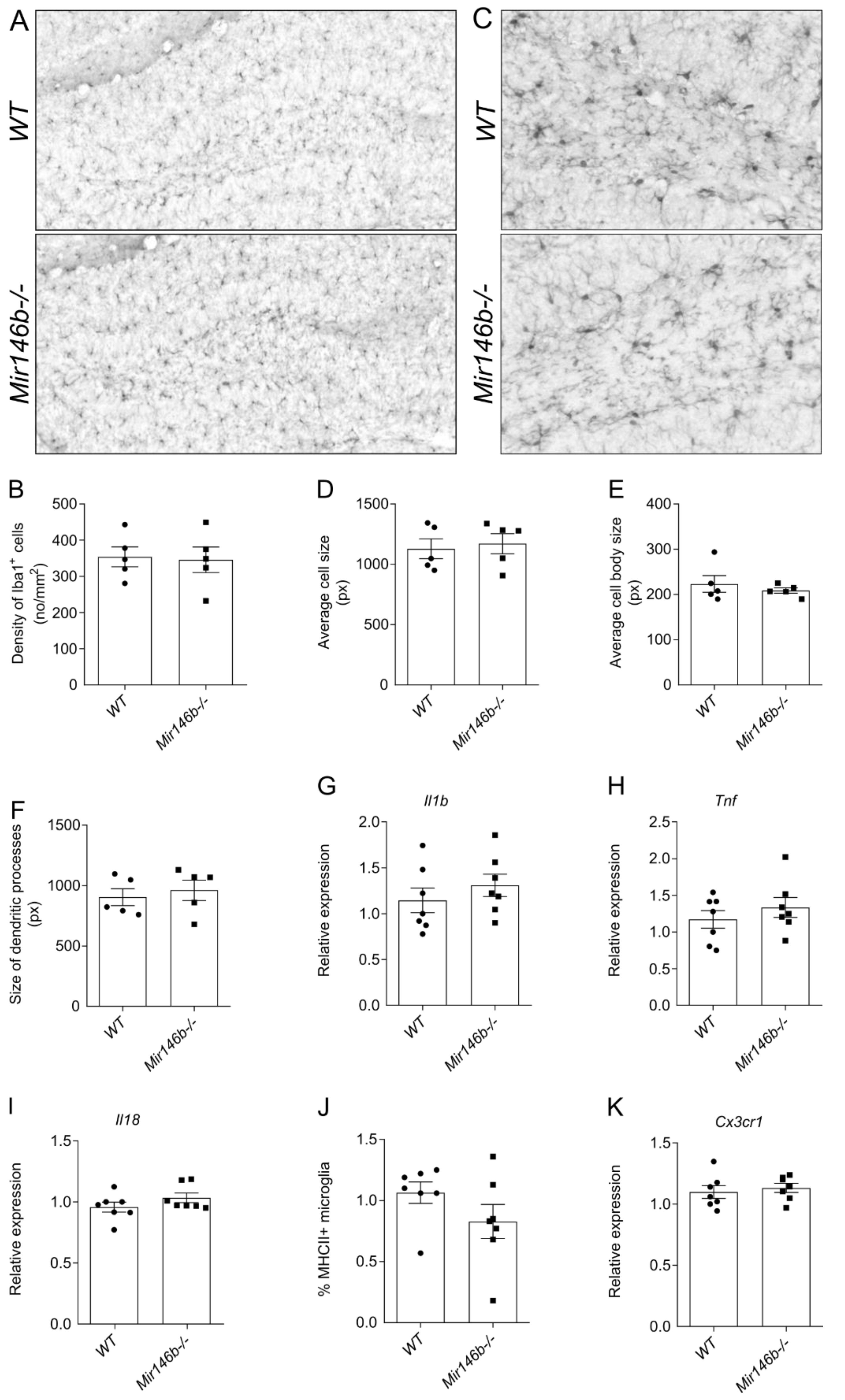
3.6. miR-146b Deficiency Does Not Cause Microglial Activation in the Hippocampus
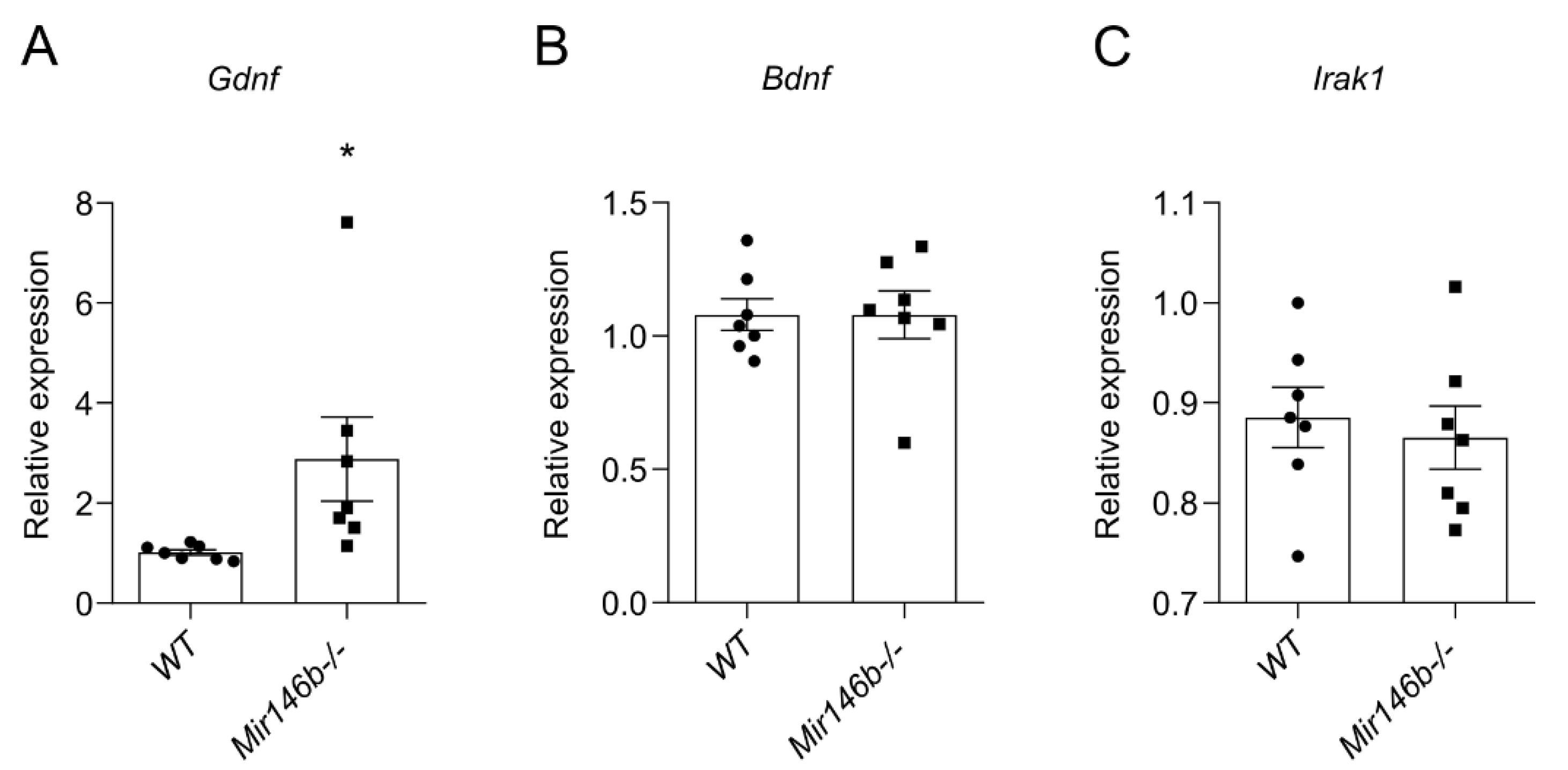
3.7. Glial Cell Line-Derived Neurotrophic Factor (Gdnf) mRNA Is Upregulated in the Hippocampus of Mir146b-/- Mice
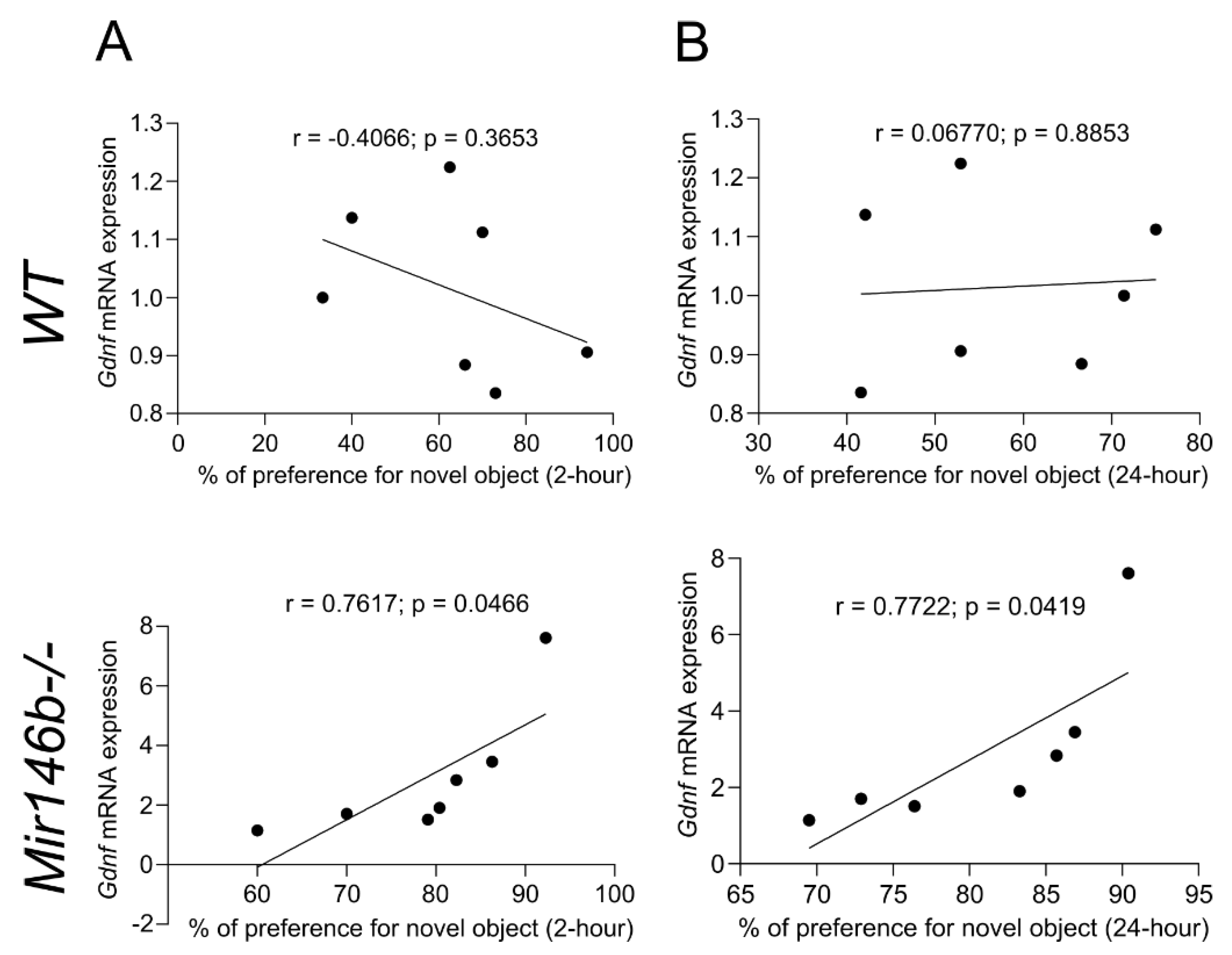
3.8. Association between Enhanced Cognition in NORT and Gdnf mRNA Expression of Mir146b-/- Mice
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Bartel, D.P. Metazoan Micrornas. Cell 2018, 173, 20–51. [Google Scholar] [CrossRef] [PubMed]
- Brennan, G.P.; Henshall, D.C. MicroRNAs as Regulators of Brain Function and Targets for Treatment of Epilepsy. Nat. Rev. Neurol. 2020, 16, 506–519. [Google Scholar] [CrossRef] [PubMed]
- Su, Z.-F.; Sun, Z.-W.; Zhang, Y.; Wang, S.; Yu, Q.-G.; Wu, Z.-B. Regulatory Effects of MiR-146a/b on the Function of Endothelial Progenitor Cells in Acute Ischemic Stroke in Mice. Kaohsiung J. Med. Sci. 2017, 33, 369–378. [Google Scholar] [CrossRef] [PubMed]
- Adlakha, Y.K.; Saini, N. Brain MicroRNAs and Insights into Biological Functions and Therapeutic Potential of Brain Enriched MiRNA-128. Mol. Cancer 2014, 13, 33. [Google Scholar] [CrossRef]
- Bak, M.; Silahtaroglu, A.; Møller, M.; Christensen, M.; Rath, M.F.; Skryabin, B.; Tommerup, N.; Kauppinen, S. MicroRNA Expression in the Adult Mouse Central Nervous System. Rna 2008, 14, 432–444. [Google Scholar] [CrossRef] [PubMed]
- He, M.; Liu, Y.; Wang, X.; Zhang, M.Q.; Hannon, G.J.; Huang, Z.J. Cell-Type-Based Analysis of MicroRNA Profiles in the Mouse Brain. Neuron 2012, 73, 35–48. [Google Scholar] [CrossRef]
- Kuwabara, T.; Hsieh, J.; Nakashima, K.; Taira, K.; Gage, F.H. A Small Modulatory DsRNA Specifies the Fate of Adult Neural Stem Cells. Cell 2004, 116, 779–793. [Google Scholar] [CrossRef]
- Schratt, G.M.; Tuebing, F.; Nigh, E.A.; Kane, C.G.; Sabatini, M.E.; Kiebler, M.; Greenberg, M.E. A Brain-Specific MicroRNA Regulates Dendritic Spine Development. Nature 2006, 439, 283–289. [Google Scholar] [CrossRef]
- Aten, S.; Hansen, K.F.; Hoyt, K.R.; Obrietan, K. The MiR-132/212 Locus: A Complex Regulator of Neuronal Plasticity, Gene Expression and Cognition. RNA Dis. 2016, 3, e1375. [Google Scholar]
- Yan, H.-L.; Sun, X.-W.; Wang, Z.-M.; Liu, P.-P.; Mi, T.-W.; Liu, C.; Wang, Y.-Y.; He, X.-C.; Du, H.-Z.; Liu, C.-M.; et al. MiR-137 Deficiency Causes Anxiety-like Behaviors in Mice. Front. Mol. Neurosci. 2019, 12, 260. [Google Scholar] [CrossRef]
- Jauhari, A.; Singh, T.; Mishra, S.; Shankar, J.; Yadav, S. Coordinated Action of MiR-146a and Parkin Gene Regulate Rotenone-Induced Neurodegeneration. Toxicol. Sci. 2020, 176, 433–445. [Google Scholar] [CrossRef] [PubMed]
- Launay, J.; Mouillet-Richard, S.; Baudry, A.; Pietri, M.; Kellermann, O. Raphe-Mediated Signals Control the Hippocampal Response to SRI Antidepressants via MiR-16. Transl. Psychiatry 2011, 1, e56. [Google Scholar] [CrossRef] [PubMed]
- Lopez, J.P.; Fiori, L.M.; Cruceanu, C.; Lin, R.; Labonte, B.; Cates, H.M.; Heller, E.A.; Vialou, V.; Ku, S.M.; Gerald, C.; et al. MicroRNAs 146a/b-5 and 425-3p and 24-3p Are Markers of Antidepressant Response and Regulate MAPK/Wnt-System Genes. Nat. Commun. 2017, 8, 15497. [Google Scholar] [CrossRef] [PubMed]
- So, A.Y.; Zhao, J.L.; Baltimore, D. The Yin and Yang of Micro RNA s: Leukemia and Immunity. Immunol. Rev. 2013, 253, 129–145. [Google Scholar] [CrossRef] [PubMed]
- Curtale, G.; Mirolo, M.; Renzi, T.A.; Rossato, M.; Bazzoni, F.; Locati, M. Negative Regulation of Toll-like Receptor 4 Signaling by IL-10–Dependent MicroRNA-146b. Proc. Natl. Acad. Sci. USA 2013, 110, 11499–11504. [Google Scholar] [CrossRef]
- Xiang, M.; Birkbak, N.J.; Vafaizadeh, V.; Walker, S.R.; Yeh, J.E.; Liu, S.; Kroll, Y.; Boldin, M.; Taganov, K.; Groner, B.; et al. STAT3 Induction of MiR-146b Forms a Feedback Loop to Inhibit the NF-ΚB to IL-6 Signaling Axis and STAT3-Driven Cancer Phenotypes. Sci. Signal. 2014, 7, ra11. [Google Scholar] [CrossRef]
- Hermann, H.; Runnel, T.; Aab, A.; Baurecht, H.; Rodriguez, E.; Magilnick, N.; Urgard, E.; Šahmatova, L.; Prans, E.; Maslovskaja, J.; et al. MiR-146b Probably Assists MiRNA-146a in the Suppression of Keratinocyte Proliferation and Inflammatory Responses in Psoriasis. J. Invest. Dermatol. 2017, 137, 1945–1954. [Google Scholar] [CrossRef]
- Paterson, M.R.; Kriegel, A.J. MiR-146a/b: A Family with Shared Seeds and Different Roots. Physiol. Genom. 2017, 49, 243–252. [Google Scholar] [CrossRef]
- Taganov, K.D.; Boldin, M.P.; Chang, K.-J.; Baltimore, D. NF-ΚB-Dependent Induction of MicroRNA MiR-146, an Inhibitor Targeted to Signaling Proteins of Innate Immune Responses. Proc. Natl. Acad. Sci. USA 2006, 103, 12481–12486. [Google Scholar] [CrossRef]
- Lee, H.-M.; Kim, T.S.; Jo, E.-K. MiR-146 and MiR-125 in the Regulation of Innate Immunity and Inflammation. BMB Rep. 2016, 49, 311. [Google Scholar] [CrossRef]
- Deng, M.; Du, G.; Zhao, J.; Du, X. MiR-146a Negatively Regulates the Induction of Proinflammatory Cytokines in Response to Japanese Encephalitis Virus Infection in Microglial Cells. Arch. Virol. 2017, 162, 1495–1505. [Google Scholar] [CrossRef] [PubMed]
- Saba, R.; Gushue, S.; Huzarewich, R.L.; Manguiat, K.; Medina, S.; Robertson, C.; Booth, S.A. MicroRNA 146a (MiR-146a) Is over-Expressed during Prion Disease and Modulates the Innate Immune Response and the Microglial Activation State. PLoS ONE 2012, 7, e30832. [Google Scholar] [CrossRef] [PubMed]
- Nguyen, L.S.; Fregeac, J.; Bole-Feysot, C.; Cagnard, N.; Iyer, A.; Anink, J.; Aronica, E.; Alibeu, O.; Nitschke, P.; Colleaux, L. Role of MiR-146a in Neural Stem Cell Differentiation and Neural Lineage Determination: Relevance for Neurodevelopmental Disorders. Mol. Autism 2018, 9, 38. [Google Scholar] [CrossRef]
- Fregeac, J.; Moriceau, S.; Poli, A.; Nguyen, L.S.; Oury, F.; Colleaux, L. Loss of the Neurodevelopmental Disease-Associated Gene MiR-146a Impairs Neural Progenitor Differentiation and Causes Learning and Memory Deficits. Mol. Autism 2020, 11, 22. [Google Scholar] [CrossRef] [PubMed]
- Yang, G.; Zhao, Y. Overexpression of MiR-146b-5p Ameliorates Neonatal Hypoxic Ischemic Encephalopathy by Inhibiting IRAK1/TRAF6/TAK1/NF-AB Signaling. Yonsei Med. J. 2020, 61, 660. [Google Scholar] [CrossRef] [PubMed]
- Dai, Y.; Chen, L.; He, X.; Lin, H.; Jia, W.; Chen, L.; Tao, J.; Liu, W. Construction of MiR-146b Overexpression Lentiviral Vector and the Effect on the Proliferation of Hippocampal Neural Stem Cells. Chin. J. Tissue Eng. Res. 2021, 25, 3024. [Google Scholar]
- Bokobza, C.; Joshi, P.; Schang, A.; Csaba, Z.; Faivre, V.; Montané, A.; Galland, A.; Benmamar-Badel, A.; Bosher, E.; Lebon, S.; et al. MiR-146b Protects the Perinatal Brain against Microglia-Induced Hypomyelination. Ann. Neurol. 2022, 91, 48–65. [Google Scholar] [CrossRef]
- Katakowski, M.; Zheng, X.; Jiang, F.; Rogers, T.; Szalad, A.; Chopp, M. MiR-146b-5p Suppresses EGFR Expression and Reduces in Vitro Migration and Invasion of Glioma. Cancer Investig. 2010, 28, 1024–1030. [Google Scholar] [CrossRef]
- Liu, J.; Xu, J.; Li, H.; Sun, C.; Yu, L.; Li, Y.; Shi, C.; Zhou, X.; Bian, X.; Ping, Y.; et al. MiR-146b-5p Functions as a Tumor Suppressor by Targeting TRAF6 and Predicts the Prognosis of Human Gliomas. Oncotarget 2015, 6, 29129. [Google Scholar] [CrossRef]
- Xia, H.; Qi, Y.; Ng, S.S.; Chen, X.; Li, D.; Chen, S.; Ge, R.; Jiang, S.; Li, G.; Chen, Y.; et al. MicroRNA-146b Inhibits Glioma Cell Migration and Invasion by Targeting MMPs. Brain Res. 2009, 1269, 158–165. [Google Scholar] [CrossRef]
- Zhang, L.; Wang, J.; Fu, Z.; Ai, Y.; Li, Y.; Wang, Y.; Wang, Y. Sevoflurane Suppresses Migration and Invasion of Glioma Cells by Regulating MiR-146b-5p and MMP16. Artif. Cells Nanomed. Biotechnol. 2019, 47, 3306–3314. [Google Scholar] [CrossRef] [PubMed]
- Jürgenson, M.; Aonurm-Helm, A.; Zharkovsky, A. Behavioral Profile of Mice with Impaired Cognition in the Elevated Plus-Maze Due to a Deficiency in Neural Cell Adhesion Molecule. Pharmacol. Biochem. Behav. 2010, 96, 461–468. [Google Scholar] [CrossRef] [PubMed]
- Tronson, N.C.; Schrick, C.; Fischer, A.; Sananbenesi, F.; Pouysségur, J.; Radulovic, J. Regulatory Mechanisms of Fear Extinction and Depression-like Behavior. Neuropsychopharmacology 2008, 33, 1570–1583. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Yan, L.; Jayaram, M.; Chithanathan, K.; Zharkovsky, A.; Tian, L. Sex-Specific Microglial Activation and SARS-CoV-2 Receptor Expression Induced by Chronic Unpredictable Stress. Front. Cell. Neurosci. 2021, 15, 750373. [Google Scholar] [CrossRef] [PubMed]
- Jürgenson, M.; Aonurm-Helm, A.; Zharkovsky, A. Partial Reduction in Neural Cell Adhesion Molecule (NCAM) in Heterozygous Mice Induces Depression-Related Behaviour without Cognitive Impairment. Brain Res. 2012, 1447, 106–118. [Google Scholar] [CrossRef]
- Piirainen, S.; Chithanathan, K.; Bisht, K.; Piirsalu, M.; Savage, J.C.; Tremblay, M.; Tian, L. Microglia Contribute to Social Behavioral Adaptation to Chronic Stress. Glia 2021, 69, 2459–2473. [Google Scholar] [CrossRef]
- Hovens, I.B.; Nyakas, C.; Schoemaker, R.G. A Novel Method for Evaluating Microglial Activation Using Ionized Calcium-Binding Adaptor Protein-1 Staining: Cell Body to Cell Size Ratio. Neuroimmunol. Neuroinflamm. 2014, 1, 82–88. [Google Scholar] [CrossRef]
- Jürgenson, M.; Zharkovskaja, T.; Noortoots, A.; Morozova, M.; Beniashvili, A.; Zapolski, M.; Zharkovsky, A. Effects of the Drug Combination Memantine and Melatonin on Impaired Memory and Brain Neuronal Deficits in an Amyloid-Predominant Mouse Model of Alzheimer’s Disease. J. Pharm. Pharmacol. 2019, 71, 1695–1705. [Google Scholar] [CrossRef]
- Somelar, K.; Jürgenson, M.; Jaako, K.; Anier, K.; Aonurm-Helm, A.; Zvejniece, L.; Zharkovsky, A. Development of Depression-like Behavior and Altered Hippocampal Neurogenesis in a Mouse Model of Chronic Neuropathic Pain. Brain Res. 2021, 1758, 147329. [Google Scholar] [CrossRef]
- Agalave, N.M.; Lane, B.T.; Mody, P.H.; Szabo-Pardi, T.A.; Burton, M.D. Isolation, Culture, and Downstream Characterization of Primary Microglia and Astrocytes from Adult Rodent Brain and Spinal Cord. J. Neurosci. Methods 2020, 340, 108742. [Google Scholar] [CrossRef]
- Agarwal, V.; Bell, G.W.; Nam, J.-W.; Bartel, D.P. Predicting Effective MicroRNA Target Sites in Mammalian MRNAs. eLife 2015, 4, e05005. [Google Scholar] [CrossRef] [PubMed]
- McGeary, S.E.; Lin, K.S.; Shi, C.Y.; Pham, T.M.; Bisaria, N.; Kelley, G.M.; Bartel, D.P. The Biochemical Basis of MicroRNA Targeting Efficacy. Science 2019, 366, eaav1741. [Google Scholar] [CrossRef] [PubMed]
- Reimand, J.; Kull, M.; Peterson, H.; Hansen, J.; Vilo, J. g:Profiler—A Web-Based Toolset for Functional Profiling of Gene Lists from Large-Scale Experiments. Nucleic Acids Res. 2007, 35 (Suppl. 2), W193–W200. [Google Scholar] [CrossRef] [PubMed]
- Ansari, A.; Maffioletti, E.; Milanesi, E.; Marizzoni, M.; Frisoni, G.B.; Blin, O.; Richardson, J.C.; Bordet, R.; Forloni, G.; Gennarelli, M.; et al. MiR-146a and MiR-181a Are Involved in the Progression of Mild Cognitive Impairment to Alzheimer’s Disease. Neurobiol. Aging 2019, 82, 102–109. [Google Scholar] [CrossRef] [PubMed]
- Cui, L.; Li, Y.; Ma, G.; Wang, Y.; Cai, Y.; Liu, S.; Chen, Y.; Li, J.; Xie, Y.; Liu, G. A Functional Polymorphism in the Promoter Region of MicroRNA-146a Is Associated with the Risk of Alzheimer Disease and the Rate of Cognitive Decline in Patients. PLoS ONE 2014, 9, e89019. [Google Scholar] [CrossRef]
- Zhan-Qiang, H.; Hai-Hua, Q.; Chi, Z.; Miao, W.; Cui, Z.; Zi-Yin, L.; Jing, H.; Yi-Wei, W. MiR-146a Aggravates Cognitive Impairment and Alzheimer Disease-like Pathology by Triggering Oxidative Stress through MAPK Signaling. Neurología 2021. [Google Scholar] [CrossRef]
- Ennaceur, A.; Delacour, J. A New One-Trial Test for Neurobiological Studies of Memory in Rats. 1: Behavioral Data. Behav. Brain Res. 1988, 31, 47–59. [Google Scholar] [CrossRef]
- Ma, L. Depression, Anxiety, and Apathy in Mild Cognitive Impairment: Current Perspectives. Front. Aging Neurosci. 2020, 12, 9. [Google Scholar] [CrossRef]
- Enatescu, V.R.; Papava, I.; Enatescu, I.; Antonescu, M.; Anghel, A.; Seclaman, E.; Sirbu, I.O.; Marian, C. Circulating Plasma Micro RNAs in Patients with Major Depressive Disorder Treated with Antidepressants: A Pilot Study. Psychiatry Investig. 2016, 13, 549. [Google Scholar] [CrossRef]
- Hung, Y.-Y.; Wu, M.-K.; Tsai, M.-C.; Huang, Y.-L.; Kang, H.-Y. Aberrant Expression of Intracellular Let-7e, MiR-146a, and MiR-155 Correlates with Severity of Depression in Patients with Major Depressive Disorder and Is Ameliorated after Antidepressant Treatment. Cells 2019, 8, 647. [Google Scholar] [CrossRef]
- Lindert, J.; Paul, K.C.; Lachman, M.E.; Ritz, B.; Seeman, T.E. Depression-, Anxiety-, and Anger and Cognitive Functions: Findings From a Longitudinal Prospective Study. Front. Psychiatry 2021, 12, 665742. [Google Scholar] [CrossRef] [PubMed]
- Von Bartheld, C.S.; Wouters, F.S. Quantitative Techniques for Imaging Cells and Tissues. Cell Tissue Res. 2015, 360, 1–4. [Google Scholar] [CrossRef] [PubMed]
- De Lucia, C.; Rinchon, A.; Olmos-Alonso, A.; Riecken, K.; Fehse, B.; Boche, D.; Perry, V.H.; Gomez-Nicola, D. Microglia Regulate Hippocampal Neurogenesis during Chronic Neurodegeneration. Brain Behav. Immun. 2016, 55, 179–190. [Google Scholar] [CrossRef]
- Bachstetter, A.D.; Morganti, J.M.; Jernberg, J.; Schlunk, A.; Mitchell, S.H.; Brewster, K.W.; Hudson, C.E.; Cole, M.J.; Harrison, J.K.; Bickford, P.C.; et al. Fractalkine and CX3CR1 Regulate Hippocampal Neurogenesis in Adult and Aged Rats. Neurobiol. Aging 2011, 32, 2030–2044. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Chen, K.; Sloan, S.A.; Bennett, M.L.; Scholze, A.R.; O’Keeffe, S.; Phatnani, H.P.; Guarnieri, P.; Caneda, C.; Ruderisch, N.; et al. An RNA-Sequencing Transcriptome and Splicing Database of Glia, Neurons, and Vascular Cells of the Cerebral Cortex. J. Neurosci. 2014, 34, 11929–11947. [Google Scholar] [CrossRef] [PubMed]
- Kumar, A.; Kopra, J.; Varendi, K.; Porokuokka, L.L.; Panhelainen, A.; Kuure, S.; Marshall, P.; Karalija, N.; Härma, M.-A.; Vilenius, C.; et al. GDNF Overexpression from the Native Locus Reveals Its Role in the Nigrostriatal Dopaminergic System Function. PLoS Genet. 2015, 11, e1005710. [Google Scholar] [CrossRef]
- Hsu, P.-K.; Xu, B.; Mukai, J.; Karayiorgou, M.; Gogos, J.A. The BDNF Val66Met Variant Affects Gene Expression through MiR-146b. Neurobiol. Dis. 2015, 77, 228–237. [Google Scholar] [CrossRef]
- Robinson, O.J.; Vytal, K.; Cornwell, B.R.; Grillon, C. The Impact of Anxiety upon Cognition: Perspectives from Human Threat of Shock Studies. Front. Hum. Neurosci. 2013, 7, 203. [Google Scholar] [CrossRef]
- Hu, K.; Bauer, A.; Padmala, S.; Pessoa, L. Threat of Bodily Harm Has Opposing Effects on Cognition. Emotion 2012, 12, 28. [Google Scholar] [CrossRef]
- Roozendaal, B.; Okuda, S.; Van der Zee, E.A.; McGaugh, J.L. Glucocorticoid Enhancement of Memory Requires Arousal-Induced Noradrenergic Activation in the Basolateral Amygdala. Proc. Natl. Acad. Sci. USA 2006, 103, 6741–6746. [Google Scholar] [CrossRef]
- Kolk, S.M.; Rakic, P. Development of Prefrontal Cortex. Neuropsychopharmacology 2022, 47, 41–57. [Google Scholar] [CrossRef] [PubMed]
- Kroon, T.; van Hugte, E.; van Linge, L.; Mansvelder, H.D.; Meredith, R.M. Early Postnatal Development of Pyramidal Neurons across Layers of the Mouse Medial Prefrontal Cortex. Sci. Rep. 2019, 9, 5037. [Google Scholar] [CrossRef] [PubMed]
- Bejoy, J.; Bijonowski, B.; Marzano, M.; Jeske, R.; Ma, T.; Li, Y. Wnt-Notch Signaling Interactions during Neural and Astroglial Patterning of Human Stem Cells. Tissue Eng. Part A 2020, 26, 419–431. [Google Scholar] [CrossRef] [PubMed]
- Lee, H.-C.; Tan, K.-L.; Cheah, P.-S.; Ling, K.-H. Potential Role of JAK-STAT Signaling Pathway in the Neurogenic-to-Gliogenic Shift in down Syndrome Brain. Neural Plast. 2016, 2016, 7434191. [Google Scholar] [CrossRef] [PubMed]
- Deng, X.; Wu, B.; Xiao, K.; Kang, J.; Xie, J.; Zhang, X.; Fan, Y. MiR-146b-5p Promotes Metastasis and Induces Epithelial-Mesenchymal Transition in Thyroid Cancer by Targeting ZNRF3. Cell. Physiol. Biochem. 2015, 35, 71–82. [Google Scholar] [CrossRef] [PubMed]
- Kee, N.; Teixeira, C.M.; Wang, A.H.; Frankland, P.W. Preferential Incorporation of Adult-Generated Granule Cells into Spatial Memory Networks in the Dentate Gyrus. Nat. Neurosci. 2007, 10, 355–362. [Google Scholar] [CrossRef]
- Vivar, C.; Van Praag, H. Functional Circuits of New Neurons in the Dentate Gyrus. Front. Neural Circuits 2013, 7, 15. [Google Scholar] [CrossRef]
- Araki, T.; Ikegaya, Y.; Koyama, R. The Effects of Microglia-and Astrocyte-derived Factors on Neurogenesis in Health and Disease. Eur. J. Neurosci. 2021, 54, 5880–5901. [Google Scholar] [CrossRef]
- Kim, S.U.; de Vellis, J. Microglia in Health and Disease. J. Neurosci. Res. 2005, 81, 302–313. [Google Scholar] [CrossRef]
- Paolicelli, R.C.; Bolasco, G.; Pagani, F.; Maggi, L.; Scianni, M.; Panzanelli, P.; Giustetto, M.; Ferreira, T.A.; Guiducci, E.; Dumas, L.; et al. Synaptic Pruning by Microglia Is Necessary for Normal Brain Development. Science 2011, 333, 1456–1458. [Google Scholar] [CrossRef]
- Gianino, S.; Grider, J.R.; Cresswell, J.; Enomoto, H.; Heuckeroth, R.O. GDNF Availability Determines Enteric Neuron Number by Controlling Precursor Proliferation. Development 2003, 130, 2187–2198. [Google Scholar] [CrossRef] [PubMed]
- Cortés, D.; Carballo-Molina, O.A.; Castellanos-Montiel, M.J.; Velasco, I. The Non-Survival Effects of Glial Cell Line-Derived Neurotrophic Factor on Neural Cells. Front. Mol. Neurosci. 2017, 10, 258. [Google Scholar] [CrossRef] [PubMed]
- Pelleymounter, M.A.; Cullen, M.J.; Baker, M.B.; Healy, D. Glial Cell Line-Derived Neurotrophic Factor (GDNF) Improves Spatial Learning in Aged Fischer 344 Rats. Psychobiology 1999, 27, 397–401. [Google Scholar] [CrossRef]
- Pertusa, M.; Garcia-Matas, S.; Mammeri, H.; Adell, A.; Rodrigo, T.; Mallet, J.; Cristofol, R.; Sarkis, C.; Sanfeliu, C. Expression of GDNF Transgene in Astrocytes Improves Cognitive Deficits in Aged Rats. Neurobiol. Aging 2008, 29, 1366–1379. [Google Scholar] [CrossRef]
- Zhang, S.; Jin, T.; Wang, L.; Liu, W.; Zhang, Y.; Zheng, Y.; Lin, Y.; Yang, M.; He, X.; Lin, H.; et al. Electro-Acupuncture Promotes the Differentiation of Endogenous Neural Stem Cells via Exosomal MicroRNA 146b after Ischemic Stroke. Front. Cell. Neurosci. 2020, 14, 223. [Google Scholar] [CrossRef]
- Li, N.; Guo, Y.; Gong, Y.; Zhang, Y.; Fan, W.; Yao, K.; Chen, Z.; Dou, B.; Lin, X.; Chen, B.; et al. The Anti-Inflammatory Actions and Mechanisms of Acupuncture from Acupoint to Target Organs via Neuro-Immune Regulation. J. Inflamm. Res. 2021, 14, 7191. [Google Scholar] [CrossRef]
- Pan, J.-A.; Tang, Y.; Yu, J.-Y.; Zhang, H.; Zhang, J.-F.; Wang, C.-Q.; Gu, J. MiR-146a Attenuates Apoptosis and Modulates Autophagy by Targeting TAF9b/P53 Pathway in Doxorubicin-Induced Cardiotoxicity. Cell Death Dis. 2019, 10, 668. [Google Scholar] [CrossRef]
- Qiu, M.; Li, T.; Wang, B.; Gong, H.; Huang, T. MiR-146a-5p Regulated Cell Proliferation and Apoptosis by Targeting SMAD3 and SMAD4. Protein Pept. Lett. 2020, 27, 411–418. [Google Scholar] [CrossRef]
- Xu, J.; Zhang, Z.; Chen, Q.; Yang, L.; Yin, J. MiR-146b Regulates Cell Proliferation and Apoptosis in Gastric Cancer by Targeting PTP1B. Dig. Dis. Sci. 2020, 65, 457–463. [Google Scholar] [CrossRef]
- Ouyang, B.; Pan, N.; Zhang, H.; Xing, C.; Ji, W. MiR-146b-5p Inhibits Tumorigenesis and Metastasis of Gallbladder Cancer by Targeting Toll-like Receptor 4 via the Nuclear Factor-κB Pathway. Oncol. Rep. 2021, 45, 15. [Google Scholar] [CrossRef]


| Time Point | Task Assigned |
|---|---|
| Cohort 1 (WT and Mir146b-/-) | |
| Day 0 | Open field test |
| Day 4 | Elevated plus maze |
| Day 8–10 | Novel object recognition test |
| Day 13–14 | Social dominance test |
| Day 17 | Tail suspension test |
| Day 22 | Sacrifice the animals and collect the tissues for flow cytometry, qPCR and immunohistochemistry |
| Cohort 2 (WT and Mir146b-/-) | |
| Day 0–7 | Contextual fear conditioning |
| Day 10 | BrdU injections (300 mg/kg) |
| Day 31 | Sacrifice animals for immunohistochemistry and qPCR |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Chithanathan, K.; Somelar, K.; Jürgenson, M.; Žarkovskaja, T.; Periyasamy, K.; Yan, L.; Magilnick, N.; Boldin, M.P.; Rebane, A.; Tian, L.; et al. Enhanced Cognition and Neurogenesis in miR-146b Deficient Mice. Cells 2022, 11, 2002. https://doi.org/10.3390/cells11132002
Chithanathan K, Somelar K, Jürgenson M, Žarkovskaja T, Periyasamy K, Yan L, Magilnick N, Boldin MP, Rebane A, Tian L, et al. Enhanced Cognition and Neurogenesis in miR-146b Deficient Mice. Cells. 2022; 11(13):2002. https://doi.org/10.3390/cells11132002
Chicago/Turabian StyleChithanathan, Keerthana, Kelli Somelar, Monika Jürgenson, Tamara Žarkovskaja, Kapilraj Periyasamy, Ling Yan, Nathaniel Magilnick, Mark P. Boldin, Ana Rebane, Li Tian, and et al. 2022. "Enhanced Cognition and Neurogenesis in miR-146b Deficient Mice" Cells 11, no. 13: 2002. https://doi.org/10.3390/cells11132002
APA StyleChithanathan, K., Somelar, K., Jürgenson, M., Žarkovskaja, T., Periyasamy, K., Yan, L., Magilnick, N., Boldin, M. P., Rebane, A., Tian, L., & Zharkovsky, A. (2022). Enhanced Cognition and Neurogenesis in miR-146b Deficient Mice. Cells, 11(13), 2002. https://doi.org/10.3390/cells11132002







