Contribution of Oxidative Stress (OS) in Calcific Aortic Valve Disease (CAVD): From Pathophysiology to Therapeutic Targets
Abstract
1. Introduction
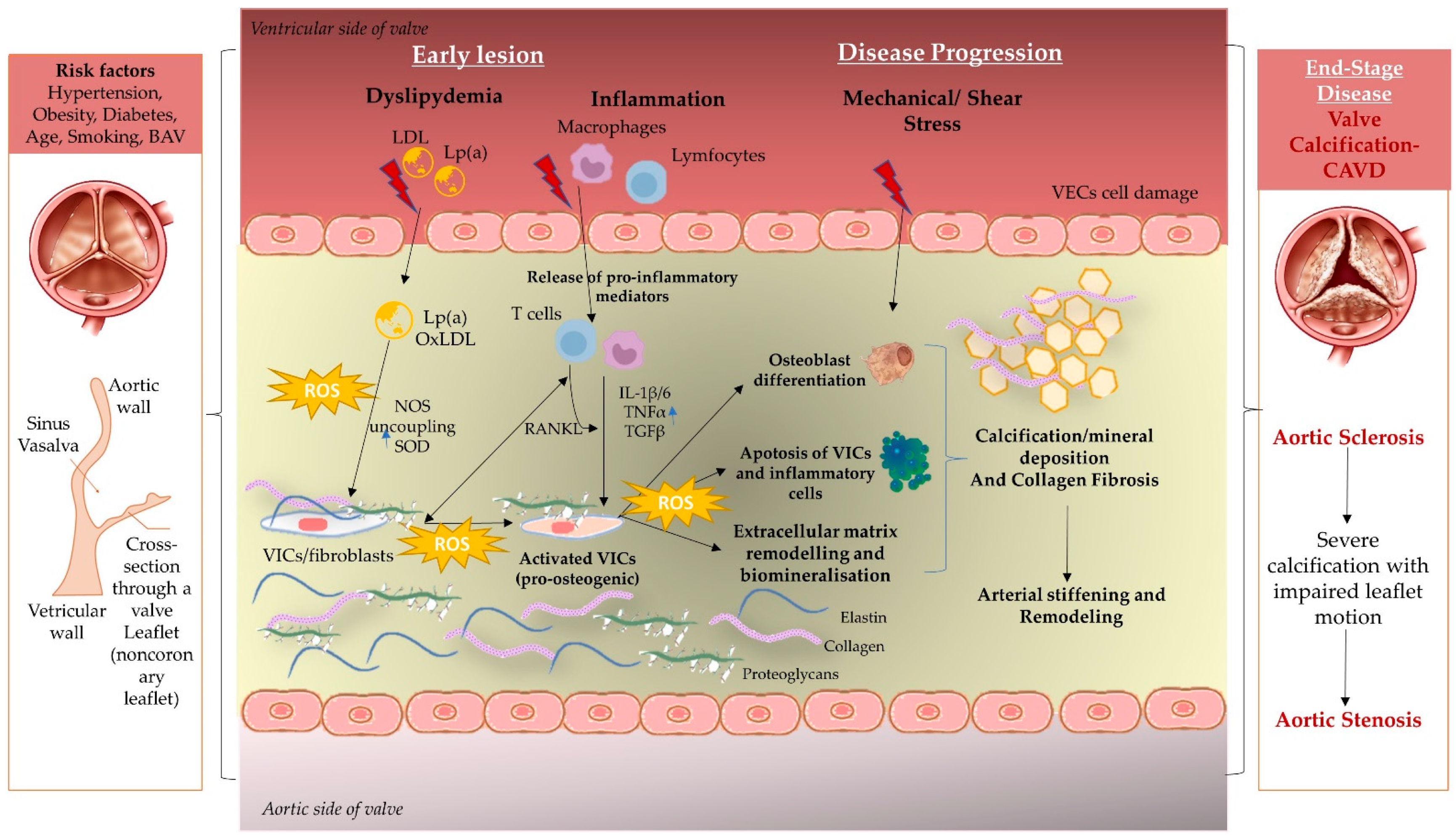
2. Immunopathogenesis behind Aortic Disease
2.1. Mechanical Stress and Endothelial Dysfunction
2.2. Lipid Deposition and Oxidative Stress in SA and BAV
2.3. Inflammation Mechanism
2.4. Extracellular Matrix Remodeling and Biomineralization
3. Therapeutic Targets of Oxidative Stress
3.1. Targeting Lipid Oxidation
3.2. Antioxidants from Natural Compounds to Targeted Experimental Therapies
3.2.1. Phenolic Acids, Flavonoids, Anthocyanins, Lignans, and Stilbenes
3.2.2. Vitamins
3.2.3. Other Antioxidant Compounds
3.2.4. Targeting mitoROS and SOD
3.3. NO Bioavailability and NOX Inhibition
| Study and Refs. | Compound | Administration and Doses/Researched Cells | Salient Findings |
|---|---|---|---|
| Clinical Trials | |||
| ASTRONOMER [101] | Rosuvastatin vs. placebo | 40 mg/day | -Lp(a) and OxPL-apoB levels are associated with faster AS progression; -OxPL-apoB levels were higher after one year in the rosuvastatin arm. |
| RAAVE [102] | Rosuvastatin vs. placebo | 20 mg/day | -Precocious statin treatment is more effective in the progression of aortic valve stenosis. |
| SALTIRE [103] | Atorvastatin vs. placebo | 80 mg/day | -Intensive lipid-lowering therapy delays the progression of calcific aortic stenosis. |
| SEAS [104] | Simvastatin + Ezetimibe vs. placebo | 40 mg + 10 mg/day | -No reduction in valvular or ischemic events in patients with aortic stenosis. |
| FOURIER [115,117] | Evolocumab | 140 mg every 2 weeks or 420 mg every month | -After 1-year of reduced LDL cholesterol levels and cardiovascular events; -Higher Lp(a) levels were associated with a higher risk of AS events. |
| GLAGOV [118] | Evolocumab | 420 mg every month | -Added statin treatment in angiographic coronary artery disease decreased atheroma volume. |
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Chen, J.; Li, W.; Xiang, M. Burden of Valvular Heart Disease, 1990–2017: Results from the Global Burden of Disease Study 2017. J. Glob. Health 2020, 10, 020404. [Google Scholar] [CrossRef]
- James, S.L.; Abate, D.; Abate, K.H.; Abay, S.M.; Abbafati, C.; Abbasi, N.; Abbastabar, H.; Abd-Allah, F.; Abdela, J.; Abdelalim, A.; et al. Global, Regional, and National Incidence, Prevalence, and Years Lived with Disability for 354 Diseases and Injuries for 195 Countries and Territories, 1990–2017: A Systematic Analysis for the Global Burden of Disease Study 2017. Lancet 2018, 392, 1789–1858. [Google Scholar] [CrossRef]
- Iung, B. A Prospective Survey of Patients with Valvular Heart Disease in Europe: The Euro Heart Survey on Valvular Heart Disease. Eur. Heart J. 2003, 24, 1231–1243. [Google Scholar] [CrossRef]
- Vahanian, A.; Beyersdorf, F.; Bauersachs, J.; Capodanno, D.; Conradi, L.; Bonis, M.D.; Paulis, R.D.; Delgado, V.; Freemantle, N.; Jeppsson, A.; et al. 2021 ESC/EACTS Guidelines for the Management of Valvular Heart Disease. Eur. Heart J. 2022, 43, 561–632. [Google Scholar] [CrossRef]
- Conte, M.; Petraglia, L.; Campana, P.; Gerundo, G.; Caruso, A.; Grimaldi, M.G.; Russo, V.; Attena, E.; Leosco, D.; Parisi, V. The Role of Inflammation and Metabolic Risk Factors in the Pathogenesis of Calcific Aortic Valve Stenosis. Aging Clin. Exp. Res. 2021, 33, 1765–1770. [Google Scholar] [CrossRef]
- Natorska, J.; Kopytek, M.; Undas, A. Aortic Valvular Stenosis: Novel Therapeutic Strategies. Eur. J. Clin. Investig. 2021, 51, e13527. [Google Scholar] [CrossRef]
- Akahori, H.; Tsujino, T.; Masuyama, T.; Ishihara, M. Mechanisms of Aortic Stenosis. J. Cardiol. 2018, 71, 215–220. [Google Scholar] [CrossRef]
- Tretjakovs, P.; Lurins, J.; Svirskis, S.; Gersone, G.; Lurina, D.; Rozenberga, U.; Blumfelds, L.; Bahs, G.; Lejnieks, A.; Mackevics, V. Thioredoxin-1 and Correlations of the Plasma Cytokines Regarding Aortic Valve Stenosis Severity. Biomedicines 2021, 9, 1041. [Google Scholar] [CrossRef]
- Phua, K.; Chew, N.W.; Kong, W.K.; Tan, R.-S.; Ye, L.; Poh, K.-K. The Mechanistic Pathways of Oxidative Stress in Aortic Stenosis and Clinical Implications. Theranostics 2022, 12, 5189–5203. [Google Scholar] [CrossRef]
- Mozzini, C.; Girelli, D.; Cominacini, L.; Soresi, M. An Exploratory Look at Bicuspid Aortic Valve (Bav) Aortopathy: Focus on Molecular and Cellular Mechanisms. Curr. Probl. Cardiol. 2021, 46, 100425. [Google Scholar] [CrossRef]
- Manno, G.; Bentivegna, R.; Morreale, P.; Nobile, D.; Santangelo, A.; Novo, S.; Novo, G. Chronic Inflammation: A Key Role in Degeneration of Bicuspid Aortic Valve. J. Mol. Cell. Cardiol. 2019, 130, 59–64. [Google Scholar] [CrossRef] [PubMed]
- Phillippi, J.A.; Hill, J.C.; Billaud, M.; Green, B.R.; Kotlarczyk, M.P.; Gleason, T.G. Bicuspid Aortic Valve Morphotype Correlates With Regional Antioxidant Gene Expression Profiles in the Proximal Ascending Aorta. Ann. Thorac. Surg. 2017, 104, 79–87. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Mathieu, P.; Bossé, Y.; Huggins, G.S.; Della Corte, A.; Pibarot, P.; Michelena, H.I.; Limongelli, G.; Boulanger, M.-C.; Evangelista, A.; Bédard, E.; et al. The Pathology and Pathobiology of Bicuspid Aortic Valve: State of the Art and Novel Research Perspectives: Pathology and Pathobiology of Bicuspid Aortic Valve. J. Pathol. Clin. Res. 2015, 1, 195–206. [Google Scholar] [CrossRef] [PubMed]
- Zheng, K.H.; Tzolos, E.; Dweck, M.R. Pathophysiology of Aortic Stenosis and Future Perspectives for Medical Therapy. Cardiol. Clin. 2020, 38, 1–12. [Google Scholar] [CrossRef]
- Farrar, E.J.; Huntley, G.D.; Butcher, J. Endothelial-Derived Oxidative Stress Drives Myofibroblastic Activation and Calcification of the Aortic Valve. PLoS ONE 2015, 10, e0123257. [Google Scholar] [CrossRef]
- Mercier, N.; Pawelzik, S.-C.; Pirault, J.; Carracedo, M.; Persson, O.; Wollensack, B.; Franco-Cereceda, A.; Bäck, M. Semicarbazide-Sensitive Amine Oxidase Increases in Calcific Aortic Valve Stenosis and Contributes to Valvular Interstitial Cell Calcification. Oxid. Med. Cell. Longev. 2020, 2020, 5197376. [Google Scholar] [CrossRef] [PubMed]
- Alushi, B.; Curini, L.; Christopher, M.R.; Grubitzch, H.; Landmesser, U.; Amedei, A.; Lauten, A. Calcific Aortic Valve Disease-Natural History and Future Therapeutic Strategies. Front. Pharmacol. 2020, 11, 685. [Google Scholar] [CrossRef]
- Hofmanis, J.; Hofmane, D.; Svirskis, S.; Mackevics, V.; Tretjakovs, P.; Lejnieks, A.; Signorelli, S.S. HDL-C Role in Acquired Aortic Valve Stenosis Patients and Its Relationship with Oxidative Stress. Medicina 2019, 55, 416. [Google Scholar] [CrossRef]
- Rysz, J.; Gluba-Brzózka, A.; Rokicki, R.; Franczyk, B. Oxidative Stress-Related Susceptibility to Aneurysm in Marfan’s Syndrome. Biomedicines 2021, 9, 1171. [Google Scholar] [CrossRef]
- Cho, K.I.; Sakuma, I.; Sohn, I.S.; Jo, S.-H.; Koh, K.K. Inflammatory and Metabolic Mechanisms Underlying the Calcific Aortic Valve Disease. Atherosclerosis 2018, 277, 60–65. [Google Scholar] [CrossRef]
- Goody, P.R.; Hosen, M.R.; Christmann, D.; Niepmann, S.T.; Zietzer, A.; Adam, M.; Bönner, F.; Zimmer, S.; Nickenig, G.; Jansen, F. Aortic Valve Stenosis: From Basic Mechanisms to Novel Therapeutic Targets. Arterioscler. Thromb. Vasc. Biol. 2020, 40, 885–900. [Google Scholar] [CrossRef] [PubMed]
- Sun, J.T.; Chen, Y.Y.; Mao, J.Y.; Wang, Y.P.; Chen, Y.F.; Hu, X.; Yang, K.; Liu, Y. Oxidized HDL, as a Novel Biomarker for Calcific Aortic Valve Disease, Promotes the Calcification of Aortic Valve Interstitial Cells. J. Cardiovasc. Transl. Res. 2019, 12, 560–568. [Google Scholar] [CrossRef] [PubMed]
- Xue, Y.; St. Hilaire, C.; Hortells, L.; Phillippi, J.A.; Sant, V.; Sant, S. Shape-Specific Nanoceria Mitigate Oxidative Stress-Induced Calcification in Primary Human Valvular Interstitial Cell Culture. Cell. Mol. Bioeng. 2017, 10, 483–500. [Google Scholar] [CrossRef]
- Branchetti, E.; Sainger, R.; Poggio, P.; Grau, J.B.; Patterson-Fortin, J.; Bavaria, J.E.; Chorny, M.; Lai, E.; Gorman, R.C.; Levy, R.J.; et al. Antioxidant Enzymes Reduce DNA Damage and Early Activation of Valvular Interstitial Cells in Aortic Valve Sclerosis. Arterioscler. Thromb. Vasc. Biol. 2013, 33, e66–e74. [Google Scholar] [CrossRef]
- Steven, S.; Frenis, K.; Oelze, M.; Kalinovic, S.; Kuntic, M.; Bayo Jimenez, M.T.; Vujacic-Mirski, K.; Helmstädter, J.; Kröller-Schön, S.; Münzel, T.; et al. Vascular Inflammation and Oxidative Stress: Major Triggers for Cardiovascular Disease. Oxid. Med. Cell. Longev. 2019, 2019, 7092151. [Google Scholar] [CrossRef] [PubMed]
- Aboonabi, A.; Singh, I.; Rose’ Meyer, R. Cytoprotective Effects of Berry Anthocyanins against Induced Oxidative Stress and Inflammation in Primary Human Diabetic Aortic Endothelial Cells. Chem. Biol. Interact. 2020, 317, 108940. [Google Scholar] [CrossRef]
- Kostyunin, A.E.; Yuzhalin, A.E.; Ovcharenko, E.A.; Kutikhin, A.G. Development of Calcific Aortic Valve Disease: Do We Know Enough for New Clinical Trials? J. Mol. Cell. Cardiol. 2019, 132, 189–209. [Google Scholar] [CrossRef]
- Honda, S.; Miyamoto, T.; Watanabe, T.; Narumi, T.; Kadowaki, S.; Honda, Y.; Otaki, Y.; Hasegawa, H.; Netsu, S.; Funayama, A.; et al. A Novel Mouse Model of Aortic Valve Stenosis Induced by Direct Wire Injury. Arterioscler. Thromb. Vasc. Biol. 2014, 34, 270–278. [Google Scholar] [CrossRef] [PubMed]
- Heistad, D.D. Endothelial Function in the Time of the Giants. J. Cardiovasc. Pharmacol. 2008, 52, 385–392. [Google Scholar] [CrossRef]
- Butcher, J.T.; Tressel, S.; Johnson, T.; Turner, D.; Sorescu, G.; Jo, H.; Nerem, R.M. Transcriptional Profiles of Valvular and Vascular Endothelial Cells Reveal Phenotypic Differences: Influence of Shear Stress. Arterioscler. Thromb. Vasc. Biol. 2006, 26, 69–77. [Google Scholar] [CrossRef]
- Martínez-Martínez, E.; Souza-Neto, F.; Jiménez-González, S.; Cachofeiro, V. Oxidative Stress and Vascular Damage in the Context of Obesity: The Hidden Guest. Antioxidants 2021, 10, 406. [Google Scholar] [CrossRef] [PubMed]
- Gioscia-Ryan, R.A.; Clayton, Z.S.; Zigler, M.C.; Richey, J.J.; Cuevas, L.M.; Rossman, M.J.; Battson, M.L.; Ziemba, B.P.; Hutton, D.A.; VanDongen, N.S.; et al. Lifelong Voluntary Aerobic Exercise Prevents Age- and Western Diet- Induced Vascular Dysfunction, Mitochondrial Oxidative Stress and Inflammation in Mice. J. Physiol. 2021, 599, 911–925. [Google Scholar] [CrossRef] [PubMed]
- Simmons, C.A.; Grant, G.R.; Manduchi, E.; Davies, P.F. Spatial Heterogeneity of Endothelial Phenotypes Correlates with Side-Specific Vulnerability to Calcification in Normal Porcine Aortic Valves. Circ. Res. 2005, 96, 792–799. [Google Scholar] [CrossRef] [PubMed]
- Hulin, A.; Hego, A.; Lancellotti, P.; Oury, C. Advances in Pathophysiology of Calcific Aortic Valve Disease Propose Novel Molecular Therapeutic Targets. Front. Cardiovasc. Med. 2018, 5, 21. [Google Scholar] [CrossRef] [PubMed]
- Theodoris, C.V.; Li, M.; White, M.P.; Liu, L.; He, D.; Pollard, K.S.; Bruneau, B.G.; Srivastava, D. Human Disease Modeling Reveals Integrated Transcriptional and Epigenetic Mechanisms of NOTCH1 Haploinsufficiency. Cell 2015, 160, 1072–1086. [Google Scholar] [CrossRef]
- Hjortnaes, J.; Shapero, K.; Goettsch, C.; Hutcheson, J.D.; Keegan, J.; Kluin, J.; Mayer, J.E.; Bischoff, J.; Aikawa, E. Valvular Interstitial Cells Suppress Calcification of Valvular Endothelial Cells. Atherosclerosis 2015, 242, 251–260. [Google Scholar] [CrossRef]
- Liu, M.; Luo, M.; Sun, H.; Ni, B.; Shao, Y. Integrated Bioinformatics Analysis Predicts the Key Genes Involved in Aortic Valve Calcification: From Hemodynamic Changes to Extracellular Remodeling. Tohoku J. Exp. Med. 2017, 243, 263–273. [Google Scholar] [CrossRef]
- Bäck, M.; Gasser, T.C.; Michel, J.-B.; Caligiuri, G. Biomechanical Factors in the Biology of Aortic Wall and Aortic Valve Diseases. Cardiovasc. Res. 2013, 99, 232–241. [Google Scholar] [CrossRef]
- Wu, C.; Huang, R.-T.; Kuo, C.-H.; Kumar, S.; Kim, C.W.; Lin, Y.-C.; Chen, Y.-J.; Birukova, A.; Birukov, K.G.; Dulin, N.O.; et al. Mechanosensitive PPAP2B Regulates Endothelial Responses to Atherorelevant Hemodynamic Forces. Circ. Res. 2015, 117. [Google Scholar] [CrossRef]
- Li, W.; Wang, H.; Kuang, C.; Zhu, J.; Yu, Y.; Qin, Z.; Liu, J.; Huang, L. An Essential Role for the Id1/PI3K/Akt/NFkB/Survivin Signalling Pathway in Promoting the Proliferation of Endothelial Progenitor Cells In Vitro. Mol. Cell. Biochem. 2012, 363, 135–145. [Google Scholar] [CrossRef]
- López-Gil, J.F.; Tárraga-López, P.J. Research on Diet and Human Health. Int. J. Environ. Res. Public. Health 2022, 19, 6526. [Google Scholar] [CrossRef] [PubMed]
- Afshin, A.; Sur, P.J.; Fay, K.A.; Cornaby, L.; Ferrara, G.; Salama, J.S.; Mullany, E.C.; Abate, K.H.; Abbafati, C.; Abebe, Z.; et al. Health Effects of Dietary Risks in 195 Countries, 1990–2017: A Systematic Analysis for the Global Burden of Disease Study 2017. Lancet 2019, 393, 1958–1972. [Google Scholar] [CrossRef]
- Donato, A.J.; Machin, D.R.; Lesniewski, L.A. Mechanisms of Dysfunction in the Aging Vasculature and Role in Age-Related Disease. Circ. Res. 2018, 123, 825–848. [Google Scholar] [CrossRef] [PubMed]
- Greenberg, H.Z.E.; Zhao, G.; Shah, A.M.; Zhang, M. Role of Oxidative Stress in Calcific Aortic Valve Disease and Its Therapeutic Implications. Cardiovasc. Res. 2021, 118, 1433–1451. [Google Scholar] [CrossRef] [PubMed]
- Chen, Q.; Wang, Q.; Zhu, J.; Xiao, Q.; Zhang, L. Reactive Oxygen Species: Key Regulators in Vascular Health and Diseases: ROS in Vascular Diseases. Br. J. Pharmacol. 2018, 175, 1279–1292. [Google Scholar] [CrossRef]
- Sies, H. Oxidative Stress: Concept and Some Practical Aspects. Antioxidants 2020, 9, 852. [Google Scholar] [CrossRef]
- Shaito, A.; Aramouni, K.; Assaf, R.; Parenti, A.; Orekhov, A.; Yazbi, A.E.; Pintus, G.; Eid, A.H. Oxidative Stress-Induced Endothelial Dysfunction in Cardiovascular Diseases. Front. Biosci.-Landmark 2022, 27, 0105. [Google Scholar] [CrossRef]
- Mahmoudi, M.; Gormaz, J.G.; Erazo, M.; Howard, M.; Baeza, C.; Feelisch, M.; Curzen, N.; Olechowski, B.; Fernandez, B.; Minnion, M.; et al. Early Oxidative Stress Response in Patients with Severe Aortic Stenosis Undergoing Transcatheter and Surgical Aortic Valve Replacement: A Transatlantic Study. Oxid. Med. Cell. Longev. 2019, 2019, 1–8. [Google Scholar] [CrossRef]
- Poznyak, A.V.; Grechko, A.V.; Orekhova, V.A.; Chegodaev, Y.S.; Wu, W.-K.; Orekhov, A.N. Oxidative Stress and Antioxidants in Atherosclerosis Development and Treatment. Biology 2020, 9, 60. [Google Scholar] [CrossRef]
- Ma, Q.; He, X.; Virginia, W. Molecular Basis of Electrophilic and Oxidative Defense: Promises and Perils of Nrf2. Pharmacol. Rev. 2012, 64, 1055–1081. [Google Scholar] [CrossRef]
- Ma, Q. Role of Nrf2 in Oxidative Stress and Toxicity. Annu. Rev. Pharmacol. Toxicol. 2013, 53, 401–426. [Google Scholar] [CrossRef] [PubMed]
- Cui, L.; Zhou, Q.; Zheng, X.; Sun, B.; Zhao, S. Mitoquinone Attenuates Vascular Calcification by Suppressing Oxidative Stress and Reducing Apoptosis of Vascular Smooth Muscle Cells via the Keap1/Nrf2 Pathway. Free Radic. Biol. Med. 2020, 161, 23–31. [Google Scholar] [CrossRef] [PubMed]
- Balogh, E.; Chowdhury, A.; Ababneh, H.; Csiki, D.M.; Tóth, A.; Jeney, V. Heme-Mediated Activation of the Nrf2/HO-1 Axis Attenuates Calcification of Valve Interstitial Cells. Biomedicines 2021, 9, 427. [Google Scholar] [CrossRef] [PubMed]
- Heistad, D.D.; Wakisaka, Y.; Miller, J.; Chu, Y.; Pena-Silva, R. Novel Aspects of Oxidative Stress in Cardiovascular Diseases. Circ. J. 2009, 73, 201–207. [Google Scholar] [CrossRef] [PubMed]
- Reustle, A.; Torzewski, M. Role of P38 MAPK in Atherosclerosis and Aortic Valve Sclerosis. Int. J. Mol. Sci. 2018, 19, 3761. [Google Scholar] [CrossRef]
- Daiber, A.; Xia, N.; Steven, S.; Oelze, M.; Hanf, A.; Kröller-Schön, S.; Münzel, T.; Li, H. New Therapeutic Implications of Endothelial Nitric Oxide Synthase (ENOS) Function/Dysfunction in Cardiovascular Disease. Int. J. Mol. Sci. 2019, 20, 187. [Google Scholar] [CrossRef]
- Miller, J.D.; Chu, Y.; Brooks, R.M.; Richenbacher, W.E.; Peña-Silva, R.; Heistad, D.D. Dysregulation of Antioxidant Mechanisms Contributes to Increased Oxidative Stress in Calcific Aortic Valvular Stenosis in Humans. J. Am. Coll. Cardiol. 2008, 52, 843–850. [Google Scholar] [CrossRef]
- Rajamannan, N.M. Bicuspid Aortic Valve Disease: The Role of Oxidative Stress in Lrp5 Bone Formation. Cardiovasc. Pathol. 2011, 20, 168–176. [Google Scholar] [CrossRef]
- Valerio, V.; Myasoedova, V.A.; Moschetta, D.; Porro, B.; Perrucci, G.L.; Cavalca, V.; Cavallotti, L.; Songia, P.; Poggio, P. Impact of Oxidative Stress and Protein S-Glutathionylation in Aortic Valve Sclerosis Patients with Overt Atherosclerosis. J. Clin. Med. 2019, 8, 552. [Google Scholar] [CrossRef]
- Junco-Vicente, A.; del Río-García, Á.; Martín, M.; Rodríguez, I. Update in Biomolecular and Genetic Bases of Bicuspid Aortopathy. Int. J. Mol. Sci. 2021, 22, 5694. [Google Scholar] [CrossRef]
- Lo Presti, F.; Guzzardi, D.G.; Bancone, C.; Fedak, P.W.M.; Della Corte, A. The Science of BAV Aortopathy. Prog. Cardiovasc. Dis. 2020, 63, 465–474. [Google Scholar] [CrossRef] [PubMed]
- Mennander, A.A. Biochemistry: On Bicuspid Aortic Valve’s Aortic Service. J. Thorac. Cardiovasc. Surg. 2017, 154, 1763. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Portelli, S.S.; Hambly, B.D.; Jeremy, R.W.; Robertson, E.N. Oxidative Stress in Genetically Triggered Thoracic Aortic Aneurysm: Role in Pathogenesis and Therapeutic Opportunities. Redox Rep. 2021, 26, 45–52. [Google Scholar] [CrossRef] [PubMed]
- Skeffington, K.; Bond, A.; Bigotti, M.G.; Abdulghani, S.; Iacobazzi, D.; Kang, S.-L.; Heesom, K.; Wilson, M.; Stoica, S.; Martin, R.; et al. Changes in Inflammation and Oxidative Stress Signalling Pathways in Coarcted Aorta Triggered by Bicuspid Aortic Valve and Growth in Young Children. Exp. Ther. Med. 2020, 20, 1. [Google Scholar] [CrossRef]
- Billaud, M.; Phillippi, J.A.; Kotlarczyk, M.P.; Hill, J.C.; Ellis, B.W.; St Croix, C.M.; Cantu-Medéllin, N.; Kelley, E.E.; Gleason, T.G. Elevated Oxidative Stress in the Aortic Media of Patients with Bicuspid Aortic Valve. J. Thorac. Cardiovasc. Surg. 2017, 154, 1756–1762. [Google Scholar] [CrossRef]
- Phillippi, J.A.; Klyachko, E.A.; Kenny, J.P.; Eskay, M.A.; Gorman, R.C.; Gleason, T.G. Basal and Oxidative Stress–Induced Expression of Metallothionein Is Decreased in Ascending Aortic Aneurysms of Bicuspid Aortic Valve Patients. Circulation 2009, 119, 2498–2506. [Google Scholar] [CrossRef]
- Phillippi, J.A.; Eskay, M.A.; Kubala, A.A.; Pitt, B.R.; Gleason, T.G. Altered Oxidative Stress Responses and Increased Type I Collagen Expression in Bicuspid Aortic Valve Patients. Ann. Thorac. Surg. 2010, 90, 1893–1898. [Google Scholar] [CrossRef]
- Förstermann, U.; Xia, N.; Li, H. Roles of Vascular Oxidative Stress and Nitric Oxide in the Pathogenesis of Atherosclerosis. Circ. Res. 2017, 120, 713–735. [Google Scholar] [CrossRef]
- Chu, Y.; Lund, D.D.; Weiss, R.M.; Brooks, R.M.; Doshi, H.; Hajj, G.P.; Sigmund, C.D.; Heistad, D.D. Pioglitazone Attenuates Valvular Calcification Induced by Hypercholesterolemia. Arterioscler. Thromb. Vasc. Biol. 2013, 33, 523–532. [Google Scholar] [CrossRef]
- Liu, H.; Wang, L.; Pan, Y.; Wang, X.; Ding, Y.; Zhou, C.; Shah, A.M.; Zhao, G.; Zhang, M. Celastrol Alleviates Aortic Valve Calcification Via Inhibition of NADPH Oxidase 2 in Valvular Interstitial Cells. JACC Basic Transl. Sci. 2019, 5, 35–49. [Google Scholar] [CrossRef]
- Liberman, M.; Bassi, E.; Martinatti, M.K.; Lario, F.C.; Wosniak, J.; Pomerantzeff, P.M.A.; Laurindo, F.R.M. Oxidant Generation Predominates Around Calcifying Foci and Enhances Progression of Aortic Valve Calcification. Arterioscler. Thromb. Vasc. Biol. 2008, 28, 463–470. [Google Scholar] [CrossRef] [PubMed]
- Canugovi, C.; Stevenson, M.D.; Vendrov, A.E.; Hayami, T.; Robidoux, J.; Xiao, H.; Zhang, Y.-Y.; Eitzman, D.T.; Runge, M.S.; Madamanchi, N.R. Increased Mitochondrial NADPH Oxidase 4 (NOX4) Expression in Aging Is a Causative Factor in Aortic Stiffening. Redox Biol. 2019, 26, 101288. [Google Scholar] [CrossRef] [PubMed]
- Tung, M.-C.; Lan, Y.-W.; Li, H.-H.; Chen, H.-L.; Chen, S.-Y.; Chen, Y.-H.; Lin, C.-C.; Tu, M.-Y.; Chen, C.-M. Kefir Peptides Alleviate High-Fat Diet-Induced Atherosclerosis by Attenuating Macrophage Accumulation and Oxidative Stress in ApoE Knockout Mice. Sci. Rep. 2020, 10, 8802. [Google Scholar] [CrossRef] [PubMed]
- Matsushima, S.; Kuroda, J.; Ago, T.; Zhai, P.; Park, J.Y.; Xie, L.-H.; Tian, B.; Sadoshima, J. Increased Oxidative Stress in the Nucleus Caused by Nox4 Mediates Oxidation of HDAC4 and Cardiac Hypertrophy. Circ. Res. 2013, 112, 651–663. [Google Scholar] [CrossRef]
- Wang, Z.; Zhang, J.; Chen, L.; Li, J.; Zhang, H.; Guo, X. Glycine Suppresses AGE/RAGE Signaling Pathway and Subsequent Oxidative Stress by Restoring Glo1 Function in the Aorta of Diabetic Rats and in HUVECs. Oxid. Med. Cell. Longev. 2019, 2019, 4628962. [Google Scholar] [CrossRef]
- Zhou, B.; Qiu, Y.; Wu, N.; Chen, A.-D.; Zhou, H.; Chen, Q.; Kang, Y.-M.; Li, Y.-H.; Zhu, G.-Q. FNDC5 Attenuates Oxidative Stress and NLRP3 Inflammasome Activation in Vascular Smooth Muscle Cells via Activating the AMPK-SIRT1 Signal Pathway. Oxid. Med. Cell. Longev. 2020, 2020, 6384803. [Google Scholar] [CrossRef]
- Doroszko, A.; Radziwon-Balicka, A.; Skomro, R. Novel Approaches for Diagnosing and Management of Cardiovascular Disorders Mediated by Oxidative Stress. Oxid. Med. Cell. Longev. 2020, 2020, 7096727. [Google Scholar] [CrossRef]
- Plunde, O.; Bäck, M. Fatty Acids and Aortic Valve Stenosis. Kardiol. Pol. 2021, 79, 614–621. [Google Scholar] [CrossRef]
- Di Vito, A.; Donato, A.; Presta, I.; Mancuso, T.; Brunetti, F.S.; Mastroroberto, P.; Amorosi, A.; Malara, N.; Donato, G. Extracellular Matrix in Calcific Aortic Valve Disease: Architecture, Dynamic and Perspectives. Int. J. Mol. Sci. 2021, 22, 913. [Google Scholar] [CrossRef]
- Kopytek, M.; Mazur, P.; Ząbczyk, M.; Undas, A.; Natorska, J. Diabetes Concomitant to Aortic Stenosis Is Associated with Increased Expression of NF-ΚB and More Pronounced Valve Calcification. Diabetologia 2021, 64, 2562–2574. [Google Scholar] [CrossRef]
- Izzo, C.; Vitillo, P.; Di Pietro, P.; Visco, V.; Strianese, A.; Virtuoso, N.; Ciccarelli, M.; Galasso, G.; Carrizzo, A.; Vecchione, C. The Role of Oxidative Stress in Cardiovascular Aging and Cardiovascular Diseases. Life 2021, 11, 60. [Google Scholar] [CrossRef] [PubMed]
- Mathieu, P.; Bouchareb, R.; Boulanger, M.-C. Innate and Adaptive Immunity in Calcific Aortic Valve Disease. J. Immunol. Res. 2015, 2015, 851945. [Google Scholar] [CrossRef] [PubMed]
- Matilla, L.; Ibarrola, J.; Arrieta, V.; Garcia-Peña, A.; Martinez-Martinez, E.; Sádaba, R.; Alvarez, V.; Navarro, A.; Fernández-Celis, A.; Gainza, A.; et al. Soluble ST2 Promotes Oxidative Stress and Inflammation in Cardiac Fibroblasts: An In Vitro and In Vivo Study in Aortic Stenosis. Clin. Sci. 2019, 133, 1537–1548. [Google Scholar] [CrossRef] [PubMed]
- Kapelouzou, A.; Tsourelis, L.; Kaklamanis, L.; Degiannis, D.; Kogerakis, N.; Cokkinos, D.V. Serum and Tissue Biomarkers in Aortic Stenosis. Glob. Cardiol. Sci. Pract. 2015, 2015, 49. [Google Scholar] [CrossRef]
- Goh, K.Y.; He, L.; Song, J.; Jinno, M.; Rogers, A.J.; Sethu, P.; Halade, G.V.; Rajasekaran, N.S.; Liu, X.; Prabhu, S.D.; et al. Mitoquinone Ameliorates Pressure Overload-Induced Cardiac Fibrosis and Left Ventricular Dysfunction in Mice. Redox Biol. 2019, 21, 101100. [Google Scholar] [CrossRef]
- Patel, V.B.; Zhong, J.-C.; Fan, D.; Basu, R.; Morton, J.S.; Parajuli, N.; McMurtry, M.S.; Davidge, S.T.; Kassiri, Z.; Oudit, G.Y. Angiotensin-Converting Enzyme 2 Is a Critical Determinant of Angiotensin II–Induced Loss of Vascular Smooth Muscle Cells and Adverse Vascular Remodeling. Hypertension 2014, 64, 157–164. [Google Scholar] [CrossRef]
- Chiang, Y.-F.; Tsai, C.-H.; Chen, H.-Y.; Wang, K.-L.; Chang, H.-Y.; Huang, Y.-J.; Hong, Y.-H.; Ali, M.; Shieh, T.-M.; Huang, T.-C.; et al. Protective Effects of Fucoxanthin on Hydrogen Peroxide-Induced Calcification of Heart Valve Interstitial Cells. Mar. Drugs 2021, 19, 307. [Google Scholar] [CrossRef]
- Henderson, B.C.; Tyagi, N.; Ovechkin, A.; Kartha, G.K.; Moshal, K.S.; Tyagi, S.C. Oxidative Remodeling in Pressure Overload Induced Chronic Heart Failure. Eur. J. Heart Fail. 2007, 9, 450–457. [Google Scholar] [CrossRef]
- Xin, M.; Jin, X.; Cui, X.; Jin, C.; Piao, L.; Wan, Y.; Xu, S.; Zhang, S.; Yue, X.; Wang, H.; et al. Dipeptidyl Peptidase-4 Inhibition Prevents Vascular Aging in Mice under Chronic Stress: Modulation of Oxidative Stress and Inflammation. Chem. Biol. Interact. 2019, 314, 108842. [Google Scholar] [CrossRef]
- Jones, J.A. Oxidative Stress in Bicuspid Aortic Valve-Related Aortopathy: Hand-Me-Downs and Yoga Pants. J. Thorac. Cardiovasc. Surg. 2017, 154, 1764–1765. [Google Scholar] [CrossRef]
- Nachar, W.; Merlet, N.; Maafi, F.; Shi, Y.; Mihalache-Avram, T.; Mecteau, M.; Ferron, M.; Rhéaume, E.; Tardif, J.-C. Cardiac Inflammation and Diastolic Dysfunction in Hypercholesterolemic Rabbits. PLoS ONE 2019, 14, e0220707. [Google Scholar] [CrossRef] [PubMed]
- Navaratnarajah, A.; Bhan, A.; Alcock, E.; Dew, T.; Monaghan, M.; Shah, A.M.; Wendler, O.; MacCarthy, P.; Dworakowski, R. Systemic Inflammation and Oxidative Stress Contribute to Acute Kidney Injury after Transcatheter Aortic Valve Implantation. Cardiol. J. 2020. [Google Scholar] [CrossRef] [PubMed]
- Heldmaier, K.; Stoppe, C.; Goetzenich, A.; Foldenauer, A.-C.; Zayat, R.; Breuer, T.; Schälte, G. Oxidation-Reduction Potential in Patients Undergoing Transcatheter or Surgical Aortic Valve Replacement. BioMed Res. Int. 2018, 2018, 8469383. [Google Scholar] [CrossRef] [PubMed]
- Komosa, A.; Perek, B.; Rzymski, P.; Lesiak, M.; Siller-Matula, J.M.; Grygier, M.; Puślecki, M.; Misterski, M.; Olasińska-Wiśniewska, A.; Ropacka-Lesiak, M.; et al. Transcatheter Aortic Valve Replacement Is Associated with Less Oxidative Stress and Faster Recovery of Antioxidant Capacity than Surgical Aortic Valve Replacement. J. Clin. Med. 2019, 8, 1364. [Google Scholar] [CrossRef] [PubMed]
- Marchandot, B.; Kibler, M.; Charles, A.L.; Trinh, A.; Petit Eisenmann, H.; Zeyons, F.; Von Hunolstein, J.J.; Reydel, A.; Matsushita, K.; Kindo, M.; et al. Does Transcatheter Aortic Valve Replacement Modulate the Kinetic of Superoxide Anion Generation? Antioxid. Redox Signal. 2019, 31, 420–426. [Google Scholar] [CrossRef]
- Marquis-Gravel, G.; Redfors, B.; Leon, M.B.; Généreux, P. Medical Treatment of Aortic Stenosis. Circulation 2016, 134, 1766–1784. [Google Scholar] [CrossRef]
- Towler, D.A. Molecular and Cellular Aspects of Calcific Aortic Valve Disease. Circ. Res. 2013, 113, 198–208. [Google Scholar] [CrossRef]
- Pedriali, G.; Morciano, G.; Patergnani, S.; Cimaglia, P.; Morelli, C.; Mikus, E.; Ferrari, R.; Gasbarro, V.; Giorgi, C.; Wieckowski, M.R.; et al. Aortic Valve Stenosis and Mitochondrial Dysfunctions: Clinical and Molecular Perspectives. Int. J. Mol. Sci. 2020, 21, 4899. [Google Scholar] [CrossRef]
- Zhong, S.; Li, L.; Shen, X.; Li, Q.; Xu, W.; Wang, X.; Tao, Y.; Yin, H. An Update on Lipid Oxidation and Inflammation in Cardiovascular Diseases. Free Radic. Biol. Med. 2019, 144, 266–278. [Google Scholar] [CrossRef]
- Miller, J.D.; Weiss, R.M.; Serrano, K.M.; Brooks, R.M.; Berry, C.J.; Zimmerman, K.; Young, S.G.; Heistad, D.D. Lowering Plasma Cholesterol Levels Halts Progression of Aortic Valve Disease in Mice. Circulation 2009, 119, 2693–2701. [Google Scholar] [CrossRef]
- Capoulade, R.; Chan, K.L.; Yeang, C.; Mathieu, P.; Bossé, Y.; Dumesnil, J.G.; Tam, J.W.; Teo, K.K.; Mahmut, A.; Yang, X.; et al. Oxidized Phospholipids, Lipoprotein(a), and Progression of Calcific Aortic Valve Stenosis. J. Am. Coll. Cardiol. 2015, 66, 1236–1246. [Google Scholar] [CrossRef] [PubMed]
- Moura, L.M.; Ramos, S.F.; Zamorano, J.L.; Barros, I.M.; Azevedo, L.F.; Rocha-Gonçalves, F.; Rajamannan, N.M. Rosuvastatin Affecting Aortic Valve Endothelium to Slow the Progression of Aortic Stenosis. J. Am. Coll. Cardiol. 2007, 49, 554–561. [Google Scholar] [CrossRef] [PubMed]
- Cowell, S.J.; Newby, D.E.; Prescott, R.J.; Bloomfield, P.; Reid, J.; Northridge, D.B.; Boon, N.A. A Randomized Trial of Intensive Lipid-Lowering Therapy in Calcific Aortic Stenosis. N. Engl. J. Med. 2005, 352, 2389–2397. [Google Scholar] [CrossRef] [PubMed]
- Rossebø, A.B.; Pedersen, T.R.; Boman, K.; Brudi, P.; Chambers, J.B.; Egstrup, K.; Gerdts, E.; Gohlke-Bärwolf, C.; Holme, I.; Kesäniemi, Y.A.; et al. Intensive Lipid Lowering with Simvastatin and Ezetimibe in Aortic Stenosis. N. Engl. J. Med. 2008, 359, 1343–1356. [Google Scholar] [CrossRef] [PubMed]
- Capoulade, R.; Yeang, C.; Chan, K.L.; Pibarot, P.; Tsimikas, S. Association of Mild to Moderate Aortic Valve Stenosis Progression With Higher Lipoprotein(a) and Oxidized Phospholipid Levels: Secondary Analysis of a Randomized Clinical Trial. JAMA Cardiol. 2018, 3, 1212. [Google Scholar] [CrossRef] [PubMed]
- Tsimikas, S.; Karwatowska-Prokopczuk, E.; Gouni-Berthold, I.; Tardif, J.-C.; Baum, S.J.; Steinhagen-Thiessen, E.; Shapiro, M.D.; Stroes, E.S.; Moriarty, P.M.; Nordestgaard, B.G.; et al. Lipoprotein(a) Reduction in Persons with Cardiovascular Disease. N. Engl. J. Med. 2020, 382, 244–255. [Google Scholar] [CrossRef]
- Yeang, C.; Hung, M.-Y.; Byun, Y.-S.; Clopton, P.; Yang, X.; Witztum, J.L.; Tsimikas, S. Effect of Therapeutic Interventions on Oxidized Phospholipids on Apolipoprotein B100 and Lipoprotein(a). J. Clin. Lipidol. 2016, 10, 594–603. [Google Scholar] [CrossRef]
- Hu, J.; Lei, H.; Liu, L.; Xu, D. Lipoprotein(a), a Lethal Player in Calcific Aortic Valve Disease. Front. Cell Dev. Biol. 2022, 10, 812368. [Google Scholar] [CrossRef]
- Guddeti, R.R.; Patil, S.; Ahmed, A.; Sharma, A.; Aboeata, A.; Lavie, C.J.; Alla, V.M. Lipoprotein(a) and Calcific Aortic Valve Stenosis: A Systematic Review. Prog. Cardiovasc. Dis. 2020, 63, 496–502. [Google Scholar] [CrossRef]
- Ljungberg, J.; Holmgren, A.; Bergdahl, I.A.; Hultdin, J.; Norberg, M.; Näslund, U.; Johansson, B.; Söderberg, S. Lipoprotein(a) and the Apolipoprotein B/A1 Ratio Independently Associate with Surgery for Aortic Stenosis Only in Patients With Concomitant Coronary Artery Disease. J. Am. Heart Assoc. 2017, 6, e007160. [Google Scholar] [CrossRef]
- Ma, G.S.; Wilkinson, M.J.; Reeves, R.R.; Yeang, C.; DeMaria, A.N.; Cotter, B.; Patel, M.; Mahmud, E.; Tsimikas, S. Lipoprotein(a) in Patients Undergoing Transcatheter Aortic Valve Replacement. Angiology 2019, 70, 332–336. [Google Scholar] [CrossRef] [PubMed]
- Pérez de Isla, L.; Watts, G.F.; Alonso, R.; Díaz-Díaz, J.L.; Muñiz-Grijalvo, O.; Zambón, D.; Fuentes, F.; de Andrés, R.; Padró, T.; López-Miranda, J.; et al. Lipoprotein(a), LDL-Cholesterol, and Hypertension: Predictors of the Need for Aortic Valve Replacement in Familial Hypercholesterolaemia. Eur. Heart J. 2021, 42, 2201–2211. [Google Scholar] [CrossRef] [PubMed]
- Capoulade, R.; Torzewski, M.; Mayr, M.; Chan, K.-L.; Mathieu, P.; Bossé, Y.; Dumesnil, J.G.; Tam, J.; Teo, K.K.; Burnap, S.A.; et al. ApoCIII-Lp(a) Complexes in Conjunction with Lp(a)-OxPL Predict Rapid Progression of Aortic Stenosis. Heart 2020, 106, 738–745. [Google Scholar] [CrossRef] [PubMed]
- Vongpromek, R.; Bos, S.; ten Kate, G.-J.R.; Yahya, R.; Verhoeven, A.J.M.; de Feyter, P.J.; Kronenberg, F.; Roeters van Lennep, J.E.; Sijbrands, E.J.G.; Mulder, M.T. Lipoprotein(a) Levels Are Associated with Aortic Valve Calcification in Asymptomatic Patients with Familial Hypercholesterolaemia. J. Intern. Med. 2015, 278, 166–173. [Google Scholar] [CrossRef]
- Sabatine, M.S.; Giugliano, R.P.; Wiviott, S.D.; Raal, F.J.; Blom, D.J.; Robinson, J.; Ballantyne, C.M.; Somaratne, R.; Legg, J.; Wasserman, S.M.; et al. Efficacy and Safety of Evolocumab in Reducing Lipids and Cardiovascular Events. N. Engl. J. Med. 2015, 372, 1500–1509. [Google Scholar] [CrossRef]
- O’Donoghue, M.L.; Fazio, S.; Giugliano, R.P.; Stroes, E.S.G.; Kanevsky, E.; Gouni-Berthold, I.; Im, K.; Lira Pineda, A.; Wasserman, S.M.; Češka, R.; et al. Lipoprotein(a), PCSK9 Inhibition, and Cardiovascular Risk: Insights from the FOURIER Trial. Circulation 2019, 139, 1483–1492. [Google Scholar] [CrossRef]
- Bergmark, B.A.; O’Donoghue, M.L.; Murphy, S.A.; Kuder, J.F.; Ezhov, M.V.; Ceška, R.; Gouni-Berthold, I.; Jensen, H.K.; Tokgozoglu, S.L.; Mach, F.; et al. An Exploratory Analysis of Proprotein Convertase Subtilisin/Kexin Type 9 Inhibition and Aortic Stenosis in the FOURIER Trial. JAMA Cardiol. 2020, 5, 709. [Google Scholar] [CrossRef]
- Nicholls, S.J.; Puri, R.; Anderson, T.; Ballantyne, C.M.; Cho, L.; Kastelein, J.J.P.; Koenig, W.; Somaratne, R.; Kassahun, H.; Yang, J.; et al. Effect of Evolocumab on Progression of Coronary Disease in Statin-Treated Patients: The GLAGOV Randomized Clinical Trial. JAMA 2016, 316, 2373. [Google Scholar] [CrossRef]
- van Capelleveen, J.C.; van der Valk, F.M.; Stroes, E.S.G. Current Therapies for Lowering Lipoprotein (a). J. Lipid Res. 2016, 57, 1612–1618. [Google Scholar] [CrossRef]
- Viney, N.J.; van Capelleveen, J.C.; Geary, R.S.; Xia, S.; Tami, J.A.; Yu, R.Z.; Marcovina, S.M.; Hughes, S.G.; Graham, M.J.; Crooke, R.M.; et al. Antisense Oligonucleotides Targeting Apolipoprotein(a) in People with Raised Lipoprotein(a): Two Randomised, Double-Blind, Placebo-Controlled, Dose-Ranging Trials. Lancet 2016, 388, 2239–2253. [Google Scholar] [CrossRef]
- Que, X.; Hung, M.-Y.; Yeang, C.; Gonen, A.; Prohaska, T.A.; Sun, X.; Diehl, C.; Määttä, A.; Gaddis, D.E.; Bowden, K.; et al. Oxidized Phospholipids Are Proinflammatory and Proatherogenic in Hypercholesterolaemic Mice. Nature 2018, 558, 301–306. [Google Scholar] [CrossRef] [PubMed]
- Lin, C.P.; Huang, P.-H.; Chen, C.-Y.; Chen, J.-S.; Chen, J.-W.; Lin, S.-J. Dipeptidyl Peptidase-4 Inhibitor Sitagliptin Attenuates Arterial Calcification By Downregulating Oxidative Stress-Induced Receptor for Advanced Glycation End Products in Low-Density Lipoprotein Receptor Knockout Mice. Res. Square. 2021. [Google Scholar] [CrossRef]
- Yang, S.; Liu, L.; Meng, L.; Hu, X. Capsaicin Is Beneficial to Hyperlipidemia, Oxidative Stress, Endothelial Dysfunction, and Atherosclerosis in Guinea Pigs Fed on a High-Fat Diet. Chem. Biol. Interact. 2019, 297, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Adhikari, R.; Shiwakoti, S.; Ko, J.-Y.; Dhakal, B.; Park, S.-H.; Choi, I.J.; Kim, H.J.; Oak, M.-H. Oxidative Stress in Calcific Aortic Valve Stenosis: Protective Role of Natural Antioxidants. Antioxidants 2022, 11, 1169. [Google Scholar] [CrossRef] [PubMed]
- Chao, C.-T.; Yeh, H.-Y.; Tsai, Y.-T.; Chuang, P.-H.; Yuan, T.-H.; Huang, J.-W.; Chen, H.-W. Natural and Non-Natural Antioxidative Compounds: Potential Candidates for Treatment of Vascular Calcification. Cell Death Discov. 2019, 5, 145. [Google Scholar] [CrossRef] [PubMed]
- Hammad, S.K.; Eissa, R.G.; Shaheen, M.A.; Younis, N.N. Resveratrol Ameliorates Aortic Calcification in Ovariectomized Rats via SIRT1 Signaling. Curr. Issues Mol. Biol. 2021, 43, 1057–1071. [Google Scholar] [CrossRef]
- Zhang, P.; Li, Y.; Du, Y.; Li, G.; Wang, L.; Zhou, F. Resveratrol Ameliorated Vascular Calcification by Regulating Sirt-1 and Nrf2. Transplant. Proc. 2016, 48, 3378–3386. [Google Scholar] [CrossRef]
- Tomayko, E.J.; Cachia, A.J.; Chung, H.R.; Wilund, K.R. Resveratrol Supplementation Reduces Aortic Atherosclerosis and Calcification and Attenuates Loss of Aerobic Capacity in a Mouse Model of Uremia. J. Med. Food 2014, 17, 278–283. [Google Scholar] [CrossRef]
- Gugliandolo, E.; Fusco, R.; Biundo, F.; D’Amico, R.; Benedetto, F.; Di Paola, R.; Cuzzocrea, S. Palmitoylethanolamide and Polydatin Combination Reduces Inflammation and Oxidative Stress in Vascular Injury. Pharmacol. Res. 2017, 123, 83–92. [Google Scholar] [CrossRef]
- Xia, N.; Daiber, A.; Förstermann, U.; Li, H. Antioxidant Effects of Resveratrol in the Cardiovascular System: Antioxidant Effects of Resveratrol. Br. J. Pharmacol. 2017, 174, 1633–1646. [Google Scholar] [CrossRef]
- Jin, Y.; Wang, Y.; Weng, Y.; Li, X.; Huang, Q.; Liu, Y.; Xiang, Y.; Li, X.; Jiang, P.; He, W.; et al. Resveratrol Exhibits Inhibition Effects on Osteogenic Differentiation of Aortic Valve Interstitial Cells by Interfering with the AKT Pathway. J. Funct. Foods 2022, 91, 105002. [Google Scholar] [CrossRef]
- van Andel, M.; Groenink, M.; Zwinderman, A.; Mulder, B.; de Waard, V. The Potential Beneficial Effects of Resveratrol on Cardiovascular Complications in Marfan Syndrome Patients–Insights from Rodent-Based Animal Studies. Int. J. Mol. Sci. 2019, 20, 1122. [Google Scholar] [CrossRef] [PubMed]
- Assis, R.; Arcaro, C.; Gutierres, V.; Oliveira, J.; Costa, P.; Baviera, A.; Brunetti, I. Combined Effects of Curcumin and Lycopene or Bixin in Yoghurt on Inhibition of LDL Oxidation and Increases in HDL and Paraoxonase Levels in Streptozotocin-Diabetic Rats. Int. J. Mol. Sci. 2017, 18, 332. [Google Scholar] [CrossRef]
- Hewlings, S.; Kalman, D. Curcumin: A Review of Its Effects on Human Health. Foods 2017, 6, 92. [Google Scholar] [CrossRef] [PubMed]
- Qin, S.; Huang, L.; Gong, J.; Shen, S.; Huang, J.; Ren, H.; Hu, H. Efficacy and Safety of Turmeric and Curcumin in Lowering Blood Lipid Levels in Patients with Cardiovascular Risk Factors: A Meta-Analysis of Randomized Controlled Trials. Nutr. J. 2017, 16, 68. [Google Scholar] [CrossRef] [PubMed]
- Zhang, S.; Zou, J.; Li, P.; Zheng, X.; Feng, D. Curcumin Protects against Atherosclerosis in Apolipoprotein E-Knockout Mice by Inhibiting Toll-like Receptor 4 Expression. J. Agric. Food Chem. 2018, 66, 449–456. [Google Scholar] [CrossRef] [PubMed]
- Zhou, T.; Wang, Y.; Liu, M.; Huang, Y.; Shi, J.; Dong, N.; Xu, K. Curcumin Inhibits Calcification of Human Aortic Valve Interstitial Cells by Interfering NF-κB, AKT, and ERK Pathways. Phyther. Res. 2020, 34, 2074–2081. [Google Scholar] [CrossRef]
- Lin, K.; Chen, H.; Chen, X.; Qian, J.; Huang, S.; Huang, W. Efficacy of Curcumin on Aortic Atherosclerosis: A Systematic Review and Meta-Analysis in Mouse Studies and Insights into Possible Mechanisms. Oxid. Med. Cell. Longev. 2020, 2020, 1520747. [Google Scholar] [CrossRef]
- Daimary, U.D.; Parama, D.; Rana, V.; Banik, K.; Kumar, A.; Harsha, C.; Kunnumakkara, A.B. Emerging Roles of Cardamonin, a Multitargeted Nutraceutical in the Prevention and Treatment of Chronic Diseases. Curr. Res. Pharmacol. Drug Discov. 2021, 2, 100008. [Google Scholar] [CrossRef]
- Gonçalves, L.M.; Valente, I.M.; Rodrigues, J.A. An Overview on Cardamonin. J. Med. Food 2014, 17, 633–640. [Google Scholar] [CrossRef]
- Wang, C.; Xia, Y.; Qu, L.; Liu, Y.; Liu, X.; Xu, K. Cardamonin Inhibits Osteogenic Differentiation of Human Valve Interstitial Cells and Ameliorates Aortic Valve Calcification via Interfering NF-ΚB/NLRP3 Inflammasome Pathway. Food Funct. 2021. [Google Scholar] [CrossRef] [PubMed]
- Sharifi-Rad, J.; Quispe, C.; Castillo, C.M.S.; Caroca, R.; Lazo-Vélez, M.A.; Antonyak, H.; Polishchuk, A.; Lysiuk, R.; Oliinyk, P.; De Masi, L.; et al. Ellagic Acid: A Review on Its Natural Sources, Chemical Stability, and Therapeutic Potential. Oxid. Med. Cell. Longev. 2022, 2022, 3848084. [Google Scholar] [CrossRef] [PubMed]
- Jordão, J.; Porto, H.; Lopes, F.; Batista, A.; Rocha, M. Protective Effects of Ellagic Acid on Cardiovascular Injuries Caused by Hypertension in Rats. Planta Med. 2017, 83, 830–836. [Google Scholar] [CrossRef] [PubMed]
- Kee, H.J.; Cho, S.-N.; Kim, G.R.; Choi, S.Y.; Ryu, Y.; Kim, I.K.; Hong, Y.J.; Park, H.W.; Ahn, Y.; Cho, J.G.; et al. Gallic Acid Inhibits Vascular Calcification through the Blockade of BMP2–Smad1/5/8 Signaling Pathway. Vascul. Pharmacol. 2014, 63, 71–78. [Google Scholar] [CrossRef] [PubMed]
- Liu, M.; Li, F.; Huang, Y.; Zhou, T.; Chen, S.; Li, G.; Shi, J.; Dong, N.; Xu, K. Caffeic Acid Phenethyl Ester Ameliorates Calcification by Inhibiting Activation of the AKT/NF-ΚB/NLRP3 Inflammasome Pathway in Human Aortic Valve Interstitial Cells. Front. Pharmacol. 2020, 11, 826. [Google Scholar] [CrossRef]
- Nie, J.; Chang, Y.; Li, Y.; Zhou, Y.; Qin, J.; Sun, Z.; Li, H. Caffeic Acid Phenethyl Ester (Propolis Extract) Ameliorates Insulin Resistance by Inhibiting JNK and NF-ΚB Inflammatory Pathways in Diabetic Mice and HepG2 Cell Models. J. Agric. Food Chem. 2017, 65, 9041–9053. [Google Scholar] [CrossRef]
- Huang, H.; Li, L.; Shi, W.; Liu, H.; Yang, J.; Yuan, X.; Wu, L. The Multifunctional Effects of Nobiletin and Its Metabolites In Vivo and In Vitro. Evid. Based Complement. Alternat. Med. 2016, 2016, 2918796. [Google Scholar] [CrossRef]
- Xu, K.; Huang, Y.; Zhou, T.; Wang, C.; Chi, Q.; Shi, J.; Zhu, P.; Dong, N. Nobiletin Exhibits Potent Inhibition on Tumor Necrosis Factor Alpha-induced Calcification of Human Aortic Valve Interstitial Cells via Targeting ABCG2 and AKR1B1. Phytother. Res. 2019, 33, 1717–1725. [Google Scholar] [CrossRef]
- Kaneda, H.; Otomo, R.; Sasaki, N.; Omi, T.; Sato, T.; Kaneda, T. Endothelium-Independent Vasodilator Effects of Nobiletin in Rat Aorta. J. Pharmacol. Sci. 2019, 140, 48–53. [Google Scholar] [CrossRef]
- Andres, S.; Pevny, S.; Ziegenhagen, R.; Bakhiya, N.; Schäfer, B.; Hirsch-Ernst, K.I.; Lampen, A. Safety Aspects of the Use of Quercetin as a Dietary Supplement. Mol. Nutr. Food Res. 2018, 62, 1700447. [Google Scholar] [CrossRef]
- Chang, X.; Cui, L.; Wang, X.; Zhang, L.; Zhu, D.; Zhou, X.; Hao, L. Quercetin Attenuates Vascular Calcification through Suppressed Oxidative Stress in Adenine-Induced Chronic Renal Failure Rats. BioMed Res. Int. 2017, 2017, 5716204. [Google Scholar] [CrossRef] [PubMed]
- Bhaskar, S.; Sudhakaran, P.R.; Helen, A. Quercetin Attenuates Atherosclerotic Inflammation and Adhesion Molecule Expression by Modulating TLR-NF-ΚB Signaling Pathway. Cell. Immunol. 2016, 310, 131–140. [Google Scholar] [CrossRef] [PubMed]
- Cao, H.; Jia, Q.; Shen, D.; Yan, L.; Chen, C.; Xing, S. Quercetin Has a Protective Effect on Atherosclerosis via Enhancement of Autophagy in ApoE-/- Mice. Exp. Ther. Med. 2019, 18, 2451–2458. [Google Scholar] [CrossRef] [PubMed]
- Hu, W.; Wang, W.; Ma, Q.; Liu, T.; Zhang, J.; Zhang, J. Blueberry Anthocyanin-enriched Extract Ameliorates Transverse Aortic Constriction-induced Myocardial Dysfunction via the DDAH1/ADMA/NO Signaling Pathway in Mice. Mol. Med. Rep. 2019, 21, 454–462. [Google Scholar] [CrossRef] [PubMed]
- Liu, H.; Zhang, X.; Zhong, X.; Li, Z.; Cai, S.; Yang, P.; Ou, C.; Chen, M. Puerarin Inhibits Vascular Calcification of Uremic Rats. Eur. J. Pharmacol. 2019, 855, 235–243. [Google Scholar] [CrossRef]
- Jiang, Z.; Cui, X.; Qu, P.; Shang, C.; Xiang, M.; Wang, J. Roles and Mechanisms of Puerarin on Cardiovascular Disease: A Review. Biomed. Pharmacother. 2022, 147, 112655. [Google Scholar] [CrossRef]
- Cai, S.-A.; Hou, N.; Zhao, G.-J.; Liu, X.-W.; He, Y.-Y.; Liu, H.-L.; Hua, Y.-Q.; Li, L.-R.; Huang, Y.; Ou, C.-W.; et al. Nrf2 Is a Key Regulator on Puerarin Preventing Cardiac Fibrosis and Upregulating Metabolic Enzymes UGT1A1 in Rats. Front. Pharmacol. 2018, 9, 540. [Google Scholar] [CrossRef]
- Hu, Y.; Li, H.; Li, R.; Wu, Z.; Yang, W.; Qu, W. Puerarin Protects Vascular Smooth Muscle Cells from Oxidized Low-density Lipoprotein-induced Reductions in Viability via Inhibition of the P38 MAPK and JNK Signaling Pathways. Exp. Ther. Med. 2020, 20, 1. [Google Scholar] [CrossRef]
- Zhu, S.; Tang, S.; Su, F. Dioscin Inhibits Ischemic Stroke-induced Inflammation through Inhibition of the TLR4/MyD88/NF-κB Signaling Pathway in a Rat Model. Mol. Med. Rep. 2017, 17, 660–666. [Google Scholar] [CrossRef]
- Pari, L.; Monisha, P.; Mohamed Jalaludeen, A. Beneficial Role of Diosgenin on Oxidative Stress in Aorta of Streptozotocin Induced Diabetic Rats. Eur. J. Pharmacol. 2012, 691, 143–150. [Google Scholar] [CrossRef]
- Manivannan, J.; Balamurugan, E.; Thangarasu, S.; Raja, B. Diosgenin Improves Vascular Function by Increasing Aortic ENOS Expression, Normalize Dyslipidemia and ACE Activity in Chronic Renal Failure Rats. Mol. Cell. Biochem. 2013, 384, s11010–s11013. [Google Scholar] [CrossRef] [PubMed]
- Elseweidy, M.M.; Mohamed, H.E.; Elrashidy, R.A.; Atteia, H.H.; Elnagar, G.M.; Ali, A.E.-M. Potential Therapeutic Roles of 10-Dehydrogingerdione and/or Pentoxifylline against Calcium Deposition in Aortic Tissues of High Dietary Cholesterol-Fed Rabbits. Mol. Cell. Biochem. 2019, 453, 131–142. [Google Scholar] [CrossRef] [PubMed]
- Böhm, V. Vitamin, E. Antioxidants 2018, 7, 44. [Google Scholar] [CrossRef] [PubMed]
- Peralta-Ramírez, A.; Montes de Oca, A.; Raya, A.I.; Pineda, C.; López, I.; Guerrero, F.; Diez, E.; Muñoz-Castañeda, J.R.; Martinez, J.; Almaden, Y.; et al. Vitamin E Protection of Obesity-Enhanced Vascular Calcification in Uremic Rats. Am. J. Physiol.-Ren. Physiol. 2014, 306, F422–F429. [Google Scholar] [CrossRef]
- Knekt, P.; Reunanen, A.; Jävinen, R.; Seppänen, R.; Heliövaara, M.; Aromaa, A. Antioxidant Vitamin Intake and Coronary Mortality in a Longitudinal Population Study. Am. J. Epidemiol. 1994, 139, 1180–1189. [Google Scholar] [CrossRef]
- Bleys, J.; Miller, E.R.; Pastor-Barriuso, R.; Appel, L.J.; Guallar, E. Vitamin-Mineral Supplementation and the Progression of Atherosclerosis: A Meta-Analysis of Randomized Controlled Trials. Am. J. Clin. Nutr. 2006, 84, 880–887. [Google Scholar] [CrossRef]
- Vivekananthan, D.P.; Penn, M.S.; Sapp, S.K.; Hsu, A.; Topol, E.J. Use of Antioxidant Vitamins for the Prevention of Cardiovascular Disease: Meta-Analysis of Randomised Trials. Lancet 2003, 361, 2017–2023. [Google Scholar] [CrossRef]
- Gammone, M.A. Prevention of Cardiovascular Diseases with Carotenoids. Front. Biosci. 2017, 9, 165–171. [Google Scholar] [CrossRef]
- Huang, Y.; Zhou, X.; Liu, M.; Zhou, T.; Shi, J.; Dong, N.; Xu, K. The Natural Compound Andrographolide Inhibits Human Aortic Valve Interstitial Cell Calcification via the NF-Kappa B/Akt/ERK Pathway. Biomed. Pharmacother. 2020, 125, 109985. [Google Scholar] [CrossRef]
- Kandanur, S.G.S.; Tamang, N.; Golakoti, N.R.; Nanduri, S. Andrographolide: A Natural Product Template for the Generation of Structurally and Biologically Diverse Diterpenes. Eur. J. Med. Chem. 2019, 176, 513–533. [Google Scholar] [CrossRef]
- Zhu, T.; Wang, D.; Zhang, W.; Liao, X.; Guan, X.; Bo, H.; Sun, J.; Huang, N.; He, J.; Zhang, Y.; et al. Andrographolide Protects against LPS-Induced Acute Lung Injury by Inactivation of NF-ΚB. PLoS ONE 2013, 8, e56407. [Google Scholar] [CrossRef]
- Feng, W.; Zhang, K.; Liu, Y.; Chen, J.; Cai, Q.; Zhang, Y.; Wang, M.; Wang, J.; Huang, H. Apocynin Attenuates Angiotensin II-Induced Vascular Smooth Muscle Cells Osteogenic Switching via Suppressing Extracellular Signal-Regulated Kinase 1/2. Oncotarget 2016, 7, 83588–83600. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Watanabe, S.; Fujii, H.; Kono, K.; Watanabe, K.; Goto, S.; Nishi, S. Influence of Oxidative Stress on Vascular Calcification in the Setting of Coexisting Chronic Kidney Disease and Diabetes Mellitus. Sci. Rep. 2020, 10, 20708. [Google Scholar] [CrossRef] [PubMed]
- Gu, L.; Bai, W.; Li, S.; Zhang, Y.; Han, Y.; Gu, Y.; Meng, G.; Xie, L.; Wang, J.; Xiao, Y.; et al. Celastrol Prevents Atherosclerosis via Inhibiting LOX-1 and Oxidative Stress. PLoS ONE 2013, 8, e65477. [Google Scholar] [CrossRef] [PubMed]
- Shi, J.; Li, J.; Xu, Z.; Chen, L.; Luo, R.; Zhang, C.; Gao, F.; Zhang, J.; Fu, C. Celastrol: A Review of Useful Strategies Overcoming Its Limitation in Anticancer Application. Front. Pharmacol. 2020, 11, 558741. [Google Scholar] [CrossRef] [PubMed]
- Li, X.; Gao, M.; Zhu, S.; Yin, L.; Zhang, B.; Qi, Y.; Zhao, Y.; Yu, Y.; Xu, L. Hengshun Aromatic Vinegar Ameliorates Vascular Endothelial Injury via Regulating PKCζ-Mediated Oxidative Stress and Apoptosis. Front. Nutr. 2021, 8, 635232. [Google Scholar] [CrossRef] [PubMed]
- Ma, S.; Chen, J.; Feng, J.; Zhang, R.; Fan, M.; Han, D.; Li, X.; Li, C.; Ren, J.; Wang, Y.; et al. Melatonin Ameliorates the Progression of Atherosclerosis via Mitophagy Activation and NLRP3 Inflammasome Inhibition. Oxid. Med. Cell. Longev. 2018, 2018, 9286458. [Google Scholar] [CrossRef]
- Li, M.; Xu, S.; Geng, Y.; Sun, L.; Wang, R.; Yan, Y.; Wang, H.; Li, Y.; Yi, Q.; Zhang, Y.; et al. The Protective Effects of L-carnitine on Myocardial Ischaemia–Reperfusion Injury in Patients with Rheumatic Valvular Heart Disease Undergoing CPB Surgery Are Associated with the Suppression of NF -κB Pathway and the Activation of Nrf2 Pathway. Clin. Exp. Pharmacol. Physiol. 2019, 46, 1001–1012. [Google Scholar] [CrossRef]
- Chai, H.; Tao, Z.; Qi, Y.; Qi, H.; Chen, W.; Xu, Y.; Zhang, L.; Chen, H.; Chen, X. IKK Epsilon Deficiency Attenuates Angiotensin II-Induced Abdominal Aortic Aneurysm Formation in Mice by Inhibiting Inflammation, Oxidative Stress, and Apoptosis. Oxid. Med. Cell. Longev. 2020, 2020, 3602824. [Google Scholar] [CrossRef]
- Nelson, B.; Johnson, M.; Walker, M.; Riley, K.; Sims, C. Antioxidant Cerium Oxide Nanoparticles in Biology and Medicine. Antioxidants 2016, 5, 15. [Google Scholar] [CrossRef]
- Xue, Y.; Balmuri, S.R.; Patel, A.; Sant, V.; Sant, S. Synthesis, Physico-Chemical Characterization, and Antioxidant Effect of PEGylated Cerium Oxide Nanoparticles. Drug Deliv. Transl. Res. 2018, 8, 357–367. [Google Scholar] [CrossRef] [PubMed]
- Anselmo, W.; Branchetti, E.; Grau, J.B.; Li, G.; Ayoub, S.; Lai, E.K.; Rioux, N.; Tovmasyan, A.; Fortier, J.H.; Sacks, M.S.; et al. Porphyrin-Based SOD Mimic MnTnBuOE-2-PyP 5+ Inhibits Mechanisms of Aortic Valve Remodeling in Human and Murine Models of Aortic Valve Sclerosis. J. Am. Heart Assoc. 2018, 7, e007861. [Google Scholar] [CrossRef] [PubMed]
- Majumdar, U.; Manivannan, S.; Basu, M.; Ueyama, Y.; Blaser, M.C.; Cameron, E.; McDermott, M.R.; Lincoln, J.; Cole, S.E.; Wood, S.; et al. Nitric Oxide Prevents Aortic Valve Calcification by S-Nitrosylation of USP9X to Activate NOTCH Signaling. Sci. Adv. 2021, 7, eabe3706. [Google Scholar] [CrossRef] [PubMed]
- Richards, J.; El-Hamamsy, I.; Chen, S.; Sarang, Z.; Sarathchandra, P.; Yacoub, M.H.; Chester, A.H.; Butcher, J.T. Side-Specific Endothelial-Dependent Regulation of Aortic Valve Calcification. Am. J. Pathol. 2013, 182, 1922–1931. [Google Scholar] [CrossRef]
- Tyagi, N.; Vacek, T. Tyagi Hydrogen Sulfide and Sodium Nitroprusside Compete to Activate/Deactivate MMPs in Bone Tissue Homogenates. Vasc. Health Risk Manag. 2013, 9, 117. [Google Scholar] [CrossRef]
- Rattazzi, M.; Donato, M.; Bertacco, E.; Millioni, R.; Franchin, C.; Mortarino, C.; Faggin, E.; Nardin, C.; Scarpa, R.; Cinetto, F.; et al. L-Arginine Prevents Inflammatory and pro-Calcific Differentiation of Interstitial Aortic Valve Cells. Atherosclerosis 2020, 298, 27–35. [Google Scholar] [CrossRef]
- Lindman, B.R.; Chakinala, M.M. Modulating the Nitric Oxide—Cyclic GMP Pathway in the Pressure-Overloaded Left Ventricle and Group II Pulmonary Hypertension: Group II PH and NO-CGMP Signalling Pathway. Int. J. Clin. Pract. 2010, 64, 15–22. [Google Scholar] [CrossRef][Green Version]
- Liu, Z.; Dong, N.; Hui, H.; Wang, Y.; Liu, F.; Xu, L.; Liu, M.; Rao, Z.; Yuan, Z.; Shang, Y.; et al. Endothelial Cell-Derived Tetrahydrobiopterin Prevents Aortic Valve Calcification. Eur. Heart J. 2022, 43, 1652–1664. [Google Scholar] [CrossRef]
- Choi, B.; Lee, S.; Kim, S.-M.; Lee, E.-J.; Lee, S.R.; Kim, D.-H.; Jang, J.Y.; Kang, S.-W.; Lee, K.-U.; Chang, E.-J.; et al. Dipeptidyl Peptidase-4 Induces Aortic Valve Calcification by Inhibiting Insulin-Like Growth Factor-1 Signaling in Valvular Interstitial Cells. Circulation 2017, 135, 1935–1950. [Google Scholar] [CrossRef]
- Lin, C.-P.; Huang, P.-H.; Chen, C.-Y.; Wu, M.-Y.; Chen, J.-S.; Chen, J.-W.; Lin, S.-J. Sitagliptin Attenuates Arterial Calcification by Downregulating Oxidative Stress-Induced Receptor for Advanced Glycation End Products in LDLR Knockout Mice. Sci. Rep. 2021, 11, 17851. [Google Scholar] [CrossRef]
- Yeang, C.; Tsimikas, S. Ancient Remedy for a Modern Disease. JACC Basic Transl. Sci. 2020, 5, 50–52. [Google Scholar] [CrossRef] [PubMed]
- Helmstädter, J.; Keppeler, K.; Aust, F.; Küster, L.; Frenis, K.; Filippou, K.; Vujacic-Mirski, K.; Tsohataridis, S.; Kalinovic, S.; Kröller-Schön, S.; et al. GLP-1 Analog Liraglutide Improves Vascular Function in Polymicrobial Sepsis by Reduction of Oxidative Stress and Inflammation. Antioxidants 2021, 10, 1175. [Google Scholar] [CrossRef]
- Li, F.; Cai, Z.; Chen, F.; Shi, X.; Zhang, Q.; Chen, S.; Shi, J.; Wang, D.W.; Dong, N. Pioglitazone Attenuates Progression of Aortic Valve Calcification via Down-Regulating Receptor for Advanced Glycation End Products. Basic Res. Cardiol. 2012, 107, 306. [Google Scholar] [CrossRef]
- Drummond, G.R.; Selemidis, S.; Griendling, K.K.; Sobey, C.G. Combating Oxidative Stress in Vascular Disease: NADPH Oxidases as Therapeutic Targets. Nat. Rev. Drug Discov. 2011, 10, 453–471. [Google Scholar] [CrossRef] [PubMed]
- Lu, W.J.; Li, J.Y.; Chen, R.J.; Huang, L.T.; Lee, T.Y.; Lin, K.H. VAS2870 and VAS3947 Attenuate Platelet Activation and Thrombus Formation via a NOX-Independent Pathway Downstream of PKC. Sci. Rep. 2019, 9, 18852. [Google Scholar] [CrossRef] [PubMed]
- Reis, J.; Massari, M.; Marchese, S.; Ceccon, M.; Aalbers, F.S.; Corana, F.; Valente, S.; Mai, A.; Magnani, F.; Mattevi, A. A Closer Look into NADPH Oxidase Inhibitors: Validation and Insight into Their Mechanism of Action. Redox Biol. 2020, 32, 101466. [Google Scholar] [CrossRef]
- Teixeira, G.; Szyndralewiez, C.; Molango, S.; Carnesecchi, S.; Heitz, F.; Wiesel, P.; Wood, J.M. Therapeutic Potential of NADPH Oxidase 1/4 Inhibitors: Pharmacology of NOX Inhibition. Br. J. Pharmacol. 2017, 174, 1647–1669. [Google Scholar] [CrossRef]
- Choi, B.; Kim, E.-Y.; Kim, J.-E.; Oh, S.; Park, S.-O.; Kim, S.-M.; Choi, H.; Song, J.-K.; Chang, E.-J. Evogliptin Suppresses Calcific Aortic Valve Disease by Attenuating Inflammation, Fibrosis, and Calcification. Cells 2021, 10, 57. [Google Scholar] [CrossRef]
| Refs. | Compound | Administration and Doses/Researched Cells | Salient Findings |
|---|---|---|---|
| Experimental Human Studies | |||
| [120] | IONIS-APO(a)Rx vs. placebo | 100 mg, 200 mg, and then 300 mg once a week for 4 weeks each /1 dose of 10–120 mg sq /multiple doses of 10 mg, 20 mg, or 40 mg sq | -Reduced Lp(a) levels in a dose-dependent manner. |
| [137] | Curcumin | hVICs | -Inhibition of NF-κB, AKT, ERK. |
| [141] | Cardamonin | In Vitro: hVICs Ex Vivo: human aortic valve leaflet | -Inhibition of VIC osteogenic differentiation through the NF-κB/NLRP3 inflammasome pathway. |
| [145] | Caffeic Acid | hVICs | -Inhibition of the ERK/AKT/NF-κB/NLRP3 inflammasome pathway. |
| [152] | Quercetin | HUVECs | -Attenuated atherosclerotic inflammation and adhesion molecule expression by the TLR-NF-κB pathway. |
| [26] | Anthocyanins | D-HAEC | -Inhibition of the NF-κB pathway. |
| [169] | Andrographolide | hVICs (from patients undergoing Bentall surgery due to acute type I aortic dissection) | -Inhibition of the NF-κB/Akt/ERK pathway. |
| [176] | HSAV | HUVECs | -Inhibited apoptosis, decreased serum Hcy, ET-1), ox-LDL levels, MDA level; -Increased NO, eNOS, SOD, GSH, and GSH-Px levels; -Downregulated the expression of PKCζ and regulated the SIRT1-mediated pathway. |
| [178] | L-carnitine | Patients with RVHD with CPB-induced MIRI (myocardial ischemia-reperfusion injury) | -Increased levels of SOD, CAT; -Suppressed activation of NF-κB and Nrf2. |
| [24] | Adenoviral SOD delivery | hVIC | -Reduced VIC osteoblastic differentiation by reducing RUNX2, MSX2, and OPN. |
| [23,181] | CNPs | hVIC | -Scavenged ROS, acted as SOD-mimetics, and reduced VIC osteoblastic differentiation. |
| Ref. | Compound | Species and/or Cells Researched | Meaningful Findings |
|---|---|---|---|
| [121] | E06 natural antibody | E06-scFv transgenic mice | -Counteracted the proinflammatory and proatherogenic OxPL effects. |
| [126] | Resveratrol | Ovariectomized rats | -Reduced RUNX2, ALP expression, and aortic calcification. |
| [127] | Resveratrol | Rat vascular smooth muscle cells (RASMCs) | -Prevents vascular calcification and mitochondria dysfunction through SIRT1 and Nrf2. |
| [128] | Resveratrol | Mouse model of uremia | -Fewer aortic atherosclerotic lesions at the site of the ascending aorta. |
| [131] | Resveratrol | Porcine aortic valve interstitial cells (pVICs) | -Inhibition of osteogenic pVIC differentiation through the AKT/SMAD1/5/8 signaling pathway. |
| [129] | PLD the natural precursor of resveratrol | Mice with complete ligatures of the left carotid arteries for 14 days | -Reduced adhesion molecule expression (ICAM-1, VCAM-1), proinflammatory cytokine production (TNF-α, IL-1β), iNOS, NF-κB expression, and BAX, Fas-Ligand activation. |
| [136] | Curcumin | Apolipoprotein E-knockout mice | -Reduced TLR4 expression, macrophage infiltration in atherosclerotic plaque, aortic IL-1β, TNF-α, VCAM-1, ICAM-1 expression, NF-κB activity, and plasma IL-1β, TNF-α, soluble VCAM-1, and ICAM-1 levels; -Reduced the extent of atherosclerotic lesions and inhibited atherosclerosis development. |
| [138] | Curcumin | Different types of mice, all treated with HF and mice fed with a normal chow diet | -Reduced serum lipid levels, TNF-α, IL-1β, and the aortic atherosclerotic lesion area. |
| [141] | Cardamonin | In Vivo: mice model fed with a HF diet | -Inhibition of VIC osteogenic differentiation through the NF-κB/NLRP3 inflammasome pathway. |
| [143] | Ellagic acid | Rat model | -Improved nitric oxide bioavailability and reduced ROS formation. |
| [144] | Gallic acid | Vascular smooth muscle cell | -Inhibition of vascular calcification through the BMP2-SMAD1/5/8 signaling pathway. |
| [149] | Nobiletin | Male Wistar rats | -Increased intracellular cGMP (activation of cGC, opening BK channels and KATP channels). |
| [151] | Quercetin | Adenine-induced chronic renal failure rats | -Modulation of vascular calcification through the iNOS/p38 MAPK pathway. |
| [154] | Anthocyanins | Tac-induced myocardial dysfunction in mice | -Ameliorated Tac-induced myocardial dysfunction, oxidative stress, and apoptosis via the DDAH1/ADMA/no pathway. |
| [155] | Puerarin | In Vitro; rat vascular smooth muscle cells In Vivo; uremic rats | -Modulated NLRP3/CASPASE1/IL-1β, NF-κB, and ER/PI3K-AK signaling pathways; -Prevents calcium deposition and inhibits the expression of RUNX2 and BMP2. |
| [158] | Puerarin | VSMCs | -Inhibited oxLDL-induced VSMC viability via inhibition of the p38 MAPK and JNK signaling pathways; -Decreased the levels of IL-6 and TNF-α and increased SOD activity. |
| [161] | Diosgenin | Adenine-induced chronic renal failure rats | -Inhibited the c/Akt/ERK, p38 pathway. |
| [162] | 10-DHGD | HCD-fed rabbits | -Alleviated calcium deposition via the downregulation of the BMP2/Wnt3a pathway, OPG/RANK modulation, and raised HDL-C levels. |
| [164] | Vitamin E | Uremic obese rats | -Prevents osteoblastic differentiation in VSMC and inhibits dephosphorylation of Akt. |
| [87] | Fucoxanthin | In Vitro; rat heart VIC In Vivo; dog model | -Inhibition of the Akt/ERK pathway. |
| [172] | Apocynin | VSMCs | -Enhanced expression of α-SMA, reduced expression of BMP2, RUNX2, OPN, suppressed the ERK1/2 pathway and phosphorylation of p47phox (cytosolic NOX2 component). |
| [70] | Celastrol | In Vitro; porcine AVIC In Vivo; rabbit CAVD model | -Inhibition of NADPH Oxidase 2 and the GSK3β/β-catenin pathway |
| [174] | Celastrol | Macrophages in mice | -Attenuated oxLDL-induced excessive expression of LOX-1; -Decreased IkB phosphorylation and degradation, reduced production of iNOS, NO, TNF-α, and IL-6; -Reduced atherosclerotic plaque size. |
| [75] | Glycine | Streptozotocin-induced diabetic rats and HUVECs | -Downregulating the AGE/RAGE signaling pathway by decreasing levels of AGEs, RAGE, NOX4, and NF-κB p65, and by restoring GLO1 function. |
| [182] | MnBuOE | hVIC and murine model of aortic valve sclerosis | -Inhibited aortic valve remodeling and α-SMA upregulation via TGF-β1; -Upregulated MnSOD via activation of Nrf2. |
| [52,85] | Mitoquinone | Male Sprague–Dawley rats and adult C57BL/6J mice | -Reduced vascular calcification through the Nrf2/Keap1 pathway and fibrosis by inhibiting the TGF-β1-NOX4-ROS axis. |
| [184] | DETA NONOate | PAVEC and aortic VIC PAVIC | -Inhibited VIC osteogenic differentiation and calcification. |
| [186] | L-arginine | Bovine aortic VICs | -Inhibited VIC osteogenic differentiation and remodeling by downregulating ADAMTSL4 and fibrillin-1. |
| [89] | Anagliptin | Eight-week-old male BALB/c mice | -Activated the PI3K/Akt signaling pathway; -Downregulated the expression of MCP-1, ICAM-1, VCAM-1; -Reduced proteolysis via MMP-2/-9 and CatS/K. |
| [122,190] | Sitagliptin | Weaned male low-density lipoprotein receptor knockout mice | -Blocked NADPH activation; -Inhibited calcification by downregulating RAGE expression and NF-κB activation. |
| [189] | Sitagliptin | Rabbit model of CAVD fed with HCD and vitamin D2 | -Reduced osteogenic transformation of VICs by reinstating IGF-1 activity. |
| [198] | Evogliptin | hVIC, endothelial nitric oxide synthase-deficient, male New Zealand white rabbits | -Reduced TNF-α, IL-1β, and IL-6 levels; -Reduced RUNX2 expression. |
| [69] | Pioglitazone | Mice fed a western-type diet | -Attenuated cusp mobility and inhibited valve calcification by reducing TNFα, IL-6, and BMP2. |
| [193] | Pioglitazone | Male New Zealand rabbits | -Reduced RAGE activation and inhibited NF-κB p65 intranuclear translocation. |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Tanase, D.M.; Valasciuc, E.; Gosav, E.M.; Floria, M.; Costea, C.F.; Dima, N.; Tudorancea, I.; Maranduca, M.A.; Serban, I.L. Contribution of Oxidative Stress (OS) in Calcific Aortic Valve Disease (CAVD): From Pathophysiology to Therapeutic Targets. Cells 2022, 11, 2663. https://doi.org/10.3390/cells11172663
Tanase DM, Valasciuc E, Gosav EM, Floria M, Costea CF, Dima N, Tudorancea I, Maranduca MA, Serban IL. Contribution of Oxidative Stress (OS) in Calcific Aortic Valve Disease (CAVD): From Pathophysiology to Therapeutic Targets. Cells. 2022; 11(17):2663. https://doi.org/10.3390/cells11172663
Chicago/Turabian StyleTanase, Daniela Maria, Emilia Valasciuc, Evelina Maria Gosav, Mariana Floria, Claudia Florida Costea, Nicoleta Dima, Ionut Tudorancea, Minela Aida Maranduca, and Ionela Lacramioara Serban. 2022. "Contribution of Oxidative Stress (OS) in Calcific Aortic Valve Disease (CAVD): From Pathophysiology to Therapeutic Targets" Cells 11, no. 17: 2663. https://doi.org/10.3390/cells11172663
APA StyleTanase, D. M., Valasciuc, E., Gosav, E. M., Floria, M., Costea, C. F., Dima, N., Tudorancea, I., Maranduca, M. A., & Serban, I. L. (2022). Contribution of Oxidative Stress (OS) in Calcific Aortic Valve Disease (CAVD): From Pathophysiology to Therapeutic Targets. Cells, 11(17), 2663. https://doi.org/10.3390/cells11172663









