1. Introduction
Angiogenesis is the development of new blood vessels from pre-existing vessels; this process is required for embryogenesis, normal tissue development, wound healing and tumor growth [
1]. However, based on accumulating evidence, vascular endothelial cells in tumors are different from normal endothelial cells [
2]. Croix et al. revealed the altered gene expression pattern in tumor endothelial cells compared to normal endothelial cells [
3]. The two cell types may also respond differently to epidermal growth factor (EGF), adrenomedullin and VEGF from the microenvironment [
4,
5]. Moreover, tumor endothelial cells directly regulate the proliferation of cancer cells [
6,
7], suggesting that tumor blood vessels might have other unexpected roles within the bulk tumor.
Nogo isoform-B (
Nogo-B), also known as reticulon 4B, is a member of the reticulon family [
8].
Nogo-B is expressed at high levels in the microvessels [
9]. In a recent study by Wälchli et al., another isoform of Nogo, Nogo-A, functioned as a negative regulator of angiogenesis in the developing central nervous system [
10].
Nogo-B knockout (
Nogo-B−/−) mice exhibit impaired arteriogenesis, indicating an essential role for
Nogo-B in physiological angiogenesis [
9].
Nogo-B knockout animals exhibit excessive repair of the intimal and medial layers of the balloon traction-injured femoral artery, which was remedied upon the restoration of
Nogo-B expression, suggesting a critical role for
Nogo-B in vascular remodeling [
11,
12]. Nevertheless, the role of vascular endothelial
Nogo-B in cancer development largely remains unknown.
Hepatocellular carcinoma (HCC) is the second leading cause of cancer-related mortality globally; nearly half of patients diagnosed with HCC reside in China [
13]. Although remarkable progress has been achieved in understanding the molecular mechanism and developing comprehensive therapies for HCC, rendering incurable HCC curable in certain patients, the prognosis of patients with HCC remains largely disappointing [
14].
Nogo-B was reported to be preferentially expressed in non-parenchymal cells in the liver, and upregulation of
Nogo-B expression has been detected in patients’ cirrhotic liver tissues [
15].
Nogo-B is potentially involved in the activation of hepatic stellate cells (HSCs), as knockdown of
Nogo-B in HSCs dramatically impaired HSC activation [
15]. Nonetheless, the role of vascular endothelial
Nogo-B in HCC progression remains unclear. In this study, we examined
Nogo-B expression in vascular endothelial cells in HCC tissues from patients and investigated the role of endothelial
Nogo-B in HCC growth. Moreover, recent studies provide additional evidence that neurovascular crosstalk is more important for understanding the molecular basis of neurological diseases than originally anticipated [
16,
17]. Here, we also confirmed our observations in a glioma model to expand our findings to another cancer type.
2. Materials and Methods
2.1. Patients and Tumor Specimens
HCC tissue specimens (
n = 167) were acquired from patients who underwent surgical resection at Eastern Hepatobiliary Surgery Hospital (EHBH) in Shanghai between January 2000 and December 2009. The demographic and baseline characteristics of the patients are shown in
Supplementary Table S1. Patients were followed at clinical visits every 2 months during the first postoperative year and at least every 4 months thereafter. Each visit included standard liver function and hematologic tests, as well as liver ultrasonography. Patients with a progressive increase in serum α-fetoprotein (AFP) levels and/or ultrasonographic detection of a new hepatic lesion were hospitalized for confirmation of the diagnosis and appropriate management. Ten fresh HCC specimens were obtained from patients undergoing hepatectomy. The acquisition of tissue specimens was approved by the Ethics Committee at Eastern Hepatobiliary Surgery Hospital and was performed in accordance with institutional and state regulations (EHBHKY2020-K-016).
2.2. Laser Capture Microdissection (LCM)
Tissue sectioning and staining were performed as previously described [
18]. LCM was performed using the Leica Laser Capture Microdissection System (Leica AS LMD, Wetzlar, Germany). Three to four 10-µm sections from each specimen were used to capture approximately 10,000 cells (approximately 4 mm
2). The time between when the tissue sections were removed from xylene to the completion of LCM and initiation of the RNA extraction process was limited to less than 45 min.
2.3. Quantitative Real-Time PCR
Total cellular RNA was isolated from cells using the RNeasy Micro kit (Qiagen, Germantown, MD, USA). First strand cDNA synthesis was performed using the Reverse Transcription System (Promega, Madison, WI, USA). Quantitative real-time PCR was performed using a SYBR Green PCR Kit (Applied Biosystems, Foster City, CA, USA) and the ViiA 7 Dx Real-Time PCR system (Applied Biosystems, Foster City, CA, USA). The sequences of the primers used in this study are: Nogo-B-F:5′-GCAGTGTTGATGTGGGTATTT-3′; Nogo-B-R:5′-CTGTGCCTGATGCCGTTC-3′; TGFβ1-F:5′-GTACCTGAACCCGTGTTGCT-3′; TGFβ1-R:5′-TGAACCCGTTGATGTCCACT-3′; β-actin-F:5′-AATCGTGCGTGACATTAAGGAG-3′; β-actin-R: 5′-ACTGTGTTGGCGTACAGGTCTT-3′. Each reaction was performed in triplicate.
2.4. Immunohistochemistry and Microarray Analysis
Immunohistochemistry of tumor sections was performed as previously described [
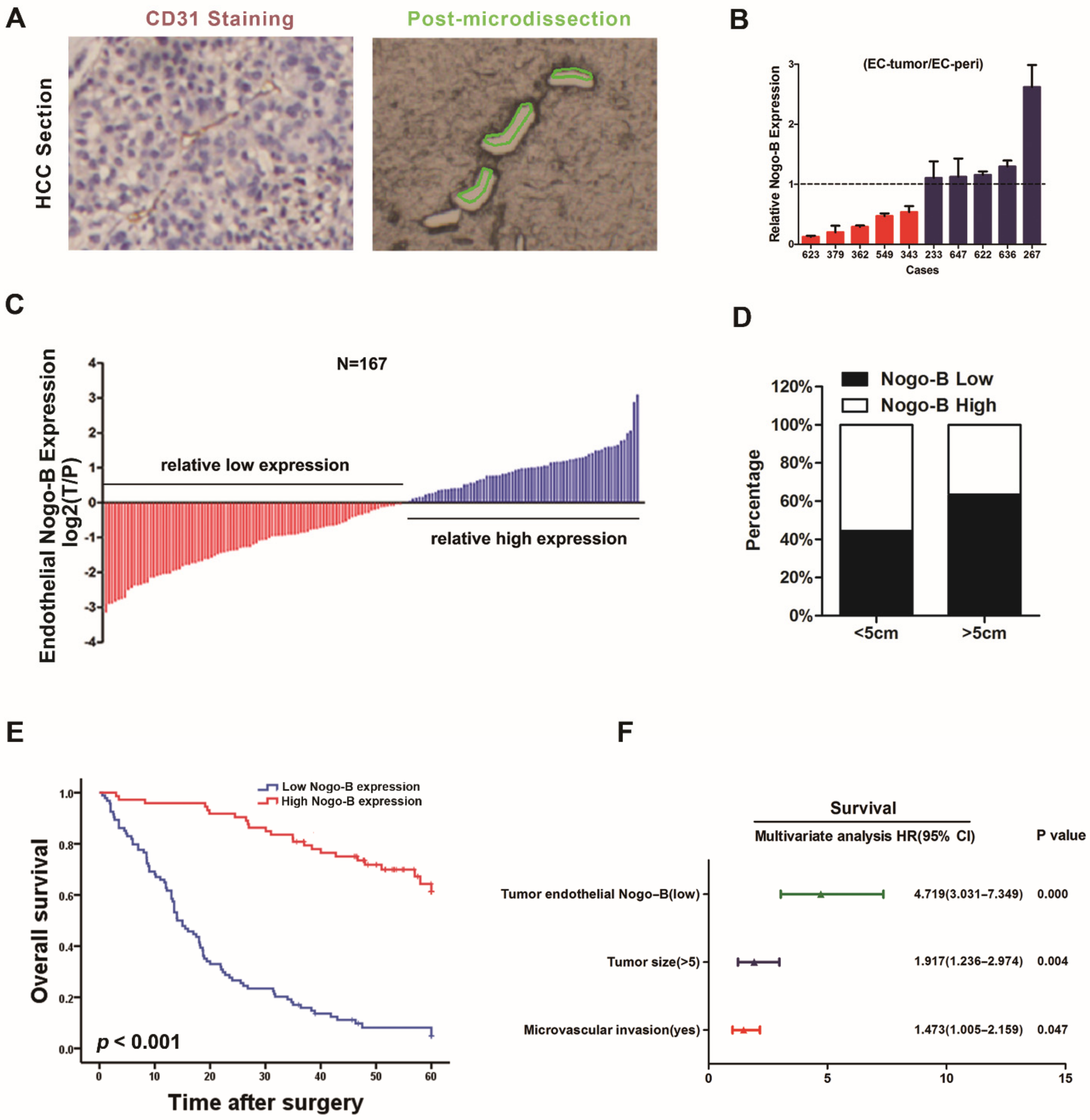
18]. Primary antibodies against the following proteins were used: CD31, 1:100 dilution (Santa Cruz Biotechnology, Heidelberg, Germany),
Nogo-B, 1:100 dilution (Abclonal, Wuhan, China) and Ki-67, 1:100 dilution (Santa Cruz Biotechnology, Heidelberg, Germany). We used serial sections of HCC tissues for microarray chips in which one section was stained with the anti-CD31 antibody and another section was stained with the anti-
Nogo-B antibody. Then, the expression of
Nogo-B in CD31-positive vascular endothelial cells was compared. All sections displaying immunohistochemical staining were observed and measured under an Olympus microscope (IX-70 OLYMPUS, Tokyo, Japan). The integrated optical density (IOD) was measured and calculated as IOD/total area of each image [
19]. High
Nogo-B expression is defined as sections in which the signal for positive staining was higher than the median value.
2.5. Lentivirus and Cell Lines
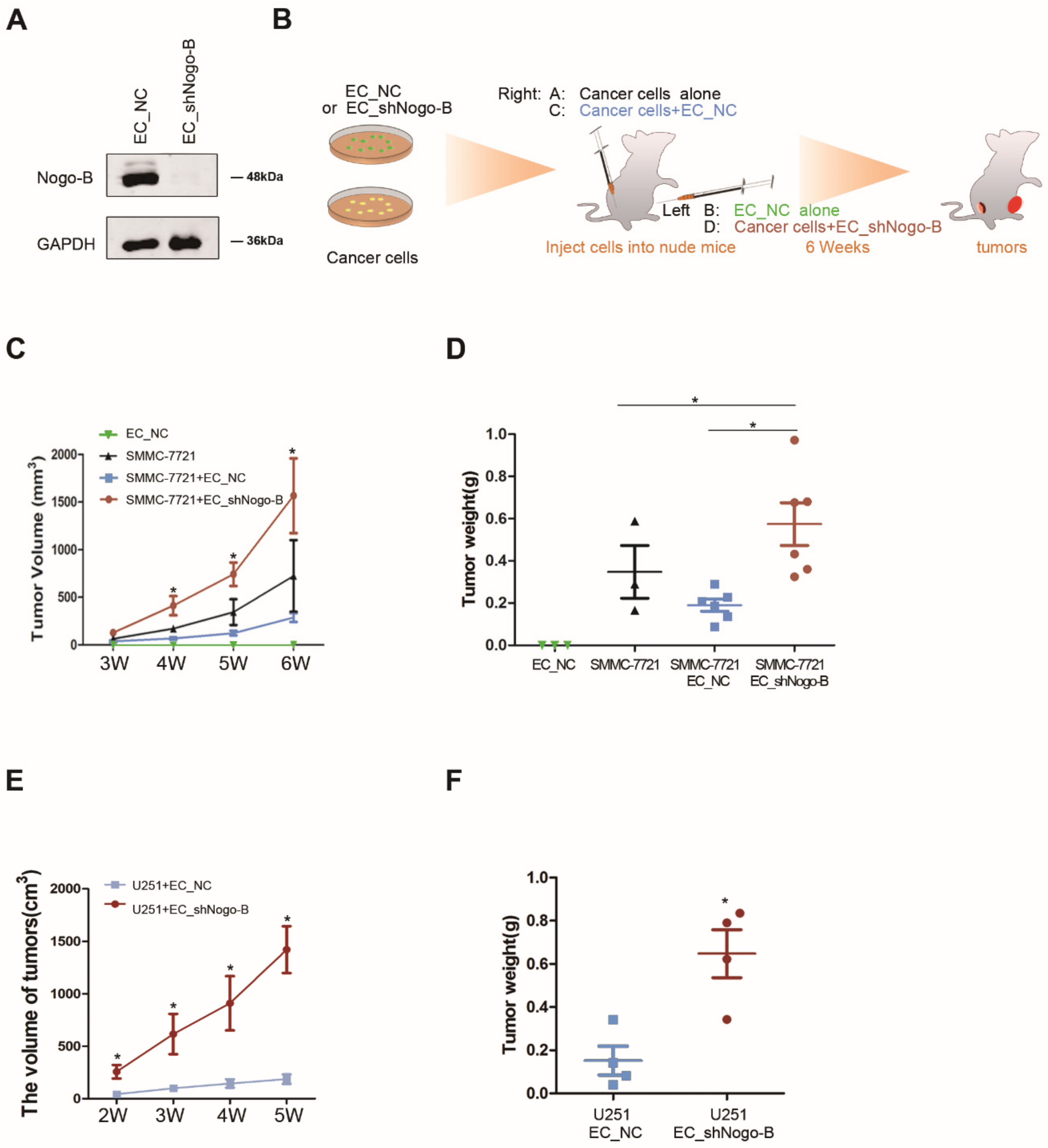
Human HCC SMMC-7721 cells, glioma U251 cells and human umbilical vein endothelial cells (HUVECs) were obtained from the American Type Culture Collection (Manassas, VA, USA) and cultured in DMEM with 10% fetal bovine serum (FBS) at 37 °C in a humidified incubator containing 5% CO2. HUVECs were infected with a lentivirus expressing a short hairpin RNA targeting Nogo-B (shRNA, target sequence: TATATCTGAGGAGTTGGT) or scrambled control, and stable transfectants were established and termed EC_shNogo-B and EC_NC, respectively. HUVECs were infected with a lentivirus expressing Nogo-B or GFP, and stable transfectants were established and named EC_GFP and EC_Nogo-B, respectively. All lentiviruses were purchased from Cyagen Biosciences Inc., Guangzhou, China.
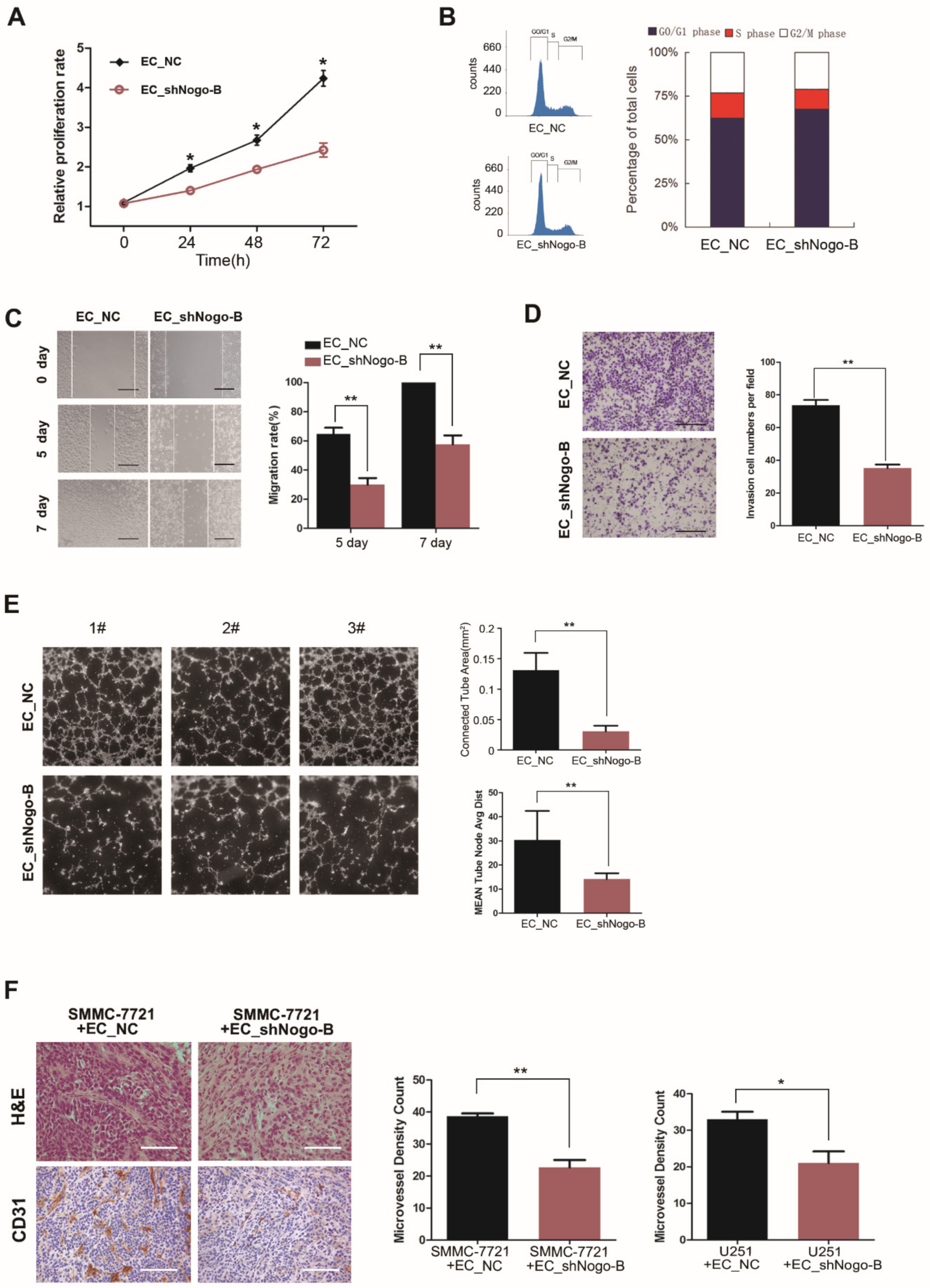
2.6. Cell Proliferation Analysis
In total, 3 × 103 cells were cultured in each well of 96-well plates in 10% FBS/DMEM. ATP activity was measured using a Cell Counting Kit-8 (CCK-8) and a Synergy 2 microplate reader at the indicated time points. The results are presented as a proliferation index relative to control cells.
2.7. In Vitro Co-Culture Assays
SMCC-7721 or U251 cells were mixed with 1 × 103 CFSE-labeled EC_NC or EC_shNogo-B, respectively, and then seeded in 96-well plates. After a 3-day incubation, the number of cancer cells and HUVECs was counted by flow cytometry.
EC_NC and EC_shNogo-B were seeded in 15-cm Petri dishes at a density of 1 × 106 cells per dish. Upon reaching 80% confluency, the medium was discarded, the monolayer was washed thrice with PBS and then media were replenished with serum-free DMEM. After a 24-h incubation, the medium was collected and filtered (0.45 µm). SMCC-7721 cells cultured in 6-well plates or 96-well plates were incubated with normal DMEM containing 10% FBS or the culture medium from EC_NC and EC_shNogo-B before analysis using a CCK-8 assay or flow cytometry assay.
2.8. In Vivo Co-Culture Assays
SMMC-7721 or U251 cells (1 × 10
6) were mixed with EC_NC or EC_shNogo-B at a ratio of 1:1 and subcutaneously implanted into nude mice. Tumor weights and volumes were calculated using previously described methods [
20]. For immunofluorescence staining, frozen sections of xenograft tumors were incubated with a rabbit anti-phospho-Smad2 antibody (1:100, Abcam, Cambridge, UK), followed by an incubation with Alexa Fluor 488-conjugated anti-mouse IgG and Alexa Fluor 555-conjugated anti-rabbit IgG antibodies (Invitrogen, Carlsbad, CA, USA). Nuclei were stained with 4,6-diamidino-2-phenylindole (DAPI). All animal experiments met the requirement of the Second Military Medical University Animal Care Facility and followed the USA National Institutes of Health guidelines.
2.9. Antibody Arrays
Soluble proteins in the medium of the stromal cell lines were measured using the Human Cytokine Array G4000 (RayBio, AAH-CYT-G4000-8, Guangzhou, China) and a Biotin Label-Based Human Antibody Array (RayBio, AAH-BLG-1-4, Guangzhou, China), each of which is capable of detecting 507 proteins. FBS-free DMEM tissue culture media were collected from 90% confluent HUVECs and filtered. Array chips treated with a serum-free medium were used for normalization. Hybridization was conducted overnight at 4 °C. All slides were scanned using a GenePix 4000B Microarray Scanner (Axon, Seattle, WA, USA) and analyzed using GenePix Pro 6.0 software (Axon, Seattle, WA, USA). The F532 median 2B532 score was used and averaged across triplicates on each array. The results were then normalized to internal controls. The KEGG analysis was performed using tools at
https://david.ncifcrf.gov/ (accessed on 3 May 2016).
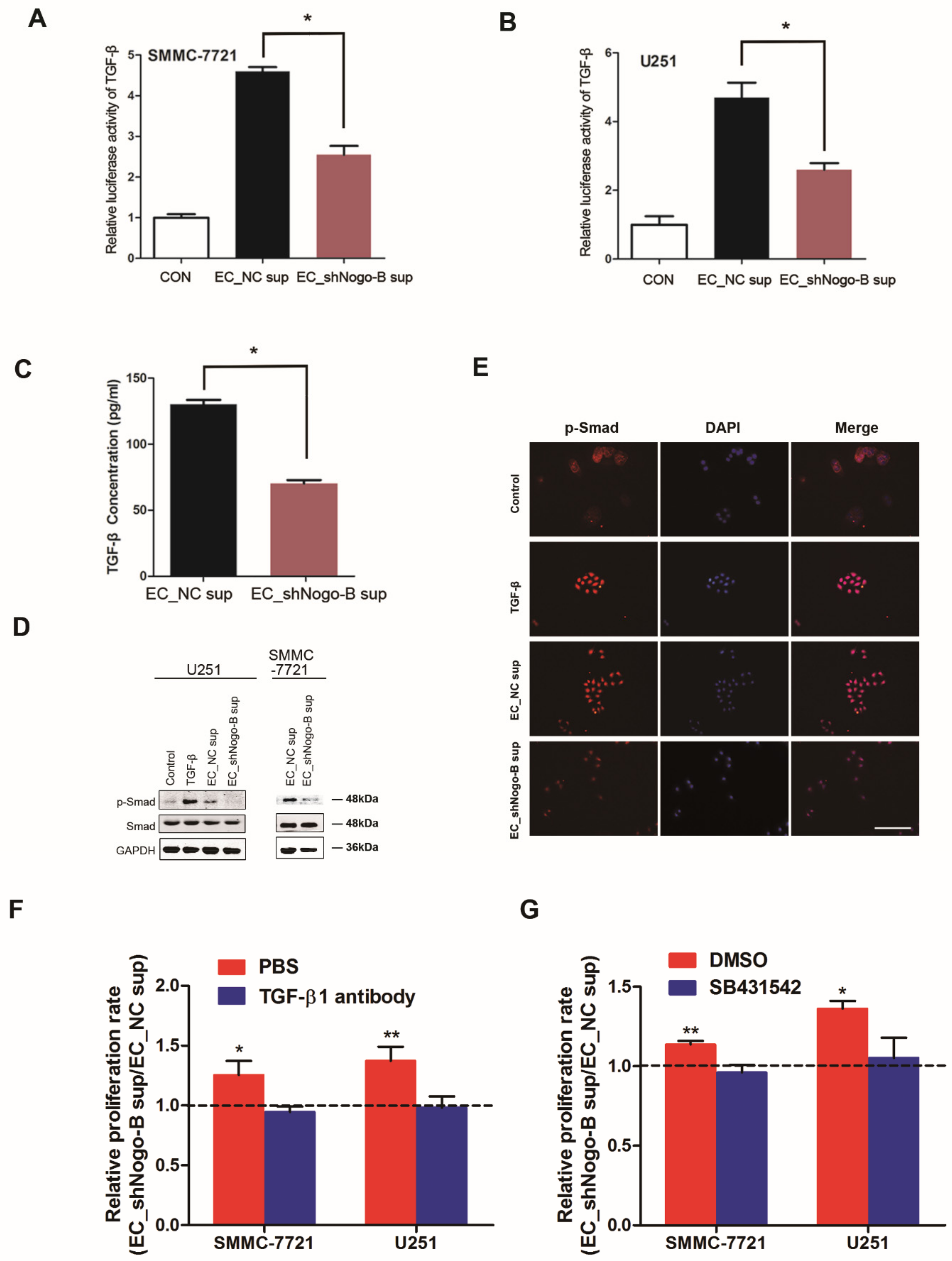
2.10. Luciferase Reporter Assays and ELISA
SMMC-7721 or U251 cells were incubated with the tissue culture media from EC_NC or EC_shNogo-B or serum-free DMEM for 48 h. SMMC-7721 or U251 cells were then transfected with the plasmids pGL-TGF-β-luc, pGL-STAT3-luc or pGL-AP-1-luc using jetPEI (Polyplus-Transfection, New York, NY, USA). Luciferase activities were measured using the Dual Luciferase Reporter Assay System (Promega, Madison, WI, USA). Luciferase activity was normalized to the activity of the Renilla luciferase control. All experiments were performed in triplicate.
TGF-β contents in the tissue culture media of EC_NC or EC_shNogo-B were measured using a commercially available kit (eBioscience, San Diego, CA, USA). The optical density was determined at 450 nm using a microplate reader.
2.11. Western Blot Assays
Cells were directly lysed in Laemmli buffer. The immunoblotting procedure was performed as previously described [
21]. Twenty micrograms of lysate were loaded in each well. The following antibodies were used for the procedure:
Nogo-B (1:1000, Abclonal, Wuhan, China), p-Smad3 (1:1000, Cell Signaling Technology, Danvers, MA, USA), Smad3 (1:1000, Cell Signaling Technology, Danvers, MA, USA) and GAPDH (1:5000, Santa Cruz Biotechnology, Heidelberg, Germany). Densitometric analyses were performed using the Quantity One analysis software (Bio-Rad, Berkeley, CA, USA). The expression of these proteins was normalized to GAPDH.
2.12. Matrigel Tube Formation Assays
For Matrigel™ tube formation assays, 96-well plates were coated with Matrigel (BD Biosciences, Heidelberg, Germany). EC_NC or EC_shNogo-B or untransfected HUVECs were seeded on a layer of previously polymerized and growth factor-reduced Matrigel™. After an 8-h incubation, Cellomics Cytoskeletal Rearrangement Kits (Thermoscientific, Millersburg, PA, USA) were used to stain the tubes, and photomicrographs of each well were captured using an Arrayscan HCS Reader (Thermoscientific, Millersburg, PA, USA). The number and line length of the circular tubules formed by the cells were calculated using the Image-Pro® Plus 4.5 software (Media CyberMetics, Rockville, MD, USA).
2.13. Scratch Wound-Healing Assay
HUVECs were seeded in a 12-well plate. Approximately 48 h later, when cells were 80% confluent, cells were incubated with serum-free DMEM overnight. A wound was generated by scraping the cell monolayer with a 10-µL pipette tip. Medium and non-adherent cells were removed, cells were washed twice with PBS and fresh media, supplemented with or without EGF (R&D Systems, Minneapolis, MN, USA), were added. Cells were permitted to migrate into the wound area for 7 days. Wound healing was photographed microscopically at days 0, 5 and 7 post-scratching (Carl Zeiss Meditec, Jena, Germany). The distance of the gap wound was measured using Photoshop software (ADOBE SYSTEMS INCORPORATED, (accessed on 15 March 2016)).
2.14. Transwell Migration Assays
The cell migration assay was performed in a 24-well Transwell migration chamber (BD Biosciences, Heidelberg, Germany) with polycarbonate filters of 6.5 mm in diameter and 8 µm in pore size. Approximately 2 × 105 EC_NC and EC_shNogo-B were resuspended in serum-free DMEM and added to the upper chamber of the well, and DMEM supplemented with 10% FBS was added to the lower chamber. Cells were allowed to migrate for 18 h at 37 °C. Non-migrated cells were removed from the upper surface with a cotton swab. Cells that had migrated were fixed with 5% paraformaldehyde and stained with 1% crystal violet in 2% ethanol. The number of cells was counted using Image-Pro® Plus 4.5 software (Media CyberMetics, Rockville, MD, USA) and expressed as the mean number of cells per field of view.
2.15. Statistical Analysis
Statistical analyses were performed using SPSS 20.0 for Windows (SPSS Inc., Chicago, IL, USA). Data are expressed as means ± SD. The significance of the difference in mean values between two groups was analyzed using two-tailed Student’s t-test. Pearson’s correlation analysis was conducted to assess correlations between two variables. Overall survival (OS) was defined as the interval from the date of surgery until death of any cause. Univariate and multivariate Cox proportional hazard regression analyses were performed to estimate crude or adjusted hazard ratios (HR) and their 95% confidence intervals (CIs). Kaplan–Meier and log-rank analyses were performed to compare OS and recurrence between subgroups. A p-value < 0.05 was considered statistically significant.
4. Discussion
Angiogenesis is a critical event in cancer development due to the supply of indispensable nutrition and oxygen to tumor cells. Most of the studies related it to decreased tumor vascularization and a concomitant inhibition of tumor growth or metastasis development [
22,
23]. However, whether endothelial cells of blood vessels may exert other functions in the tumor mass was not well known. In the present study,
Nogo-B was differentially expressed in the tumor vasculature, and endothelial
Nogo-B silencing promoted endothelial cell proliferation but suppressed tumor growth via a paracrine TGFβ/Smad signaling, suggesting an extremely complex interplay between tumor blood vessels and tumor cells.
Nogo-B is mainly expressed in hepatic non-parenchymal cells, and its expression is upregulated in patients with cirrhosis [
24]. However, the expression pattern of endothelia
Nogo-B in cancer and its correlation with the clinical outcomes of patients remain poorly understood. The current study presents evidence that endothelial
Nogo-B was differentially expressed in patients’ tumors. More interestingly, the median OS was significantly longer in patients with HCC who presented high endothelial
Nogo-B expression than in patients with low endothelial
Nogo-B levels. According to the results of our multivariate analysis, endothelial
Nogo-B expression in the tumor is an independent prognostic determinant of patient survival. Discovery of novel biomarkers that incorporate well with traditional cancer staging may improve the prognostic predictions and beneficial effects of therapies on patients. Thus, endothelial
Nogo-B expression in the tumor represents a potential diagnostic and therapeutic biomarker.
Angiogenesis plays an essential role in tumor growth and has become an attractive target for cancer therapy [
25]. Emerging evidence has indicated a role for endothelial cells in carcinogenesis and cancer therapy [
26,
27]. In the present study,
Nogo-B silencing suppressed the proliferation, migration and tube formation ability of HUVECs in vitro. Consistent with these results, endothelial
Nogo-B silencing reduced the microvessel density in mouse xenograft tumors, suggesting a positive effect of
Nogo-B on angiogenesis. Intriguingly, knockdown of endothelial
Nogo-B promoted the proliferation of co-cultured cancer cells, which was validated in our in vivo xenograft study. Based on these observations, we believe that the interaction between cancer cells and vascular endothelial cells in the tumor is rather complicated. The disappointing effects of current anti-angiogenesis therapy may be at least partially due to the much more complex than anticipated effects of anti-angiogenesis treatment. In addition, anti-angiogenesis therapy usually affects the function of normal endothelial cells and sometimes interrupts physiological angiogenesis. Here,
Nogo-B delivery did not affect the proliferation and migration of HUVECs, which express high levels of endogenous
Nogo-B, suggesting that endothelial cell-specific
Nogo-B delivery might be a novel anti-tumor therapy to suppress cancer growth without causing adverse effects.
The crosstalk between cancer cells and stromal cells is critical during tumor progression. Stromal cell-secreted factors have been shown to remarkably alter the characteristics of tumor cells [
28]. Here, the preliminary screen using a protein array showed that
Nogo-B silencing reduced the secretion of TGF-β signaling-associated molecules in HUVECs. In a subsequent study, we clarified that
Nogo-B increased TGF-β production in endothelial cells. As a cytokine, TGF-β is known to maintain cell morphology and restrict cell proliferation [
29]. Nevertheless, complicated and even conflicting roles of TGF-β have been observed in various tumor models. As shown in the study by Krishnan et al., TGF-β induces the expression of VEGF and placental growth factor (PlGF) under normoxic and hypoxic conditions, defining a potential indirect proangiogenic activity of TGF-β in glioblastoma [
30]. In the present study,
Nogo-B-enhanced TGF-β secretion in endothelial cells activated Smad signaling in neighboring tumor cells and, thus, suppressed tumor growth.
Nogo-B is a component of the endoplasmic reticulum, which is required for protein synthesis. Loss of
Nogo-B is sufficient to affect ER morphology [
31], which might at least partially explain why
Nogo-B depletion in endothelial cells reduced the TGF-β secretion. Additional mechanistic studies are warranted to delineate the detailed molecular mechanism.
In summary, our study reported the differential expression of endothelial Nogo-B in tumors and the correlation between endothelial Nogo-B expression and patient survival, providing the first evidence for the clinical significance of endothelial Nogo-B. Furthermore, the functional studies revealed that endothelial Nogo-B enhanced TGF-β secretion and, thus, suppressed cancer growth by activating Smad signaling in neighboring cancer cells (Graphical Abstract). These data further unraveled the complexity of the tumor microenvironment and raised necessary concerns regarding anti-angiogenesis therapy.