Distinct Dynamics of Migratory Response to PD-1 and CTLA-4 Blockade Reveals New Mechanistic Insights for Potential T-Cell Reinvigoration following Immune Checkpoint Blockade
Abstract
1. Introduction
2. Materials and Methods
3. Results
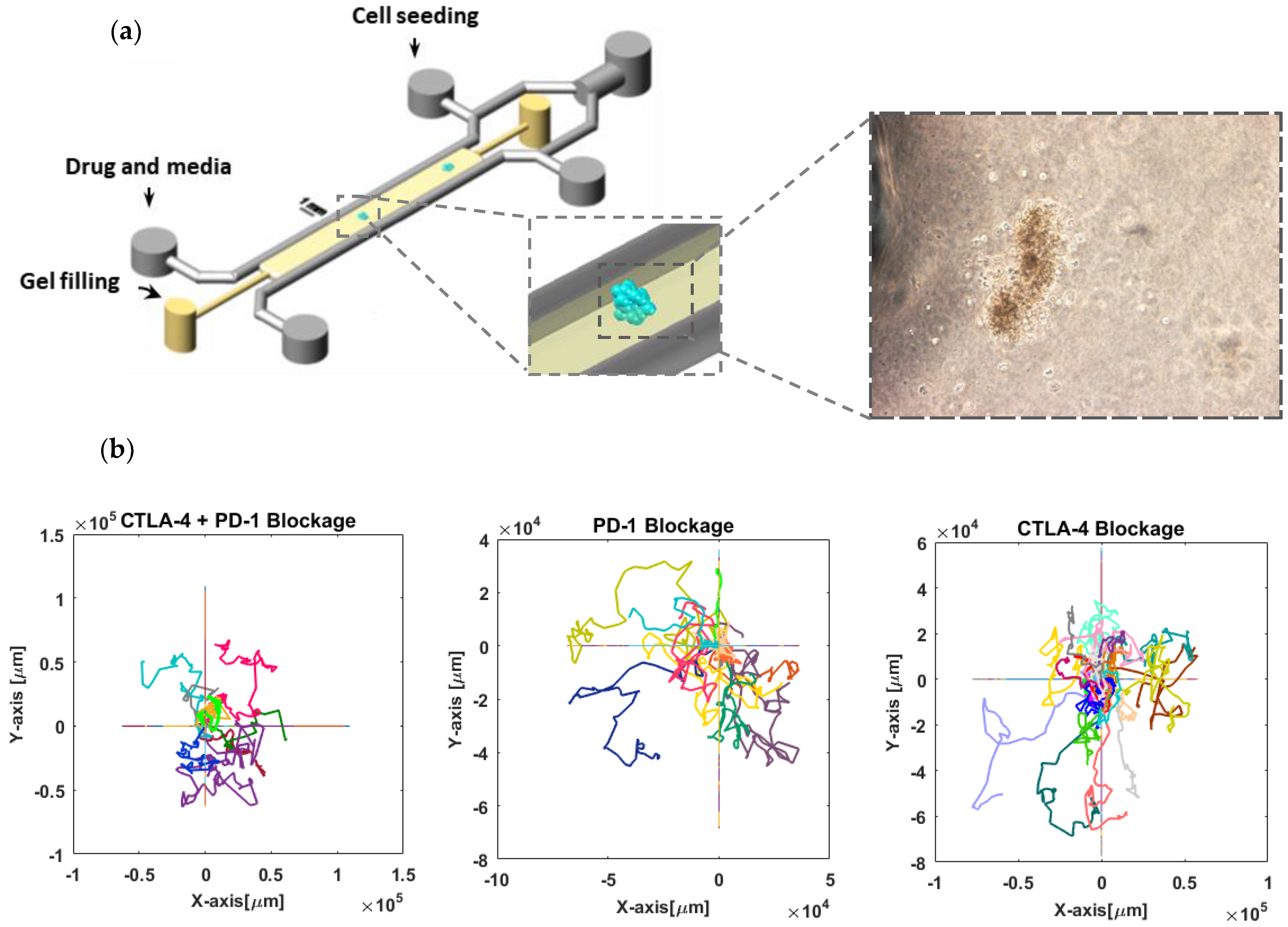
3.1. Stochastic Modeling of T-Cell Migration
3.2. Dynamical Modeling of Lymphocyte Activity in the Presence of Checkpoint Inhibitors
4. Discussion
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Appendix A
Experimental Background

| Experiment | Spheroid Population | Tumor Cell Ratio * | Tumor Death Ratio ** |
|---|---|---|---|
| Control | 0.22 | 0.090 | |
| PD-1 | 0.16 | 0.196 | |
| CTLA4 | 0.16 | 0.483 | |
| Combo | 0.13 | 0.433 |
Appendix B
Evaluation of Parameter Estimation for AR-1 Model of T-Cell Migration


Appendix C
Appendix C.1. Parameter Inference for the Random Walk Model of Lymphocyte Migration
Appendix C.2. Estimation of Parameters for the Dynamical Model of Checkpoint-Induced Lymphocyte Inactivation
Appendix C.3. Improving the Initial Values of Lymphocyte Subpopulations (Correction of α and β Parameters)
References
- Wei, S.C.; Duffy, C.R.; Allison, J.P. Fundamental Mechanisms of Immune Checkpoint Blockade Therapy. Cancer Discov. 2018, 8, 1069–1086. [Google Scholar] [CrossRef]
- Boomer, J.S.; Green, J.M. An Enigmatic Tail of CD28 Signaling. Csh. Perspect. Biol. 2010, 2. [Google Scholar] [CrossRef]
- Walker, L.S.K.; Sansom, D.M. Confusing Signals: Recent Progress in CTLA-4 Biology. Trends Immunol. 2015, 36, 63–70. [Google Scholar] [CrossRef]
- Henderson, V.; Smith, B.; Burton, L.J.; Randle, D.; Morris, M.; Odero-Marah, V.A. Snail Promotes Cell Migration through PI3K/AKT-Dependent Rac1 Activation as Well as PI3K/AKT-Independent Pathways during Prostate Cancer Progression. Cell Adhes. Migr. 2015, 9, 255–264. [Google Scholar] [CrossRef]
- Carlier, M.F.; Nioche, P.; Broutin-L’hermite, I.; Boujemaa, R.; Le, C.C.; Egile, C.; Garbay, C.; Ducruix, A.; Sansonetti, P.; Pantaloni, D. GRB2 Links Signaling to Actin Assembly by Enhancing Interaction of Neural Wiskott-Aldrich Syndrome Protein (N-WASp) with Actin-Related Protein (ARP2/3) Complex. J. Biol. Chem. 2000, 275, 21946–21952. [Google Scholar] [CrossRef]
- Salazar-Fontana, L.I.; Barr, V.; Samelson, L.E.; Bierer, B.E. CD28 Engagement Promotes Actin Polymerization Through the Activation of the Small Rho GTPase Cdc42 in Human T Cells. J. Immunol. 2003, 171, 2225–2232. [Google Scholar] [CrossRef]
- Schneider, H.; Valk, E.; Leung, R.; Rudd, C.E. CTLA-4 Activation of Phosphatidylinositol 3-Kinase (PI 3-K) and Protein Kinase B (PKB/AKT) Sustains T-Cell Anergy without Cell Death. PLoS ONE 2008, 3, e3842. [Google Scholar] [CrossRef]
- Xue, G.; Hemmings, B.A. PKB/Akt-Dependent Regulation of Cell Motility. J. Natl. Cancer. 2013, 105, 393–404. [Google Scholar] [CrossRef]
- García-Aranda, M.; Redondo, M. Targeting Protein Kinases to Enhance the Response to Anti-PD-1/PD-L1 Immunotherapy. Int. J. Mol. Sci. 2019, 20, 2296. [Google Scholar] [CrossRef]
- Huang, C.; Jacobson, K.; Schaller, M.D. MAP Kinases and Cell Migration. J. Cell Sci. 2004, 117, 4619–4628. [Google Scholar] [CrossRef]
- Patsoukis, N.; Bardhan, K.; Chatterjee, P.; Sari, D.; Liu, B.; Bell, L.N.; Karoly, E.D.; Freeman, G.J.; Petkova, V.; Seth, P.; et al. PD-1 Alters T-Cell Metabolic Reprogramming by Inhibiting Glycolysis and Promoting Lipolysis and Fatty Acid Oxidation. Nat. Commun. 2015, 6, 1–13. [Google Scholar] [CrossRef] [PubMed]
- Dempsey, L.A. PD-1 Targets CD28. Nat. Immunol. 2017, 18, 487. [Google Scholar] [CrossRef] [PubMed]
- Ganesan, S.; Mehnert, J. Biomarkers for Response to Immune Checkpoint Blockade. Annu. Rev. Cancer Biol. 2020, 4, 331–351. [Google Scholar] [CrossRef]
- Huang, R.Y.; Francois, A.; McGray, A.R.; Miliotto, A.; Odunsi, K. Compensatory Upregulation of PD-1, LAG-3, and CTLA-4 Limits the Efficacy of Single-Agent Checkpoint Blockade in Metastatic Ovarian Cancer. OncoImmunology 2017, 6, 1–13. [Google Scholar] [CrossRef] [PubMed]
- Wherry, E.J. T Cell Exhaustion. Nat. Immunol. 2011, 12, 492–499. [Google Scholar] [CrossRef]
- Koyama, S.; Akbay, E.A.; Li, Y.Y.; Herter-Sprie, G.S.; Buczkowski, K.A.; Richards, W.G.; Gandhi, L.; Redig, A.J.; Rodig, S.J.; Asahina, H.; et al. Adaptive Resistance to Therapeutic PD-1 Blockade Is Associated with Upregulation of Alternative Immune Checkpoints. Nat. Commun. 2016, 7, 10501. [Google Scholar] [CrossRef] [PubMed]
- Gide, T.N.; Quek, C.; Menzies, A.M.; Tasker, A.T.; Shang, P.; Holst, J.; Madore, J.; Lim, S.Y.; Velickovic, R.; Wongchenko, M.; et al. Distinct Immune Cell Populations Define Response to Anti-PD-1 Monotherapy and Anti-PD-1/Anti-CTLA-4 Combined Therapy. Cancer Cell 2019, 35, 238–255.e6. [Google Scholar] [CrossRef]
- Wei, S.C.; Levine, J.H.; Wargo, J.A.; Pe, D.; Allison, J.P.; Wei, S.C.; Levine, J.H.; Cogdill, A.P.; Zhao, Y.; Anang, N.A.S.; et al. Distinct Cellular Mechanisms Underlie Anti-CTLA-4 and Anti-PD-1 Checkpoint Blockade Distinct Cellular Mechanisms Underlie. Cell 2017, 170, 1120–1125.e17. [Google Scholar] [CrossRef]
- Parry, R.V.; Chemnitz, J.M.; Frauwirth, K.A.; Lanfranco, A.R.; Braunstein, I.; Kobayashi, S.V.; Linsley, P.S.; Thompson, C.B.; Riley, J.L. CTLA-4 and PD-1 Receptors Inhibit T-Cell Activation by Distinct Mechanisms. Mol. Cell. Biol. 2005, 25, 9543–9553. [Google Scholar] [CrossRef]
- Robert, L.; Harview, C.; Emerson, R.; Wang, X.; Mok, S.; Homet, B.; Comin-Anduix, B.; Koya, R.C.; Robins, H.; Tumeh, P.C.; et al. Distinct Immunological Mechanisms of CTLA-4 and PD-1 Blockade Revealed by Analyzing TCR Usage in Blood Lymphocytes. OncoImmunology 2014, 3, 3–6. [Google Scholar] [CrossRef]
- Bally, A.P.R.; Austin, J.W.; Boss, J.M. Genetic and Epigenetic Regulation of PD-1 Expression. J. Immunol. 2016, 196, 2431. [Google Scholar] [CrossRef] [PubMed]
- Schneider, H.; Rudd, C.E. Diverse Mechanisms Regulate the Surface Expression of Immunotherapeutic Target CTLA-4. Front. Immunol. 2014, 5, 619. [Google Scholar] [CrossRef] [PubMed]
- Series, W.P.; Sterman, J.D. System Dynamics: Systems Thinking and Modeling for a Complex World. Eur. J. Comput. Sci. 2003, 21, 35–39. [Google Scholar]
- Serre, R.; Benzekry, S.; Padovani, L.; Meille, C.; Andre, N.; Ciccolini, J.; Barlesi, F.; Muracciole, X.; Barbolosi, D. Mathematical Modeling of Cancer Immunotherapy and Its Synergy with Radiotherapy. Cancer Res. 2016, 76, 4931–4940. [Google Scholar] [CrossRef]
- Kosinsky, Y.; Dovedi, S.J.; Peskov, K.; Voronova, V.; Chu, L.; Tomkinson, H.; Al-Huniti, N.; Stanski, D.R.; Helmlinger, G. Radiation and PD-(L)1 Treatment Combinations: Immune Response and Dose Optimization via a Predictive Systems Model. J. ImmunoTherapy Cancer 2018, 6. [Google Scholar] [CrossRef] [PubMed]
- Lai, X.; Stiff, A.; Duggan, M.; Wesolowski, R.; Carson, W.E.; Friedman, A. Modeling Combination Therapy for Breast Cancer with BET and Immune Checkpoint Inhibitors. Proc. Natl. Acad. Sci. USA 2018, 115, 5534–5539. [Google Scholar] [CrossRef]
- Lai, X.; Friedman, A. Combination Therapy for Melanoma with BRAF/MEK Inhibitor and Immune Checkpoint Inhibitor: A Mathematical Model. BMC Syst. Biol. 2017, 11, 1–18. [Google Scholar] [CrossRef]
- Butner, J.D.; Wang, Z.; Elganainy, D.; Feghali, K.A.A.; Plodinec, M.; Calin, G.A.; Dogra, P.; Nizzero, S.; Ramírez, J.R.; Martin, G.V.; et al. A Mathematical Model for the Quantification of a Patient’s Sensitivity to Checkpoint Inhibitors and Long-Term Tumour Burden. Nat. Biomed. Eng. 2021, 5, 297. [Google Scholar] [CrossRef]
- Butner, J.D.; Martin, G.V.; Wang, Z.; Corradetti, B.; Ferrari, M.; Esnaola, N.; Chung, C.; Hong, D.S.; Welsh, J.W.; Hasegawa, N.; et al. Early Prediction of Clinical Response to Checkpoint Inhibitor Therapy in Human Solid Tumors through Mathematical Modeling. eLife 2021, 10. [Google Scholar] [CrossRef]
- Wang, H.; Milberg, O.; Bartelink, I.H.; Vicini, P.; Wang, B.; Narwal, R.; Roskos, L.; Santa-Maria, C.A.; Popel, A.S. In Silico Simulation of a Clinical Trial with Anti-CTLA-4 and Anti-PD-L1 Immunotherapies in Metastatic Breast Cancer Using a Systems Pharmacology Model. R. Soc. Open Sci. 2019, 6, 190366. [Google Scholar] [CrossRef]
- Khajanchi, S.; Banerjee, S. Influence of Multiple Delays in Brain Tumor and Immune System Interaction with T11 Target Structure as a Potent Stimulator. Math. Biosci. 2018, 302, 116–130. [Google Scholar] [CrossRef] [PubMed]
- Khajanchi, S. The Impact of Immunotherapy on a Glioma Immune Interaction Model. Chaos Solitons Fractals 2021, 152, 111346. [Google Scholar] [CrossRef]
- Khajanchi, S.; Banerjee, S. Quantifying the Role of Immunotherapeutic Drug T11 Target Structure in Progression of Malignant Gliomas: Mathematical Modeling and Dynamical Perspective. Math. Biosci. 2017, 289, 69–77. [Google Scholar] [CrossRef] [PubMed]
- Nishino, M.; Ramaiya, N.H.; Hatabu, H.; Hodi, F.S. Monitoring Immune-Checkpoint Blockade: Response Evaluation and Biomarker Development. Nat. Rev. Clin. Oncol. 2017, 14, 655–668. [Google Scholar] [CrossRef]
- Robert, L.; Tsoi, J.; Wang, X.; Emerson, R.; Homet, B.; Chodon, T.; Mok, S.; Huang, R.R.; Cochran, A.J.; Comin-Anduix, B.; et al. CTLA4 Blockade Broadens the Peripheral T-Cell Receptor Repertoire. Clin. Cancer Res. 2014, 20, 2424–2432. [Google Scholar] [CrossRef]
- Pai, C.C.S.; Simons, D.M.; Lu, X.; Evans, M.; Wei, J.; Wang, Y.H.; Chen, M.; Huang, J.; Park, C.; Chang, A.; et al. Tumor-Conditional Anti-CTLA4 Uncouples Antitumor Efficacy from Immunotherapy-Related Toxicity. J. Clin. Investig. 2019, 129, 349–363. [Google Scholar] [CrossRef]
- Huang, R.R.; Jalil, J.; Economou, J.S.; Chmielowski, B.; Koya, R.C.; Mok, S.; Sazegar, H.; Seja, E.; Villanueva, A.; Gomez-Navarro, J.; et al. CTLA4 Blockade Induces Frequent Tumor Infiltration by Activated Lymphocytes Regardless of Clinical Responses in Humans. Clin. Cancer Res. 2011, 17, 4101–4109. [Google Scholar] [CrossRef]
- Gangaev, A.; Rozeman, E.A.; Rohaan, M.W.; Isaeva, O.I.; Patiwael, S.; van den Berg, J.H.; Ribas, A.; Schadendorf, D.; Schilling, B.; Philips, D.; et al. Differential Effects of PD-1 and CTLA-4 Blockade on the Melanoma-Reactive CD8 T Cell Response. Proc. Natl. Acad. Sci. USA 2021, 118, e2102849118. [Google Scholar] [CrossRef]
- Seidel, J.A.; Otsuka, A.; Kabashima, K. Anti-PD-1 and Anti-CTLA-4 Therapies in Cancer: Mechanisms of Action, Efficacy, and Limitations. Front. Oncol. 2018, 8, 1–14. [Google Scholar] [CrossRef]
- Farahat, W.A.; Wood, L.B.; Zervantonakis, I.K.; Schor, A.; Ong, S.; Neal, D.; Kamm, R.D.; Asada, H.H. Ensemble Analysis of Angiogenic Growth in Three-Dimensional Microfluidic Cell Cultures. PLoS ONE 2012, 7, e37333. [Google Scholar] [CrossRef]
- Aref, A.R.; Huang, R.Y.-J.; Yu, W.; Chua, K.-N.; Sun, W.; Tu, T.-Y.; Bai, J.; Sim, W.-J.; Zervantonakis, I.K.; Thiery, J.P.; et al. Screening Therapeutic EMT Blocking Agents in a Three-Dimensional Microenvironment. Integr. Biol. 2013, 5, 381–389. [Google Scholar] [CrossRef] [PubMed]
- Jenkins, R.W.; Aref, A.R.; Lizotte, P.H.; Ivanova, E.; Stinson, S.; Zhou, C.W.; Bowden, M.; Deng, J.; Liu, H.; Miao, D.; et al. Ex Vivo Profiling of PD-1 Blockade Using Organotypic Tumor Spheroids. Cancer Discov. 2018, 8, 196–215. [Google Scholar] [CrossRef] [PubMed]
- Metzner, C.; Mark, C.; Steinwachs, J.; Lautscham, L.; Stadler, F.; Fabry, B. Superstatistical Analysis and Modelling of Heterogeneous Random Walks. Nat. Commun. 2015, 6, 7516. [Google Scholar] [CrossRef]
- Binnewies, M.; Roberts, E.W.; Kersten, K.; Chan, V.; Fearon, D.F.; Merad, M.; Coussens, L.M.; Gabrilovich, D.I.; Ostrand-Rosenberg, S.; Hedrick, C.C.; et al. Understanding the Tumor Immune Microenvironment (TIME) for Effective Therapy. Nat. Med. 2018, 24, 541–550. [Google Scholar] [CrossRef]
- Shariatpanahi, S.P.; Shariatpanahi, S.P.; Madjidzadeh, K.; Hassan, M.; Abedi-Valugerdi, M. Mathematical Modeling of Tumor-Induced Immunosuppression by Myeloid-Derived Suppressor Cells: Implications for Therapeutic Targeting Strategies. J. Theor. Biol. 2018, 442, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Valk, E.; Rudd, C.E.; Schneider, H. CTLA-4 Trafficking and Surface Expression. Trends Immunol. 2008, 29, 272–279. [Google Scholar] [CrossRef]
- Tormoen, G.W.; Crittenden, M.R.; Gough, M.J. Role of the Immunosuppressive Microenvironment in Immunotherapy. Adv. Radiat. Oncol. 2018, 3, 520–526. [Google Scholar] [CrossRef]
- Khailaie, S.; Rowshanravan, B.; Robert, P.A.; Waters, E.; Halliday, N.; Badillo Herrera, J.D.; Walker, L.S.K.; Sansom, D.M.; Meyer-Hermann, M. Characterization of CTLA4 Trafficking and Implications for Its Function. Biophys. J. 2018, 115, 1330–1343. [Google Scholar] [CrossRef]
- Kansy, B.A.; Concha-Benavente, F.; Srivastava, R.M.; Jie, H.B.; Shayan, G.; Lei, Y.; Moskovitz, J.; Moy, J.; Li, J.; Brandau, S.; et al. PD-1 Status in CD8+ T Cells Associates with Survival and Anti-PD-1 Therapeutic Outcomes in Head and Neck Cancer. Cancer Res. 2017, 77, 6353–6364. [Google Scholar] [CrossRef]
- Huang, A.C.; Postow, M.A.; Orlowski, R.J.; Mick, R.; Bengsch, B.; Manne, S.; Xu, W.; Harmon, S.; Giles, J.R.; Wenz, B.; et al. T-Cell Invigoration to Tumour Burden Ratio Associated with Anti-PD-1 Response. Nat. Publ. Group 2017, 545, 60–65. [Google Scholar] [CrossRef]
- Joseph, R.W.; Elassaiss-Schaap, J.; Kefford, R.; Hwu, W.J.; Wolchok, J.D.; Joshua, A.M.; Ribas, A.; Hodi, F.S.; Hamid, O.; Robert, C.; et al. Baseline Tumor Size Is an Independent Prognostic Factor for Overall Survival in Patients with Melanoma Treated with Pembrolizumab. Clin. Cancer Res. 2018, 24, 4960–4967. [Google Scholar] [CrossRef] [PubMed]
- Borcoman, E.; Nandikolla, A.; Long, G.; Goel, S.; Le Tourneau, C. Patterns of Response and Progression to Immunotherapy. Am. Soc. Clin. Oncol. Educ. Book 2018, 38, 169–178. [Google Scholar] [CrossRef] [PubMed]
- Kamada, T.; Togashi, Y.; Tay, C.; Ha, D.; Sasaki, A.; Nakamura, Y.; Sato, E.; Fukuoka, S.; Tada, Y.; Tanaka, A.; et al. PD-1 + Regulatory T Cells Amplified by PD-1 Blockade Promote Hyperprogression of Cancer. Proc. Natl. Acad. Sci. USA 2019, 116, 9999–10008. [Google Scholar] [CrossRef] [PubMed]
- Manjarrez-Orduño, N.; Menard, L.C.; Kansal, S.; Fischer, P.; Kakrecha, B.; Jiang, C.; Cunningham, M.; Greenawalt, D.; Patel, V.; Yang, M.; et al. Circulating T Cell Subpopulations Correlate with Immune Responses at the Tumor Site and Clinical Response to PD1 Inhibition in Non-Small Cell Lung Cancer. Front. Immunol. 2018, 9, 1–9. [Google Scholar] [CrossRef]
- Durham, J.N.; Rytlewski, J.A.; Le, D.T.; Robins, H.S.; Jaffee, E.M.; Laheru, D.A.; Hopkins, A.C.; Yarchoan, M.; Lutz, E.R.; Yusko, E.C. T Cell Receptor Repertoire Features Associated with Survival in Immunotherapy-Treated Pancreatic Ductal Adenocarcinoma. JCI Insight 2018, 3, 1–10. [Google Scholar] [CrossRef]
- Ho, W.J.; Yarchoan, M.; Hopkins, A.; Mehra, R.; Grossman, S.; Kang, H. Association between Pretreatment Lymphocyte Count and Response to PD1 Inhibitors in Head and Neck Squamous Cell Carcinomas. J. ImmunoTherapy Cancer 2018, 6, 1–8. [Google Scholar] [CrossRef]
- Banerjee, S.; Khajanchi, S.; Chaudhuri, S. A Mathematical Model to Elucidate Brain Tumor Abrogation by Immunotherapy with T11 Target Structure. PLoS ONE 2015, 10. [Google Scholar] [CrossRef]
- Khajanchi, S.; Nieto, J.J. Spatiotemporal Dynamics of a Glioma Immune Interaction Model. Sci. Rep. 2021, 11. [Google Scholar] [CrossRef]
- Branco, J.R.; Ferreira, J.A.; de Oliveira, P. Mathematical Modeling of Efficient Protocols to Control Glioma Growth. Math. Biosci. 2014, 255, 83–90. [Google Scholar] [CrossRef]



| Parameter | Definition | OptimalValue * | Unit ** |
|---|---|---|---|
| σ | Tumor cell death rate | ||
| CTLA-4-induced immune cell inactivation rate | |||
| β | PD-1-induced immune cell inactivation rate | ||
| ξ | Non-checkpoint-induced immune cell inactivation rate | ||
| α | PD-1 expression rate | 0.22 |
| Type of Treatment | Spheroids’ Active Cell Number (Average) |
|---|---|
| Anti-CTLA-4 | 65.6 ± 9.38 |
| Anti-PD-1 | 19.3 ± 1.64 |
| Combo | 61.6 ± 7.75 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Safaeifard, F.; Goliaei, B.; Aref, A.R.; Foroughmand-Araabi, M.-H.; Goliaei, S.; Lorch, J.; Jenkins, R.W.; Barbie, D.A.; Shariatpanahi, S.P.; Rüegg, C. Distinct Dynamics of Migratory Response to PD-1 and CTLA-4 Blockade Reveals New Mechanistic Insights for Potential T-Cell Reinvigoration following Immune Checkpoint Blockade. Cells 2022, 11, 3534. https://doi.org/10.3390/cells11223534
Safaeifard F, Goliaei B, Aref AR, Foroughmand-Araabi M-H, Goliaei S, Lorch J, Jenkins RW, Barbie DA, Shariatpanahi SP, Rüegg C. Distinct Dynamics of Migratory Response to PD-1 and CTLA-4 Blockade Reveals New Mechanistic Insights for Potential T-Cell Reinvigoration following Immune Checkpoint Blockade. Cells. 2022; 11(22):3534. https://doi.org/10.3390/cells11223534
Chicago/Turabian StyleSafaeifard, Fateme, Bahram Goliaei, Amir R. Aref, Mohammad-Hadi Foroughmand-Araabi, Sama Goliaei, Jochen Lorch, Russell W. Jenkins, David A. Barbie, Seyed Peyman Shariatpanahi, and Curzio Rüegg. 2022. "Distinct Dynamics of Migratory Response to PD-1 and CTLA-4 Blockade Reveals New Mechanistic Insights for Potential T-Cell Reinvigoration following Immune Checkpoint Blockade" Cells 11, no. 22: 3534. https://doi.org/10.3390/cells11223534
APA StyleSafaeifard, F., Goliaei, B., Aref, A. R., Foroughmand-Araabi, M.-H., Goliaei, S., Lorch, J., Jenkins, R. W., Barbie, D. A., Shariatpanahi, S. P., & Rüegg, C. (2022). Distinct Dynamics of Migratory Response to PD-1 and CTLA-4 Blockade Reveals New Mechanistic Insights for Potential T-Cell Reinvigoration following Immune Checkpoint Blockade. Cells, 11(22), 3534. https://doi.org/10.3390/cells11223534









