The Impact of CC16 on Pulmonary Epithelial-Driven Host Responses during Mycoplasma pneumoniae Infection in Mouse Tracheal Epithelial Cells
Abstract
:1. Introduction
2. Results
2.1. WT MTECs Have Decreased Apical Proteins Secretions during Mp Infection
2.2. WT MTECs Have Decreased Secretion of Antioxidant Proteins during Mp Infection
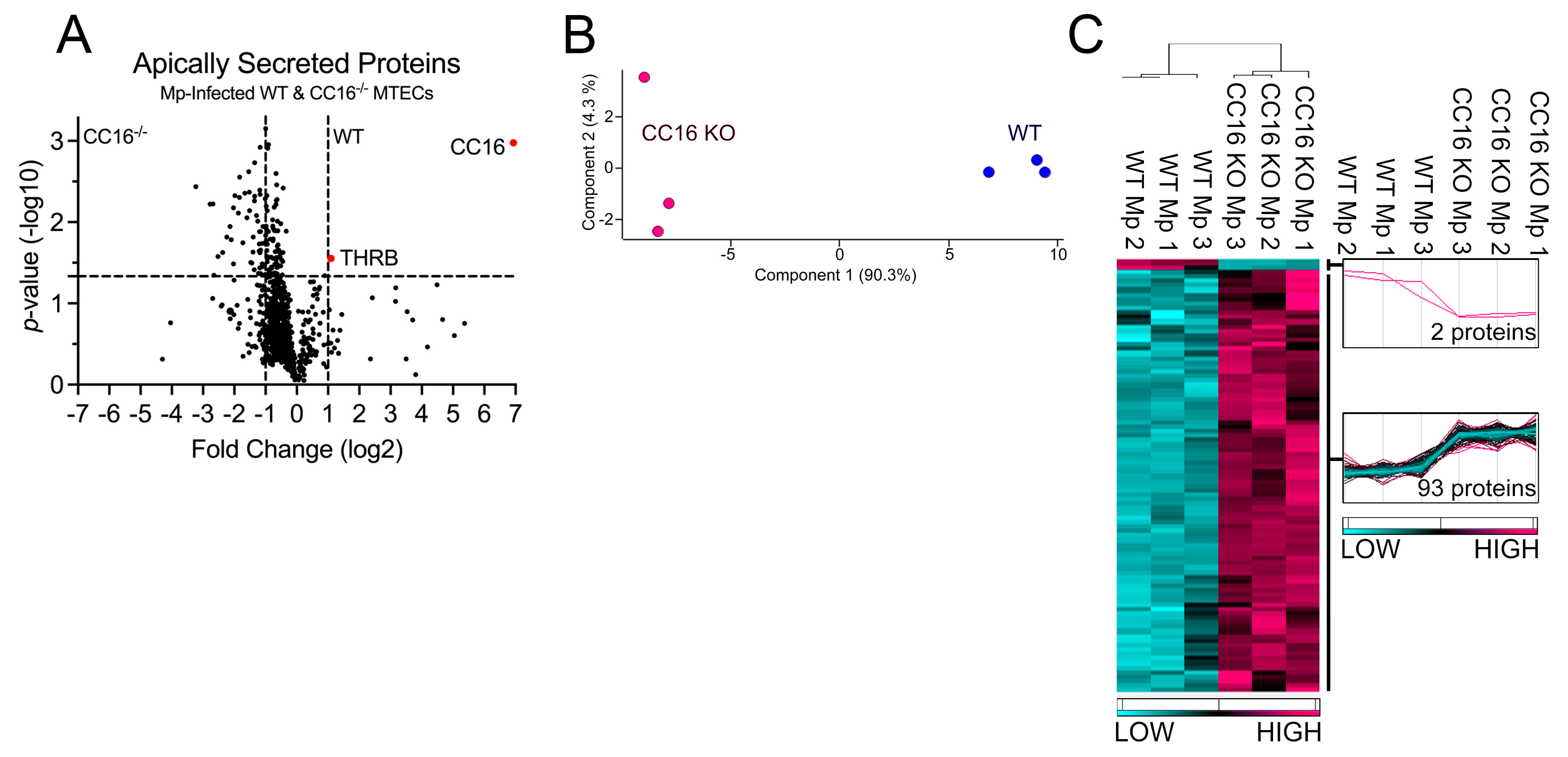
2.3. CC16−/− MTECs Have Increased Apical Protein Secretions during Mp Infection
2.4. CC16−/− MTECs Have Increased Secretion of Airway Remodeling and Bacterial Invasion Proteins during Mp Infection
2.5. CC16 Mediates Pulmonary Epithelial Cell Apical Protein Secretion during Mp Infection
2.6. CC16 Deficiency Increases Pulmonary Epithelial-Driven Airway Remodeling during Mp Infection
2.7. Validation of Apically Secreted Proteins in MTECs and Mouse Lung Tissue by Western Blot
3. Discussion
4. Conclusions
5. Methods
5.1. WT and CC16−/− Mice
5.2. Mouse Tracheal Epithelial Cell (MTEC) Isolation and Culturing
5.3. Mp Infection for WT and CC16−/− MTECs and Mice
5.4. Collection of Apically Secreted Proteins from MTECs
5.5. In-Solution Tryptic Digestion
5.6. Mass Spectrometry and Spectrum Count Data Processing
5.7. Label-Free Quantitative Proteomics
5.8. Determination of GRB2 and LAMB3 Expression by Western Blotting
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Broeckaert, F.; Bernard, A. Clara cell secretory protein (CC16): Characteristics and perspectives as lung peripheral biomarker. Clin. Exp. Allergy 2000, 30, 469–475. [Google Scholar] [CrossRef] [Green Version]
- Mukherjee, A.B.; Kundu, G.C.; Mantile-Selvaggi, G.; Yuan, C.J.; Mandal, A.K.; Chattopadhyay, S.; Zheng, F.; Pattabiraman, N.; Zhang, Z. Uteroglobin: A novel cytokine? Cell. Mol. Life Sci. CMLS 1999, 55, 771–787. [Google Scholar] [CrossRef]
- Mukherjee, A.B.; Zhang, Z.; Chilton, B.S. Uteroglobin: A Steroid-Inducible Immunomodulatory Protein That Founded the Secretoglobin Superfamily. Endocr. Rev. 2007, 28, 707–725. [Google Scholar] [CrossRef] [PubMed]
- Laucho-Contreras, M.E.; Polverino, F.; Gupta, K.; Taylor, K.L.; Kelly, E.; Pinto-Plata, V.; Divo, M.; Ashfaq, N.; Petersen, H.; Stripp, B.; et al. Protective role for club cell secretory protein-16 (CC16) in the development of COPD. Eur. Respir. J. 2015, 45, 1544. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kraft, M.; Ledford, J.; Gozdz, J.; Li, X.A.; Francisco, D.; Manne, A.; Guerra, S.; Martinez, F.; Kaminski, N.; Wenzel, S.; et al. Club Cell Protein-16 modifies airway inflammation in asthma and is associated with significant clinical asthma outcomes. Eur. Respir. J. 2018, 52, PA5496. [Google Scholar] [CrossRef]
- Lin, J.; Li, J.; Shu, M.; Wu, W.; Zhang, W.; Dou, Q.; Wu, J.; Zeng, X. The rCC16 Protein Protects Against LPS-Induced Cell Apoptosis and Inflammatory Responses in Human Lung Pneumocytes. Front. Pharmacol. 2020, 11, 1060. [Google Scholar] [CrossRef] [PubMed]
- Zhou, R.; Qu, Y.; Huang, Q.; Sun, X.; Mu, D.; Li, X. Recombinant CC16 regulates inflammation, oxidative stress, apoptosis and autophagy via the inhibition of the p38MAPK signaling pathway in the brain of neonatal rats with sepsis. Brain Res. 2019, 1725, 146473. [Google Scholar] [CrossRef]
- Arsalane, K.; Broeckaert, F.; Knoops, B.; Wiedig, M.; Toubeau, G.; Bernard, A. Clara Cell Specific Protein (CC16) Expression after Acute Lung Inflammation Induced by Intratracheal Lipopolysaccharide Administration. Am. J. Respir. Crit. Care Med. 2000, 161, 1624–1630. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Mango, G.W.; Johnston, C.J.; Reynolds, S.D.; Finkelstein, J.N.; Plopper, C.G.; Stripp, B.R. Clara cell secretory protein deficiency increases oxidant stress response in conducting airways. Am. J. Physiol. 1998, 275, L348–L356. [Google Scholar] [CrossRef]
- Chowdhury, B.; Mantile-Selvaggi, G.; Kundu, G.C.; Miele, L.; Cordella-Miele, E.; Zhang, Z.; Mukherjee, A.B. Amino acid residues in alpha-helix-3 of human uteroglobin are critical for its phospholipase A2 inhibitory activity. Ann. N. Y. Acad. Sci. 2000, 923, 307–311. [Google Scholar] [CrossRef]
- Facchiano, A.; Cordella-Miele, E.; Miele, L.; Mukherjee, A.B. Inhibition of pancreatic phospholipase A2 activity by uteroglobin and antiflammin peptides: Possible mechanism of action. Life Sci. 1991, 48, 453–464. [Google Scholar] [CrossRef]
- Pang, M.; Yuan, Y.; Wang, D.; Li, T.; Wang, D.; Shi, X.; Guo, M.; Wang, C.; Zhang, X.; Zheng, G.; et al. Recombinant CC16 protein inhibits the production of pro-inflammatory cytokines via NF-κB and p38 MAPK pathways in LPS-activated RAW264.7 macrophages. Acta Biochim. Biophys. Sin. 2017, 49, 435–443. [Google Scholar] [CrossRef] [Green Version]
- Laucho-Contreras, M.E.; Polverino, F.; Rojas-Quintero, J.; Wang, X.; Owen, C.A. Club cell protein 16 (Cc16) deficiency increases inflamm-aging in the lungs of mice. Physiol. Rep. 2018, 6, e13797. [Google Scholar] [CrossRef] [Green Version]
- Pang, M.; Liu, H.Y.; Li, T.; Wang, D.; Hu, X.Y.; Zhang, X.R.; Yu, B.F.; Guo, R.; Wang, H.L. Recombinant club cell protein 16 (CC16) ameliorates cigarette smoke-induced lung inflammation in a murine disease model of COPD. Mol. Med. Rep. 2018, 18, 2198–2206. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hermans, C.; Bernard, A. Clara cell protein (CC16): Characteristics and potential applications as biomarker of lung toxicity. Biomarkers 1996, 1, 3–8. [Google Scholar] [CrossRef]
- Waites, K.B.; Xiao, L.; Liu, Y.; Balish, M.F.; Atkinson, T.P. Mycoplasma pneumoniae from the Respiratory Tract and Beyond. Clin. Microbiol. Rev. 2017, 30, 747–809. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Jiang, Z.; Li, S.; Zhu, C.; Zhou, R.; Leung, P.H.M. Mycoplasma pneumoniae Infections: Pathogenesis and Vaccine Development. Pathogens 2021, 10, 119. [Google Scholar] [CrossRef]
- Waites, K.B.; Talkington, D.F. Mycoplasma pneumoniae and Its Role as a Human Pathogen. Clin. Microbiol. Rev. 2004, 17, 697–728. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- He, J.; Liu, M.; Ye, Z.; Tan, T.; Liu, X.; You, X.; Zeng, Y.; Wu, Y. Insights into the pathogenesis of Mycoplasma pneumoniae (Review). Mol. Med. Rep. 2016, 14, 4030–4036. [Google Scholar] [CrossRef] [Green Version]
- Iannuzo, N.; Insel, M.; Marshall, C.; Pederson, W.P.; Addison, K.J.; Polverino, F.; Guerra, S.; Ledford, J.G. CC16 Deficiency in the Context of Early-Life Mycoplasma pneumoniae Infection Results in Augmented Airway Responses in Adult Mice. Infect. Immun. 2022, 90, e0054821. [Google Scholar] [CrossRef]
- Johnson, M.D.L.; Younis, U.S.; Menghani, S.V.; Addison, K.J.; Whalen, M.; Pilon, A.L.; Cress, A.E.; Polverino, F.; Romanoski, C.E.; Kraft, M.; et al. CC16 Binding to α(4)β(1) Integrin Protects against Mycoplasma pneumoniae Infection. Am. J. Respir. Crit. Care Med. 2021, 203, 1410–1418. [Google Scholar] [CrossRef] [PubMed]
- Guerra, S.; Halonen, M.; Vasquez, M.M.; Spangenberg, A.; Stern, D.A.; Morgan, W.J.; Wright, A.L.; Lavi, I.; Tares, L.; Carsin, A.E.; et al. Relation between circulating CC16 concentrations, lung function, and development of chronic obstructive pulmonary disease across the lifespan: A prospective study. Lancet Respir. Med. 2015, 3, 613–620. [Google Scholar] [CrossRef] [PubMed]
- Guerra, S.; Vasquez, M.M.; Spangenberg, A.; Halonen, M.; Martin, R.J. Club cell secretory protein in serum and bronchoalveolar lavage of patients with asthma. J. Allergy Clin. Immunol. 2016, 138, 932–934.e1. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Li, X.; Guerra, S.; Ledford, J.G.; Kraft, M.; Li, H.; Hastie, A.T.; Castro, M.; Denlinger, L.C.; Erzurum, S.C.; Fahy, J.V.; et al. Low CC16 mRNA Expression Levels in Bronchial Epithelial Cells Are Associated with Asthma Severity. Am. J. Respir. Crit. Care Med. 2023, 207, 438–451. [Google Scholar] [CrossRef]
- Rava, M.; Tares, L.; Lavi, I.; Barreiro, E.; Zock, J.P.; Ferrer, A.; Muniozguren, N.; Nadif, R.; Cazzoletti, L.; Kauffmann, F.; et al. Serum levels of Clara cell secretory protein, asthma, and lung function in the adult general population. J. Allergy Clin. Immunol. 2013, 132, 230–232. [Google Scholar] [CrossRef] [Green Version]
- Silva, G.E.; Sherrill, D.L.; Guerra, S.; Barbee, R.A. Asthma as a Risk Factor for COPD in a Longitudinal Study. Chest 2004, 126, 59–65. [Google Scholar] [CrossRef]
- Zhai, J.; Emond, M.J.; Spangenberg, A.; Stern, D.A.; Vasquez, M.M.; Blue, E.E.; Buckingham, K.J.; Sherrill, D.L.; Halonen, M.; Gibson, R.L.; et al. Club cell secretory protein and lung function in children with cystic fibrosis. J. Cyst. Fibros. 2022, 21, 811–820. [Google Scholar] [CrossRef]
- Rojas-Quintero, J.; Laucho-Contreras, M.E.; Wang, X.; Fucci, Q.-A.; Burkett, P.R.; Kim, S.-J.; Zhang, D.; Tesfaigzi, Y.; Li, Y.; Bhashyam, A.R.; et al. CC16 augmentation reduces exaggerated COPD-like disease in Cc16-deficient mice. JCI Insight 2023, 8, e130771. [Google Scholar] [CrossRef]
- Jung, C.-G.; Cao, T.B.T.; Quoc, Q.L.; Yang, E.-M.; Ban, G.-Y.; Park, H.-S. Role of club cell 16-kDa secretory protein in asthmatic airways. Clin. Exp. Allergy 2023, 53, 648–658. [Google Scholar] [CrossRef]
- Chen, M.; Xu, K.; He, Y.; Jin, J.; Mao, R.; Gao, L.; Zhang, Y.; Wang, G.; Gao, P.; Xie, M.; et al. CC16 as an Inflammatory Biomarker in Induced Sputum Reflects Chronic Obstructive Pulmonary Disease (COPD) Severity. Int. J. Chron. Obstruct. Pulmon. Dis. 2023, 18, 705–717. [Google Scholar] [CrossRef]
- Zhang, H.; Lipinski, A.A.; Liktor-Busa, E.; Smith, A.F.; Moutal, A.; Khanna, R.; Langlais, P.R.; Largent-Milnes, T.M.; Vanderah, T.W. The Effects of Repeated Morphine Treatment on the Endogenous Cannabinoid System in the Ventral Tegmental Area. Front. Pharmacol. 2021, 12, 632757. [Google Scholar] [CrossRef] [PubMed]
- Zheng, Z.; Huang, D.; Wang, J.; Zhao, K.; Zhou, Y.; Guo, Z.; Zhai, S.; Xu, H.; Cui, H.; Yao, H.; et al. QTLbase: An integrative resource for quantitative trait loci across multiple human molecular phenotypes. Nucleic Acids Res. 2019, 48, D983–D991. [Google Scholar] [CrossRef] [Green Version]
- Han, Y.; Jia, Q.; Jahani, P.S.; Hurrell, B.P.; Pan, C.; Huang, P.; Gukasyan, J.; Woodward, N.C.; Eskin, E.; Gilliland, F.D.; et al. Genome-wide analysis highlights contribution of immune system pathways to the genetic architecture of asthma. Nat. Commun. 2020, 11, 1776. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Shrine, N.; Guyatt, A.L.; Erzurumluoglu, A.M.; Jackson, V.E.; Hobbs, B.D.; Melbourne, C.A.; Batini, C.; Fawcett, K.A.; Song, K.; Sakornsakolpat, P.; et al. New genetic signals for lung function highlight pathways and chronic obstructive pulmonary disease associations across multiple ancestries. Nat. Genet. 2019, 51, 481–493. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Levin, S.W.; Butler, J.D.; Schumacher, U.K.; Wightman, P.D.; Mukherjee, A.B. Uteroglobin inhibits phospholipase A2 activity. Life Sci. 1986, 38, 1813–1819. [Google Scholar] [CrossRef]
- Miele, L.; Cordella-Miele, E.; Facchiano, A.; Mukherjee, A.B. Novel anti-inflammatory peptides from the region of highest similarity between uteroglobin and lipocortin I. Nature 1988, 335, 726–730. [Google Scholar] [CrossRef]
- Plopper, C.G.; Mango, G.W.; Hatch, G.E.; Wong, V.J.; Toskala, E.; Reynolds, S.D.; Tarkington, B.K.; Stripp, B.R. Elevation of susceptibility to ozone-induced acute tracheobronchial injury in transgenic mice deficient in Clara cell secretory protein. Toxicol. Appl. Pharmacol. 2006, 213, 74–85. [Google Scholar] [CrossRef]
- Zhai, J.; Insel, M.; Addison, K.J.; Stern, D.A.; Pederson, W.; Dy, A.; Rojas-Quintero, J.; Owen, C.A.; Sherrill, D.L.; Morgan, W.; et al. Club Cell Secretory Protein Deficiency Leads to Altered Lung Function. Am. J. Respir. Crit. Care Med. 2019, 199, 302–312. [Google Scholar] [CrossRef]
- Braido, F.; Riccio, A.M.; Guerra, L.; Gamalero, C.; Zolezzi, A.; Tarantini, F.; Giovanni, B.D.; Folli, C.; Descalzi, D.; Canonica, G.W. Clara cell 16 protein in COPD sputum: A marker of small airways damage? Respir. Med. 2007, 101, 2119–2124. [Google Scholar] [CrossRef] [Green Version]
- Wang, S.-Z.; Rosenberger, C.L.; Bao, Y.-X.; Stark, J.M.; Harrod, K.S. Clara Cell Secretory Protein Modulates Lung Inflammatory and Immune Responses to Respiratory Syncytial Virus Infection. J. Immunol. 2003, 171, 1051–1060. [Google Scholar] [CrossRef] [Green Version]
- Nomori, H.; Horio, H.; Fuyuno, G.; Kobayashi, R.; Morinaga, S.; Hirabayashi, Y. Protein 1 (Clara cell protein) serum levels in healthy subjects and patients with bacterial pneumonia. Am. J. Respir. Crit. Care Med. 1995, 152, 746–750. [Google Scholar] [CrossRef] [PubMed]
- Almuntashiri, S.; Zhu, Y.; Han, Y.; Wang, X.; Somanath, P.R.; Zhang, D. Club Cell Secreted Protein CC16: Potential Applications in Prognosis and Therapy for Pulmonary Diseases. J. Clin. Med. 2020, 9, 4039. [Google Scholar] [CrossRef]
- Periayah, M.H.; Halim, A.S.; Mat Saad, A.Z. Mechanism Action of Platelets and Crucial Blood Coagulation Pathways in Hemostasis. Int. J. Hematol. Oncol. Stem Cell Res. 2017, 11, 319–327. [Google Scholar] [PubMed]
- Terada, M.; Kelly, E.A.; Jarjour, N.N. Increased thrombin activity after allergen challenge: A potential link to airway remodeling? Am. J. Respir. Crit. Care Med. 2004, 169, 373–377. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Yuliani, F.S.; Chen, J.-Y.; Cheng, W.-H.; Wen, H.-C.; Chen, B.-C.; Lin, C.-H. Thrombin induces IL-8/CXCL8 expression by DCLK1-dependent RhoA and YAP activation in human lung epithelial cells. J. Biomed. Sci. 2022, 29, 95. [Google Scholar] [CrossRef]
- Paulsson, M.; Su, Y.C.; Ringwood, T.; Uddén, F.; Riesbeck, K. Pseudomonas aeruginosa uses multiple receptors for adherence to laminin during infection of the respiratory tract and skin wounds. Sci. Rep. 2019, 9, 18168. [Google Scholar] [CrossRef] [Green Version]
- Prince, O.A.; Krunkosky, T.M.; Sheppard, E.S.; Krause, D.C. Modelling persistent Mycoplasma pneumoniae infection of human airway epithelium. Cell. Microbiol. 2018, 20, e12810. [Google Scholar] [CrossRef]
- Elkhal, C.K.; Kean, K.M.; Parsonage, D.; Maenpuen, S.; Chaiyen, P.; Claiborne, A.; Karplus, P.A. Structure and proposed mechanism of L-α-glycerophosphate oxidase from Mycoplasma pneumoniae. FEBS J. 2015, 282, 3030–3042. [Google Scholar] [CrossRef]
- Chu, H.W.; Rino, J.G.; Wexler, R.B.; Campbell, K.; Harbeck, R.J.; Martin, R.J. Mycoplasma pneumoniae infection increases airway collagen deposition in a murine model of allergic airway inflammation. Am. J. Physiol. Lung Cell Mol. Physiol. 2005, 289, L125–L133. [Google Scholar] [CrossRef] [Green Version]
- Hong, S.J. The Role of Mycoplasma pneumoniae Infection in Asthma. Allergy Asthma Immunol. Res. 2012, 4, 59–61. [Google Scholar] [CrossRef] [Green Version]
- Ledford, J.; Guerra, S.; Martinez, F.; Kraft, M. Club cell secretory protein: A key mediator in Mycoplasma pneumoniae infection. Eur. Respir. J. 2018, 52, PA4972. [Google Scholar] [CrossRef]
- Kraft, M.; Adler, K.B.; Ingram, J.L.; Crews, A.L.; Atkinson, T.P.; Cairns, C.B.; Krause, D.C.; Chu, H.W. Mycoplasma pneumoniae induces airway epithelial cell expression of MUC5AC in asthma. Eur. Respir. J. 2008, 31, 43–46. [Google Scholar] [CrossRef] [PubMed]
- Biscardi, S.; Lorrot, M.; Marc, E.; Moulin, F.; Boutonnat-Faucher, B.; Heilbronner, C.; Iniguez, J.L.; Chaussain, M.; Nicand, E.; Raymond, J.; et al. Mycoplasma pneumoniae and Asthma in Children. Clin. Infect. Dis. 2004, 38, 1341–1346. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Nisar, N.; Guleria, R.; Kumar, S.; Chand Chawla, T.; Ranjan Biswas, N. Mycoplasma pneumoniae and its role in asthma. Postgrad. Med. J. 2007, 83, 100–104. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Benamar, M.; Harb, H.; Chen, Q.; Wang, M.; Chan, T.M.F.; Fong, J.; Phipatanakul, W.; Cunningham, A.; Ertem, D.; Petty, C.R.; et al. A common IL-4 receptor variant promotes asthma severity via a T(reg) cell GRB2-IL-6-Notch4 circuit. Allergy 2022, 77, 3377–3387. [Google Scholar] [CrossRef] [PubMed]
- Athari, S.S. Targeting cell signaling in allergic asthma. Signal Transduct. Target. Ther. 2019, 4, 45. [Google Scholar] [CrossRef] [Green Version]
- Wang, Q.; Hong, L.; Chen, M.; Shi, J.; Lin, X.; Huang, L.; Tang, T.; Guo, Y.; Yuan, X.; Jiang, S. Targeting M2 Macrophages Alleviates Airway Inflammation and Remodeling in Asthmatic Mice via miR-378a-3p/GRB2 Pathway. Front. Mol. Biosci. 2021, 8, 717969. [Google Scholar] [CrossRef]
- Bates, M.E.; Busse, W.W.; Bertics, P.J. Interleukin 5 Signals through Shc and Grb2 in Human Eosinophils. Am. J. Respir. Cell Mol. Biol. 1998, 18, 75–83. [Google Scholar] [CrossRef]
- Eisener-Dorman, A.F.; Lawrence, D.A.; Bolivar, V.J. Cautionary insights on knockout mouse studies: The gene or not the gene? Brain Behav. Immun. 2009, 23, 318–324. [Google Scholar] [CrossRef] [Green Version]
- Stripp, B.R.; Lund, J.; Mango, G.W.; Doyen, K.C.; Johnston, C.; Hultenby, K.; Nord, M.; Whitsett, J.A. Clara cell secretory protein: A determinant of PCB bioaccumulation in mammals. Am. J. Physiol. 1996, 271, L656–L664. [Google Scholar] [CrossRef]
- Eenjes, E.; Mertens, T.C.J.; Buscop-Van Kempen, M.J.; Van Wijck, Y.; Taube, C.; Rottier, R.J.; Hiemstra, P.S. A novel method for expansion and differentiation of mouse tracheal epithelial cells in culture. Sci. Rep. 2018, 8, 7349. [Google Scholar] [CrossRef] [Green Version]
- James, J.; Valuparampil Varghese, M.; Vasilyev, M.; Langlais, P.R.; Tofovic, S.P.; Rafikova, O.; Rafikov, R. Complex III Inhibition-Induced Pulmonary Hypertension Affects the Mitochondrial Proteomic Landscape. Int. J. Mol. Sci. 2020, 21, 5683. [Google Scholar] [CrossRef] [PubMed]
- Parker, S.S.; Krantz, J.; Kwak, E.A.; Barker, N.K.; Deer, C.G.; Lee, N.Y.; Mouneimne, G.; Langlais, P.R. Insulin Induces Microtubule Stabilization and Regulates the Microtubule Plus-End Tracking Protein Network in Adipocytes. Mol. Cell. Proteom. 2019, 18, 1363–1381. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sangam, S.; Sun, X.; Schwantes-An, T.H.; Yegambaram, M.; Lu, Q.; Shi, Y.; Cook, T.; Fisher, A.; Frump, A.L.; Coleman, A.; et al. SOX17 Deficiency Mediates Pulmonary Hypertension: At the Crossroads of Sex, Metabolism, and Genetics. Am. J. Respir. Crit. Care Med. 2023, 207, 1055–1069. [Google Scholar] [CrossRef] [PubMed]
- Wahl, J.R.; Vivek, A.; Palomino, S.M.; Almuslim, M.; Cottier, K.E.; Langlais, P.R.; Streicher, J.M.; Vanderah, T.W.; Liktor-Busa, E.; Largent-Milnes, T.M. Extracellular Alterations in pH and K+ Modify the Murine Brain Endothelial Cell Total and Phospho-Proteome. Pharmaceutics 2022, 14, 1469. [Google Scholar] [CrossRef] [PubMed]
- Kwak, E.A.; Pan, C.C.; Ramonett, A.; Kumar, S.; Cruz-Flores, P.; Ahmed, T.; Ortiz, H.R.; Lochhead, J.J.; Ellis, N.A.; Mouneimne, G.; et al. β(IV)-spectrin as a stalk cell-intrinsic regulator of VEGF signaling. Nat. Commun. 2022, 13, 1326. [Google Scholar] [CrossRef]
- Tyanova, S.; Temu, T.; Sinitcyn, P.; Carlson, A.; Hein, M.Y.; Geiger, T.; Mann, M.; Cox, J. The Perseus computational platform for comprehensive analysis of (prote)omics data. Nat. Methods 2016, 13, 731–740. [Google Scholar] [CrossRef]
- Tyanova, S.; Cox, J. Perseus: A Bioinformatics Platform for Integrative Analysis of Proteomics Data in Cancer Research. Methods Mol. Biol. 2018, 1711, 133–148. [Google Scholar] [CrossRef] [Green Version]
- Huang, D.W.; Sherman, B.T.; Lempicki, R.A. Systematic and integrative analysis of large gene lists using DAVID bioinformatics resources. Nat. Protoc. 2009, 4, 44–57. [Google Scholar] [CrossRef]






| Protein Name | Protein Abbreviation | Gene Abbreviation | SNP IDs in Lung Tissue a | Effective Allele | p-Value | Predicted Expression Change Due to SNP within Lung Tissue b |
|---|---|---|---|---|---|---|
| Heparin Sulfate Proteoglycan 2 | PGBM | HSPG2 | rs2229475 | T | 5.58 × 10−14 | Increase |
| rs115963344 | T | 4.84 × 10−13 | Increase | |||
| rs12724454 | A | 4.84 × 10−13 | Increase | |||
| rs12725566 | A | 4.84 × 10−13 | Increase | |||
| rs12737091 | T | 4.84 × 10−13 | Increase | |||
| rs12741617 | A | 4.84 × 10−13 | Increase | |||
| rs12742444 | T | 4.84 × 10−13 | Increase | |||
| Adapter Molecule CRK | CRK | CRK | rs16946807 | A | 3.74 × 10−8 | Decrease |
| rs34490967 | C | 5.86 × 10−8 | Decrease | |||
| rs7208768 | A | 8.46 × 10−8 | Decrease | |||
| rs11652967 | G | 9.72 × 10−8 | Decrease | |||
| rs35316912 | C | 1.16 × 10−7 | Decrease | |||
| Ras GTPase-Activating-Like Protein | IQGA2 | IQGAP2 | rs112872685 | A | 1.84 × 10−7 | Decrease |
| rs875541 | A | 9.95 × 10−6 | Decrease | |||
| rs10075621 | T | 1.40 × 10−5 | Decrease | |||
| rs4296785 | T | 1.42 × 10−5 | Decrease | |||
| rs4501323 | C | 1.42 × 10−5 | Decrease | |||
| rs4501324 | C | 1.42 × 10−5 | Decrease | |||
| Syndecan-4 | SDC4 | SDC4 | rs2267867 | A | 3.10 × 10−8 | Increase |
| Laminin Subunit Beta 3 | LAMB3 | LAMB3 | rs1534962 | A | 5.22 × 10−14 | Decrease |
| rs9429823 | G | 1.08 × 10−12 | Increase | |||
| rs2298928 | T | 2.05 × 10−12 | Increase | |||
| rs2298926 | A | 3.74 × 10−12 | Increase | |||
| rs1534962 | A | 5.63 × 10−12 | Decrease | |||
| rs9429823 | G | 5.12 × 10−9 | Increase | |||
| Plexin-B2 | PLXB2 | PLXNB2 | rs74933135 | A | 3.36 × 10−8 | Increase |
| rs117289563 | T | 1.05 × 10−7 | Increase | |||
| rs118067161 | T | 1.05 × 10−7 | Increase | |||
| rs142227764 | T | 1.05 × 10−7 | Increase | |||
| rs78854007 | A | 1.05 × 10−7 | Increase | |||
| rs146310944 | T | 1.05 × 10−7 | Increase | |||
| rs183166435 | C | 1.05 × 10−7 | Increase | |||
| rs117199604 | A | 2.33 × 10−7 | Increase | |||
| Growth Factor Receptor-Bound Protein 2 | GRB2 | GRB2 | rs12451296 | A | 3.33 × 10−5 | Increase |
| rs11649784 | T | 3.54 × 10−5 | Increase | |||
| rs12604003 | A | 3.54 × 10−5 | Increase | |||
| rs4789187 | G | 3.54 × 10−5 | Increase | |||
| rs9895739 | T | 3.66 × 10−5 | Increase | |||
| rs9909443 | T | 4.02 × 10−5 | Decrease | |||
| rs9901434 | G | 4.18 × 10−5 | Increase | |||
| Dynactin Subunit 1 | DCTN1 | DCTN1 | rs34215278 | T | 5.19 × 10−16 | Decrease |
| rs11555696 | A | 6.86 × 10−16 | Decrease | |||
| rs71418733 | T | 6.86 × 10−16 | Decrease | |||
| rs71418738 | G | 1.56 × 10−15 | Decrease | |||
| rs13021268 | T | 9.16 × 10−15 | Decrease | |||
| rs34988074 | C | 1.12 × 10−14 | Decrease | |||
| rs34522471 | G | 1.10 × 10−11 | Decrease | |||
| rs35241844 | C | 1.00 × 10−9 | Decrease | |||
| Protein BRICK1 | BRK1 | BRK1 | rs67342818 | A | 8.10 × 10−9 | Decrease |
| rs111284032 | T | 8.10 × 10−9 | Decrease | |||
| rs112180322 | T | 8.10 × 10−9 | Decrease | |||
| rs113378528 | A | 8.10 × 10−9 | Decrease | |||
| rs3774208 | G | 8.10 × 10−9 | Decrease | |||
| rs41464050 | G | 8.10 × 10−9 | Decrease | |||
| rs58862481 | A | 8.10 × 10−9 | Decrease | |||
| Clathrin Light Chain B | CLCB | CLTB | rs13181024 | T | 2.97 × 10−32 | Increase |
| rs13168842 | T | 2.97 × 10−32 | Increase | |||
| rs13154366 | A | 2.19 × 10−25 | Increase | |||
| rs13175787 | G | 2.19 × 10−25 | Increase | |||
| rs13168952 | A | 5.90 × 10−25 | Increase | |||
| rs13180938 | A | 2.47 × 10−23 | Increase | |||
| rs13181024 | T | 1.94 × 10−22 | Increase | |||
| rs13168842 | T | 1.94 × 10−22 | Increase | |||
| Integrin-Linked Protein Kinase | ILK | ILK | rs11826498 | C | 4.89 × 10−15 | Increase |
| rs11602107 | A | 1.17 × 10−14 | Increase | |||
| rs11605114 | A | 1.17 × 10−14 | Increase | |||
| rs2255405 | A | 1.56 × 10−14 | Increase | |||
| rs12451 | T | 2.62 × 10−14 | Increase | |||
| rs3741271 | C | 4.63 × 10−14 | Increase | |||
| rs2255538 | A | 9.88 × 10−14 | Increase | |||
| rs2292195 | T | 1.80 × 10−13 | Increase |
| Gene | SNP ID | Risk Allele | Expression Change | Effect of SNP on Phenotype | p-Value |
|---|---|---|---|---|---|
| GRB2 | rs9901434 | G | ↑ | ↓ FEV1 | 6.53 × 10−6 |
| ↑ asthma risk | 2.33 × 10−2 | ||||
| rs9895739 | T | ↑ | ↓ FEV1 | 4.26 × 10−6 | |
| ↑ asthma risk | 3.10 × 10−2 | ||||
| rs11649784 | T | ↑ | ↑ asthma risk | 2.22 × 10−2 | |
| rs12604003 | A | ↑ | ↑ asthma risk | 2.13 × 10−2 | |
| rs4789187 | G | ↑ | ↑ asthma risk | 2.22 × 10−2 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Iannuzo, N.; Dy, A.B.C.; Guerra, S.; Langlais, P.R.; Ledford, J.G. The Impact of CC16 on Pulmonary Epithelial-Driven Host Responses during Mycoplasma pneumoniae Infection in Mouse Tracheal Epithelial Cells. Cells 2023, 12, 1984. https://doi.org/10.3390/cells12151984
Iannuzo N, Dy ABC, Guerra S, Langlais PR, Ledford JG. The Impact of CC16 on Pulmonary Epithelial-Driven Host Responses during Mycoplasma pneumoniae Infection in Mouse Tracheal Epithelial Cells. Cells. 2023; 12(15):1984. https://doi.org/10.3390/cells12151984
Chicago/Turabian StyleIannuzo, Natalie, Alane Blythe C. Dy, Stefano Guerra, Paul R. Langlais, and Julie G. Ledford. 2023. "The Impact of CC16 on Pulmonary Epithelial-Driven Host Responses during Mycoplasma pneumoniae Infection in Mouse Tracheal Epithelial Cells" Cells 12, no. 15: 1984. https://doi.org/10.3390/cells12151984







