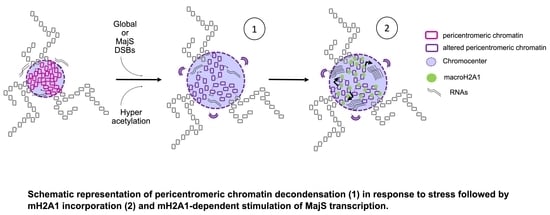
Recruitment of the Histone Variant MacroH2A1 to the Pericentric Region Occurs upon Chromatin Relaxation and Is Responsible for Major Satellite Transcriptional Regulation
Abstract
:1. Introduction
2. Materials and Methods
3. Results
3.1. The Histone Variant mH2A1 Accumulates at Pericentric Heterochromatin in Mouse Senescent Cells
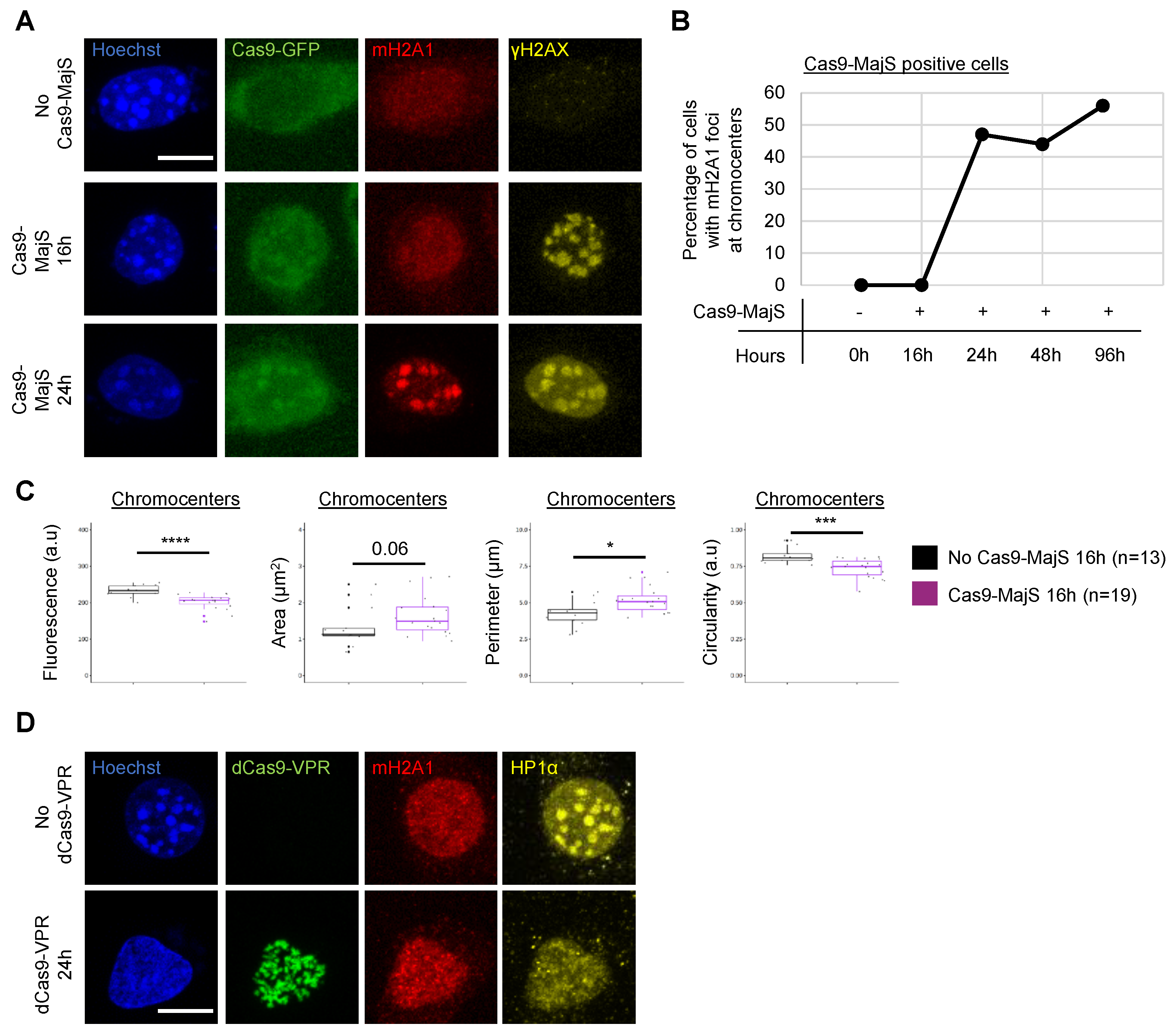
3.2. Cas9-Mediated Induction of DSBs at Pericentric Heterochromatin Triggers Recruitment of mH2A1
3.3. mH2A1 Recruitment to Pericentric Regions Is Not Cell Type-Dependent
3.4. DSBs Are Not Necessary for mH2A1 Recruitment to Pericentric Regions
3.5. Recruitment of mH2A1 Proteins to Pericentric Heterochromatin Depends on Chromocenter Partial Decondensation
3.6. The “H2A-like” Domain of mH2A1 Is Sufficient to Recruit mH2A1 Proteins to Pericentric Heterochromatin
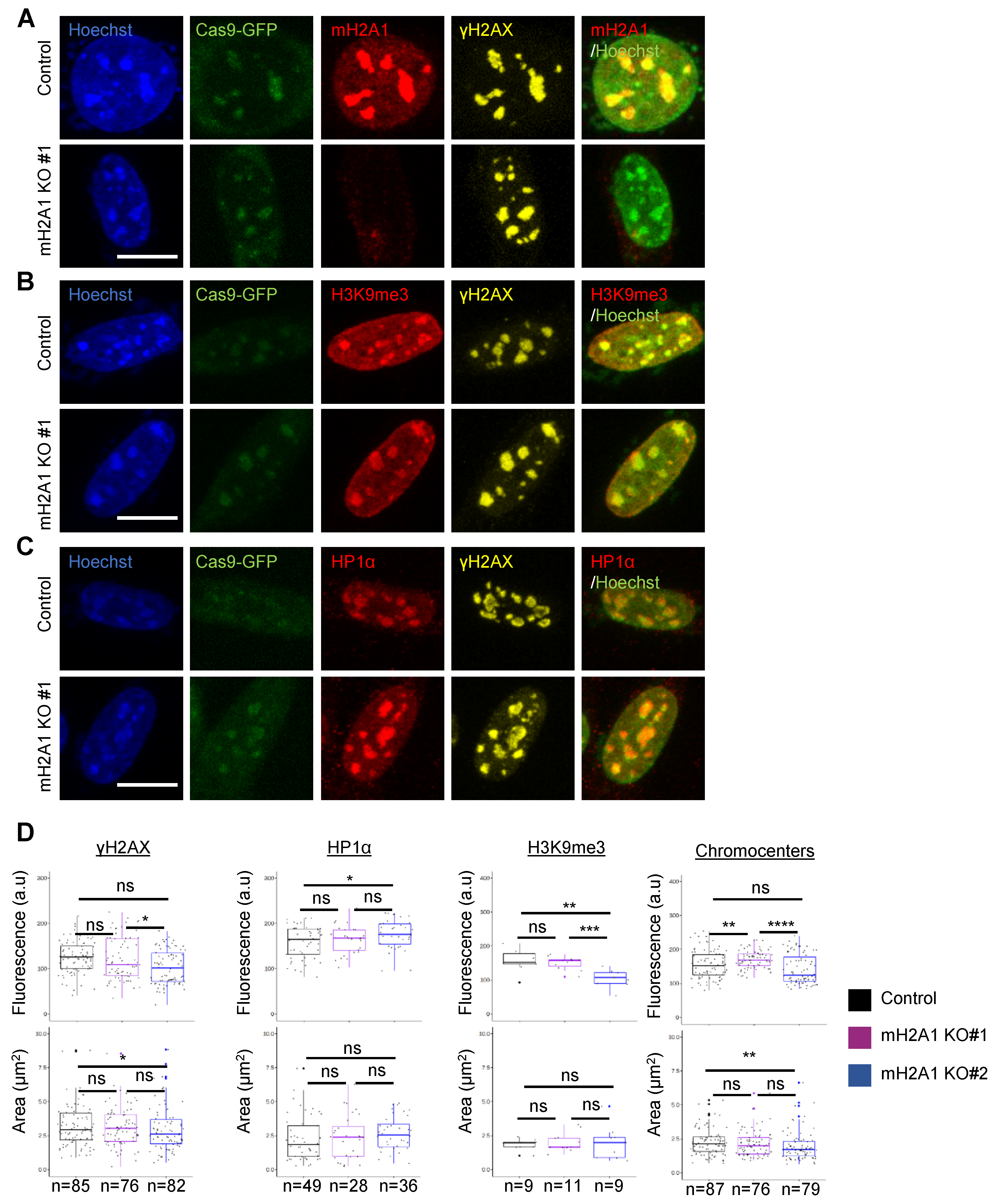
3.7. mH2A1 Is Not Required for Pericentric Heterochromatin Organization
3.8. mH2A1 Regulates Pericentromeric RNA Transcription
4. Discussion
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Acknowledgments
Conflicts of Interest
Appendix A
| gRNAs | gRNA Sequence | PAM |
|---|---|---|
| gRNA 1 | ATTCGGCAACACGCCCCCGC | TGG |
| gRNA 2 | CACGCCTCCGCCGGCCAAAA | AGG |
| Cas9 | pSpCas9(BB)-2A-Puro (PX459) V2.0 (Plasmid #62988, addgene) |
| Cas9-GFP | pSpCas9(BB)-2A-GFP (PX458) (Plasmid #48138, addgene) |
| Cas | pSpCas9(BB)-2A-Puro gRNA 1 ex4 H2AFY MS |
| Cas9-GF | pSpCas9(BB)-2A-GFP (PX458) gRNA 2 ex4 H2AFY Ms |
| MajS gRNA | pEX-A-U6-MaSgRN (Plasmid #84780, addgene) |
| MinS gRNA | pEX-A-U6-MiSgRN (Plasmid #84781, addgene) |
| Telo gRNA | pEX-A-U6-TelgRN (Plasmid #84782, addgene) |
| EGFP | pLVX-EGFP |
| EGFP-mH2A1.1 | pLVX-EGFP-mH2A1.1 |
| EGFP-mH2A1 delMD (aa1-179) | pLVX-EGFP-mH2A1 delMD (aa1-179) |
| EGFP-mH2A1 delLMD (aa1-123) | pLVX-EGFP-mH2A1 delLMD (aa1-123) |
| H2B-EGFP | pBOS-H2B-GFP (BD Pharmingen) |
| Flag-mH2A1.1 | Given by Marcus Buschbeck |
| Flag-mH2A1.2 | Given by Marcus Buschbeck |
| dCas9-VPR | Given by Fabian Erdel [43] |
| siRNA | Sequence |
|---|---|
| HIRA (5’ -> 3’) | GGAGAUGACAAACUGAUUAUU |
| Target | Forward Primer | Reverse Primer |
|---|---|---|
| 18S mRNA | CCCTATCAACTTTCGATGGTAGTCG | CCAATGGATCCTCGTTAAAGGATTT |
| ANKRD1 | AGTAGAGGAACTGGTCACTGG | TGGGCTAGAAGTGTCTTCAGAT |
| CDKN1A (p21) | GACACCACTGGAGGGTGACT | CAGGTCCACATGGTCTTCCT |
| CXCL1 | GAAAGCTTGCCTCAATCCTG | CACCAGTGAGCTTCCTCCTC |
| EDN1 | CAGCAGTCTTAGGCGCTGAG | ACTCTTTATCCATCAGGGACGAG |
| IL6 | CCGGGAACGAAAGAGAAGCT | GCGCTTGTGGAGAAGGAGTT |
| IL8 | CTTTCCACCCCAAATTTATCAAAG | CAGACAGAGCTCTCTTCCATCAGA |
| MajS | GACGACTTGAAAAATGACGAAATC | CATATTCCAGGTCCTTCAGTGTGC |
| GAPDH | AACTTTGGCATTGTGGAAGG | ACACATTGGGGGTAGGAACA |
| gRNA 1 | CACCGATTCGGCAACACGCCCCCGC | AAACGCGGGGGCGTGTTGCCGAAT |
| gRNA 2 | CACCGCACGCCTCCGCCGGCCAAAA | AAACTTTTGGCCGGCGGAGGCGTG |
| Antibody | Supplier (Reference) | Dilution (Use) * |
|---|---|---|
| HIRA | Cell signaling (D2A5E) | 1/1000 (WB) |
| γH2AX | Abcam Ab26350 [9F3] | 1/1000 (WB & IF) |
| γH2AX | Abcam Ab81299 | 1/1000 (IF) |
| mH2A1.2 | Millipore #MABE61 Clone 14GT | 1/1000 |
| mH2A1 | Millipore #AbE215 | 1/1000 (WB & IF) |
| H3K9me3 | Abcam Ab8898 | 1/1000 (IF) |
| H3K9me3 | Abcam (Ab1991) | 1/1000 (WB) |
| Flag | Sigma (F3165) M2 | 1/1000 (WB & IF) |
| βactin | Abcam (Ab8227) | 1/1000 (WB) |
| HP1α | Upstate #05-689 | 1/1000 (WB & IF) |
| H3K9ac | Upstate #06-942 | 1/1000 (WB) |
| LaminB1 | Abcam Ab16048 | 1/1000 (WB) |
| GFP | Roche 1814460001 | 1/1000 (WB) |
| mH2A1-Nter | Abcam (Ab137117) | 1/1000 (IF) |
| mH2A1.1 | Ab αmH2A1.1 Home-made | 1/1000 (IF) |
| Anti-mouse-Peroxidase | Sigma A2304 | 1/10.000 (WB) |
| Anti-Rabbit-Peroxidase | Sigma A0545 | 1/10.000 (WB) |
| Alexa Fluor 488 Anti-mouse | Invitrogen A11029 | 1/1000 (IF) |
| Alexa Fluor 647 Anti-Rabbit | Invitrogen A21245 | 1/1000 (IF) |
References
- Ferrand, J.; Rondinelli, B.; Polo, S.E. Histone Variants: Guardians of Genome Integrity. Cells 2020, 9, 2424. [Google Scholar] [CrossRef]
- Martire, S.; Banaszynski, L.A. The roles of histone variants in fine-tuning chromatin organization and function. Nat. Rev. Mol. Cell Biol. 2020, 21, 522–541. [Google Scholar] [CrossRef] [PubMed]
- Buschbeck, M.; Hake, S.B. Variants of core histones and their roles in cell fate decisions, development and cancer. Nat. Rev. Mol. Cell Biol. 2017, 18, 299–314. [Google Scholar] [CrossRef]
- Pehrson, J.R.; Fuji, R.N. Evolutionary conservation of histone macroH2A subtypes and domains. Nucleic Acids Res. 1998, 26, 2837–2842. [Google Scholar] [CrossRef]
- Rivera-Casas, C.; Gonzalez-Romero, R.; Cheema, M.S.; Ausió, J.; Eirín-López, J.M. The characterization of macroH2A beyond vertebrates supports an ancestral origin and conserved role for histone variants in chromatin. Epigenetics 2016, 11, 415–425. [Google Scholar] [CrossRef] [PubMed]
- Gamble, M.J.; Kraus, W.L. Multiple facets of the unique histone variant macroH2A: From genomics to cell biology. Cell Cycle 2010, 9, 2568–2574. [Google Scholar] [CrossRef]
- Costanzi, C.; Pehrson, J.R. Histone macroH2A1 is concentrated in the inactive X chromosome of female mammals. Nature 1998, 393, 599–601. [Google Scholar] [CrossRef]
- Galupa, R.; Heard, E. X-chromosome inactivation: A crossroads between chromosome architecture and gene regulation. Annu. Rev. Genet. 2018, 52, 535–566. [Google Scholar] [CrossRef]
- Gamble, M.J.; Frizzell, K.M.; Yang, C.; Krishnakumar, R.; Kraus, W.L. The histone variant macroH2A1 marks repressed autosomal chromatin, but protects a subset of its target genes from silencing. Genes Dev. 2010, 24, 21–32. [Google Scholar] [CrossRef]
- Recoules, L.; Heurteau, A.; Raynal, F.; Karasu, N.; Moutahir, F.; Bejjani, F.; Jariel-Encontre, I.; Cuvier, O.; Sexton, T.; Lavigne, A.C.; et al. The histone variant macroH2A1.1 regulates RNA polymerase II-paused genes within defined chromatin interaction landscapes. J. Cell Sci. 2022, 135, jcs259456. [Google Scholar] [CrossRef]
- Sun, Z.; Filipescu, D.; Andrade, J.; Gaspar-Maia, A.; Ueberheide, B.; Bernstein, E. Transcription-associated histone pruning demarcates macroH2A chromatin domains. Nat. Struct. Mol. Biol. 2018, 25, 958–970. [Google Scholar] [CrossRef] [PubMed]
- Douet, J.; Corujo, D.; Malinverni, R.; Renauld, J.; Sansoni, V.; Marjanović, M.P.; Cantariño, N.; Valero, V.; Mongelard, F.; Bouvet, P.; et al. MacroH2A histone variants maintain nuclear organization and heterochromatin architecture. J. Cell Sci. 2017, 130, 1570–1582. [Google Scholar] [CrossRef] [PubMed]
- Cong, R.; Das, S.; Douet, J.; Wong, J.; Buschbeck, M.; Mongelard, F.; Bouvet, P. MacroH2A1 histone variant represses rDNA transcription. Nucleic Acids Res. 2014, 42, 181–192. [Google Scholar] [CrossRef] [PubMed]
- Zhang, R.; Poustovoitov, M.V.; Ye, X.; Santos, H.A.; Chen, W.; Daganzo, S.M.; Erzberger, J.P.; Serebriiskii, I.G.; Canutescu, A.A.; Dunbrack, R.L.; et al. Formation of macroH2A-containing senescence-associated heterochromatin foci and senescence driven by ASF1a and HIRA. Dev. Cell 2005, 8, 19–30. [Google Scholar] [CrossRef] [PubMed]
- Chandra, T.; Kirschner, K.; Thuret, J.Y.; Pope, B.D.; Ryba, T.; Newman, S.; Ahmed, K.; Samarajiwa, S.A.; Salama, R.; Carroll, T.; et al. Independence of Repressive Histone Marks and Chromatin Compaction during Senescent Heterochromatic Layer Formation. Mol. Cell 2012, 47, 203–214. [Google Scholar] [CrossRef]
- Narita, M.; Nũnez, S.; Heard, E.; Narita, M.; Lin, A.W.; Hearn, S.A.; Spector, D.L.; Hannon, G.J.; Lowe, S.W. Rb-mediated heterochromatin formation and silencing of E2F target genes during cellular senescence. Cell 2003, 113, 703–716. [Google Scholar] [CrossRef]
- Grigoryev, S.A.; Nikitina, T.; Pehrson, J.R.; Singh, P.B.; Woodcock, C.L. Dynamic relocation of epigenetic chromatin markers reveals an active role of constitutive heterochromatin in the transition from proliferation to quiescence. J. Cell Sci. 2004, 117, 6153–6162. [Google Scholar] [CrossRef]
- Zhang, R.; Liu, S.; Chen, W.; Bonner, M.; Pehrson, J.; Yen, T.J.; Adams, P.D. HP1 Proteins Are Essential for a Dynamic Nuclear Response That Rescues the Function of Perturbed Heterochromatin in Primary Human Cells. Mol. Cell. Biol. 2007, 27, 949–962. [Google Scholar] [CrossRef]
- Changolkar, L.N.; Singh, G.; Cui, K.; Berletch, J.B.; Zhao, K.; Disteche, C.M.; Pehrson, J.R. Genome-Wide Distribution of MacroH2A1 Histone Variants in Mouse Liver Chromatin. Mol. Cell. Biol. 2010, 30, 5473–5483. [Google Scholar] [CrossRef]
- Mermoud, J.E.; Costanzi, C.; Pehrson, J.R.; Brockdorff, N. Histone macroH2A1.2 relocates to the inactive X chromosome after initiation and propagation of X-inactivation. J. Cell Biol. 1999, 147, 1399–1408. [Google Scholar] [CrossRef]
- Pasque, V.; Gillich, A.; Garrett, N.; Gurdon, J.B. Histone variant macroH2A confers resistance to nuclear reprogramming. EMBO J. 2011, 30, 2373–2387. [Google Scholar] [CrossRef] [PubMed]
- Perche, P.Y.; Vourc’h, C.; Konecny, L.; Souchier, C.; Robert-Nicoud, M.; Dimitrov, S.; Khochbin, S. Higher concentrations of histone macroH2A in the Barr body are correlated with higher nucleosome density. Curr. Biol. 2000, 10, 1531–1534. [Google Scholar] [CrossRef] [PubMed]
- Hernández-Muñoz, I.; Lund, A.H.; Stoop, P.V.D.; Boutsma, E.; Muijrers, I.; Verhoeven, E.; Nusinow, D.A.; Panning, B.; Marahrens, Y.; Lohuizen, M.V. Stable X chromosome inactivation involves the PRC1 Polycomb complex and requires histone MACROH2A1 and the CULLIN3/SPOP ubiquitin E3 ligase. Proc. Natl. Acad. Sci. USA 2005, 102, 7635–7640. [Google Scholar] [CrossRef] [PubMed]
- Chen, H.; Ruiz, P.D.; Novikov, L.; Casill, A.D.; Park, J.W.; Gamble, M.J. MacroH2A1.1 and PARP-1 cooperate to regulate transcription by promoting CBP-mediated H2B acetylation. Nat. Struct. Mol. Biol. 2014, 21, 981–989. [Google Scholar] [CrossRef]
- Dell’Orso, S.; Wang, A.H.; Shih, H.Y.; Saso, K.; Berghella, L.; Gutierrez-Cruz, G.; Ladurner, A.G.; O’Shea, J.J.; Sartorelli, V.; Zare, H. The Histone Variant MacroH2A1.2 Is Necessary for the Activation of Muscle Enhancers and Recruitment of the Transcription Factor Pbx1. Cell Rep. 2016, 14, 1156–1168. [Google Scholar] [CrossRef] [PubMed]
- Gaspar-Maia, A.; Qadeer, Z.A.; Hasson, D.; Ratnakumar, K.; Leu, N.A.; Leroy, G.; Liu, S.; Costanzi, C.; Valle-Garcia, D.; Schaniel, C.; et al. MacroH2A histone variants act as a barrier upon reprogramming towards pluripotency. Nat. Commun. 2013, 4, 1565. [Google Scholar] [CrossRef]
- Johnson, W.L.; Yewdell, W.T.; Bell, J.C.; McNulty, S.M.; Duda, Z.; O, R.J.; Sullivan, B.A.; Straight, A.F. RNA-dependent stabilization of SUV39H1 at constitutive heterochromatin. eLife 2017, 6, e25299. [Google Scholar] [CrossRef]
- Lozano-Torres, B.; Estepa-Fernández, A.; Rovira, M.; Orzáez, M.; Serrano, M.; Martínez-Máñez, R.; Sancenón, F. The chemistry of senescence. Nat. Rev. Chem. 2019, 3, 426–441. [Google Scholar] [CrossRef]
- Nagano, T.; Nakano, M.; Nakashima, A.; Onishi, K.; Yamao, S.; Enari, M.; Kikkawa, U.; Kamada, S. Identification of cellular senescence-specific genes by comparative transcriptomics. Sci. Rep. 2016, 6, 31758. [Google Scholar] [CrossRef]
- Giuliano, C.J.; Lin, A.; Girish, V.; Sheltzer, J.M. Generating Single Cell–Derived Knockout Clones in Mammalian Cells with CRISPR/Cas9. Curr. Protoc. Mol. Biol. 2019, 128, e100. [Google Scholar] [CrossRef]
- Mahmood, T.; Yang, P.C. Western blot: Technique, theory, and trouble shooting. N. Am. J. Med. Sci. 2012, 4, 429–434. [Google Scholar] [CrossRef] [PubMed]
- Brändle, F.; Frühbauer, B.; Jagannathan, M. Principles and functions of pericentromeric satellite DNA clustering into chromocenters. Semin. Cell Dev. Biol. 2022, 128, 26–39. [Google Scholar] [CrossRef]
- Ostromyshenskii, D.I.; Chernyaeva, E.N.; Kuznetsova, I.S.; Podgornaya, O.I. Mouse chromocenters DNA content: Sequencing and in silico analysis. BMC Genom. 2018, 19, 151. [Google Scholar] [CrossRef] [PubMed]
- Fumagalli, M.; Rossiello, F.; Mondello, C.; d’Adda di Fagagna, F. Stable cellular senescence is associated with persistent DDR activation. PLoS ONE 2014, 9, e11096. [Google Scholar] [CrossRef] [PubMed]
- Hernandez-Segura, A.; Nehme, J.; Demaria, M. Hallmarks of Cellular Senescence. Trends Cell Biol. 2018, 28, 436–453. [Google Scholar] [CrossRef] [PubMed]
- Chiolo, I.; Minoda, A.; Colmenares, S.U.; Polyzos, A.; Costes, S.V.; Karpen, G.H. Double-strand breaks in heterochromatin move outside of a dynamic HP1a domain to complete recombinational repair. Cell 2011, 144, 732–744. [Google Scholar] [CrossRef]
- Ryu, T.; Spatola, B.; Delabaere, L.; Bowlin, K.; Hopp, H.; Kunitake, R.; Karpen, G.H.; Chiolo, I. Heterochromatic breaks move to the nuclear periphery to continue recombinational repair. Nat. Cell Biol. 2015, 17, 1401. [Google Scholar] [CrossRef] [PubMed]
- Tsouroula, K.; Furst, A.; Rogier, M.; Heyer, V.; Maglott-Roth, A.; Ferrand, A.; Reina-San-Martin, B.; Soutoglou, E. Temporal and Spatial Uncoupling of DNA Double Strand Break Repair Pathways within Mammalian Heterochromatin. Mol. Cell 2016, 63, 293–305. [Google Scholar] [CrossRef]
- Anton, T.; Bultmann, S.; Leonhardt, H.; Markaki, Y. Visualization of specific DNA sequences in living mouse embryonic stem cells with a programmable fluorescent CRISPR/Cas system. Nucleus 2014, 5, 163–172. [Google Scholar] [CrossRef]
- Li, X.; Tyler, J.K. Nucleosome disassembly during human non-homologous end joining followed by concerted HIRA- and CAF-1-dependent reassembly. eLife 2016, 5, e15129. [Google Scholar] [CrossRef]
- Hooten, N.N.; Evans, M.K. Techniques to induce and quantify cellular senescence. J. Vis. Exp. 2017, 123, 55533. [Google Scholar] [CrossRef]
- Chavez, A.; Scheiman, J.; Vora, S.; Pruitt, B.W.; Tuttle, M.; Iyer, E.P.R.; Lin, S.; Kiani, S.; Guzman, C.D.; Wiegand, D.J.; et al. Highly efficient Cas9-mediated transcriptional programming. Nat. Methods 2015, 12, 326–328. [Google Scholar] [CrossRef] [PubMed]
- Erdel, F.; Rademacher, A.; Vlijm, R.; Tünnermann, J.; Frank, L.; Weinmann, R.; Schweigert, E.; Yserentant, K.; Hummert, J.; Bauer, C.; et al. Mouse Heterochromatin Adopts Digital Compaction States without Showing Hallmarks of HP1-Driven Liquid-Liquid Phase Separation. Mol. Cell 2020, 78, 236–249.e7. [Google Scholar] [CrossRef] [PubMed]
- Changolkar, L.N.; Costanzi, C.; Leu, N.A.; Chen, D.; McLaughlin, K.J.; Pehrson, J.R. Developmental Changes in Histone macroH2A1-Mediated Gene Regulation. Mol. Cell. Biol. 2007, 27, 2758–2764. [Google Scholar] [CrossRef] [PubMed]
- Tanasijevic, B.; Rasmussen, T.P. X Chromosome Inactivation and Differentiation Occur Readily in ES Cells Doubly-Deficient for MacroH2A1 and MacroH2A2. PLoS ONE 2011, 6, e21512. [Google Scholar] [CrossRef] [PubMed]
- Kennedy, A.L.; McBryan, T.; Enders, G.H.; Johnson, F.B.; Zhang, R.; Adams, P.D. Senescent mouse cells fail to overtly regulate the HIRA histone chaperone and do not form robust Senescence Associated Heterochromatin Foci. Cell Div. 2010, 5, 16. [Google Scholar] [CrossRef]
- Kudlova, N.; Sanctis, J.B.D.; Hajduch, M. Cellular Senescence: Molecular Targets, Biomarkers, and Senolytic Drugs. Int. J. Mol. Sci. 2022, 23, 4168. [Google Scholar] [CrossRef]
- Swanson, E.C.; Manning, B.; Zhang, H.; Lawrence, J.B. Higher-order unfolding of satellite heterochromatin is a consistent and early event in cell senescence. J. Cell Biol. 2013, 203, 929–942. [Google Scholar] [CrossRef]
- Clouaire, T.; Rocher, V.; Lashgari, A.; Arnould, C.; Aguirrebengoa, M.; Biernacka, A.; Skrzypczak, M.; Aymard, F.; Fongang, B.; Dojer, N.; et al. Comprehensive Mapping of Histone Modifications at DNA Double-Strand Breaks Deciphers Repair Pathway Chromatin Signatures. Mol. Cell 2018, 72, 250–262.e6. [Google Scholar] [CrossRef]
- Timinszky, G.; Till, S.; Hassa, P.O.; Hothorn, M.; Kustatscher, G.; Nijmeijer, B.; Colombelli, J.; Altmeyer, M.; Stelzer, E.H.; Scheffzek, K.; et al. A macrodomain-containing histone rearranges chromatin upon sensing PARP1 activation. Nat. Struct. Mol. Biol. 2009, 16, 923–929. [Google Scholar] [CrossRef]
- Khurana, S.; Kruhlak, M.J.; Kim, J.; Tran, A.D.; Liu, J.; Nyswaner, K.; Shi, L.; Jailwala, P.; Sung, M.H.; Hakim, O.; et al. A macrohistone variant links dynamic chromatin compaction to BRCA1-dependent genome maintenance. Cell Rep. 2014, 8, 1049–1062. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.; Sturgill, D.; Sebastian, R.; Khurana, S.; Tran, A.D.; Edwards, G.B.; Kruswick, A.; Burkett, S.; Hosogane, E.K.; Hannon, W.W.; et al. Replication Stress Shapes a Protective Chromatin Environment across Fragile Genomic Regions. Mol. Cell 2018, 69, 36–47.e7. [Google Scholar] [CrossRef] [PubMed]
- Mehrotra, P.V.; Ahel, D.; Ryan, D.P.; Weston, R.; Wiechens, N.; Kraehenbuehl, R.; Owen-Hughes, T.; Ahel, I. DNA repair factor APLF Is a histone chaperone. Mol. Cell 2011, 41, 46–55. [Google Scholar] [CrossRef] [PubMed]
- Xu, C.; Xu, Y.; Gursoy-Yuzugullu, O.; Price, B.D. The histone variant macroH2A1.1 is recruited to DSBs through a mechanism involving PARP1. FEBS Lett. 2012, 586, 3920–3925. [Google Scholar] [CrossRef]
- Lu, J.; Gilbert, D.M. Proliferation-dependent and cell cycle regulated transcription of mouse pericentric heterochromatin. J. Cell Biol. 2007, 179, 411–421. [Google Scholar] [CrossRef]
- Probst, A.V.; Okamoto, I.; Casanova, M.; Marjou, F.E.; Baccon, P.L.; Almouzni, G. A strand-specific burst in transcription of pericentric satellites is required for chromocenter formation and early mouse development. Dev. Cell 2010, 19, 625–638. [Google Scholar] [CrossRef]
- Valgardsdottir, R.; Chiodi, I.; Giordano, M.; Rossi, A.; Bazzini, S.; Ghigna, C.; Riva, S.; Biamonti, G. Transcription of Satellite III non-coding RNAs is a general stress response in human cells. Nucleic Acids Res. 2008, 36, 423–434. [Google Scholar] [CrossRef]
- Ting, D.T.; Lipson, D.; Paul, S.; Brannigan, B.W.; Akhavanfard, S.; Coffman, E.J.; Contino, G.; Deshpande, V.; Iafrate, A.J.; Letovsky, S.; et al. Aberrant overexpression of satellite repeats in pancreatic and other epithelial cancers. Science 2011, 331, 593–596. [Google Scholar] [CrossRef]
- Lejeune, E.; Bayne, E.H.; Allshire, R.C. On the connection between RNAi and heterochromatin at centromeres. Cold Spring Harb. Symp. Quant. Biol. 2010, 75, 275–283. [Google Scholar] [CrossRef]




Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Recoules, L.; Tanguy Le Gac, N.; Moutahir, F.; Bystricky, K.; Lavigne, A.-C. Recruitment of the Histone Variant MacroH2A1 to the Pericentric Region Occurs upon Chromatin Relaxation and Is Responsible for Major Satellite Transcriptional Regulation. Cells 2023, 12, 2175. https://doi.org/10.3390/cells12172175
Recoules L, Tanguy Le Gac N, Moutahir F, Bystricky K, Lavigne A-C. Recruitment of the Histone Variant MacroH2A1 to the Pericentric Region Occurs upon Chromatin Relaxation and Is Responsible for Major Satellite Transcriptional Regulation. Cells. 2023; 12(17):2175. https://doi.org/10.3390/cells12172175
Chicago/Turabian StyleRecoules, Ludmila, Nicolas Tanguy Le Gac, Fatima Moutahir, Kerstin Bystricky, and Anne-Claire Lavigne. 2023. "Recruitment of the Histone Variant MacroH2A1 to the Pericentric Region Occurs upon Chromatin Relaxation and Is Responsible for Major Satellite Transcriptional Regulation" Cells 12, no. 17: 2175. https://doi.org/10.3390/cells12172175
APA StyleRecoules, L., Tanguy Le Gac, N., Moutahir, F., Bystricky, K., & Lavigne, A.-C. (2023). Recruitment of the Histone Variant MacroH2A1 to the Pericentric Region Occurs upon Chromatin Relaxation and Is Responsible for Major Satellite Transcriptional Regulation. Cells, 12(17), 2175. https://doi.org/10.3390/cells12172175