The Combined Effects of Topography and Stiffness on Neuronal Differentiation and Maturation Using a Hydrogel Platform
Abstract
:1. Introduction
2. Materials and Methods
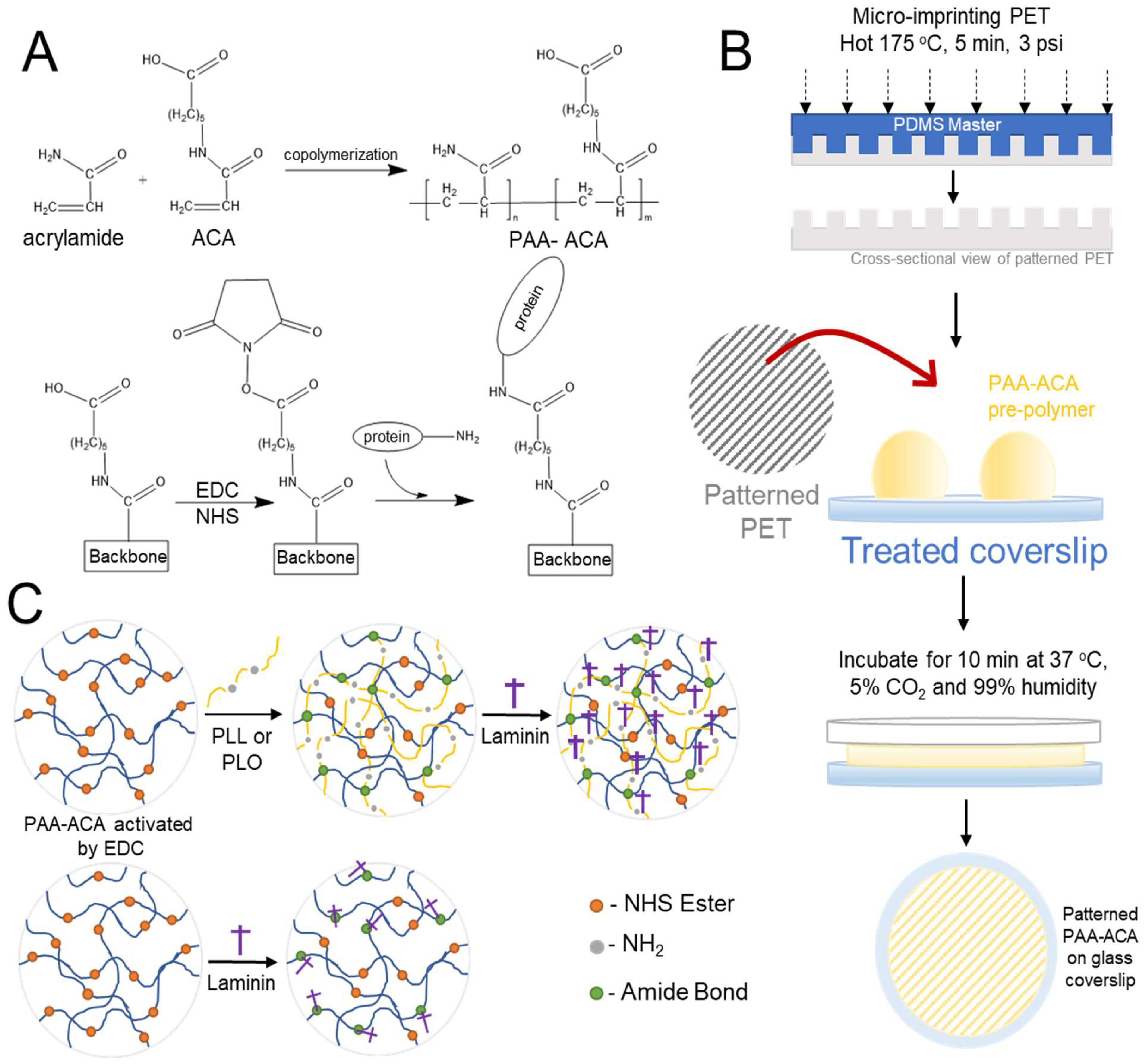
2.1. PAA-ACA Gel Fabrication Overview
2.2. Activation of Glass Coverslips
2.3. Hot Embossing of PET Molds
2.4. Co-Polymerization of Polyacrylamide and N-Acryloyl-6-Aminoproic Acid and Micropatterning with PET Molds
2.5. Conjugation of Polypeptides and Extracellular Matrix to the PAA-ACA Gels
2.6. Mechanical Characterization and Optical Profilometry of the PAA-ACA Gels
2.7. Neural Progenitor Cell Cutlure, Maintenance, and Differentiation
2.8. Cell Seeding on PAA-ACA Hydrogels
2.9. Immunofluoresence Staining and Imaging
2.10. RNA Isolation and RT-qPCR
2.11. Statistics
3. Results
3.1. Physical Characterization of the PAA-ACA Gels
3.2. Extended Attachment of mNPCs and hNPCs to the ECM-Coated PAA-ACA Gels
3.3. The Effect of Topography and Stiffness on mNPC Commitment to the Neuronal Lineage
3.4. The Effect of Topography and Stiffness on mNPC Maturation
3.5. The Effect of Topography and Stiffness on mNPC Morphology: Neurite Length and Branching
3.6. The Effect of Topography and Stiffness on hNPC Maturation
4. Discussion
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Long, K.R.; Huttner, W.B. How the Extracellular Matrix Shapes Neural Development. Open Biol. 2019, 9, 180216. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Jain, D.; Mattiassi, S.; Goh, E.; Yim, E. Extracellular Matrix and Biomimetic Engineering Microenvironment for Neuronal Differentiation. Neural. Regen. Res. 2020, 15, 573–585. [Google Scholar] [CrossRef] [PubMed]
- Yim, E.K.F.; Pang, S.W.; Leong, K.W. Synthetic Nanostructures Inducing Differentiation of Human Mesenchymal Stem Cells into Neuronal Lineage. Exp. Cell Res. 2007, 313, 1820–1829. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Moe, A.A.K.; Suryana, M.; Marcy, G.; Lim, S.K.; Ankam, S.; Goh, J.Z.W.; Jin, J.; Teo, B.K.K.; Law, J.B.K.; Low, H.Y.; et al. Microarray with Micro- and Nano-Topographies Enables Identification of the Optimal Topography for Directing the Differentiation of Primary Murine Neural Progenitor Cells. Small 2012, 8, 3050–3061. [Google Scholar] [CrossRef]
- Ankam, S.; Suryana, M.; Chan, L.Y.; Moe, A.A.K.; Teo, B.K.K.; Law, J.B.K.; Sheetz, M.P.; Low, H.Y.; Yim, E.K.F. Substrate Topography and Size Determine the Fate of Human Embryonic Stem Cells to Neuronal or Glial Lineage. Acta Biomater. 2013, 9, 4535–4545. [Google Scholar] [CrossRef] [PubMed]
- Chan, L.Y.; Birch, W.R.; Yim, E.K.F.; Choo, A.B.H. Temporal Application of Topography to Increase the Rate of Neural Differentiation from Human Pluripotent Stem Cells. Biomaterials 2013, 34, 382–392. [Google Scholar] [CrossRef]
- Chua, J.S.; Chng, C.P.; Moe, A.A.K.; Tann, J.Y.; Goh, E.L.K.; Chiam, K.H.; Yim, E.K.F. Extending Neurites Sense the Depth of the Underlying Topography during Neuronal Differentiation and Contact Guidance. Biomaterials 2014, 35, 7750–7761. [Google Scholar] [CrossRef]
- Tan, K.K.B.; Tann, J.Y.; Sathe, S.R.; Goh, S.H.; Ma, D.; Goh, E.L.K.; Yim, E.K.F. Enhanced Differentiation of Neural Progenitor Cells into Neurons of the Mesencephalic Dopaminergic Subtype on Topographical Patterns. Biomaterials 2015, 43, 32–43. [Google Scholar] [CrossRef]
- Sathe, S.; Chan, X.Q.; Jin, J.; Bernitt, E.; Döbereiner, H.-G.; Yim, E.K.F. Correlation and Comparison of Cortical and Hippocampal Neural Progenitor Morphology and Differentiation through the Use of Micro- and Nano-Topographies. J. Funct. Biomater. 2017, 8, 35. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Tan, K.K.B.; Lim, W.W.M.; Chai, C.; Kukumberg, M.; Lim, K.L.; Goh, E.L.K.; Yim, E.K.F. Sequential Application of Discrete Topographical Patterns Enhances Derivation of Functional Mesencephalic Dopaminergic Neurons from Human Induced Pluripotent Stem Cells. Sci. Rep. 2018, 8, 9567. [Google Scholar] [CrossRef] [Green Version]
- Song, L.; Wang, K.; Li, Y.; Yang, Y. Nanotopography Promoted Neuronal Differentiation of Human Induced Pluripotent Stem Cells. Colloids Surf. B Biointerfaces 2016, 148, 49–58. [Google Scholar] [CrossRef] [PubMed]
- Qi, L.; Li, N.; Huang, R.; Song, Q.; Wang, L.; Zhang, Q.; Su, R.; Kong, T.; Tang, M.; Cheng, G. The Effects of Topographical Patterns and Sizes on Neural Stem Cell Behavior. PLoS ONE 2013, 8, 59022. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Tonazzini, I.; Masciullo, C.; Savi, E.; Sonato, A.; Romanato, F.; Cecchini, M. Neuronal Contact Guidance and YAP Signaling on Ultra-Small Nanogratings. Sci. Rep. 2020, 10, 3742. [Google Scholar] [CrossRef] [Green Version]
- Budday, S.; Sommer, G.; Birkl, C.; Langkammer, C.; Haybaeck, J.; Kohnert, J.; Bauer, M.; Paulsen, F.; Steinmann, P.; Kuhl, E.; et al. Mechanical Characterization of Human Brain Tissue. Acta Biomater. 2017, 48, 319–340. [Google Scholar] [CrossRef] [PubMed]
- Georges, P.C.; Miller, W.J.; Meaney, D.F.; Sawyer, E.S.; Janmey, P.A. Matrices with Compliance Comparable to That of Brain Tissue Select Neuronal over Glial Growth in Mixed Cortical Cultures. Biophys. J. 2006, 90, 3012–3018. [Google Scholar] [CrossRef] [Green Version]
- Saha, K.; Keung, A.J.; Irwin, E.F.; Li, Y.; Little, L.; Schaffer, D.v.; Healy, K.E. Substrate Modulus Directs Neural Stem Cell Behavior. Biophys. J. 2008, 95, 4426. [Google Scholar] [CrossRef] [Green Version]
- Leipzig, N.D.; Shoichet, M.S. The Effect of Substrate Stiffness on Adult Neural Stem Cell Behavior. Biomaterials 2009, 30, 6867–6878. [Google Scholar] [CrossRef]
- Ali, S.; Wall, I.B.; Mason, C.; Pelling, A.E.; Veraitch, F.S. The Effect of Young’s Modulus on the Neuronal Differentiation of Mouse Embryonic Stem Cells. Acta Biomater. 2015, 25, 253–267. [Google Scholar] [CrossRef] [Green Version]
- Pless, D.D.; Lee, Y.C.; Roseman, S.; Schnaar, R.L. Specific Cell Adhesion to Immobilized Glycoproteins Demonstrated Using New Reagents for Protein and Glycoprotein Immobilization. J. Biol. Chem. 1983, 258, 2340–2349. [Google Scholar] [CrossRef]
- Yip, A.K.; Iwasaki, K.; Ursekar, C.; MacHiyama, H.; Saxena, M.; Chen, H.; Harada, I.; Chiam, K.H.; Sawada, Y. Cellular Response to Substrate Rigidity Is Governed by Either Stress or Strain. Biophys. J. 2013, 104, 19–29. [Google Scholar] [CrossRef] [Green Version]
- Yip, A.K.; Nguyen, A.T.; Rizwan, M.; Wong, S.T.; Chiam, K.H.; Yim, E.K.F. Anisotropic Traction Stresses and Focal Adhesion Polarization Mediates Topography-Induced Cell Elongation. Biomaterials 2018, 181, 103–112. [Google Scholar] [CrossRef] [PubMed]
- Soni, A.; Klütsch, D.; Hu, X.; Houtman, J.; Rund, N.; McCloskey, A.; Mertens, J.; Schafer, S.T.; Amin, H.; Toda, T. Improved Method for Efficient Generation of Functional Neurons from Murine Neural Progenitor Cells. Cells 2021, 10, 1894. [Google Scholar] [CrossRef]
- Vera, E.; Studer, L. When Rejuvenation Is a Problem: Challenges of Modeling Late-Onset Neurodegenerative Disease. Development 2015, 142, 3085–3089. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Berry, B.J.; Smith, A.S.T.; Young, J.E.; Mack, D.L. Advances and Current Challenges Associated with the Use of Human Induced Pluripotent Stem Cells in Modeling Neurodegenerative Disease. Cells Tissues Organs 2018, 205, 331–349. [Google Scholar] [CrossRef] [PubMed]
- Shivaraj, M.C.; Marcy, G.; Low, G.; Ryu, J.R.; Zhao, X.; Rosales, F.J.; Goh, E.L.K. Taurine Induces Proliferation of Neural Stem Cells and Synapse Development in the Developing Mouse Brain. PLoS ONE 2012, 7, e42935. [Google Scholar] [CrossRef]
- Chin, E.W.M.; Marcy, G.; Yoon, S.I.; Ma, D.; Rosales, F.J.; Augustine, G.J.; Goh, E.L.K. Choline Ameliorates Disease Phenotypes in Human IPSC Models of Rett Syndrome. Neuromol. Med. 2016, 18, 364–377. [Google Scholar] [CrossRef]
- Su, C.T.E.; Yoon, S.I.; Marcy, G.; Chin, E.W.M.; Augustine, G.J.; Goh, E.L.K. An Optogenetic Approach for Assessing Formation of Neuronal Connections in a Co-Culture System. J. Vis. Exp. 2015, 96, 52408. [Google Scholar] [CrossRef] [Green Version]
- Menezes, J.R.L.; Luskin, M.B. Expression of Neuron-Specific Tubulin Defines a Novel Population in the Proliferative Layers of the Developing Telencephalon. J. Neurosci. 1994, 14, 5399–5416. [Google Scholar] [CrossRef]
- Korzhevskii, D.E.; Karpenko, M.N.; Kirik, O.V. Microtubule-Associated Proteins as Indicators of Differentiation and the Functional State of Nerve Cells. Neurosci. Behav. Physiol. 2012, 42, 215–222. [Google Scholar] [CrossRef]
- Yang, Z.; Wang, K.K.W. Glial Fibrillary Acidic Protein: From Intermediate Filament Assembly and Gliosis to Neurobiomarker. Trends Neurosci. 2015, 38, 364. [Google Scholar] [CrossRef] [Green Version]
- Song, H.J.; Poo, M.M. The Cell Biology of Neuronal Navigation. Nat. Cell Biol. 2001, 3, E81–E88. [Google Scholar] [CrossRef] [PubMed]
- Axpe, E.; Orive, G.; Franze, K.; Appel, E.A. Towards Brain-Tissue-like Biomaterials. Nat. Commun. 2020, 11, 3423. [Google Scholar] [CrossRef] [PubMed]
- Tzvetkova-Chevolleau, T.; Stéphanou, A.; Fuard, D.; Ohayon, J.; Schiavone, P.; Tracqui, P. The Motility of Normal and Cancer Cells in Response to the Combined Influence of the Substrate Rigidity and Anisotropic Microstructure. Biomaterials 2008, 29, 1541–1551. [Google Scholar] [CrossRef] [Green Version]
- Parandakh, A.; Anbarlou, A.; Tafazzoli-Shadpour, M.; Ardeshirylajimi, A.; Khani, M.M. Substrate Topography Interacts with Substrate Stiffness and Culture Time to Regulate Mechanical Properties and Smooth Muscle Differentiation of Mesenchymal Stem Cells. Colloids Surf. B Biointerfaces 2019, 173, 194–201. [Google Scholar] [CrossRef]
- Sung, B.; Kim, D.H.; Kim, M.H.; Vigolo, D. Combined Effect of Matrix Topography and Stiffness on Neutrophil Shape and Motility. Adv. Biol. 2022, 6, 2101312. [Google Scholar] [CrossRef] [PubMed]
- Shi, Y.; Kirwan, P.; Livesey, F.J. Directed Differentiation of Human Pluripotent Stem Cells to Cerebral Cortex Neurons and Neural Networks. Nat. Protoc. 2012, 7, 1836–1846. [Google Scholar] [CrossRef]
- Rungsiwiwut, R.; Manolertthewan, C.; Numchaisrika, P.; Ahnonkitpanit, V.; Virutamasen, P.; Techakumphu, M.; Pruksananonda, K. The ROCK Inhibitor Y-26732 Enhances the Survival and Proliferation of Human Embryonic Stem Cell-Derived Neural Progenitor Cells upon Dissociation. Cells Tissues Organs 2013, 198, 127–138. [Google Scholar] [CrossRef]
- Ohashi, K.; Fujiwara, S.; Mizuno, K. Roles of the Cytoskeleton, Cell Adhesion and Rho Signalling in Mechanosensing and Mechanotransduction. J. Biochem. 2017, 161, 245–254. [Google Scholar] [CrossRef] [Green Version]
- Ishizaki, T.; Uehata, M.; Tamechika, I.; Keel, J.; Nonomura, K.; Maekawa, M.; Narumiya, S. Pharmacological Properties of Y-27632, a Specific Inhibitor of Rho-Associated Kinases. Mol. Pharmacol. 2000, 57, 976–983. [Google Scholar]
- Wu, Y.; Shu, J.; He, C.; Li, M.; Wang, Y.; Ou, W.; He, Y. ROCK Inhibitor Y27632 Promotes Proliferation and Diminishes Apoptosis of Marmoset Induced Pluripotent Stem Cells by Suppressing Expression and Activity of Caspase 3. Theriogenology 2016, 85, 302–314. [Google Scholar] [CrossRef]
- Rao, P.V.; Deng, P.-F.; Kumar, J.; Epstein, D.L. Modulation of Aqueous Humor Outflow Facility by the Rho Kinase-Specific Inhibitor Y-27632. Investig. Ophthalmol. Vis. Sci. 2001, 42, 1029–1037. [Google Scholar]
- Rees, R.W.; Ralph, D.J.; Royle, M.; Moncada, S.; Cellek, S. Y-27632, an Inhibitor of Rho-Kinase, Antagonizes Noradrenergic Contractions in the Rabbit and Human Penile Corpus Cavernosum. Br. J. Pharmacol. 2001, 133, 455. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zeng, Y.; Wong, S.T.; Teo, S.K.; Leong, K.W.; Chiam, K.H.; Yim, E.K.F. Human Mesenchymal Stem Cell Basal Membrane Bending on Gratings Is Dependent on Both Grating Width and Curvature. Sci. Rep. 2018, 8, 6444. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wong, S.T.; Teo, S.K.; Park, S.; Chiam, K.H.; Yim, E.K.F. Anisotropic Rigidity Sensing on Grating Topography Directs Human Mesenchymal Stem Cell Elongation. Biomech. Model. Mechanobiol. 2014, 13, 27–39. [Google Scholar] [CrossRef]
- Tahirovic, S.; Bradke, F. Neuronal Polarity. Cold Spring Harb. Perspect. Biol. 2009, 1, a001644. [Google Scholar] [CrossRef] [Green Version]
- Ankam, S.; Teo, B.K.K.; Pohan, G.; Ho, S.W.L.; Lim, C.K.; Yim, E.K.F. Temporal Changes in Nucleus Morphology, Lamin A/C and Histone Methylation during Nanotopography-Induced Neuronal Differentiation of Stem Cells. Front. Bioeng. Biotechnol. 2018, 6, 69. [Google Scholar] [CrossRef] [Green Version]
- Athamneh, A.I.M.; Cartagena-Rivera, A.X.; Raman, A.; Suter, D.M. Substrate Deformation Predicts Neuronal Growth Cone Advance. Biophys. J. 2015, 109, 1358–1371. [Google Scholar] [CrossRef] [Green Version]
- Jiang, F.X.; Yurke, B.; Firestein, B.L.; Langrana, N.A. Neurite Outgrowth on a DNA Crosslinked Hydrogel with Tunable Stiffnesses. Ann. Biomed. Eng. 2008, 36, 1565–1579. [Google Scholar] [CrossRef]
- Jiang, F.X.; Yurke, B.; Schloss, R.S.; Firestein, B.L.; Langrana, N.A. Effect of Dynamic Stiffness of the Substrates on Neurite Outgrowth by Using a DNA-Crosslinked Hydrogel. Tissue Eng. Part A 2010, 16, 1873–1889. [Google Scholar] [CrossRef]
- Ferrari, A.; Cecchini, M.; Serresi, M.; Faraci, P.; Pisignano, D.; Beltram, F. Neuronal Polarity Selection by Topography-Induced Focal Adhesion Control. Biomaterials 2010, 31, 4682–4694. [Google Scholar] [CrossRef]
- Teo, B.K.K.; Wong, S.T.; Lim, C.K.; Kung, T.Y.S.; Yap, C.H.; Ramagopal, Y.; Romer, L.H.; Yim, E.K.F. Nanotopography Modulates Mechanotransduction of Stem Cells and Induces Differentiation through Focal Adhesion Kinase. ACS Nano 2013, 7, 4785–4798. [Google Scholar] [CrossRef] [PubMed]
- Keung, A.J.; de Juan-Pardo, E.M.; Schaffer, D.v.; Kumar, S. Rho GTPases Mediate the Mechanosensitive Lineage Commitment of Neural Stem Cells. Stem Cells 2011, 29, 1886. [Google Scholar] [CrossRef] [PubMed]










Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Mattiassi, S.; Conner, A.A.; Feng, F.; Goh, E.L.K.; Yim, E.K.F. The Combined Effects of Topography and Stiffness on Neuronal Differentiation and Maturation Using a Hydrogel Platform. Cells 2023, 12, 934. https://doi.org/10.3390/cells12060934
Mattiassi S, Conner AA, Feng F, Goh ELK, Yim EKF. The Combined Effects of Topography and Stiffness on Neuronal Differentiation and Maturation Using a Hydrogel Platform. Cells. 2023; 12(6):934. https://doi.org/10.3390/cells12060934
Chicago/Turabian StyleMattiassi, Sabrina, Abigail A. Conner, Fan Feng, Eyleen L. K. Goh, and Evelyn K. F. Yim. 2023. "The Combined Effects of Topography and Stiffness on Neuronal Differentiation and Maturation Using a Hydrogel Platform" Cells 12, no. 6: 934. https://doi.org/10.3390/cells12060934






