Novel Therapeutic Target Critical for SARS-CoV-2 Infectivity and Induction of the Cytokine Release Syndrome
Abstract
1. Introduction
2. Material and Methods
2.1. Cell Lines
2.2. Ligands and Reagents
2.3. Inhibitors
2.4. NF-κB-Dependent Secretory Alkaline Phosphatase (SEAP) Assay
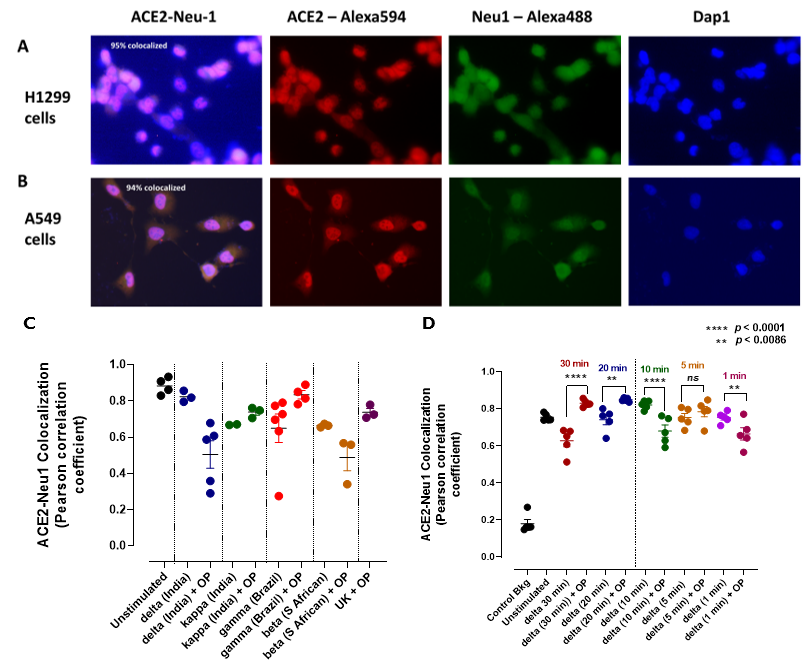
2.5. Neu-1 Colocalization with ACE2
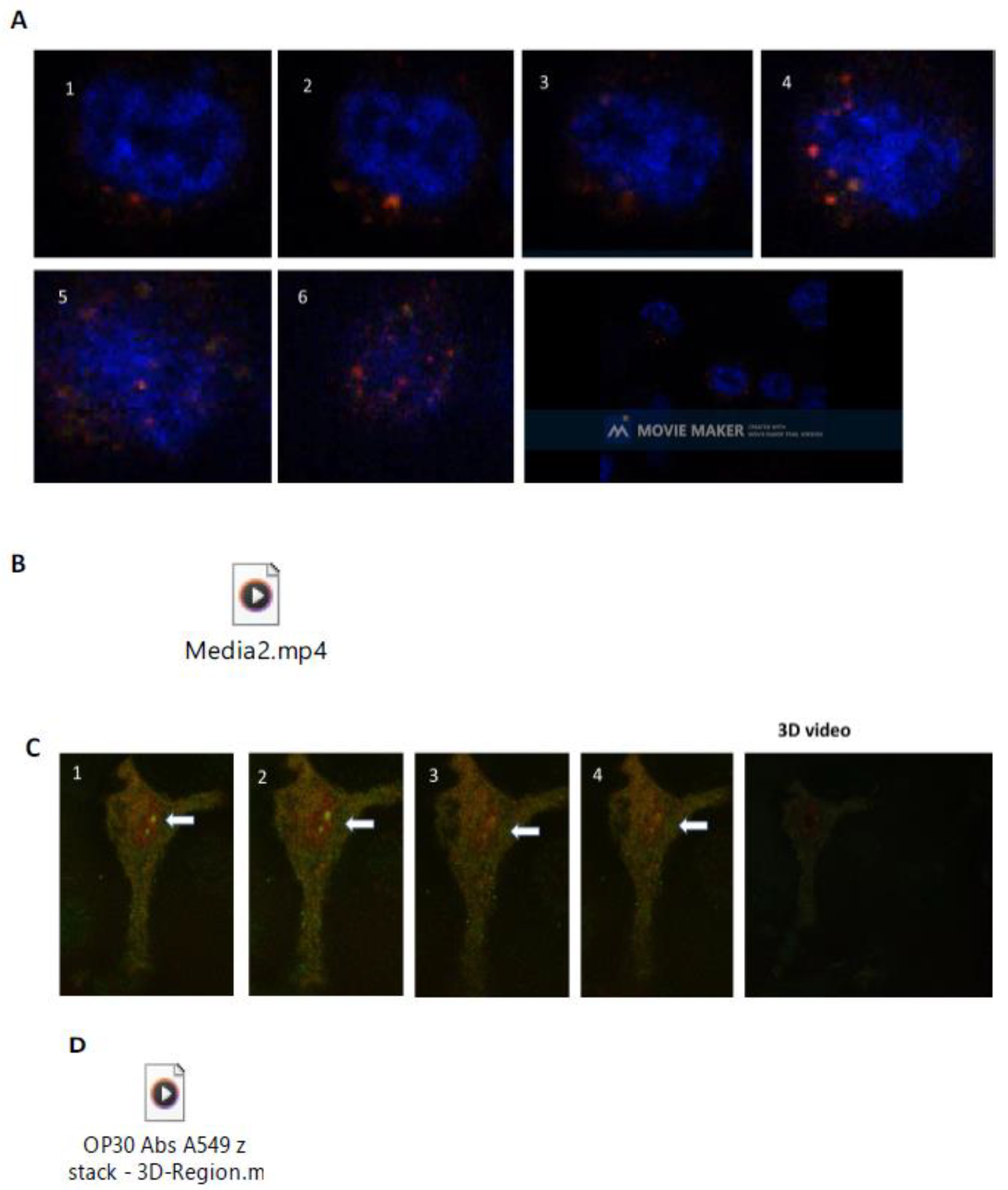
2.6. Live Cell Microscopy
2.7. Mouse Cytokine ELISA Array III (Colorimetric) Sandwich Assay
2.8. MILLIPLEX® Luminex® 200™ xMAP® Flow Cytometry-Based Instrumentation
2.9. Supplementary Material for Live Cell Video
2.10. Statistical Analysis
3. Results
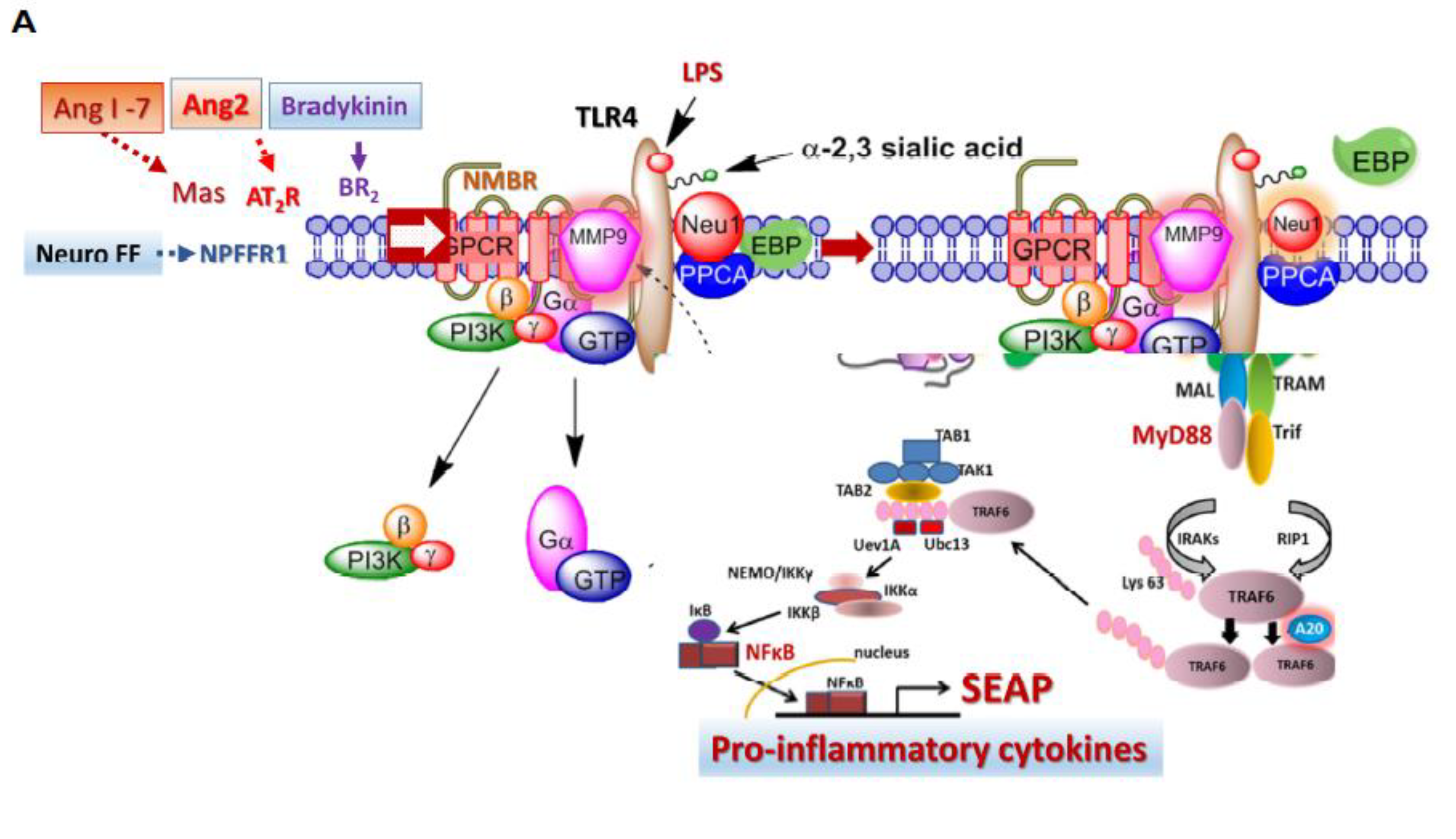
3.1. Neu-1 Sialidase Activity Is Activated by SARS-CoV-2 Spike Protein S1 Stimulation in Murine Macrophages and Human Epithelial Cells
3.2. Mammalian Neu-1 Is Tethered to Naive ACE2 Receptors
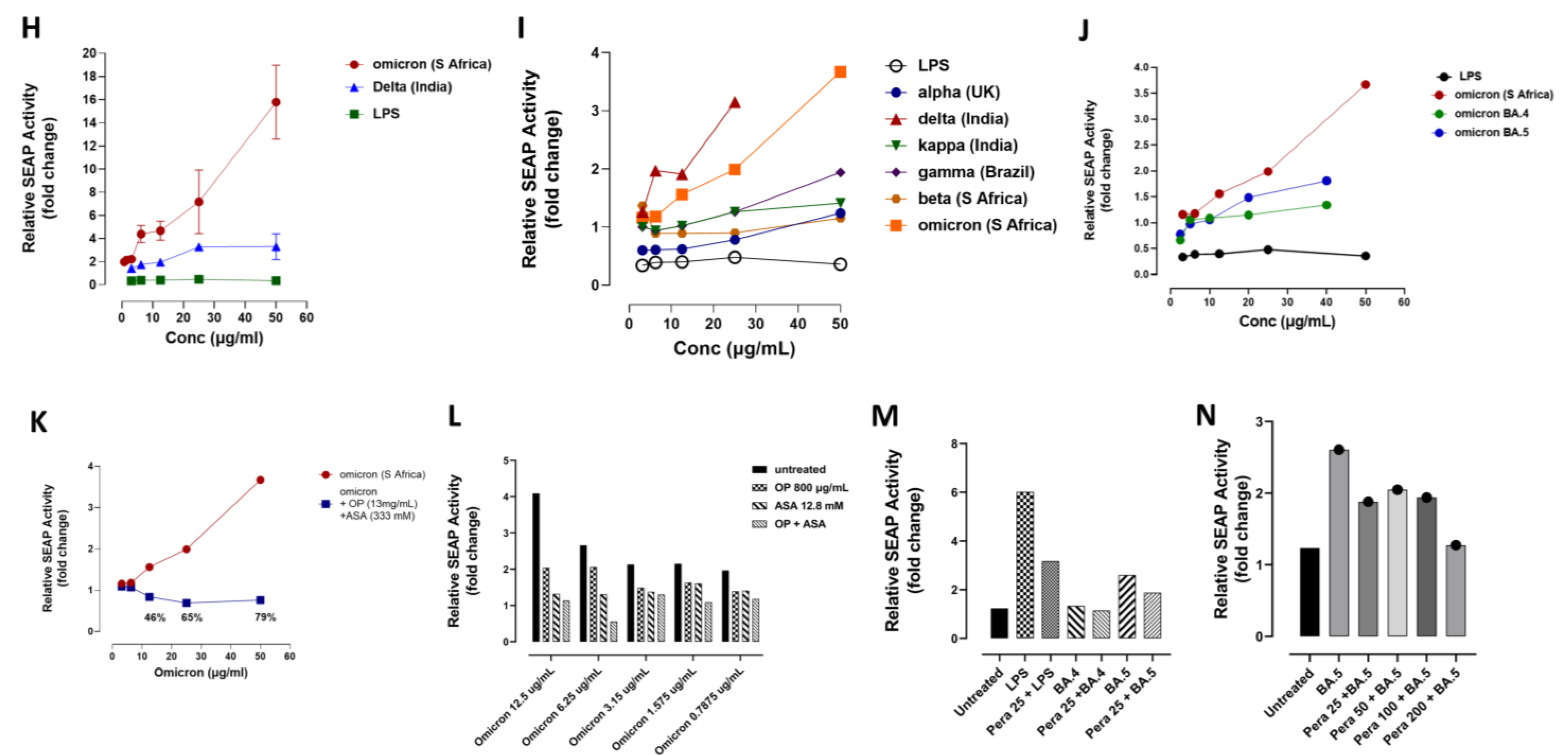
3.3. Recombinant SARS-CoV-2 S Protein Induces Upregulation of NF-kB That Is Downregulated by Selective Inhibitors of Neu-1 Oseltamivir Phosphate (OP) and Aspirin (ASA)
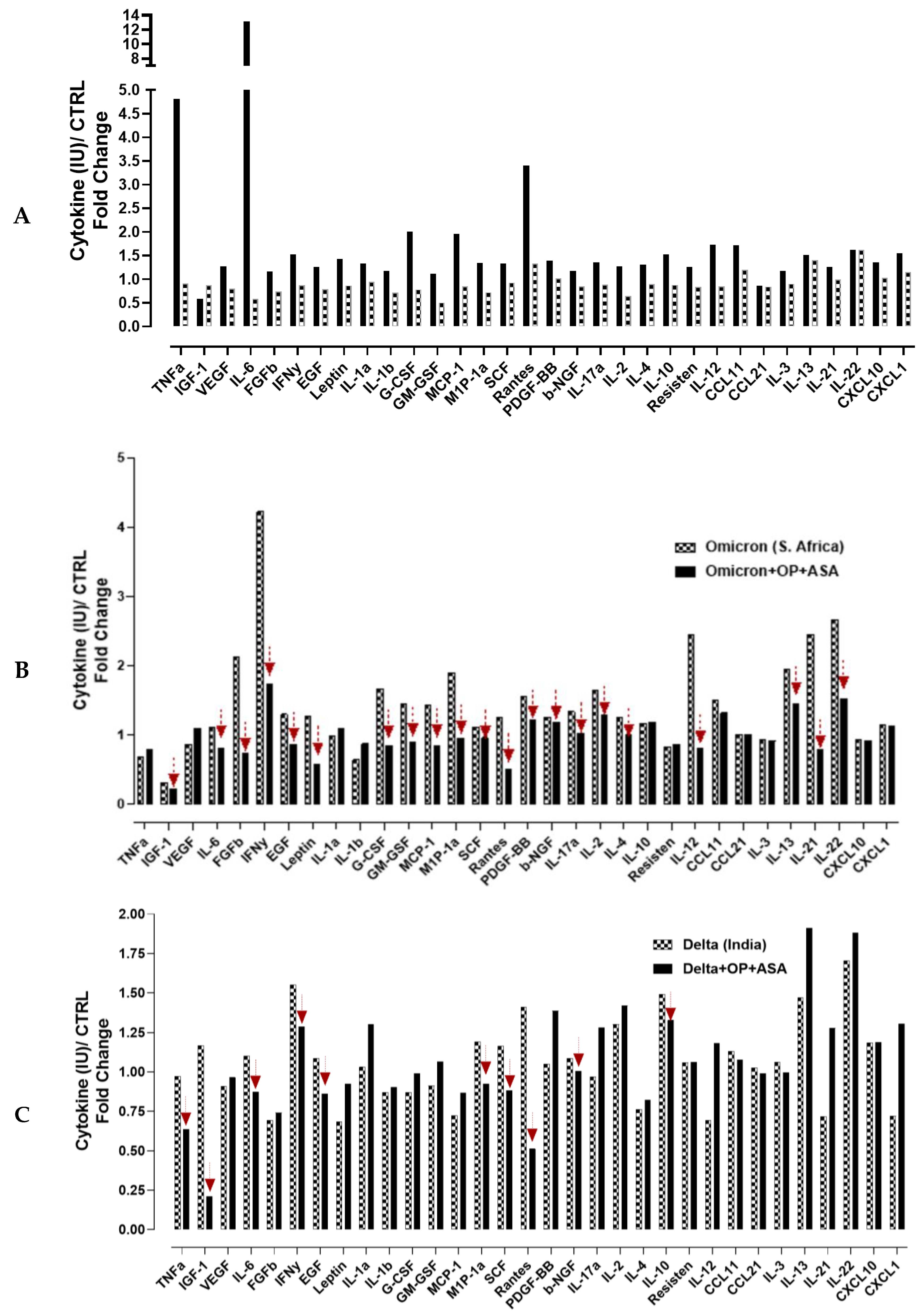
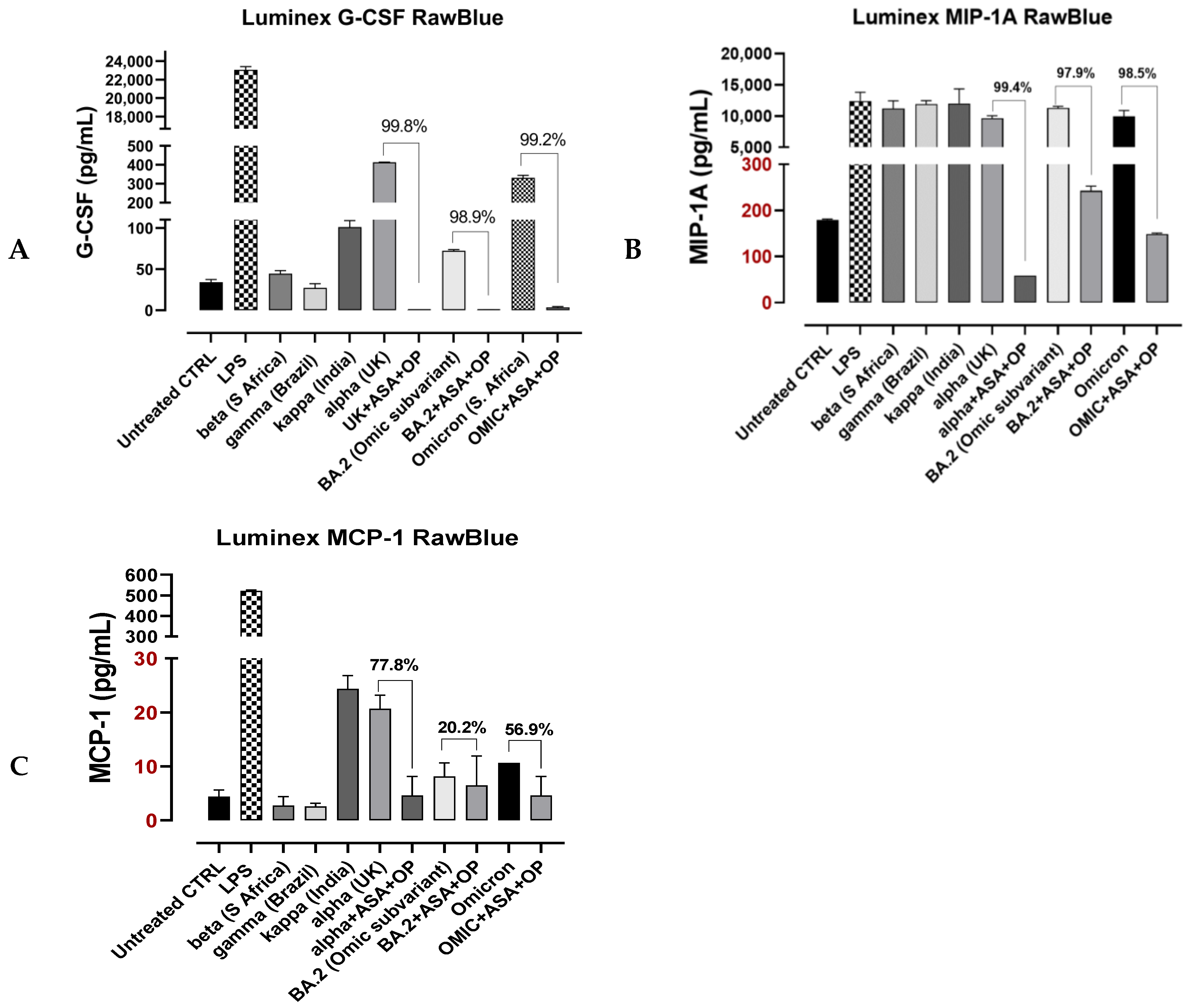
3.4. Recombinant SARS-CoV-2 S Protein Stimulation of RawBlue Macrophages Triggers the Upregulation of Distinct Cytokines Implicated in SARS-CoV-2 Pathogenesis
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Patents
References
- Shang, J.; Ye, G.; Shi, K.; Wan, Y.; Luo, C.; Aihara, H.; Geng, Q.; Auerbach, A.; Li, F. Structural basis of receptor recognition by SARS-CoV-2. Nature 2020, 581, 221–224. [Google Scholar] [CrossRef] [PubMed]
- Otsuka, R.; Seino, K.I. Macrophage activation syndrome and COVID-19. Inflamm. Regen 2020, 40, 19. [Google Scholar] [CrossRef] [PubMed]
- Gibson, P.G.; Qin, L.; Puah, S.H. COVID-19 acute respiratory distress syndrome (ARDS): Clinical features and differences from typical pre-COVID-19 ARDS. Med. J. Aust. 2020, 213, 54–56.e1. [Google Scholar] [CrossRef] [PubMed]
- Khanmohammadi, S.; Rezaei, N. Role of Toll-like receptors in the pathogenesis of COVID-19. J. Med. Virol. 2021, 93, 2735–2739. [Google Scholar] [CrossRef] [PubMed]
- Huang, C.; Tan, Z.; Zhao, K.; Zou, W.; Wang, H.; Gao, H.; Sun, S.; Bu, D.; Chai, W.; Li, Y. The effect of N-glycosylation of SARS-CoV-2 spike protein on the virus interaction with the host cell ACE2 receptor. iScience 2021, 24, 103272. [Google Scholar] [CrossRef] [PubMed]
- Duan, T.; Du, Y.; Xing, C.; Wang, H.Y.; Wang, R.F. Toll-Like Receptor Signaling and Its Role in Cell-Mediated Immunity. Front. Immunol. 2022, 13, 812774. [Google Scholar] [CrossRef]
- Takeuchi, O.; Akira, S. Pattern recognition receptors and inflammation. Cell 2010, 140, 805–820. [Google Scholar] [CrossRef]
- Roger, T.; Froidevaux, C.; Le Roy, D.; Reymond, M.K.; Chanson, A.L.; Mauri, D.; Burns, K.; Riederer, B.M.; Akira, S.; Calandra, T. Protection from lethal gram-negative bacterial sepsis by targeting Toll-like receptor 4. Proc. Natl. Acad. Sci. USA 2009, 106, 2348–2352. [Google Scholar] [CrossRef]
- Kayesh, M.E.H.; Kohara, M.; Tsukiyama-Kohara, K. An Overview of Recent Insights into the Response of TLR to SARS-CoV-2 Infection and the Potential of TLR Agonists as SARS-CoV-2 Vaccine Adjuvants. Viruses 2021, 13, 2302. [Google Scholar] [CrossRef]
- Shirato, K.; Kizaki, T. SARS-CoV-2 spike protein S1 subunit induces pro-inflammatory responses via Toll-like receptor 4 signaling in murine and human macrophages. Heliyon 2021, 7, e06187. [Google Scholar] [CrossRef]
- Zheng, M.; Karki, R.; Williams, E.P.; Yang, D.; Fitzpatrick, E.; Vogel, P.; Jonsson, C.B.; Kanneganti, T.D. TLR2 senses the SARS-CoV-2 envelope protein to produce inflammatory cytokines. Nat. Immunol. 2021, 22, 829–838. [Google Scholar] [CrossRef] [PubMed]
- Safaei, S.; Karimi-Googheri, M. Letter to the Editor: Toll-Like Receptor Antagonists as a Potential Therapeutic Strategy Against Cytokine Storm in COVID-19-Infected Patients. Viral. Immunol. 2021, 34, 361–362. [Google Scholar] [CrossRef] [PubMed]
- Cohen, M.; Zhang, X.-Q.; Senaati, H.P.; Chen, H.-W.; Varki, N.M.; Schooley, R.T.; Gagneux, P. Influenza A penetrates host mucus by cleaving sialic acids with neuraminidase. Virol. J. 2013, 10, 321. [Google Scholar] [CrossRef] [PubMed]
- Dridi, L.; Seyrantepe, V.; Fougerat, A.; Pan, X.; Bonneil, E.; Thibault, P.; Moreau, A.; Mitchell, G.A.; Heveker, N.; Cairo, C.W.; et al. Positive regulation of insulin signaling by neuraminidase 1. Diabetes 2013, 62, 2338–2346. [Google Scholar] [CrossRef] [PubMed]
- Abdulkhalek, S.; Amith, S.R.; Franchuk, S.L.; Jayanth, P.; Guo, M.; Finlay, T.; Gilmour, A.; Guzzo, C.; Gee, K.; Beyaert, R.; et al. Neu1 sialidase and matrix metalloproteinase-9 cross-talk is essential for Toll-like receptor activation and cellular signaling. J. Biol. Chem. 2011, 286, 36532–36549. [Google Scholar] [CrossRef] [PubMed]
- Abdulkhalek, S.; Hrynyk, M.; Szewczuk, M.R. A novel G-protein-coupled receptor-signaling platform and its targeted translation in human disease. Res. Rep. Biochem. 2013, 3, 17–30. [Google Scholar]
- Abdulkhalek, S.; Guo, M.; Amith, S.R.; Jayanth, P.; Szewczuk, M.R. G-protein coupled receptor agonists mediate Neu1 sialidase and matrix metalloproteinase-9 cross-talk to induce transactivation of Toll-like receptors and cellular signaling. Cell. Signal. 2012, 24, 2035–2042. [Google Scholar] [CrossRef]
- Abdulkhalek, S.; Szewczuk, M.R. Neu1 sialidase and matrix metalloproteinase-9 cross-talk regulates nucleic acid-induced endosomal Toll-like receptor-7 and -9 activation, cellular signaling and pro-inflammatory responses. Cell. Signal. 2013, 25, 2093–2105. [Google Scholar] [CrossRef]
- Amith, S.R.; Jayanth, P.; Franchuk, S.; Siddiqui, S.; Seyrantepe, V.; Gee, K.; Basta, S.; Beyaert, R.; Pshezhetsky, A.V.; Szewczuk, M.R. Dependence of pathogen molecule-induced Toll-like receptor activation and cell function on Neu1 sialidase. Glycoconj. J. 2009, 26, 1197–1212. [Google Scholar] [CrossRef]
- Gilmour, A.M.; Abdulkhalek, S.; Cheng, T.S.; Alghamdi, F.; Jayanth, P.; O’Shea, L.K.; Geen, O.; Arvizu, L.A.; Szewczuk, M.R. A novel epidermal growth factor receptor-signaling platform and its targeted translation in pancreatic cancer. Cell. Signal. 2013, 25, 2587–2603. [Google Scholar] [CrossRef]
- Alghamdi, F.; Guo, M.; Abdulkhalek, S.; Crawford, N.; Amith, S.R.; Szewczuk, M.R. A novel insulin receptor-signaling platform and its link to insulin resistance and type 2 diabetes. Cell. Signal. 2014, 26, 1355–1368. [Google Scholar] [CrossRef] [PubMed]
- Haxho, F.; Haq, S.; Szewczuk, M.R. Biased G protein-coupled receptor agonism mediates Neu1 sialidase and matrix metalloproteinase-9 crosstalk to induce transactivation of insulin receptor signaling. Cell. Signal. 2018, 43, 71–84. [Google Scholar] [CrossRef] [PubMed]
- Haxho, F.; Neufeld, R.J.; Szewczuk, M.R. Neuraminidase-1: A novel therapeutic target in multistage tumorigenesis. Oncotarget 2016, 7, 40860–40881. [Google Scholar] [CrossRef] [PubMed]
- Haxho, F.; Alghamdi, F.; Neufeld, R.J.; Szewczuk, M.R. Novel Insulin Receptor-Signaling Platform. Int. J. Diabetes Clin. Res. 2014, 1, 1–10. [Google Scholar] [CrossRef]
- Jayanth, P.; Amith, S.R.; Gee, K.; Szewczuk, M.R. Neu1 sialidase and matrix metalloproteinase-9 cross-talk is essential for neurotrophin activation of Trk receptors and cellular signaling. Cell. Signal. 2010, 22, 1193–1205. [Google Scholar] [CrossRef]
- Woronowicz, A.; Amith, S.R.; Davis, V.W.; Jayanth, P.; De Vusser, K.; Laroy, W.; Contreras, R.; Meakin, S.O.; Szewczuk, M.R. Trypanosome trans-sialidase mediates neuroprotection against oxidative stress, serum/glucose deprivation, and hypoxia-induced neurite retraction in Trk-expressing PC12 cells. Glycobiology 2007, 17, 725–734. [Google Scholar] [CrossRef]
- Woronowicz, A.; Amith, S.R.; De Vusser, K.; Laroy, W.; Contreras, R.; Basta, S.; Szewczuk, M.R. Dependence of neurotrophic factor activation of Trk tyrosine kinase receptors on cellular sialidase. Glycobiology 2007, 17, 10–24. [Google Scholar] [CrossRef] [PubMed]
- Woronowicz, A.; De Vusser, K.; Laroy, W.; Contreras, R.; Meakin, S.O.; Ross, G.M.; Szewczuk, M.R. Trypanosome trans-sialidase targets TrkA tyrosine kinase receptor and induces receptor internalization and activation. Glycobiology 2004, 14, 987–998. [Google Scholar] [CrossRef]
- Yan, R.; Zhang, Y.; Li, Y.; Xia, L.; Guo, Y.; Zhou, Q. Structural basis for the recognition of SARS-CoV-2 by full-length human ACE2. Science 2020, 367, 1444–1448. [Google Scholar] [CrossRef]
- Wrapp, D.; Wang, N.; Corbett, K.S.; Goldsmith, J.A.; Hsieh, C.-L.; Abiona, O.; Graham, B.S.; McLellan, J.S. Cryo-EM structure of the 2019-nCoV spike in the prefusion conformation. Science 2020, 367, 1260–1263. [Google Scholar] [CrossRef]
- Pyne, N.J.; Pyne, S. Sphingosine 1-phosphate, lysophosphatidic acid and growth factor signaling and termination. Biochim. Biophys. Acta (BBA)—Mol. Cell Biol. Lipids 2008, 1781, 467–476. [Google Scholar] [CrossRef] [PubMed]
- Slieker, L.J.; Lane, M.D. Post-translational processing of the epidermal growth factor receptor. Glycosylation-dependent acquisition of ligand-binding capacity. J. Biol. Chem. 1985, 260, 687–690. [Google Scholar] [CrossRef] [PubMed]
- Soderquist, A.M.; Carpenter, G. Glycosylation of the epidermal growth factor receptor in A-431 cells. The contribution of carbohydrate to receptor function. J. Biol. Chem. 1984, 259, 12586–12594. [Google Scholar] [CrossRef] [PubMed]
- Fernandes, H.; Cohen, S.; Bishayee, S. Glycosylation-induced Conformational Modification Positively Regulates Receptor-Receptor Association: A Study with An Aberrant Epidermal Growth Factor Receptor (EGFRvIII/ΔEGFR) EXPRESSED In Cancer Cells. J. Biol. Chem. 2001, 276, 5375–5383. [Google Scholar] [CrossRef] [PubMed]
- Salton, F.; Confalonieri, P.; Campisciano, G.; Cifaldi, R.; Rizzardi, C.; Generali, D.; Pozzan, R.; Tavano, S.; Bozzi, C.; Lapadula, G.; et al. Cytokine Profiles as Potential Prognostic and Therapeutic Markers in SARS-CoV-2-Induced ARDS. J. Clin. Med. 2022, 11, 2951. [Google Scholar] [CrossRef] [PubMed]
- Wang, H.; Paulson, K.R.; Pease, S.A.; Watson, S.; Comfort, H.; Zheng, P.; Aravkin, A.Y.; Bisignano, C.; Barber, R.M.; Alam, T.; et al. Estimating excess mortality due to the COVID-19 pandemic: A systematic analysis of COVID-19-related mortality. Lancet 2022, 399, 1513–1536. [Google Scholar] [CrossRef]
- Merad, M.; Martin, J.C. Pathological inflammation in patients with COVID-19: A key role for monocytes and macrophages. Nat. Rev. Immunol. 2020, 20, 355–362. [Google Scholar] [CrossRef]
- Channappanavar, R.; Perlman, S. Pathogenic human coronavirus infections: Causes and consequences of cytokine storm and immunopathology. Semin. Immunopathol. 2017, 39, 529–539. [Google Scholar] [CrossRef]
- Buchan, S.A.; Chung, H.; Brown, K.A.; Austin, P.C.; Fell, D.B.; Gubbay, J.B.; Nasreen, S.; Schwartz, K.L.; Sundaram, M.E.; Tadrous, M.; et al. Estimated Effectiveness of COVID-19 Vaccines Against Omicron or Delta Symptomatic Infection and Severe Outcomes. JAMA Netw. Open 2022, 5, e2232760. [Google Scholar] [CrossRef]
- Mutoh, Y.; Umemura, T.; Ota, A.; Okuda, K.; Moriya, R.; Tago, M.; Soejima, K.; Noguchi, Y.; Bando, T.; Ota, S.; et al. Effectiveness of monoclonal antibody therapy for COVID-19 patients using a risk scoring system. J. Infect. Chemother. 2022, 28, 352–355. [Google Scholar] [CrossRef]
- Janik, E.; Niemcewicz, M.; Podogrocki, M.; Majsterek, I.; Bijak, M. The Emerging Concern and Interest SARS-CoV-2 Variants. Pathogens 2021, 10, 633. [Google Scholar] [CrossRef] [PubMed]
- Chen, R.E.; Zhang, X.; Case, J.B.; Winkler, E.S.; Liu, Y.; VanBlargan, L.A.; Liu, J.; Errico, J.M.; Xie, X.; Suryadevara, N.; et al. Resistance of SARS-CoV-2 variants to neutralization by monoclonal and serum-derived polyclonal antibodies. Nat. Med. 2021, 27, 717–726. [Google Scholar] [CrossRef] [PubMed]
- Harvey, W.T.; Carabelli, A.M.; Jackson, B.; Gupta, R.K.; Thomson, E.C.; Harrison, E.M.; Ludden, C.; Reeve, R.; Rambaut, A.; Peacock, S.J.; et al. SARS-CoV-2 variants, spike mutations and immune escape. Nat. Rev. Microbiol. 2021, 19, 409–424. [Google Scholar] [CrossRef]
- Guarner, J. Three Emerging Coronaviruses in Two Decades. Am. J. Clin. Pathol. 2020, 153, 420–421. [Google Scholar] [CrossRef] [PubMed]
- Hui, D.S.; Azhar, E.I.; Kim, Y.J.; Memish, Z.A.; Oh, M.D.; Zumla, A. Middle East respiratory syndrome coronavirus: Risk factors and determinants of primary, household, and nosocomial transmission. Lancet Infect. Dis. 2018, 18, e217–e227. [Google Scholar] [CrossRef] [PubMed]
- Li, W.; Moore, M.J.; Vasilieva, N.; Sui, J.; Wong, S.K.; Berne, M.A.; Somasundaran, M.; Sullivan, J.L.; Luzuriaga, K.; Greenough, T.C.; et al. Angiotensin-converting enzyme 2 is a functional receptor for the SARS coronavirus. Nature 2003, 426, 450–454. [Google Scholar] [CrossRef]
- Brown, E.E.F.; Rezaei, R.; Jamieson, T.R.; Dave, J.; Martin, N.T.; Singaravelu, R.; Crupi, M.J.F.; Boulton, S.; Tucker, S.; Duong, J.; et al. Characterization of Critical Determinants of ACE2-SARS-CoV-2 RBD Interaction. Int. J. Mol. Sci. 2021, 22, 2268. [Google Scholar] [CrossRef]
- Wu, J.; Zhao, M.; Wei, H.; Li, C.; Hu, D.; Zheng, L.; Wang, D.W. Neuraminidase inhibitor treatment is associated with decreased mortality in COVID-19 patients: A retrospective analysis. Eur. Heart J. Cardiovasc. Pharmacother. 2022, 8, 392–401. [Google Scholar] [CrossRef]
- Diaz, G.A.; Christensen, A.B.; Pusch, T.; Goulet, D.; Chang, S.C.; Grunkemeier, G.L.; McKelvey, P.A.; Robicsek, A.; French, T.; Parsons, G.T.; et al. Remdesivir and Mortality in Patients with Coronavirus Disease 2019. Clin. Infect. Dis. 2022, 74, 1812–1820. [Google Scholar] [CrossRef]
- Chokkalingam, A.P.; Hayden, J.; Goldman, J.D.; Li, H.; Asubonteng, J.; Mozaffari, E.; Bush, C.; Wang, J.R.; Kong, A.; Osinusi, A.O.; et al. Association of Remdesivir Treatment with Mortality Among Hospitalized Adults with COVID-19 in the United States. JAMA Netw. Open 2022, 5, e2244505. [Google Scholar] [CrossRef]
- Ma, S.; Su, W.; Sun, C.; Lowe, S.; Zhou, Z.; Liu, H.; Qu, G.; Xia, W.; Xie, P.; Wu, B.; et al. Does aspirin have an effect on risk of death in patients with COVID-19? A meta-analysis. Eur. J. Clin. Pharmacol. 2022, 78, 1403–1420. [Google Scholar] [CrossRef] [PubMed]
- Chow, J.H.; Rahnavard, A.; Gomberg-Maitland, M.; Chatterjee, R.; Patodi, P.; Yamane, D.P.; Levine, A.R.; Davison, D.; Hawkins, K.; Jackson, A.M.; et al. Association of Early Aspirin Use with In-Hospital Mortality in Patients with Moderate COVID-19. JAMA Netw. Open 2022, 5, e223890. [Google Scholar] [CrossRef] [PubMed]
- Qorri, B.; Harless, W.; Szewczuk, M.R. Novel Molecular Mechanism of Aspirin and Celecoxib Targeting Mammalian Neuraminidase-1 Impedes Epidermal Growth Factor Receptor Signaling Axis and Induces Apoptosis in Pancreatic Cancer Cells. Drug Des. Devel. Ther. 2020, 14, 4149–4167. [Google Scholar] [CrossRef] [PubMed]
- Yang, D.; Wu, Y.; Turan, I.; Keil, J.; Li, K.; Chen, M.H.; Liu, R.; Wang, L.; Sun, X.L.; Chen, G.Y. Targeting intracellular Neu1 for coronavirus infection treatment. iScience 2023, 26, 106037. [Google Scholar] [CrossRef] [PubMed]


Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Harless, W.W.; Lewis, B.; Qorri, B.; Abdulkhalek, S.; Szewczuk, M.R. Novel Therapeutic Target Critical for SARS-CoV-2 Infectivity and Induction of the Cytokine Release Syndrome. Cells 2023, 12, 1332. https://doi.org/10.3390/cells12091332
Harless WW, Lewis B, Qorri B, Abdulkhalek S, Szewczuk MR. Novel Therapeutic Target Critical for SARS-CoV-2 Infectivity and Induction of the Cytokine Release Syndrome. Cells. 2023; 12(9):1332. https://doi.org/10.3390/cells12091332
Chicago/Turabian StyleHarless, William W., Beth Lewis, Bessi Qorri, Samar Abdulkhalek, and Myron R. Szewczuk. 2023. "Novel Therapeutic Target Critical for SARS-CoV-2 Infectivity and Induction of the Cytokine Release Syndrome" Cells 12, no. 9: 1332. https://doi.org/10.3390/cells12091332
APA StyleHarless, W. W., Lewis, B., Qorri, B., Abdulkhalek, S., & Szewczuk, M. R. (2023). Novel Therapeutic Target Critical for SARS-CoV-2 Infectivity and Induction of the Cytokine Release Syndrome. Cells, 12(9), 1332. https://doi.org/10.3390/cells12091332






