The Role of TLR-2 in Lethal COVID-19 Disease Involving Medullary and Resident Lung Megakaryocyte Up-Regulation in the Microthrombosis Mechanism
Abstract
:1. Introduction
2. Materials and Methods
2.1. Clinicopathological Data of Selected Patients
2.2. Autopsy Protocol
2.3. Methods
2.3.1. Histological Analysis
2.3.2. Immunohistochemistry
2.3.3. RT-PCR
RNA Was Extracted Using 30 μL Buffer and Used for RT-PCR Analysis
2.3.4. In Situ Hybridization (ISH)
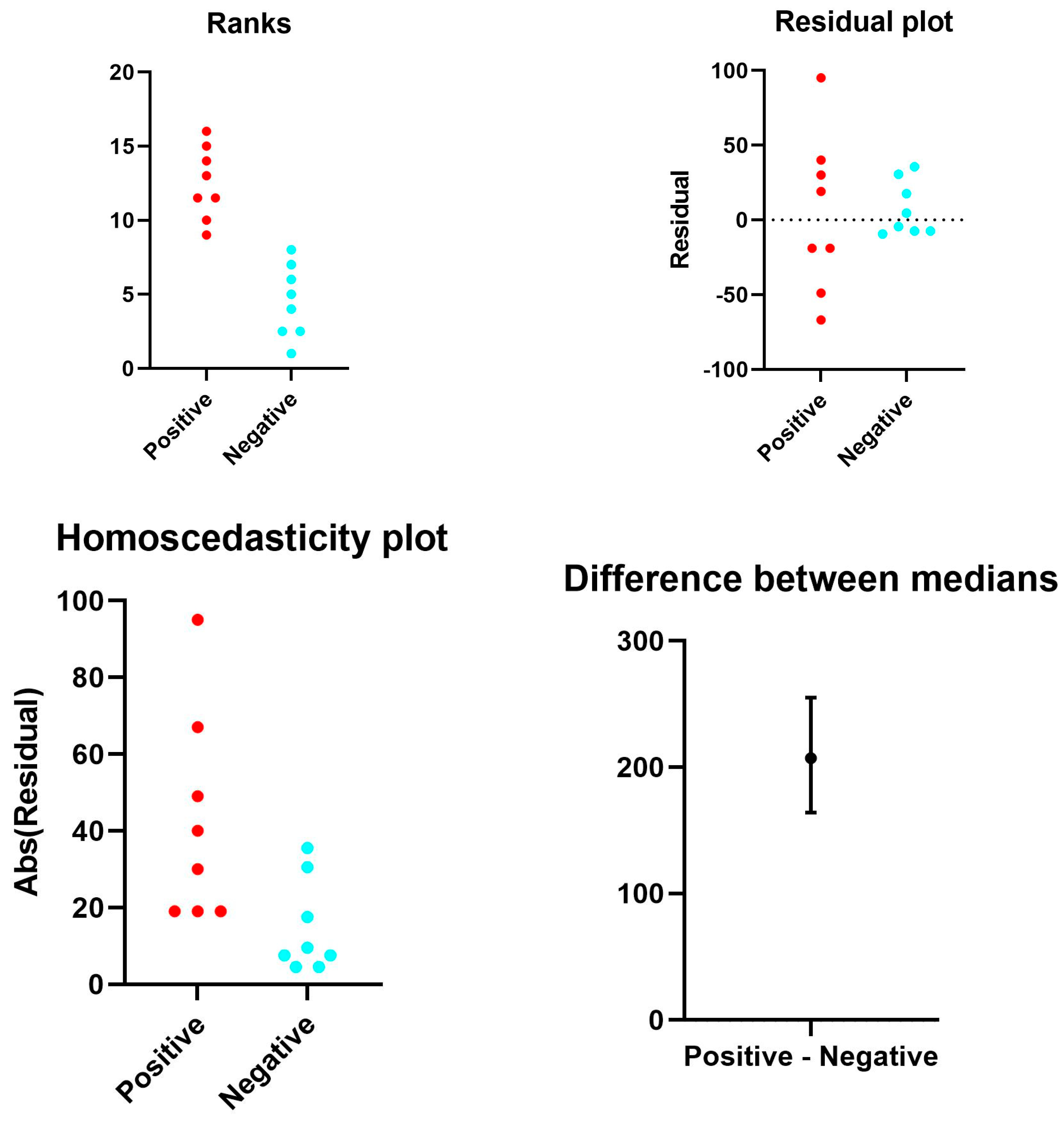
2.3.5. Statistical Analyses
2.3.6. Digital Pathology and Software AI
3. Results
3.1. Main Histopathological Findings
3.2. Outcomes of RT-PCR and In Situ Hybridization (ISH)
3.3. A Subgroup of Patients with Fatal COVID-19 Lung Disease Had Elevated TLR-2 Expression
3.4. A Subset of Individuals with an Extremely Fatal COVID-19 Lung Illness Had Elevated Levels of Platelet Membrane Glycoprotein IIIa (CD61)
3.5. Results of RT-PCR and ISH
3.6. Results Evaluated by Digital Pathology Analysis
4. Discussion
Formation of Fibrin-Rich Thrombi Is a Key Aspect of DAD
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Menter, D.G.; Kopetz, S.; Hawk, E.; Sood, A.K.; Loree, J.M.; Gresle, P.; Honn, V.K. Platelet “first responders” in wound response, cancer, and metastasis. Cancer Metastasis Rev. 2017, 36, 199–213. [Google Scholar] [CrossRef] [PubMed]
- Yin, S.; Huang, M.; Li, D.; Tang, N. Difference of coagulation features between severe pneumonia induced by SARS-CoV2 and non-SARS-CoV2. J. Thromb. Thrombolysis 2020, 51, 1107–1110. [Google Scholar] [CrossRef] [PubMed]
- Lippi, G.; Plebani, M.; Henry, B.M. Thrombocytopenia is associated with severe coronavirus disease 2019 (COVID-19) infections: A meta-analysis. Clin. Chim. Acta 2020, 506, 145–148. [Google Scholar] [CrossRef]
- Yang, X.; Yang, Q.; Wang, Y.; Wu, Y.; Xu, J.; Yu, Y.; Shang, Y. Thrombocytopenia and its Association with Mortality in Patients with COVID-19. J. Thromb. Haemost. 2020, 18, 1469–1472. [Google Scholar] [CrossRef]
- D’Atri, L.P.; Schattner, M. Platelet toll-like receptors in thromboinflammation. Front. Biosci. 2017, 22, 1867–1883. [Google Scholar]
- Hamzeh-Cognasse, H.; Berthelot, P.; Tardy, B.; Pozzetto, B.; Bourlet, T.; Laradi, S.; Garraud, O.; Cognasse, F. Platelet toll-like receptors are crucial sensors of infectious danger moieties. Platelets 2018, 29, 533–540. [Google Scholar] [CrossRef] [PubMed]
- Semeraro, F.; Ammollo, C.T.; Morrissey, J.H.; Dale, G.L.; Friese, P.; Esmon, N.L.; Esmon, C.T. Extracellular histones promotethrombin generation through platelet-dependent mechanisms: Involvement of platelet TLR2 and TLR4. Blood 2011, 118, 1952–1961. [Google Scholar] [CrossRef] [PubMed]
- Into, T.; Kanno, Y.; Dohkan, J.I.; Nakashima, M.; Inomata, M.; Shibata, K.I.; Lowenstein, C.J.; Matsushita, K. Pathogen Recognition by Toll-like Receptor 2 Activates Weibel-Palade Body Exocytosis in Human Aortic Endothelial Cells. J. Biol. Chem. 2007, 282, 8134–8141. [Google Scholar] [CrossRef]
- Singh, B.; Biswas, I.; Bhagat, S.; Surya Kumari, S.; Khan, G.A. HMGB1 facilitates hypoxia-induced vWF upregulation through TLR2-MYD88-SP1 pathway. Eur. J. Immunol. 2016, 46, 2388–2400. [Google Scholar] [CrossRef]
- Carestia, A.; Kaufman, T.; Rivadeneyra, L.; Landoni, V.I.; Pozner, R.G.; Negrotto, S.; D’Atri, L.P.; Gómez, R.M.; Schattner, M. Mediators and molecular pathways involved in the regulation of neutrophil extracellular trap formation mediated by activated platelets. J. Leukoc. Biol. 2016, 99, 153–162. [Google Scholar] [CrossRef]
- Fujimura, Y.; Holland, L.Z. COVID-19 microthrombosis: Unusually large VWF multimers are a platform for activation of the alternative complement pathway under cytokine storm. Int. J. Hematol. 2022, 115, 457–469. [Google Scholar] [CrossRef] [PubMed]
- Mussbacher, M.; Salzmann, M.; Brostjan, C.; Hoesel, B.; Schoergenhofer, C.; Datler, H.; Hohensinner, P.; Basílio, J.; Petzelbauer, P.; Assinger, A.; et al. Cell Type-Specific Roles of NF-κB Linking Inflammation and Thrombosis. Front. Immunol. 2019, 10, 85. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Calabrese, F.; Pezzuto, F.; Fortarezza, F.; Hofman, P.; Ker, I.; Panizo, A.; Thusen, V.V.d.; Timofeev, S.; Gorkiewicz, G.; Lunardi, F. Pulmonary pathology and COVID-19: Lessons from autopsy: The experience of European Pulmonary Pathologists. Virchows Arch. 2020, 477, 359–372. [Google Scholar] [CrossRef] [PubMed]
- Pedicillo, M.C.; De Stefano, I.S.; Zamparese, R.; Barile, R.; Meccariello, M.; Agostinone, A.; Villani, G.; Colangelo, T.; Serviddio, G.; Cassano, T.; et al. The Role of Toll-Like receptor-4 in Macrophage Imbalance in Lethal COVID19 Lung Disease, and Its Correlation with Galectina-3. Int. J. Mol. Sci. 2023, 24, 13259. [Google Scholar] [CrossRef] [PubMed]
- Basso, C.; Calabrese, F.; Sbaraglia, M.; Del Vecchio, C.; Carretta, G.; Saieva, A.; Donato, D.; Flor, L.; Crisanti, A.; Tos, A.P.D. Feasibility of postmortem examination in the era of COVID-19 pandemic: The experience of a Northeast Italy University Hospital. Virchows Arch. 2020, 477, 341–347. [Google Scholar] [CrossRef] [PubMed]
- Ronchi, A.; Marino, F.Z.; Carraturo, E.; La Mantia, E.; Campobasso, C.P.; De Micco, F.; Mascolo, P.; Municinò, M.; Mucininò, E.; Vestini, F.; et al. PD-L1 Overexpression in the Lungs of Subjects Who Died from COVID-19: Are We on the Way to Understandingthe Immune System Exhaustion Induced by SARS-CoV-2? Crit. Rev. Eukaryot. Gene Expr. 2022, 32, 9–20. [Google Scholar] [CrossRef] [PubMed]
- Hooper, J.E.; Padera, R.F.; Dolhnikoff, M.; da Silva, L.; Duarte-Neto, A.N.; Kapp, M.E.; Lacy, J.M.; Mauad, T.; Saldiva, P.; Rapkiewicz, A.V.; et al. A Postmortem Portrait of the Coronavirus Disease 2019 (COVID-19) Pandemic: A Large Multi-institutional Autopsy Survey Study. Arch. Pathol. Lab. Med. 2021, 145, 529–535. [Google Scholar] [CrossRef] [PubMed]
- Massoth, L.R.; Desai, N.; Szabolcs, A.; Harris, C.K.; Neyaz, A.; Crotty, R.; Chebib, I.; Rivera, M.N.; Sholl, L.M.; Stone, J.R.; et al. Comparison of RNA In Situ Hybridization and Immunohistochemistry Techniques for the Detection and Localization ofSARS-CoV-2 in Human Tissues. Am. J. Surg. Pathol. 2021, 45, 14–24. [Google Scholar] [CrossRef]
- Roden, A.C.; Vrana, J.A.; Koepplin, J.W.; Hudson, A.E.; Norgan, A.P.; Jenkinson, G.; Yamaoka, S.; Ebihara, H.; Monroe, R.; Szabolcs, M.J.; et al. Comparison of In Situ Hybridization, Immunohistochemistry, and Reverse Transcription-Droplet Digital Polymerase Chain Reaction for Severe Acute Respiratory Syndrome Coronavirus 2 (SARS-CoV-2) Testing in Tissue. Arch. Pathol. Lab. Med. 2021, 145, 785–796. [Google Scholar] [CrossRef]
- Zaid, Y.; Guessous, F. The ongoing enigma of SARS-CoV-2 and platelet interaction. Res. Pract. Thromb. Haemost. 2022, 6, e12642. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Zuniga, E.I.; Macal, M.; Lewis, G.M.; Harker, J.A. Innate and Adaptive Immune Regulation during Chronic Viral Infections. Annu. Rev. Virol. 2015, 2, 573–597. [Google Scholar] [CrossRef] [PubMed]
- Takeuchi, O.; Akira, S. Pattern Recognition Receptors and Inflammation. Cell 2010, 140, 805–820. [Google Scholar] [CrossRef]
- Weyrich, A.S.; Zimmerman, G.A. Platelets in lung biology. Annu. Rev. Physiol. 2013, 75, 569–591. [Google Scholar] [CrossRef] [PubMed]
- Lefrançais, E.; Ortiz-Muñoz, G.; Caudrillier, A.; Mallavia, B.; Liu, F.; Sayah, D.M.; Thornton, E.E.; Headley, M.B.; David, T.; Coughlin, S.R.; et al. The lung is a site of platelet biogenesis and a reservoir for haematopoietic progenitors. Nature 2017, 544, 105–109. [Google Scholar] [CrossRef] [PubMed]
- Gordon, M.S.; Hoffman, R. Growth factors affecting human thrombocytopoiesis: Potential agents for the treatment of thrombocytopenia. Blood 1992, 80, 302–307. [Google Scholar] [CrossRef] [PubMed]
- Pang, L.; Weiss, M.J.; Poncz, M. Megakaryocyte biology and related disorders. J. Clin. Investig. 2005, 115, 3332–3338. [Google Scholar] [CrossRef]
- Nurden, A.T. The biology of the platelet with special reference to inflammation, wound healing and immunity. Front. Biosci. (Landmark Ed) 2018, 23, 726–751. [Google Scholar] [CrossRef]
- Noetzli, L.J.; French, S.L.; Machlus, K.R. New Insights Into the Differentiation of Megakaryocytes From Hematopoietic Progenitors. Arterioscler. Thromb. Vasc. Biol. 2019, 39, 1288–1300. [Google Scholar] [CrossRef]
- Burstein, S.A. Effects of interleukin 6 on megakaryocytes and on canine platelet function. Stem Cells 1994, 12, 386–393. [Google Scholar] [CrossRef]
- Viksne, V.; Strumfa, I.; Sperga, M.; Ziemelis, J.; Abolins, J. Pathological Changes in the Lungs of Patients with a Lethal COVID-19 Clinical Course. Diagnostics 2022, 12, 2808. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Maiese, A.; Manetti, A.C.; LaRussa, R.; Di Paolo, M.; Turillazi, E.; Frati, P.; Fineschi, V. Autopsy findings in COVID-19 related deaths: A literature review. Forensic Sci. Med. Pathol. 2021, 17, 279–296. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Gray-Rodriguez, S.; Jensen, M.P.; Otero-Jimenez, M.; Hanley, B.; Swann, O.C.; Ward, P.A.; Saguero, F.J.; Querido, N.; Farkas, I.; Velentza-Almpani, E.; et al. Multisystem screening reveals SARS-CoV-2 in neurons of the myenteric plexus and in megakaryocytes. J. Pathol. 2022, 257, 198–217, Epub ahead of print. [Google Scholar] [CrossRef] [PubMed]
- Shen, S.; Zhang, J.; Fang, Y.; Lu, S.; Zheng, X.; Deng, F. SARS-CoV-2 interacts with platelets and megakaryocytes via ACE2-independent mechanism. J. Hematol. Oncol. 2021, 14, 72. [Google Scholar] [CrossRef]
- Battina, H.L.; Alentado, V.J.; Srour, E.F.; Moliterno, A.R.; Kacena, M.A. Interaction of the inflammatory response and megakaryocytes in COVID-19 infection. Exp. Hematol. 2021, 104, 32–39. [Google Scholar] [CrossRef] [PubMed]








| COVID-19 Status | Immunohistochemistry | Spike-1 (ISH) Positive/Total, (%) | |
|---|---|---|---|
| CD61 Mean ± SEM | TLR2 Mean ± SEM | ||
| Positive | 108.43 ± 35.22 | 163.33 ± 38.50 | Spike-1-positive 12/25(48) |
| 35.23 ± 17.46 | 95.54 ± 19.66 | Spike-1-negative 13/25(52) | |
| Negative | 26.50 ± 7.41 | 16.33 ± 5.29 | |
| SARS-CoV2 ISH | |||
|---|---|---|---|
| Spike + | Spike − | RT-PCR | |
| 12 (48%) | 12 (48%) | + | S |
| 0 | 1 (4%) | − | |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Pannone, G.; Pedicillo, M.C.; De Stefano, I.S.; Angelillis, F.; Barile, R.; Pannone, C.; Villani, G.; Miele, F.; Municinò, M.; Ronchi, A.; et al. The Role of TLR-2 in Lethal COVID-19 Disease Involving Medullary and Resident Lung Megakaryocyte Up-Regulation in the Microthrombosis Mechanism. Cells 2024, 13, 854. https://doi.org/10.3390/cells13100854
Pannone G, Pedicillo MC, De Stefano IS, Angelillis F, Barile R, Pannone C, Villani G, Miele F, Municinò M, Ronchi A, et al. The Role of TLR-2 in Lethal COVID-19 Disease Involving Medullary and Resident Lung Megakaryocyte Up-Regulation in the Microthrombosis Mechanism. Cells. 2024; 13(10):854. https://doi.org/10.3390/cells13100854
Chicago/Turabian StylePannone, Giuseppe, Maria Carmela Pedicillo, Ilenia Sara De Stefano, Francesco Angelillis, Raffaele Barile, Chiara Pannone, Giuliana Villani, Francesco Miele, Maurizio Municinò, Andrea Ronchi, and et al. 2024. "The Role of TLR-2 in Lethal COVID-19 Disease Involving Medullary and Resident Lung Megakaryocyte Up-Regulation in the Microthrombosis Mechanism" Cells 13, no. 10: 854. https://doi.org/10.3390/cells13100854
APA StylePannone, G., Pedicillo, M. C., De Stefano, I. S., Angelillis, F., Barile, R., Pannone, C., Villani, G., Miele, F., Municinò, M., Ronchi, A., Serviddio, G., Zito Marino, F., Franco, R., Colangelo, T., & Zamparese, R. (2024). The Role of TLR-2 in Lethal COVID-19 Disease Involving Medullary and Resident Lung Megakaryocyte Up-Regulation in the Microthrombosis Mechanism. Cells, 13(10), 854. https://doi.org/10.3390/cells13100854








