Characterization of Organic Anion and Cation Transport in Three Human Renal Proximal Tubular Epithelial Models
Abstract
1. Introduction
2. Materials and Methods
2.1. Cell Culture
2.2. Culturing of Cells on Transwells and Assessment of Barrier Permeability
2.3. Resazurin Reduction Assay
2.4. Polymerase Chain Reaction (PCR)
2.5. Immunofluorescence
2.6. Western Blotting
2.7. Functional Transport Assays
2.8. Statistical Analysis
3. Results
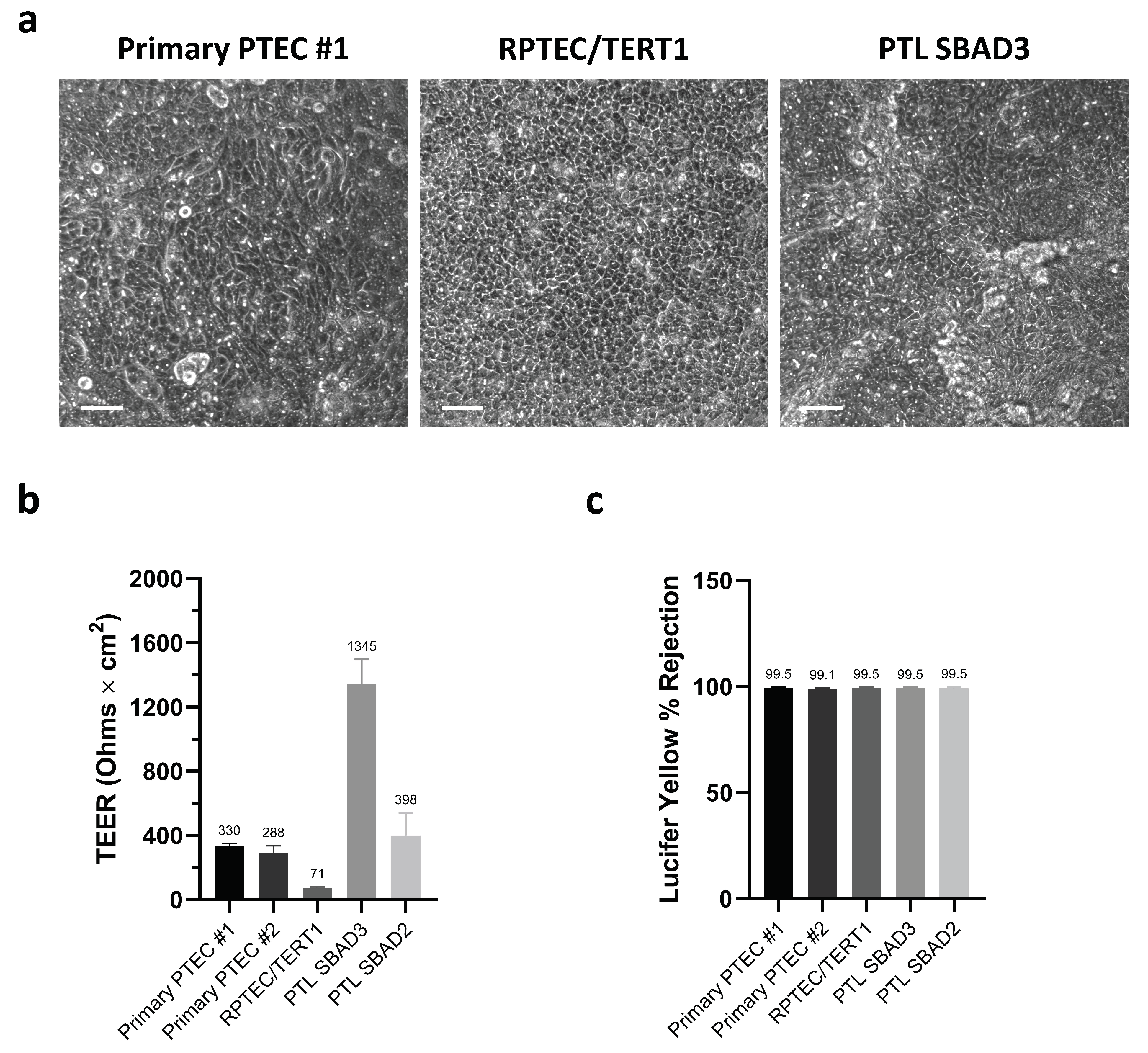
3.1. Barrier Integrity
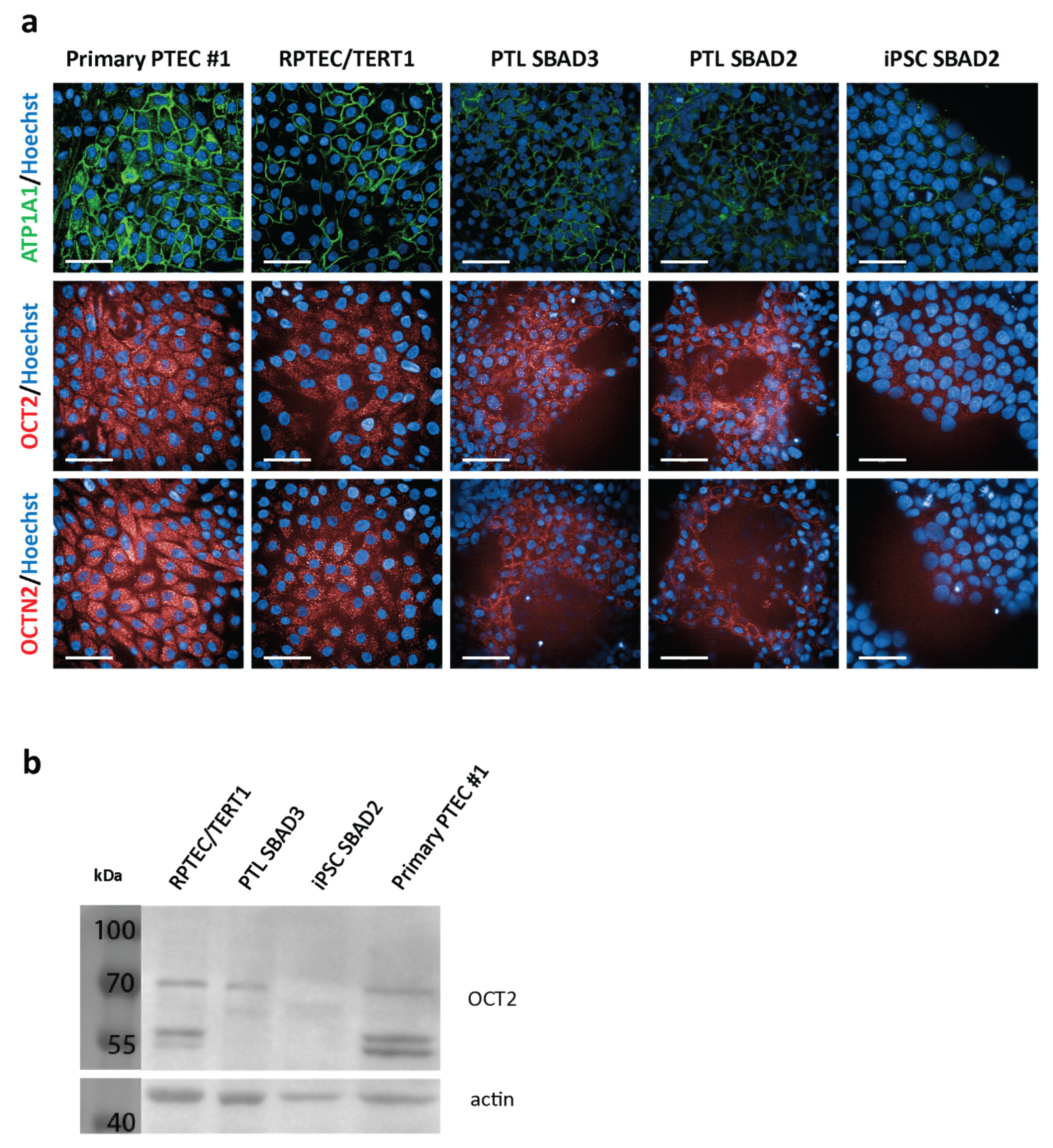
3.2. Expression of Organic Cation Transporters
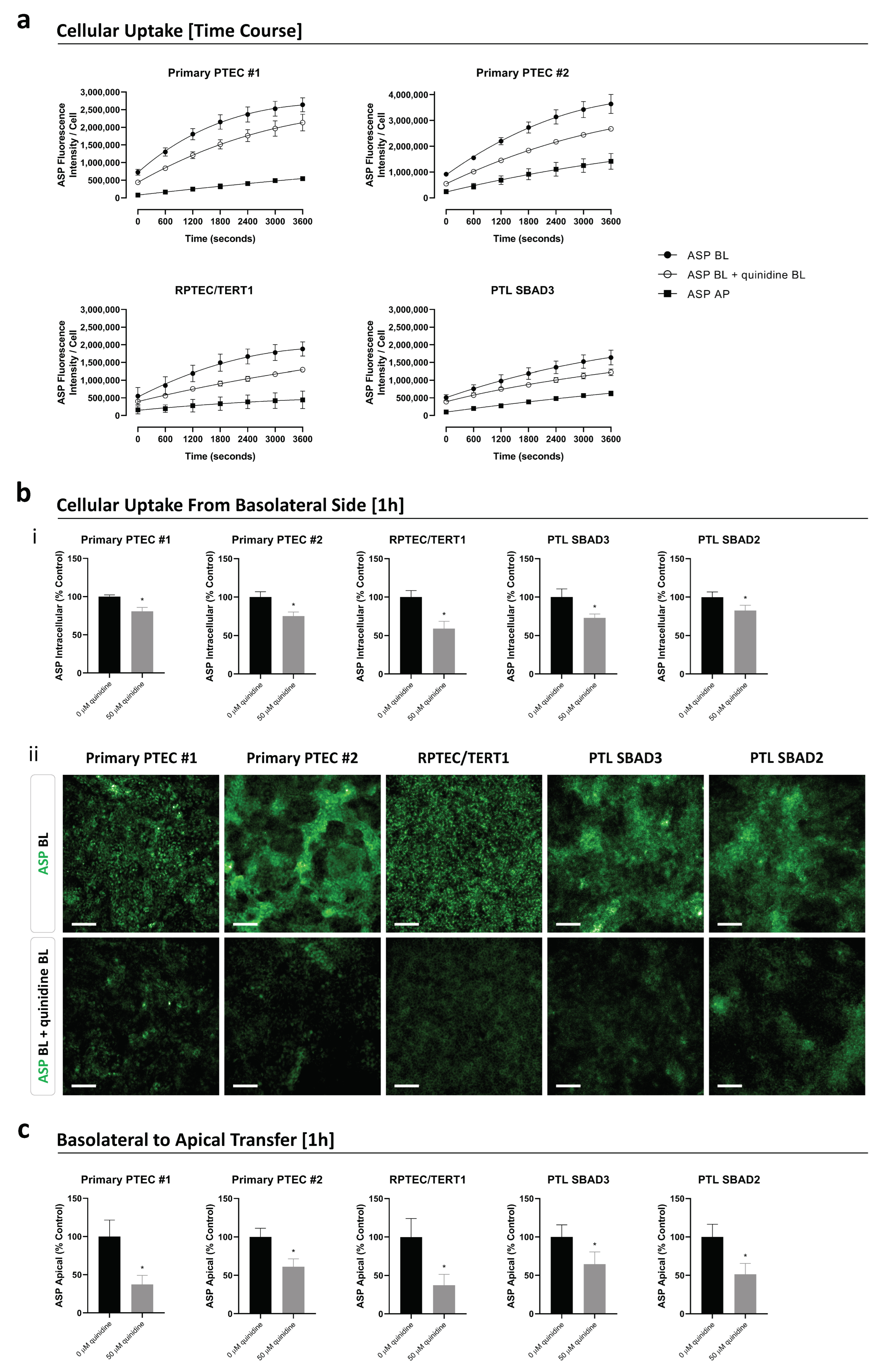
3.3. Organic Cation Transport Activity
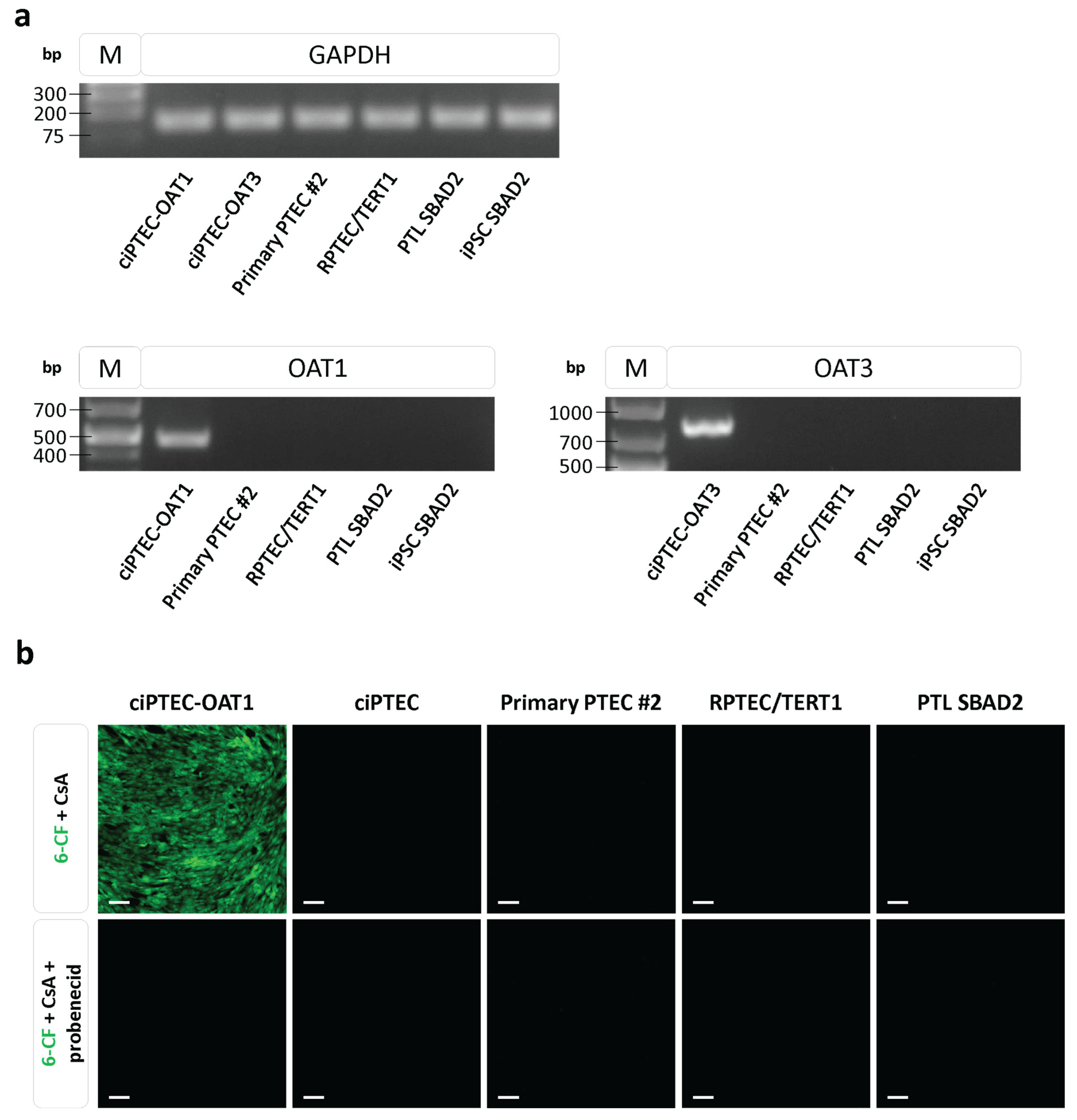
3.4. Expression and Functionality of Organic Anion Transporters
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Morrissey, K.M.; Stocker, S.L.; Wittwer, M.B.; Xu, L.; Giacomini, K.M. Renal Transporters in Drug Development. Annu. Rev. Pharmacol. Toxicol. 2013, 53, 503–529. [Google Scholar] [CrossRef]
- Wright, S.H.; Dantzler, W.H. Molecular and Cellular Physiology of Renal Organic Cation and Anion Transport. Physiol. Rev. 2004, 84, 987–1049. [Google Scholar] [CrossRef]
- Lohr, J.W.; Willsky, G.R.; Acara, M.A. Renal Drug Metabolism. Pharmacol. Rev. 1998, 50, 107–141. [Google Scholar]
- George, B.; You, D.; Joy, M.S.; Aleksunes, L.M. Xenobiotic Transporters and Kidney Injury. Adv. Drug Deliv. Rev. 2017, 116, 73–91. [Google Scholar] [CrossRef]
- Pfaller, W.; Gstraunthaler, G. Nephrotoxicity Testing in Vitro--What We Know and What We Need to Know. Environ. Health Perspect. 1998, 106, 559–569. [Google Scholar] [CrossRef]
- Awdishu, L.; Mehta, R.L. The 6R’s of Drug Induced Nephrotoxicity. BMC Nephrol. 2017, 18, 124. [Google Scholar] [CrossRef]
- Kovesdy, C.P. Epidemiology of Chronic Kidney Disease: An Update 2022. Kidney Int. Suppl. 2022, 12, 7–11. [Google Scholar] [CrossRef]
- Bikbov, B.; Purcell, C.A.; Levey, A.S.; Smith, M.; Abdoli, A.; Abebe, M.; Adebayo, O.M.; Afarideh, M.; Agarwal, S.K.; Agudelo-Botero, M.; et al. Global, Regional, and National Burden of Chronic Kidney Disease, 1990–2017: A Systematic Analysis for the Global Burden of Disease Study 2017. Lancet 2020, 395, 709–733. [Google Scholar] [CrossRef]
- Tiong, H.Y.; Huang, P.; Xiong, S.; Li, Y.; Vathsala, A.; Zink, D. Drug-Induced Nephrotoxicity: Clinical Impact and Preclinical in Vitro Models. Mol. Pharm. 2014, 11, 1933–1948. [Google Scholar] [CrossRef]
- Chu, X.; Bleasby, K.; Evers, R. Species Differences in Drug Transporters and Implications for Translating Preclinical Findings to Humans. Expert Opin. Drug Metab. Toxicol. 2013, 9, 237–252. [Google Scholar] [CrossRef]
- Martignoni, M.; Groothuis, G.M.M.; de Kanter, R. Species Differences between Mouse, Rat, Dog, Monkey and Human CYP-Mediated Drug Metabolism, Inhibition and Induction. Expert Opin. Drug Metab. Toxicol. 2006, 2, 875–894. [Google Scholar] [CrossRef] [PubMed]
- Schmeisser, S.; Miccoli, A.; von Bergen, M.; Berggren, E.; Braeuning, A.; Busch, W.; Desaintes, C.; Gourmelon, A.; Grafström, R.; Harrill, J.; et al. New Approach Methodologies in Human Regulatory Toxicology–Not If, but How and When! Environ. Int. 2023, 178, 108082. [Google Scholar] [CrossRef] [PubMed]
- Bhattacharya, S.; Zhang, Q.; Carmichael, P.L.; Boekelheide, K.; Andersen, M.E. Toxicity Testing in the 21st Century: Defining New Risk Assessment Approaches Based on Perturbation of Intracellular Toxicity Pathways. PLoS ONE 2011, 6, e20887. [Google Scholar] [CrossRef] [PubMed]
- Giacomini, K.M.; Huang, S.-M.; Tweedie, D.J.; Benet, L.Z.; Brouwer, K.L.R.; Chu, X.; Dahlin, A.; Evers, R.; Fischer, V.; Hillgren, K.M.; et al. Membrane Transporters in Drug Development. Nat. Rev. Drug Discov. 2010, 9, 215–236. [Google Scholar] [CrossRef] [PubMed]
- Yin, J.; Wang, J. Renal Drug Transporters and Their Significance in Drug–Drug Interactions. Acta Pharm. Sin. B 2016, 6, 363–373. [Google Scholar] [CrossRef] [PubMed]
- Wright, S.H. Molecular and Cellular Physiology of Organic Cation Transporter 2. Am. J. Physiol. Physiol. 2019, 317, F1669–F1679. [Google Scholar] [CrossRef]
- Yonezawa, A.; Inui, K. Importance of the Multidrug and Toxin Extrusion MATE/SLC47A Family to Pharmacokinetics, Pharmacodynamics/Toxicodynamics and Pharmacogenomics. Br. J. Pharmacol. 2011, 164, 1817–1825. [Google Scholar] [CrossRef]
- El-Sheikh, A.A.K.; Masereeuw, R.; Russel, F.G.M. Mechanisms of Renal Anionic Drug Transport. Eur. J. Pharmacol. 2008, 585, 245–255. [Google Scholar] [CrossRef]
- Huls, M.; Brown, C.D.A.; Windass, A.S.; Sayer, R.; van den Heuvel, J.J.M.W.; Heemskerk, S.; Russel, F.G.M.; Masereeuw, R. The Breast Cancer Resistance Protein Transporter ABCG2 Is Expressed in the Human Kidney Proximal Tubule Apical Membrane. Kidney Int. 2008, 73, 220–225. [Google Scholar] [CrossRef]
- Wieser, M.; Stadler, G.; Jennings, P.; Streubel, B.; Pfaller, W.; Ambros, P.; Riedl, C.; Katinger, H.; Grillari, J.; Grillari-Voglauer, R. HTERT Alone Immortalizes Epithelial Cells of Renal Proximal Tubules without Changing Their Functional Characteristics. Am. J. Physiol. Ren.-Physiol. 2008, 295, F1365–F1375. [Google Scholar] [CrossRef]
- Capinha, L.; Jennings, P.; Commandeur, J.N.M. Exposure to Cis- and Trans-Regioisomers of S-(1,2-Dichlorovinyl)-L-Cysteine and S-(1,2-Dichlorovinyl)-Glutathione Result in Quantitatively and Qualitatively Different Cellular Effects in RPTEC/TERT1 Cells. Toxicol. Lett. 2023, 383, 75–88. [Google Scholar] [CrossRef] [PubMed]
- Morrison, M.; Klein, C.; Clemann, N.; Collier, D.A.; Hardy, J.; Heiβerer, B.; Cader, M.Z.; Graf, M.; Kaye, J. StemBANCC: Governing Access to Material and Data in a Large Stem Cell Research Consortium. Stem Cell Rev. Rep. 2015, 11, 681–687. [Google Scholar] [CrossRef]
- Meijer, T.; Naderlinger, E.; Jennings, P.; Wilmes, A. Differentiation and Subculturing of Renal Proximal Tubular-like Cells Derived from Human IPSC. Curr. Protoc. 2023, 3, e850. [Google Scholar] [CrossRef] [PubMed]
- Wilmer, M.J.; Saleem, M.A.; Masereeuw, R.; Ni, L.; van der Velden, T.J.; Russel, F.G.; Mathieson, P.W.; Monnens, L.A.; van den Heuvel, L.P.; Levtchenko, E.N. Novel Conditionally Immortalized Human Proximal Tubule Cell Line Expressing Functional Influx and Efflux Transporters. Cell Tissue Res. 2010, 339, 449–457. [Google Scholar] [CrossRef]
- Nieskens, T.T.G.; Peters, J.G.P.; Schreurs, M.J.; Smits, N.; Woestenenk, R.; Jansen, K.; van der Made, T.K.; Röring, M.; Hilgendorf, C.; Wilmer, M.J.; et al. A Human Renal Proximal Tubule Cell Line with Stable Organic Anion Transporter 1 and 3 Expression Predictive for Antiviral-Induced Toxicity. AAPS J. 2016, 18, 465–475. [Google Scholar] [CrossRef] [PubMed]
- van der Stel, W.; Carta, G.; Eakins, J.; Darici, S.; Delp, J.; Forsby, A.; Bennekou, S.H.; Gardner, I.; Leist, M.; Danen, E.H.J.; et al. Multiparametric Assessment of Mitochondrial Respiratory Inhibition in HepG2 and RPTEC/TERT1 Cells Using a Panel of Mitochondrial Targeting Agrochemicals. Arch. Toxicol. 2020, 94, 2707–2729. [Google Scholar] [CrossRef] [PubMed]
- Aschauer, L.; Carta, G.; Vogelsang, N.; Schlatter, E.; Jennings, P. Expression of Xenobiotic Transporters in the Human Renal Proximal Tubule Cell Line RPTEC/TERT1. Toxicol. Vitr. 2015, 30, 95–105. [Google Scholar] [CrossRef] [PubMed]
- Pelis, R.M.; Suhre, W.M.; Wright, S.H. Functional Influence of N-Glycosylation in OCT2-Mediated Tetraethylammonium Transport. Am. J. Physiol. Physiol. 2006, 290, F1118–F1126. [Google Scholar] [CrossRef] [PubMed]
- Gupta, A.; Dai, Y.; Vethanayagam, R.R.; Hebert, M.F.; Thummel, K.E.; Unadkat, J.D.; Ross, D.D.; Mao, Q. Cyclosporin A, Tacrolimus and Sirolimus Are Potent Inhibitors of the Human Breast Cancer Resistance Protein (ABCG2) and Reverse Resistance to Mitoxantrone and Topotecan. Cancer Chemother. Pharmacol. 2006, 58, 374–383. [Google Scholar] [CrossRef]
- Ryan, M.J.; Johnson, G.; Kirk, J.; Fuerstenberg, S.M.; Zager, R.A.; Torok-Storb, B. HK-2: An Immortalized Proximal Tubule Epithelial Cell Line from Normal Adult Human Kidney. Kidney Int. 1994, 45, 48–57. [Google Scholar] [CrossRef]
- Jennings, P.; Koppelstaetter, C.; Aydin, S.; Abberger, T.; Wolf, A.M.; Mayer, G.; Pfaller, W. Cyclosporine A Induces Senescence in Renal Tubular Epithelial Cells. Am. J. Physiol. Ren.-Physiol. 2007, 293, F831–F838. [Google Scholar] [CrossRef] [PubMed]
- Aschauer, L.; Gruber, L.N.; Pfaller, W.; Limonciel, A.; Athersuch, T.J.; Cavill, R.; Khan, A.; Gstraunthaler, G.; Grillari, J.; Grillari, R.; et al. Delineation of the Key Aspects in the Regulation of Epithelial Monolayer Formation. Mol. Cell. Biol. 2013, 33, 2535–2550. [Google Scholar] [CrossRef]
- Chandrasekaran, V.; Carta, G.; da Costa Pereira, D.; Gupta, R.; Murphy, C.; Feifel, E.; Kern, G.; Lechner, J.; Cavallo, A.L.; Gupta, S.; et al. Generation and Characterization of IPSC-Derived Renal Proximal Tubule-like Cells with Extended Stability. Sci. Rep. 2021, 11, 11575. [Google Scholar] [CrossRef] [PubMed]
- Wilmes, A.; Aschauer, L.; Limonciel, A.; Pfaller, W.; Jennings, P. Evidence for a Role of Claudin 2 as a Proximal Tubular Stress Responsive Paracellular Water Channel. Toxicol. Appl. Pharmacol. 2014, 279, 163–172. [Google Scholar] [CrossRef] [PubMed]
- Takahashi, K.; Yamanaka, S. Induction of Pluripotent Stem Cells from Mouse Embryonic and Adult Fibroblast Cultures by Defined Factors. Cell 2006, 126, 663–676. [Google Scholar] [CrossRef]
- Takahashi, K.; Tanabe, K.; Ohnuki, M.; Narita, M.; Ichisaka, T.; Tomoda, K.; Yamanaka, S. Induction of Pluripotent Stem Cells from Adult Human Fibroblasts by Defined Factors. Cell 2007, 131, 861–872. [Google Scholar] [CrossRef] [PubMed]
- Jennings, P.; Carta, G.; Singh, P.; da Costa Pereira, D.; Feher, A.; Dinnyes, A.; Exner, T.E.; Wilmes, A. Capturing Time-Dependent Activation of Genes and Stress-Response Pathways Using Transcriptomics in IPSC-Derived Renal Proximal Tubule Cells. Cell Biol. Toxicol. 2023, 39, 1773–1793. [Google Scholar] [CrossRef] [PubMed]
- Nunes, C.; Singh, P.; Mazidi, Z.; Murphy, C.; Bourguignon, A.; Wellens, S.; Chandrasekaran, V.; Ghosh, S.; Zana, M.; Pamies, D.; et al. An in Vitro Strategy Using Multiple Human Induced Pluripotent Stem Cell-Derived Models to Assess the Toxicity of Chemicals: A Case Study on Paraquat. Toxicol. Vitr. 2022, 81, 105333. [Google Scholar] [CrossRef] [PubMed]
- Singh, P.; Chandrasekaran, V.; Hardy, B.; Wilmes, A.; Jennings, P.; Exner, T.E. Temporal Transcriptomic Alterations of Cadmium Exposed Human IPSC-Derived Renal Proximal Tubule-like Cells. Toxicol. Vitr. 2021, 76, 105229. [Google Scholar] [CrossRef]
- Snijders, K.E.; Fehér, A.; Táncos, Z.; Bock, I.; Téglási, A.; van den Berk, L.; Niemeijer, M.; Bouwman, P.; Le Dévédec, S.E.; Moné, M.J.; et al. Fluorescent Tagging of Endogenous Heme Oxygenase-1 in Human Induced Pluripotent Stem Cells for High Content Imaging of Oxidative Stress in Various Differentiated Lineages. Arch. Toxicol. 2021, 95, 3285–3302. [Google Scholar] [CrossRef]
- Koepsell, H.; Lips, K.; Volk, C. Polyspecific Organic Cation Transporters: Structure, Function, Physiological Roles, and Biopharmaceutical Implications. Pharm. Res. 2007, 24, 1227–1251. [Google Scholar] [CrossRef] [PubMed]
- Caetano-Pinto, P.; Nordell, P.; Nieskens, T.; Haughan, K.; Fenner, K.S.; Stahl, S.H. Amplifying the Impact of Kidney Microphysiological Systems: Predicting Renal Clearance Using Mechanistic Modelling Based on Reconstructed Drug Secretion. ALTEX Altern. Anim. Exp. 2023, 40, 408–424. [Google Scholar] [CrossRef] [PubMed]
- Zhang, X.; Wang, R.; Piotrowski, M.; Zhang, H.; Leach, K.L. Intracellular Concentrations Determine the Cytotoxicity of Adefovir, Cidofovir and Tenofovir. Toxicol. Vitr. 2015, 29, 251–258. [Google Scholar] [CrossRef] [PubMed]
- Van der Hauwaert, C.; Savary, G.; Buob, D.; Leroy, X.; Aubert, S.; Flamand, V.; Hennino, M.-F.; Perrais, M.; Lo-Guidice, J.-M.; Broly, F.; et al. Expression Profiles of Genes Involved in Xenobiotic Metabolism and Disposition in Human Renal Tissues and Renal Cell Models. Toxicol. Appl. Pharmacol. 2014, 279, 409–418. [Google Scholar] [CrossRef] [PubMed]
- Jansen, J.; Schophuizen, C.M.S.; Wilmer, M.J.; Lahham, S.H.M.; Mutsaers, H.A.M.; Wetzels, J.F.M.; Bank, R.A.; van den Heuvel, L.P.; Hoenderop, J.G.; Masereeuw, R. A Morphological and Functional Comparison of Proximal Tubule Cell Lines Established from Human Urine and Kidney Tissue. Exp. Cell Res. 2014, 323, 87–99. [Google Scholar] [CrossRef] [PubMed]
- Lash, L.H.; Putt, D.A.; Cai, H. Membrane Transport Function in Primary Cultures of Human Proximal Tubular Cells. Toxicology 2006, 228, 200–218. [Google Scholar] [CrossRef] [PubMed]
- Brown, C.D.A.; Sayer, R.; Windass, A.S.; Haslam, I.S.; De Broe, M.E.; D’Haese, P.C.; Verhulst, A. Characterisation of Human Tubular Cell Monolayers as a Model of Proximal Tubular Xenobiotic Handling. Toxicol. Appl. Pharmacol. 2008, 233, 428–438. [Google Scholar] [CrossRef] [PubMed]
- Bajaj, P.; Chung, G.; Pye, K.; Yukawa, T.; Imanishi, A.; Takai, Y.; Brown, C.; Wagoner, M.P. Freshly Isolated Primary Human Proximal Tubule Cells as an in Vitro Model for the Detection of Renal Tubular Toxicity. Toxicology 2020, 442, 152535. [Google Scholar] [CrossRef] [PubMed]
- Chapron, A.; Chapron, B.D.; Hailey, D.W.; Chang, S.-Y.; Imaoka, T.; Thummel, K.E.; Kelly, E.; Himmelfarb, J.; Shen, D.; Yeung, C.K. An Improved Vascularized, Dual-Channel Microphysiological System Facilitates Modeling of Proximal Tubular Solute Secretion. ACS Pharmacol. Transl. Sci. 2020, 3, 496–508. [Google Scholar] [CrossRef]
- Martovetsky, G.; Tee, J.B.; Nigam, S.K. Hepatocyte Nuclear Factors 4 α and 1 α Regulate Kidney Developmental Expression of Drug-Metabolizing Enzymes and Drug Transporters. Mol. Pharmacol. 2013, 84, 808–823. [Google Scholar] [CrossRef]
- Kikuchi, R.; Kusuhara, H.; Hattori, N.; Shiota, K.; Kim, I.; Gonzalez, F.J.; Sugiyama, Y. Regulation of the Expression of Human Organic Anion Transporter 3 by Hepatocyte Nuclear Factor 1α/β and DNA Methylation. Mol. Pharmacol. 2006, 70, 887–896. [Google Scholar] [CrossRef] [PubMed]
- Weber, E.J.; Chapron, A.; Chapron, B.D.; Voellinger, J.L.; Lidberg, K.A.; Yeung, C.K.; Wang, Z.; Yamaura, Y.; Hailey, D.W.; Neumann, T.; et al. Development of a Microphysiological Model of Human Kidney Proximal Tubule Function. Kidney Int. 2016, 90, 627–637. [Google Scholar] [CrossRef] [PubMed]
- Sakolish, C.; Moyer, H.L.; Tsai, H.-H.D.; Ford, L.C.; Dickey, A.N.; Wright, F.A.; Han, G.; Bajaj, P.; Baltazar, M.T.; Carmichael, P.L.; et al. Analysis of Reproducibility and Robustness of a Renal Proximal Tubule Microphysiological System OrganoPlate 3-Lane 40 for in Vitro Studies of Drug Transport and Toxicity. Toxicol. Sci. 2023, 196, 52–70. [Google Scholar] [CrossRef] [PubMed]
- Freedman, B.S.; Brooks, C.R.; Lam, A.Q.; Fu, H.; Morizane, R.; Agrawal, V.; Saad, A.F.; Li, M.K.; Hughes, M.R.; Werff, R.V.; et al. Modelling Kidney Disease with CRISPR-Mutant Kidney Organoids Derived from Human Pluripotent Epiblast Spheroids. Nat. Commun. 2015, 6, 8715. [Google Scholar] [CrossRef] [PubMed]
- Morizane, R.; Lam, A.Q.; Freedman, B.S.; Kishi, S.; Valerius, M.T.; Bonventre, J. V Nephron Organoids Derived from Human Pluripotent Stem Cells Model Kidney Development and Injury. Nat. Biotechnol. 2015, 33, 1193–1200. [Google Scholar] [CrossRef] [PubMed]
- Czerniecki, S.M.; Cruz, N.M.; Harder, J.L.; Menon, R.; Annis, J.; Otto, E.A.; Gulieva, R.E.; Islas, L.V.; Kim, Y.K.; Tran, L.M.; et al. High-Throughput Screening Enhances Kidney Organoid Differentiation from Human Pluripotent Stem Cells and Enables Automated Multidimensional Phenotyping. Cell Stem Cell 2018, 22, 929–940. [Google Scholar] [CrossRef] [PubMed]
- Takasato, M.; Er, P.X.; Chiu, H.S.; Little, M.H. Generation of Kidney Organoids from Human Pluripotent Stem Cells. Nat. Protoc. 2016, 11, 1681–1692. [Google Scholar] [CrossRef] [PubMed]
- Morizane, R.; Bonventre, J. V Generation of Nephron Progenitor Cells and Kidney Organoids from Human Pluripotent Stem Cells. Nat. Protoc. 2017, 12, 195–207. [Google Scholar] [CrossRef]
- Lawrence, M.L.; Elhendawi, M.; Morlock, M.; Liu, W.; Liu, S.; Palakkan, A.; Seidl, L.F.; Hohenstein, P.; Sjögren, A.K.; Davies, J.A. Human IPSC-Derived Renal Organoids Engineered to Report Oxidative Stress Can Predict Drug-Induced Toxicity. iScience 2022, 25, 103884. [Google Scholar] [CrossRef]
- Shankar, A.S.; Du, Z.; Mora, H.T.; van den Bosch, T.P.P.; Korevaar, S.S.; Van den Berg-Garrelds, I.M.; Bindels, E.; Lopez-Iglesias, C.; Clahsen-van Groningen, M.C.; Gribnau, J.; et al. Human Kidney Organoids Produce Functional Renin. Kidney Int. 2021, 99, 134–147. [Google Scholar] [CrossRef]
- Takasato, M.; Er, P.X.; Chiu, H.S.; Maier, B.; Baillie, G.J.; Ferguson, C.; Parton, R.G.; Wolvetang, E.J.; Roost, M.S.; Chuva de Sousa Lopes, S.M.; et al. Kidney Organoids from Human IPS Cells Contain Multiple Lineages and Model Human Nephrogenesis. Nature 2015, 526, 564–568. [Google Scholar] [CrossRef] [PubMed]
- Wu, H.; Uchimura, K.; Donnelly, E.L.; Kirita, Y.; Morris, S.A.; Humphreys, B.D. Comparative Analysis and Refinement of Human PSC-Derived Kidney Organoid Differentiation with Single-Cell Transcriptomics. Cell Stem Cell 2018, 23, 869–881. [Google Scholar] [CrossRef] [PubMed]
- Susa, K.; Kobayashi, K.; Galichon, P.; Matsumoto, T.; Tamura, A.; Hiratsuka, K.; Gupta, N.R.; Yazdi, I.K.; Bonventre, J.V.; Morizane, R. ATP/ADP Biosensor Organoids for Drug Nephrotoxicity Assessment. Front. Cell Dev. Biol. 2023, 11, 1138504. [Google Scholar] [CrossRef] [PubMed]
- Aceves, J.O.; Heja, S.; Kobayashi, K.; Robinson, S.S.; Miyoshi, T.; Matsumoto, T.; Schäffers, O.J.M.; Morizane, R.; Lewis, J.A. 3D Proximal Tubule-on-Chip Model Derived from Kidney Organoids with Improved Drug Uptake. Sci. Rep. 2022, 12, 14997. [Google Scholar] [CrossRef]
- Freedman, B.S. Better Being Single? Omics Improves Kidney Organoids. Nephron 2019, 141, 128–132. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Meijer, T.; da Costa Pereira, D.; Klatt, O.C.; Buitenhuis, J.; Jennings, P.; Wilmes, A. Characterization of Organic Anion and Cation Transport in Three Human Renal Proximal Tubular Epithelial Models. Cells 2024, 13, 1008. https://doi.org/10.3390/cells13121008
Meijer T, da Costa Pereira D, Klatt OC, Buitenhuis J, Jennings P, Wilmes A. Characterization of Organic Anion and Cation Transport in Three Human Renal Proximal Tubular Epithelial Models. Cells. 2024; 13(12):1008. https://doi.org/10.3390/cells13121008
Chicago/Turabian StyleMeijer, Tamara, Daniel da Costa Pereira, Olivia C. Klatt, Joanne Buitenhuis, Paul Jennings, and Anja Wilmes. 2024. "Characterization of Organic Anion and Cation Transport in Three Human Renal Proximal Tubular Epithelial Models" Cells 13, no. 12: 1008. https://doi.org/10.3390/cells13121008
APA StyleMeijer, T., da Costa Pereira, D., Klatt, O. C., Buitenhuis, J., Jennings, P., & Wilmes, A. (2024). Characterization of Organic Anion and Cation Transport in Three Human Renal Proximal Tubular Epithelial Models. Cells, 13(12), 1008. https://doi.org/10.3390/cells13121008






