Effect of Oak Powdery Mildew on Ascorbate–Glutathione Cycle and Other Antioxidants in Plant—Erysiphe alphitoides Interaction
Abstract
:1. Introduction
2. Materials and Methods
2.1. Collecting Materials and Designing Experiments
2.2. Leaves Moisture Content Measurements
2.3. Metal Content Determination
2.4. Determination of Ascorbate–Glutathione Cycle Enzymes Activities
2.5. Protein Content Assay
2.6. Ascorbate Pool Determination
2.7. Glutathione Pool Determination
2.8. Determination of Different Phenolic Compounds Content
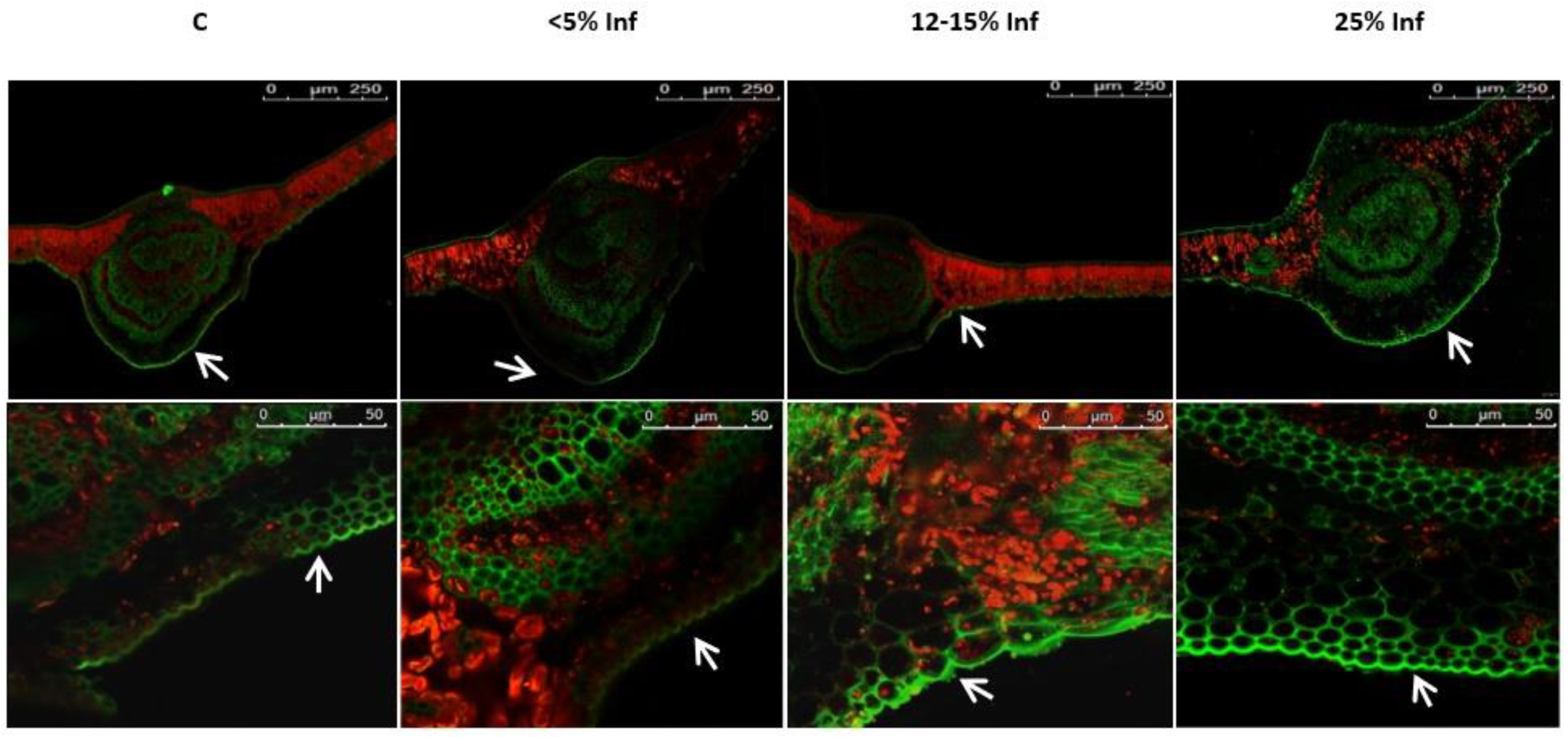
2.9. Determination of Lignin Content and Histochemical Detection
2.10. Statistical Analysis
3. Results
3.1. Effect of Oak Powdery Mildew on the Moisture Content of Oak Leaves
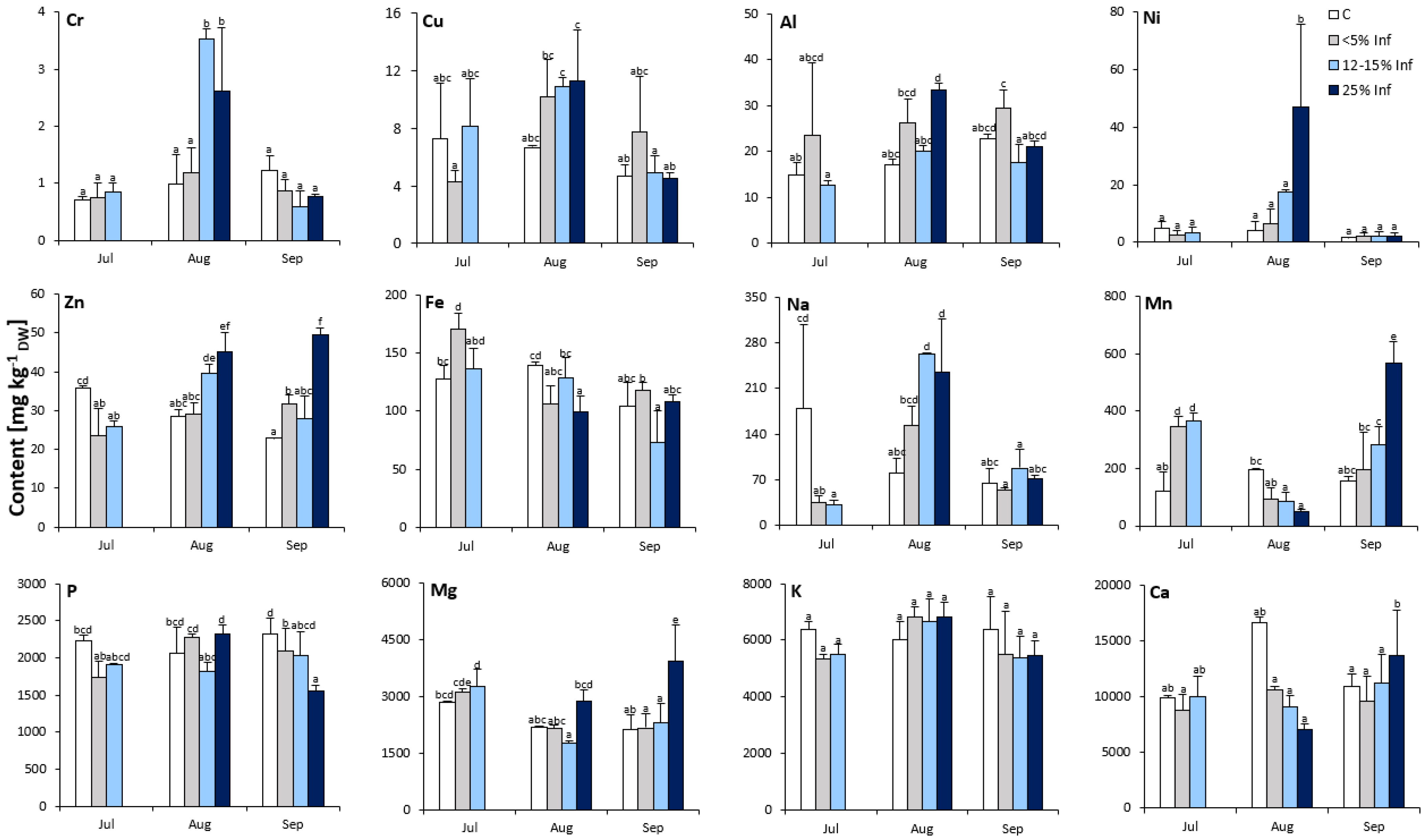
3.2. Changes in Metal Contents
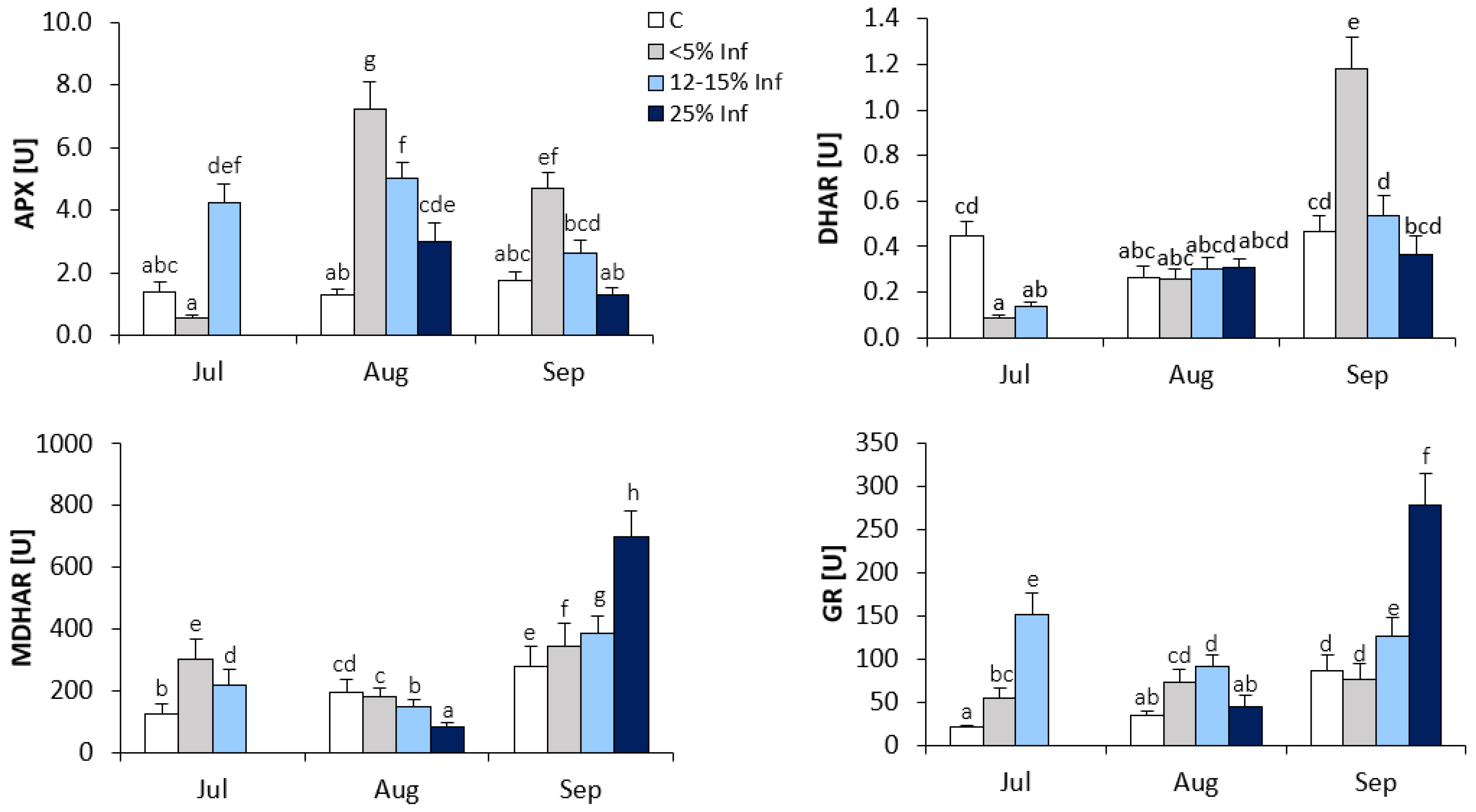
3.3. Effect of E. alphitoides Infection on the Activities of Ascorbate-Glutathione Cycle Enzymes
3.4. Infection-Related Changes in the Ascorbate and Glutathione Pools
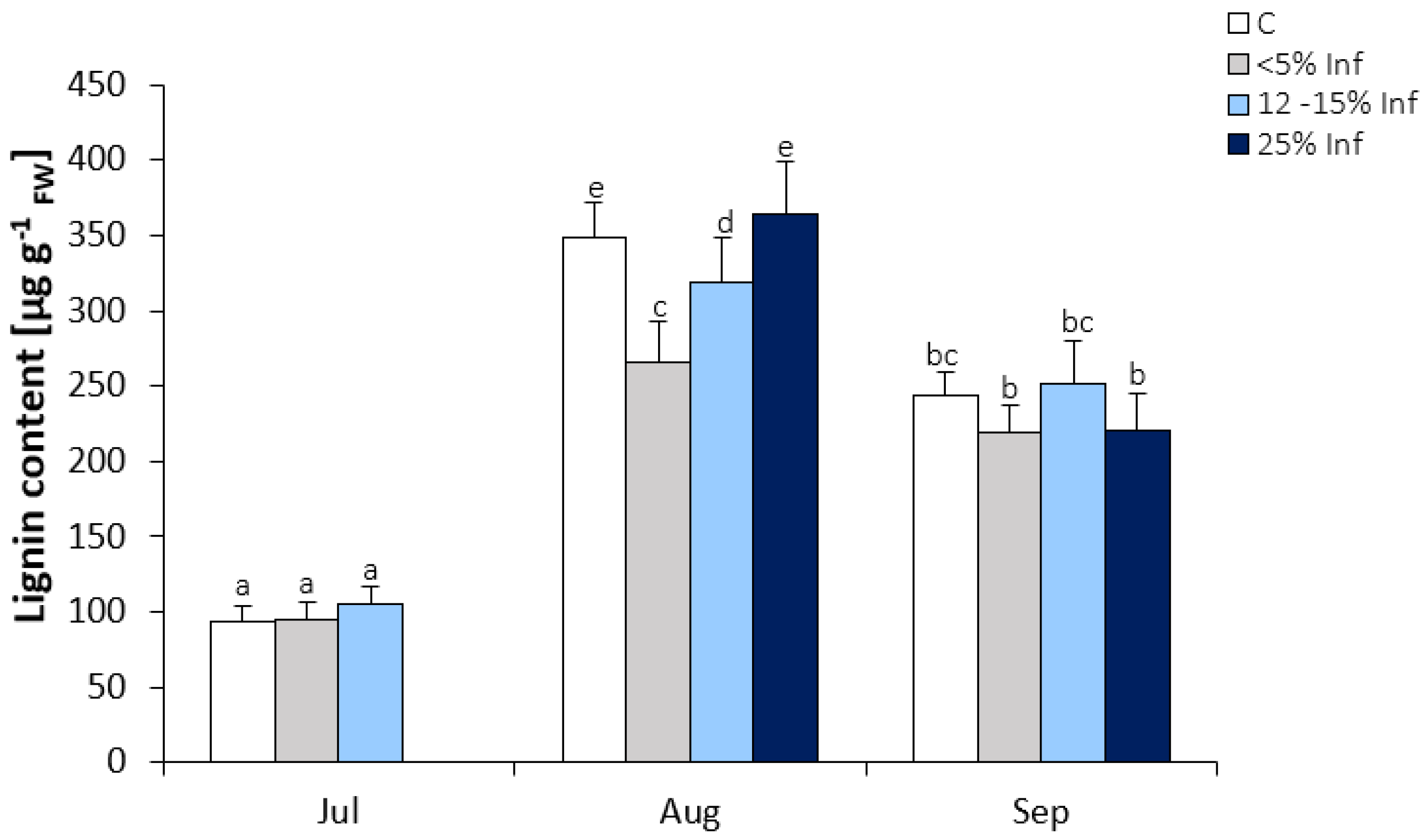
3.5. Changes in Lignin Content
3.6. Changes in Different Phenolic Contents
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Desprez-Loustau, M.L.; Feau, N.; Mougou-Hamdane, A.; Dutech, C. Interspecific and intraspecific diversity in oak powdery mildews in Europe: Coevolution history and adaptation to their hosts. Mycoscience 2011, 52, 165–173. [Google Scholar] [CrossRef]
- Foex, M.E. L’invasion Des Chenes d’Europe Par Le Blanc Ou Oidium. Rev. Eaux. For. 1941, 79, 338–349. [Google Scholar]
- G.C.H.; Lange, M. Nordic Journal of Botany. Mycologia 1982, 74, 354. [Google Scholar] [CrossRef]
- Kasprzyk, W.; Baranowska, M.; Korzeniewicz, R.; Behnke-Borowczyk, J.; Kowalkowski, W. Effect of Irrigation Dose on Powdery Mildew Incidence and Root Biomass of Sessile Oaks (Quercus petraea (Matt.) Liebl.). Plants 2022, 11, 1248. [Google Scholar] [CrossRef]
- Gross, A.; Petitcollin, C.; Dutech, C.; Ly, B.; Massot, M.; Faivre d’Arcier, J.; Dubois, L.; Saint-Jean, G.; Desprez-Loustau, M.L. Hidden invasion and niche contraction revealed by herbaria specimens in the fungal complex causing oak powdery mildew in Europe. Biol. Invasions 2021, 23, 885–901. [Google Scholar] [CrossRef]
- Sucharzewska, E. The development of Erysiphe alphitoides and E. hypophylla in the urban environment. Acta Mycol. 2009, 44, 109–123. [Google Scholar] [CrossRef]
- Roszak, R.; Baranowska, M.; Belka, M.; Behnke-Borowczyk, J. Zastosowanie Technik Biologii Molekularnej Do Detekcji Erysiphe alphitoides (Griffon & Maubl.) U. Braun and S. Takam. w Organach Roślinnych. Sylwan 2019, 163, 1–6. [Google Scholar]
- Marçais, B.; Desprez-Loustau, M.L. European oak powdery mildew: Impact on trees, effects of environmental factors, and potential effects of climate change. Ann. For. Sci. 2014, 71, 633–642. [Google Scholar] [CrossRef]
- Marković, Č.; Kanjevac, B.; Perišić, U.; Dobrosavljević, J. The effect of the oak powdery mildew, oak lace bug, and other foliofagous insects on the growth of young pedunculate oak trees. Front. For. Glob. Chang. 2024, 6, 1297560. [Google Scholar] [CrossRef]
- Marçais, B.; Kavkova, M.; Desprez-Loustau, M.L. Phenotypic variation in the phenology of ascospore production between European populations of oak powdery mildew. Ann. For. Sci. 2009, 66, 814. [Google Scholar] [CrossRef]
- Dillen, M.; Smit, C.; Buyse, M.; Höfte, M.; De Clercq, P.; Verheyen, K. Stronger diversity effects with increased environmental stress: A study of multitrophic interactions between oak, powdery mildew and ladybirds. PLoS ONE 2017, 12, e0176104. [Google Scholar] [CrossRef] [PubMed]
- Iqbal, Z.; Iqbal, M.S.; Hashem, A.; Abd_Allah, E.F.; Ansari, M.I. Plant Defense Responses to Biotic Stress and Its Interplay With Fluctuating Dark/Light Conditions. Front. Plant Sci. 2021, 12, 631810. [Google Scholar] [CrossRef] [PubMed]
- Bhuiyan, N.H.; Selvaraj, G.; Wei, Y.; King, J. Gene expression profiling and silencing reveal that monolignol biosynthesis plays a critical role in penetration defence in wheat against powdery mildew invasion, J. Exp. Bot. 2009, 60, 509–521. [Google Scholar] [CrossRef]
- Zeng, Y.; Song, H.; Xia, L.; Yang, L.; Zhang, S. The responses of poplars to fungal pathogens: A review of the defensive pathway. Front. Plant Sci. 2023, 14, 1107583. [Google Scholar] [CrossRef]
- Feechan, A.; Kwon, E.; Yun, B.W.; Wang, Y.; Pallas, J.A.; Loake, G.J. A central role for S-nitrosothiols in plant disease resistance, Proc. Natl. Acad. Sci. USA 2005, 102, 8054–8059. [Google Scholar] [CrossRef] [PubMed]
- Nawrocka, J.; Szymczak, K.; Maćkowiak, A.; Skwarek-Fadecka, M.; Małolepsza, U. Determination of Reactive Oxygen or Nitrogen Species and Novel Volatile Organic Compounds in the Defense Responses of Tomato Plants against Botrytis cinerea Induced by Trichoderma virens TRS 106. Cells 2022, 11, 3051. [Google Scholar] [CrossRef] [PubMed]
- Nogues, I.; Grenni, P.; Di Lenola, M.; Passatore, L.; Guerriero, E.; Benedetti, P.; Massacci, A.; Rauseo, J.; Barra Caracciolo, A. Microcosm Experiment to Assess the Capacity of a Poplar Clone to Grow in a PCB-Contaminated Soil. Water 2019, 11, 2220. [Google Scholar] [CrossRef]
- Kaur, S.; Samota, M.K.; Choudhary, M.; Choudhary, M.; Pandey, A.K.; Sharma, A.; Thakur, J. How do plants defend themselves against pathogens-Biochemical mechanisms and genetic interventions. Physiol. Mol. Biol. Plants 2022, 28, 485–504. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Turczański, K.; Bełka, M.; Spychalski, M.; Kukawka, R.; Prasad, R.; Smiglak, M. Resistance Inducers for the Protection of Pedunculate Oak (Quercus robur L.) Seedlings against Powdery Mildew Erysiphe alphitoides. Plants 2023, 12, 635. [Google Scholar] [CrossRef]
- Morkunas, I.; Woźniak, A.; Mai, V.C.; Rucińska-Sobkowiak, R.; Jeandet, P. The Role of Heavy Metals in Plant Response to Biotic Stress. Molecules 2018, 23, 2320. [Google Scholar] [CrossRef]
- Liu, Y.; Zheng, X.; Zhou, P.; Song, Y.; Li, L. Reactive Oxygen Species and Salicylic Acid Mediate the Responses of Pear to Venturia nashicola Infection. Agronomy 2024, 14, 877. [Google Scholar] [CrossRef]
- Kebert, M.; Kostić, S.; Zlatković, M.; Stojnic, S.; Čapelja, E.; Zorić, M.; Kiprovski, B.; Budakov, D.; Orlović, S. Ectomycorrhizal Fungi Modulate Biochemical Response against Powdery Mildew Disease in Quercus robur L. Forests 2022, 13, 1491. [Google Scholar] [CrossRef]
- Clark, K.R.; Goldberg Oppenheimer, P. Vibrational spectroscopic profiling of biomolecular interactions between oak powdery mildew and oak leaves. Soft Matter. 2024, 20, 959. [Google Scholar] [CrossRef] [PubMed]
- Skwarek-Fadecka, M.; Patykowski, J.; Nawrocka, J.; Szymczak, K.; Kaźmierczak, A.; Wielanek, M.; Ruszkiewicz-Michalska, M. Biochemical changes in Quercus robur L. leaves after Erysiphe alphitoides infection. For. Pathol. 2022, 52, e12756. [Google Scholar] [CrossRef]
- Petřivalský, M.; Vaníčková, P.; Ryzí, M.; Navrátilová, B.; Piterková, J.; Sedlářová, M.; Luhová, L. The effects of reactive nitro gen species and oxygen species in the regeneration and growth of cucumber cells from isolated protoplasts. PCTOC 2012, 108, 237–249. [Google Scholar] [CrossRef]
- Rajput, V.D.; Harish; Singh, R.K.; Verma, K.K.; Sharma, L.; Quiroz-Figueroa, F.R.; Meena, M.; Gour, V.S.; Minkina, T.; Sushkova, S.; et al. Recent Developments in Enzymatic Antioxidant Defence Mechanism in Plants with Special Reference to Abiotic Stress. Biology 2021, 10, 267. [Google Scholar] [CrossRef] [PubMed]
- Mittler, R.; Vanderauwera, S.; Gollery, M.; Van Breusegem, F. Reactive oxygen gene network of plants. Trends Plant Sci. 2004, 9, 490–498. [Google Scholar] [CrossRef]
- Guo, W.; Xing, Y.; Luo, X.; Li, F.; Ren, M.; Liang, Y. Reactive Oxygen Species: A Crosslink between Plant and Human Eukaryotic Cell Systems. Int. J. Mol. Sci. 2023, 24, 13052. [Google Scholar] [CrossRef]
- Braun, U.; Cook, R.T.A. Taxonomic manual of the Erysiphales (powdery mildews). In CBS Biodiversity Series; CBS-KNAW Fungal Biodiversity Centre: Utrecht, The Netherlands, 2012; Volume 11, pp. 1–707. ISBN 978-90-70351-89-2. [Google Scholar]
- Takamatsu, S.; Braun, U.; Limkaisang, S.; Kom-Un, S.; Sato, Y.; Cunnington, J.H. Phylogeny and taxonomy of the oak powdery mildew Erysiphe alphitoides sensu lato. Mycol. Res. 2007, 111, 809–826. [Google Scholar] [CrossRef]
- Copolovici, L.; Väärtnõu, F.; Estrada, M.P.; Niinemets, Ü. Oak powdery mildew (Erysiphe alphitoides)-induced volatile emissions scale with the degree of infection in Quercus robur. Tree Physiol. 2014, 34, 1399–1410. [Google Scholar] [CrossRef]
- Ellis, R.H.; Hong, T.D.; Roberts, E.H. Handbook of Seeds Technology for Gene Banks; Principles and Methodology International Board for Plant Genetic Resources: Rome, Italy, 1985; Volume 1, pp. 121–137. [Google Scholar]
- Skwarek, M.; Wala, M.; Kołodziejek, J.; Sieczyńska, K.; Lasoń-Rydel, M.; Ławińska, K.; Obraniak, A. Seed Coating with Biowaste Materials and Biocides—Environment-Friendly Biostimulation or Threat? Agronomy 2021, 11, 1034. [Google Scholar] [CrossRef]
- Nakano, Y.; Asada, K. Hydrogen peroxide is scavenged by ascorbate-specific peroxidase in spinach chloroplasts. Plant Cell Physiol. 1981, 22, 867–880. [Google Scholar] [CrossRef]
- Dalton, D.; Russell, S.; Hanus, F.; Pascoe, G.; Evans, H. Enzymatic reactions of ascorbate and glutathione that prevent peroxide damage in soy bean root nodules. Proc. Natl. Acad. Sci. USA 1986, 83, 3811–3815. [Google Scholar] [CrossRef]
- Hossain, M.A.; Nakano, Y.; Asada, K. Monodehydroascorbate reductase in spinach chloroplast and its participation in regeneration of ascorbate for scavenging hydrogen peroxide. Plant Cell Physiol. 1984, 25, 388–395. [Google Scholar] [CrossRef]
- Schaedle, M.; Bassham, J.A. Chloroplast Glutathione Reductase. Plant Physiol. 1977, 59, 1011–1012. [Google Scholar] [CrossRef]
- Bradford, M.M. A rapid and sensitive method for the quantitation of microgramq uantities of protein utilizing the principle of protein-dye binding. Anal. Biochem. 1976, 72, 248–254. [Google Scholar] [CrossRef]
- Okamura, M. An improved method for determination of L-ascorbic acid and L-dehydroascorbic acid in blood plasma, Clin. Chim. Acta. 1980, 103, 259–268. [Google Scholar] [CrossRef]
- Knȍrzer, O.C.; Durner, J.; Bȍger, P. Alterations of the antioxidative system of suspension-cultured soybean cells (Glycine max) induced by oxidative stress. Physiol. Plant. 1996, 97, 388–396. [Google Scholar] [CrossRef]
- Brehe, J.E.; Burch, H.B. Enzymatic assay for glutathione. Anal. Biochem. 1976, 74, 189–197. [Google Scholar] [CrossRef]
- Singleton, V.L.; Rossi, J.A. Colorimetry of Total Phenolics with Phosphomolybdic-Phosphotungstic Acid Reagents. Am. J. Enol. Vitic. 1965, 16, 144–158. [Google Scholar] [CrossRef]
- Glories, Y. Recherches sur la Matière Colorante des Vins Rouge. Doctoral Thesis, Université de Bordeaux, Bordeaux, France, 1978. [Google Scholar]
- Chang, C.C.; Yang, M.H.; Wen, H.M.; Chern, J.C. Estimation of total flavonoid content in propolis by two complementary colorimetric methods. J. Food Drug Anal. 2002, 10, 178–182. [Google Scholar]
- Hodges, D.M.; Nozzoillo, C. Anthocyanin and anthocyanoplast content of Cruciferous seedlings subjected to mineral nutrient deficiencies. J. Plant Physiol. 1996, 147, 749–754. [Google Scholar] [CrossRef]
- Bruce, R.J.; West, C.A. Elicitation of lignin biosynthesis and isoperoxidase activity by pectic fragments in suspension cultures of castor Bean. Plant Physiol. 1989, 91, 889–897. [Google Scholar] [CrossRef] [PubMed]
- Mandal, S.; Chakraborty, D.; Dey, S. Phenolic acids act as signaling molecules in plant-microbe plant-microbe symbioses. Plant Sign Behav. 2010, 5, 359–368. [Google Scholar] [CrossRef] [PubMed]
- Drnovšek, T.; Perdih, A.; Perdih, M. Fiber surface characteristics evaluated by principal component analysis. J. Wood Sci. 2005, 51, 507–513. [Google Scholar] [CrossRef]
- Arab, L.; Seegmueller, S.; Kreuzwieser, J.; Eiblmeier, M.; Dannenmann, M.; Rennenberg, H. Significance of current weather conditions for foliar traits of old-growth sessile oak (Quercus petraea Liebl) trees. Trees 2022, 36, 777–791. [Google Scholar] [CrossRef]
- Oszako, T.; Voitka, D.; Stocki, M.; Stocka, N.; Nowakowska, J.A.; Linkiewicz, A.; Malewski, T. Trichoderma asperellum efficiently protects Quercus robur leaves against Erysiphe alphitoides. Eur. J. Plant Pathol. 2021, 159, 295–308. [Google Scholar] [CrossRef]
- Prosyannikova, I.B. Influence of oak powdery dew on water exchange undergrowth Quercus petraea L. ex Liebl. (Fagaceae) [Влияние мучнистoй рoсы дуба на вoдooбмен пoдрoста Quercus petraea L. ex Liebl. (Fagaceae)]. Ukr. Bot. J. 2002, 59, 628–631. [Google Scholar]
- Poschenrieder, C.; Tolrà, R.; Barceló, J. Can metals defend plants against biotic stress? Trends Plant Sci. 2006, 11, 288–295. [Google Scholar] [CrossRef]
- Freeman, J.L.; Garcia, D.; Kim, D.; Hopf, A.; Salt, D.E. Constitutively elevated salicylic acid signals glutathione-mediated nickel tolerance in Thlaspi nickel hyperaccumulators. Plant Physiol. 2005, 137, 1082–1091. [Google Scholar] [CrossRef]
- Cabot, C.; Martos, S.; Llugany, M.; Gallego, B.; Tolrà, R.; Poschenrieder, C. A Role for Zinc in Plant Defense Against Pathogens and Herbivores. Front. Plant Sci. 2019, 10, 1171. [Google Scholar] [CrossRef] [PubMed]
- Fernando, D.R.; Baker, A.J.M.; Woodrow, I.E. Physiological responses in Macadamia integrifolia on exposure to Mn treatment. Austral. J. Bot. 2009, 57, 406–413. [Google Scholar] [CrossRef]
- Eskandari, S.; Khoshgoftarmanesh, A.H.; Sharifnabi, B. The Effect of Foliar-Applied Manganese in Mineral and Complex Forms with Amino Acids on Certain Defense Mechanisms of Cucumber (Cucumis sativus L.) Against Powdery Mildew. J. Plant Growth Regul. 2018, 37, 481–490. [Google Scholar] [CrossRef]
- Eskandari, S.; Hofte, H.; Zhang, T. Foliar manganese spray induces the resistance of cucumber to Colletotrichum lagenarium. J. Plant Physiol. 2020, 246–247, 153129. [Google Scholar] [CrossRef] [PubMed]
- Fabiano, C.C.; Tezotto, T.; Favarin, J.L.; Polacco, J.C.; Mazzafera, P. Essentiality of nickel in plants: A role in plant stresses. Front. Plant Sci. 2015, 6754, 15. [Google Scholar] [CrossRef] [PubMed]
- Greenshields, D.L.; Liu, G.; Wei, Y. Roles of iron in plant defence and fungal virulence. Plant Sig. Beh. 2007, 2, 300–302. [Google Scholar] [CrossRef]
- Huber, D.M.; Jones, J.B. The role of magnesium in plant disease. Plant Soil 2013, 368, 73–85. [Google Scholar] [CrossRef]
- Zhang, L.; Du, L.; Poovaiah, B.W. Calcium signaling and biotic defense responses in plants. Plant Signal Behav. 2014, 9, 11. [Google Scholar] [CrossRef] [PubMed]
- Gupta, S.K.; Rai, A.K.; Kanwar, S.S.; Sharma, T.R. Comparative analysis of zinc finger proteins involved in plant disease resistance. PLoS ONE 2012, 7, e42578. [Google Scholar] [CrossRef]
- Bastakoti, S. Role of zinc in management of plant diseases: A review. Cogent Food Agric. 2023, 9, 2194483. [Google Scholar] [CrossRef]
- Kopczewski, T.; Kuźniak, E.; Kornaś, A.; Rut, G.; Nosek, M.; Ciereszko, I.; Szczepaniak, L. Local and Systemic Changes in Photosynthetic Parameters and Antioxidant Activity in Cucumber Challenged with Pseudomonas syringae pv lachrymans. Int. J. Mol. Sci. 2020, 21, 6378. [Google Scholar] [CrossRef]
- Ghanbary, E.; Kouchaksaraei, M.T.; Guidi, L.; Mirabolfathy, M.; Etemad, V.; Sanavi, S.A.M.M.; Struve, D. Change in biochemical parameters of Persian oak (Quercus brantii Lindl.)’s seedlings inoculated by pathogens of charcoal disease under water deficit conditions. Trees 2018, 32, 1595–1608. [Google Scholar] [CrossRef]
- Anjum, N.A.; Gill, S.S.; Gill, R.; Hasanuzzaman, M.; Duarte, A.C.; Pereira, E.; Ahmad, I.; Tuteja, R.; Tuteja, N. Metal/metalloid stress tolerance in plants: Role of ascorbate, its redox couple, and associated enzymes. Protoplasma 2014, 251, 1265–1283. [Google Scholar] [CrossRef]
- Kesawat, M.S.; Satheesh, N.; Kherawat, B.S.; Kumar, A.; Kim, H.-U.; Chung, S.-M.; Kumar, M. Regulation of Reactive Oxygen Species during Salt Stress in Plants and Their Crosstalk with Other Signaling Molecules—Current Perspectives and Future Directions. Plants 2023, 12, 864. [Google Scholar] [CrossRef]
- Noctor, G.; Foyer, C.H. Intracellular redox compartmentation and ROS-related communication in regulation and signaling. Plant Physiol. 2016, 171, 1581–1592. [Google Scholar] [CrossRef] [PubMed]
- Treutter, D. Significance of flavonoids in plant resistance: A review. Environ. Chem. Lett. 2006, 4, 147–157. [Google Scholar] [CrossRef]
- Ruuhola, T.; Yang, S.; Ossipov, V.; Haukioja, E. Foliar oxidases as mediators of the rapidly induced resistance of mountain birch against Epirrita autumnata. Oecologia 2008, 154, 725–730. [Google Scholar] [CrossRef]
- Kumar, K.; Debnath, P.; Singh, S.; Kumar, N. An Overview of Plant Phenolics and Their Involvement in Abiotic Stress Tolerance. Stresses 2023, 3, 570–585. [Google Scholar] [CrossRef]
- Perez, F.J.; Villegas, D.; Mejia, N. Ascorbic acid and flavonoid-peroxidase reaction as a detoxifying system of H2O2 in grapevine leaves. Phytochemistry 2002, 60, 573–580. [Google Scholar] [CrossRef]
- Zandi, P.; Schnug, E. Reactive Oxygen Species, Antioxidant Responses and Implications from a Microbial Modulation Perspective. Biology 2022, 11, 155. [Google Scholar] [CrossRef]
- Sakihama, Y.; Cohen, M.F.; Grace, S.C.; Yamasaki, H. Plant phenolic antioxidant and prooxidant activities: Phenolics-induced oxidative damage mediated by metals in plants. Toxicology 2002, 177, 67–80. [Google Scholar] [CrossRef] [PubMed]
- Miedes, E.; Vanholme, R.; Boerjan, W.; Molina, A. The role of the secondary cell wall in plant resistance to pathogens. Front. Plant Sci. 2014, 5, 100584. [Google Scholar] [CrossRef] [PubMed]
- Pratyusha, S. Phenolic Compounds in the Plant Development and Defense: An Overview. IntechOpen 2022. [CrossRef]
- Smith, A.H.; Gill, W.M.; Pinkard, E.A.; Mohammed, C.L. Anatomical and histochemical defence responses induced in juvenile leaves of Eucalyptus globulus and Eucalyptus nitens by Mycosphaerella infection. For. Pathol. 2007, 37, 361–373. [Google Scholar] [CrossRef]
- Bonello, P.; Blodgett, J.T. Pinus nigra–Sphaeropsis sapinea as a model pathosystem to investigate local and systemic effects of fungal infection of pines. Physiol. Mol. Plant P. 2003, 63, 249–261. [Google Scholar] [CrossRef]
- Bucciarelli, B.; Ostry, M.E.; Fulcher, R.G.; Andersson, N.A.; Vance, C.P. Histochemical and microspectrophotometric analyses of early wound responses of resistant and susceptible Populus tremuloides inoculated with Entoleuca mammata (Hypoxylon mammatum). Can. J. Bot. 1999, 77, 548–555. [Google Scholar]

| Attribute | Value | Analytical Method | Analytical Procedure |
|---|---|---|---|
| pH in H2O | 5.6 | Potentiometric | PB 01 ed. 1 (14 May 2004) |
| Acidity in NaCl (g dm3 soil) | 0.8 | Conductometric | PB 02 ed. 2 (10 May 2016) |
| Available nutrients [mg dm3 soil] | |||
| N-NO3 | 41.0 | Potentiometric | PB 02 ed. 1 (14 May 2004) |
| N-NH4 | 21.0 | Potentiometric | PB 02 ed. 1 (14 May 2004) |
| P (P2O5) | 43.6 | Spectrophotometric | PB 02 ed. 1 (14 May 2004) |
| K (K2O) | 146 | Atomic emission spectroscopy | PB 04 ed. 1 (21 May 2004) |
| Mg | 492 | Atomic absorption spectroscopy | PB 05 ed. 1 (28 May 2004) |
| Mn | 3.5 | Atomic absorption spectroscopy | PB 05 ed. 1 (28 May 2004) |
| Cu | 2.6 | Atomic absorption spectroscopy | PB 09 ed. 1 (5 May 2004) |
| Zn | 4.6 | Atomic absorption spectroscopy | PB 09 ed. 1 (5 May 2004) |
| Fe | 41.4 | Atomic absorption spectroscopy | PB 09 ed. 1 (5 May 2004) |
| Ca | 679 | Atomic emission spectroscopy | PB 04 ed. 1 (21 May 2004) |
| Ascorbate Pool | Time | Plant Variants | |||
|---|---|---|---|---|---|
| [μmol g−1FW] | Month | C | <5% Inf | 12–15% Inf | 25% Inf |
| AsA + DHA | July | 26.1 ± 1.2 a | 27.4 ± 1.0 a | 31.6 ± 1.0 bc | ˗ |
| August | 44.7 ± 3.1 e | 34.3 ± 2.1 c | 47.7 ± 1.8 f | 59.3 ± 2.3 g | |
| September | 31.3 ± 2.0 b | 39.3 ± 1.5 d | 28.3 ± 1.0 a | 33.5 ± 2.9 bc | |
| AsA | July | 20.6 ± 3.6 ab | 20.7 ± 1.6 ab | 25.72 ± 3.2 c | ˗ |
| August | 32.5 ± 1.0 e | 25.4 ± 2.5 c | 28.97 ± 2.2 d | 32.8 ± 2.1 e | |
| September | 21.1 ± 2.5 ab | 22.9 ± 3.1 bc | 19.58 ± 1.22 a | 20.2 ± 1.7 ab | |
| DHA | July | 5.5 ± 0.7 a | 6.7 ± 1.2 ab | 5.9 ± 1.9 ab | ˗ |
| August | 12.2 ± 1.0 de | 8.8 ± 2.1 bc | 18.7 ± 1.2 f | 26.5 ± 1.6 g | |
| September | 10.2 ± 1.5 cd | 16.4 ± 1.2 f | 8.7 ± 0.4 bc | 13.4 ± 0.8 e | |
| Glutathione Pool | Time | Plant Variants | |||
|---|---|---|---|---|---|
| [μmol g−1FW] | Month | C | <5% Inf | 12–15% Inf | 25% Inf |
| GSH + GSSG | July | 30.5 ± 0.5 c | 40.7 ± 2.8 d | 51.1 ± 0.6 e | ˗ |
| August | 33.1 ± 0.7 c | 30.0 ± 0.8 bc | 61.4 ± 0.6 f | 21.2 ± 0.6 a | |
| September | 26.8 ± 2.8 abc | 23.7 ± 1.2 ab | 70.1 ± 5.9 g | 52.0 ± 1.3 e | |
| GSH | July | 28.8 ± 0.4 cd | 33.3 ± 2.8 def | 47.8 ± 0.6 g | ˗ |
| August | 30.8 ± 0.7 cde | 26.2 ± 0.7 bc | 36.1 ± 0.5 ef | 20.5 ± 0.5 ab | |
| September | 17.9 ± 2.9 a | 19.9 ± 1.3 ab | 54.5 ± 5.9 h | 37.4 ± 1.4 f | |
| GSSG | July | 1.6 ± 0.1 b | 7.4 ± 0.1 f | 3.3 ± 0.1 d | ˗ |
| August | 2.3 ± 0.1 c | 3.9 ± 0.1 e | 25.3 ± 0.1 j | 0.7 ± 0.1 a | |
| September | 8.9 ± 0.1 g | 3.8 ± 0.1 e | 15.7 ± 0.1 i | 14.5 ± 0.1 h | |
| Phenolic Compounds | Time | Plant Variants | |||
|---|---|---|---|---|---|
| Month | C | <5% Inf | 12–15% Inf | 25% Inf | |
| Total phenols | July | 18.5 ± 1.0 a | 19.7 ± 1.2 ab | 22.8 ± 1.2 bc | ˗ |
| [mg g−1FW] | August | 30.4 ± 1.0 e | 24.6 ± 1.1 cd | 18.5 ± 1.1 a | 24.6 ± 1.1 cd |
| September | 24.9 ± 1.3 d | 24.6 ± 1.4 cd | 17.6 ± 1.1 a | 22.7 ± 1.2 bc | |
| Phenylpropanoids | July | 16.4 ± 1.0 abc | 14.7 ± 1.2 a | 20.9 ± 1.4 e | ˗ |
| [mg g−1FW] | August | 21.5 ± 1.1 de | 20.5 ± 0.7 de | 16.5 ± 1.4 abc | 17.5 ± 1.7 abc |
| September | 19.0 ± 0.7 cde | 17.9 ± 1.4 bcd | 15.0 ± 0.8 ab | 16.1 ± 0.8 ab | |
| Flavonoids | July | 160 ± 4 a | 140 ± 7 a | 330 ± 18 b | ˗ |
| [μg g−1FW] | August | 330 ± 74 b | 180 ± 10 a | 190 ± 10 a | 200 ± 11 a |
| September | 180 ± 12 a | 170 ± 10 a | 110 ± 13 a | 130 ± 10 a | |
| Anthocyanin | July | 5.9 ± 1.3 ab | 7.4 ± 1.0 bc | 4.5 ± 1.0 a | ˗ |
| [μg g−1FW] | August | 5.6 ± 1.1 ab | 5.2 ± 0.9 ab | 7.9 ± 1.3 bc | 7.9 ± 1.3 bc |
| September | 5.5 ± 1.1 ab | 8.9 ± 1.3 c | 5.8 ± 1.3 ab | 7.1 ± 0.8 abc | |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Skwarek-Fadecka, M.; Nawrocka, J.; Sieczyńska, K.; Patykowski, J.; Posmyk, M.M. Effect of Oak Powdery Mildew on Ascorbate–Glutathione Cycle and Other Antioxidants in Plant—Erysiphe alphitoides Interaction. Cells 2024, 13, 1035. https://doi.org/10.3390/cells13121035
Skwarek-Fadecka M, Nawrocka J, Sieczyńska K, Patykowski J, Posmyk MM. Effect of Oak Powdery Mildew on Ascorbate–Glutathione Cycle and Other Antioxidants in Plant—Erysiphe alphitoides Interaction. Cells. 2024; 13(12):1035. https://doi.org/10.3390/cells13121035
Chicago/Turabian StyleSkwarek-Fadecka, Monika, Justyna Nawrocka, Katarzyna Sieczyńska, Jacek Patykowski, and Małgorzata Maria Posmyk. 2024. "Effect of Oak Powdery Mildew on Ascorbate–Glutathione Cycle and Other Antioxidants in Plant—Erysiphe alphitoides Interaction" Cells 13, no. 12: 1035. https://doi.org/10.3390/cells13121035







