Novel Small Molecules with Anti-Inflammatory and Anti-Angiogenic Activity in a Mouse Model of Oxygen-Induced Retinopathy
Abstract
:1. Introduction
2. Materials and Methods
2.1. Chemicals, Reagents, Media, and ELISA Kits
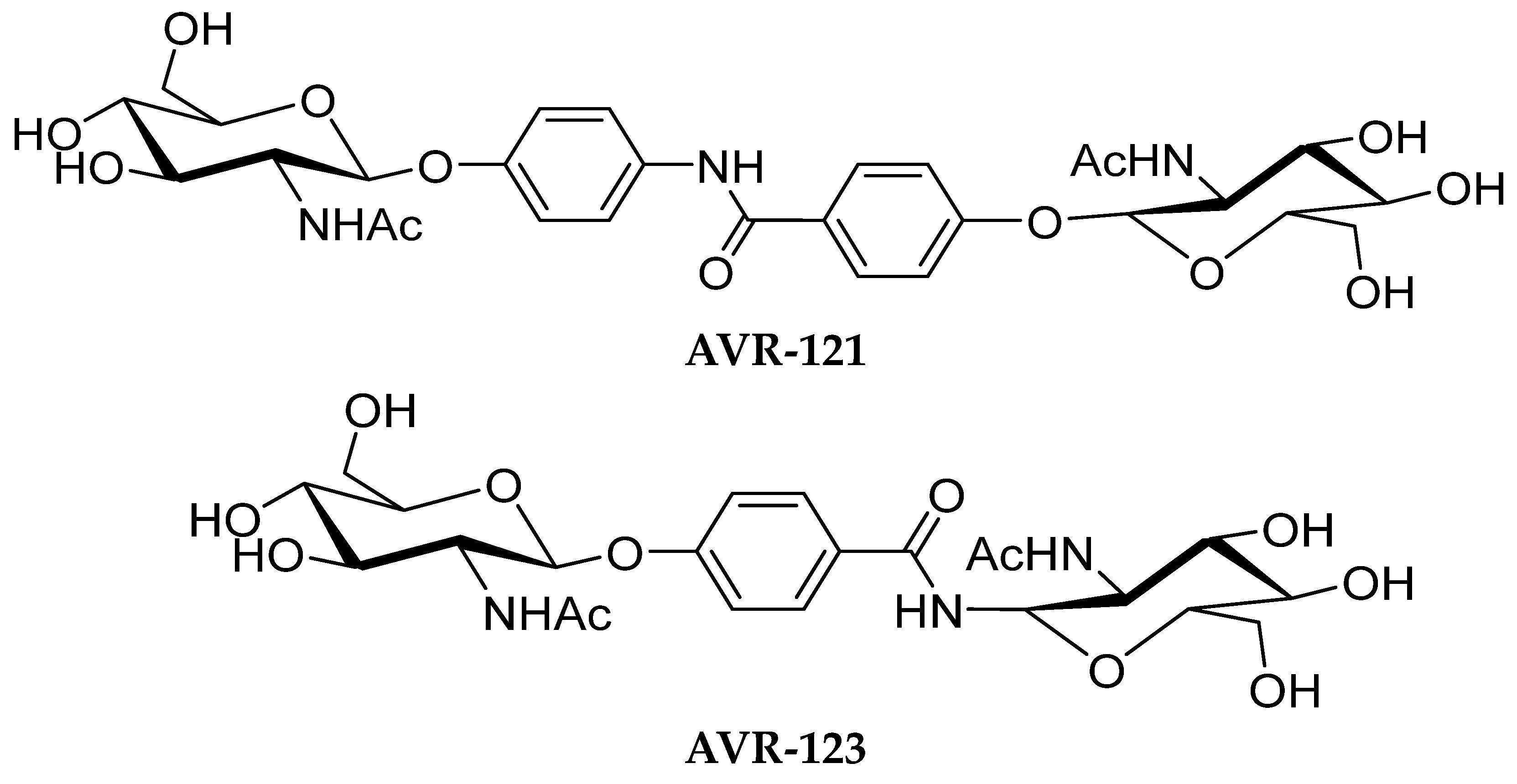
2.2. Formulations of AVR-121 and AVR-123
2.3. Animals
2.4. Cell Proliferation Assay
2.5. Cytokines, TLR2 and TLR4 Protein Quantification Using ELISA
2.6. In Vitro Tube Formation Assay
2.7. In Vitro Cell Migration Assay
2.8. Murine Oxygen-Induced Retinopathy Study
2.9. Lectin Staining and Flat Mount in Murine Retinas
2.10. qPCR Analysis of Mouse Retinas
2.11. Flow Analysis
2.12. Statistical Analysis
3. Results
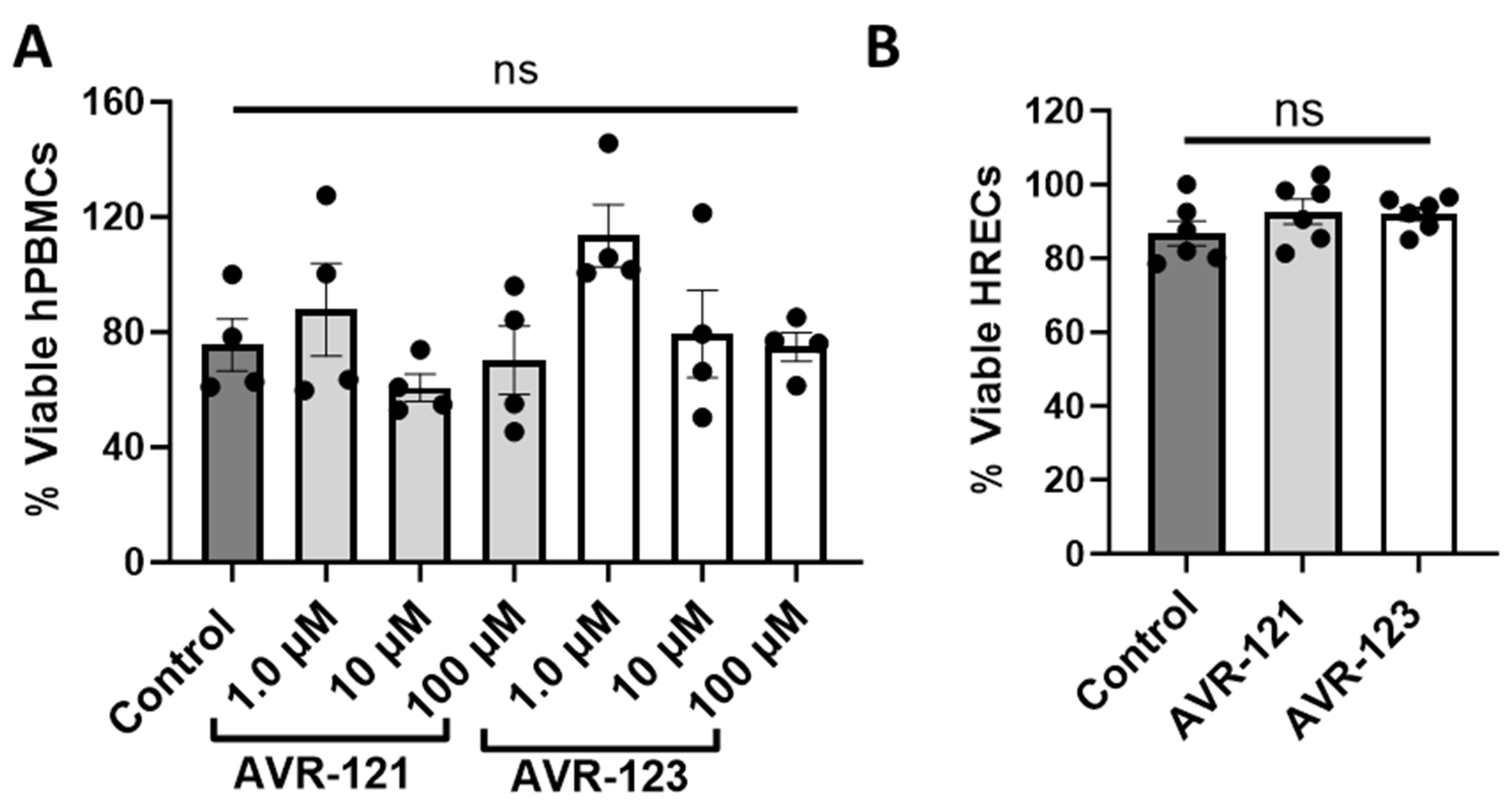
3.1. AVR-121 and AVR-123 Are Not Cytotoxic to Human Peripheral Blood Mononuclear Cells and Human Retinal Endothelial Cells (HRECs)
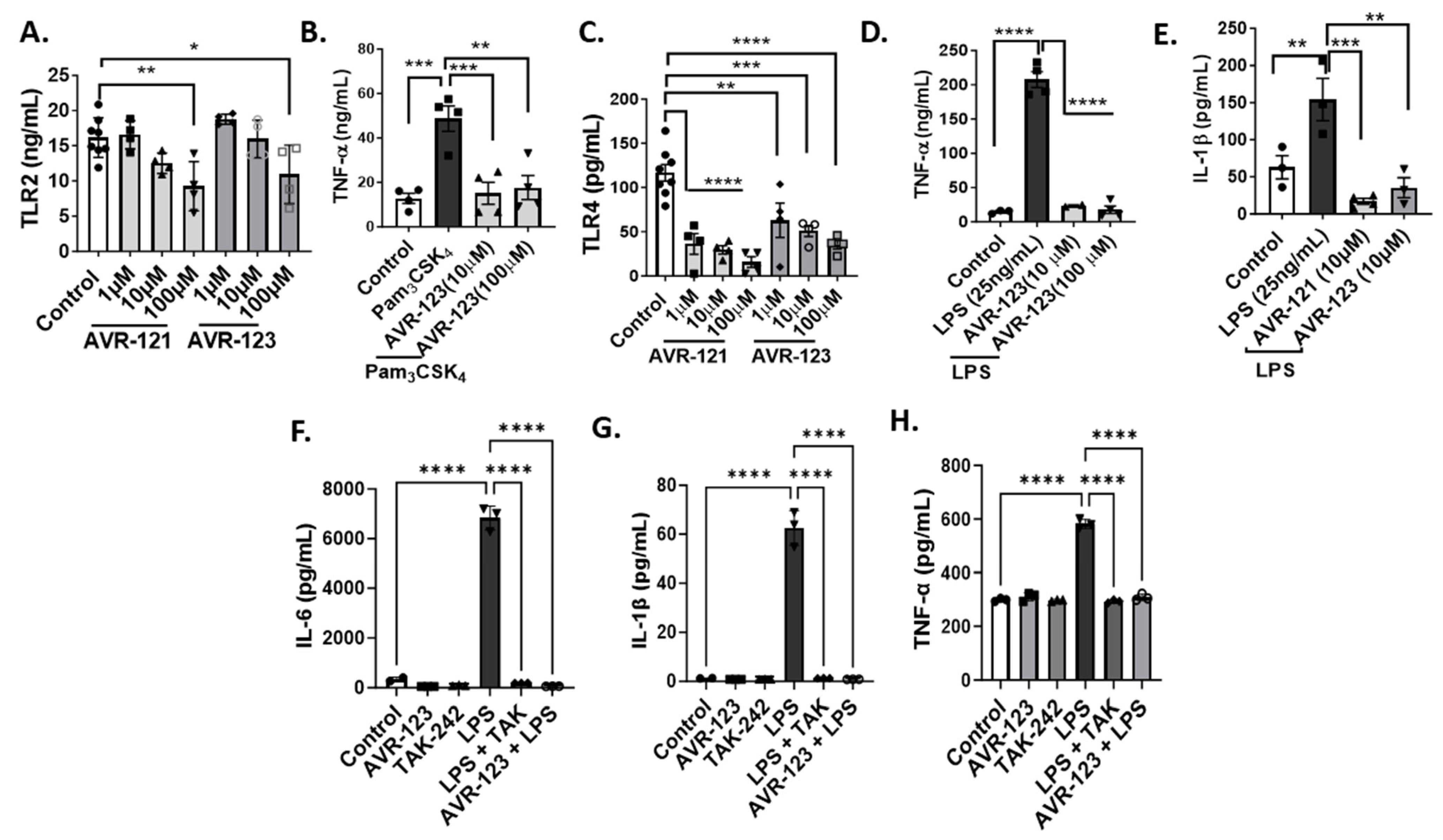
3.2. Treatment of AVR-121 and AVR-123 Inhibited Inflammatory Cytokines Via Toll-Like Receptor-2 and -4 Inhibition In Vitro
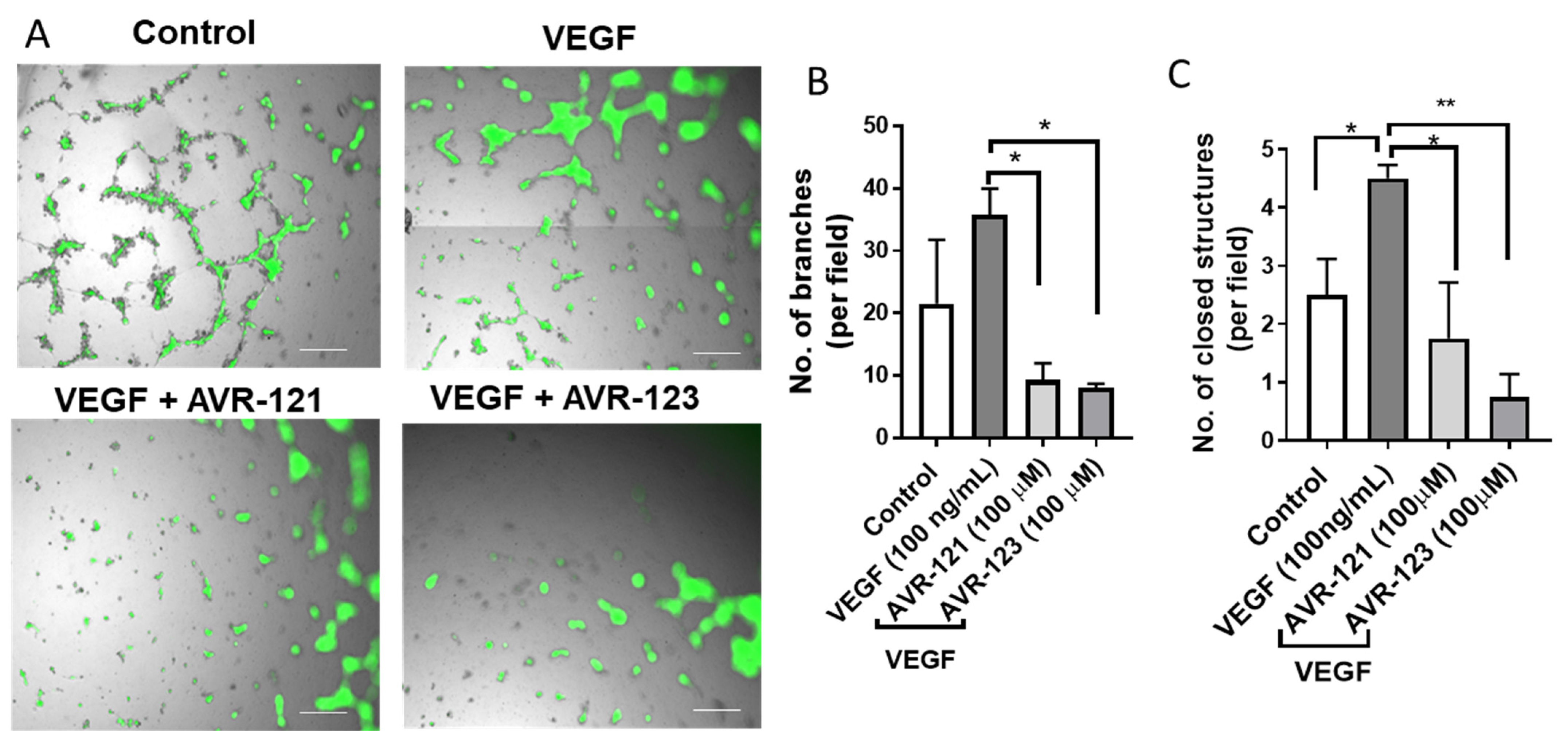
3.3. AVR-123 Inhibits VEGF-Induced Vascular Tube and Microcapillary Formation
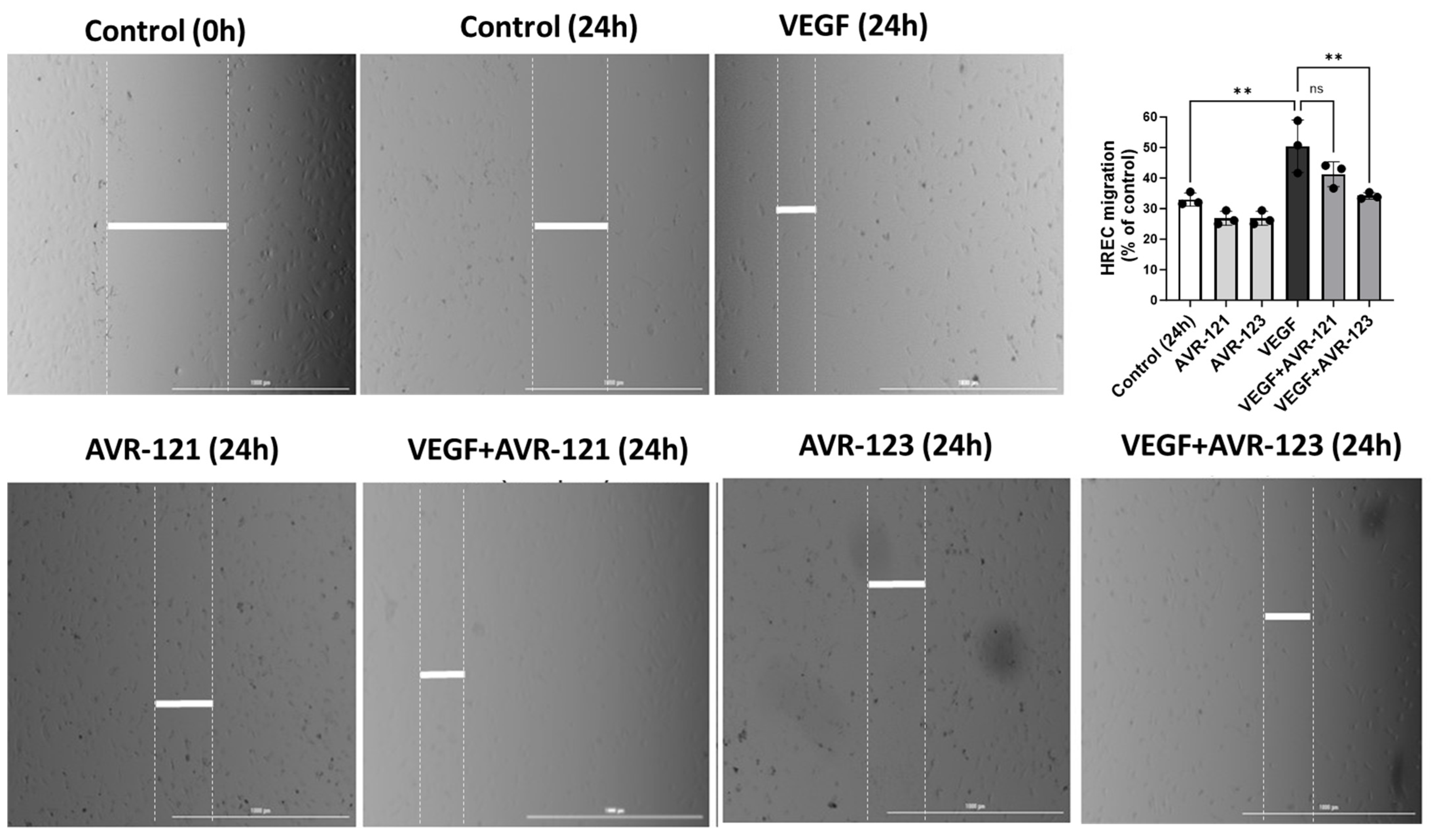
3.4. AVR-123 Inhibits VEGF-Induced Endothelial Cell Migration
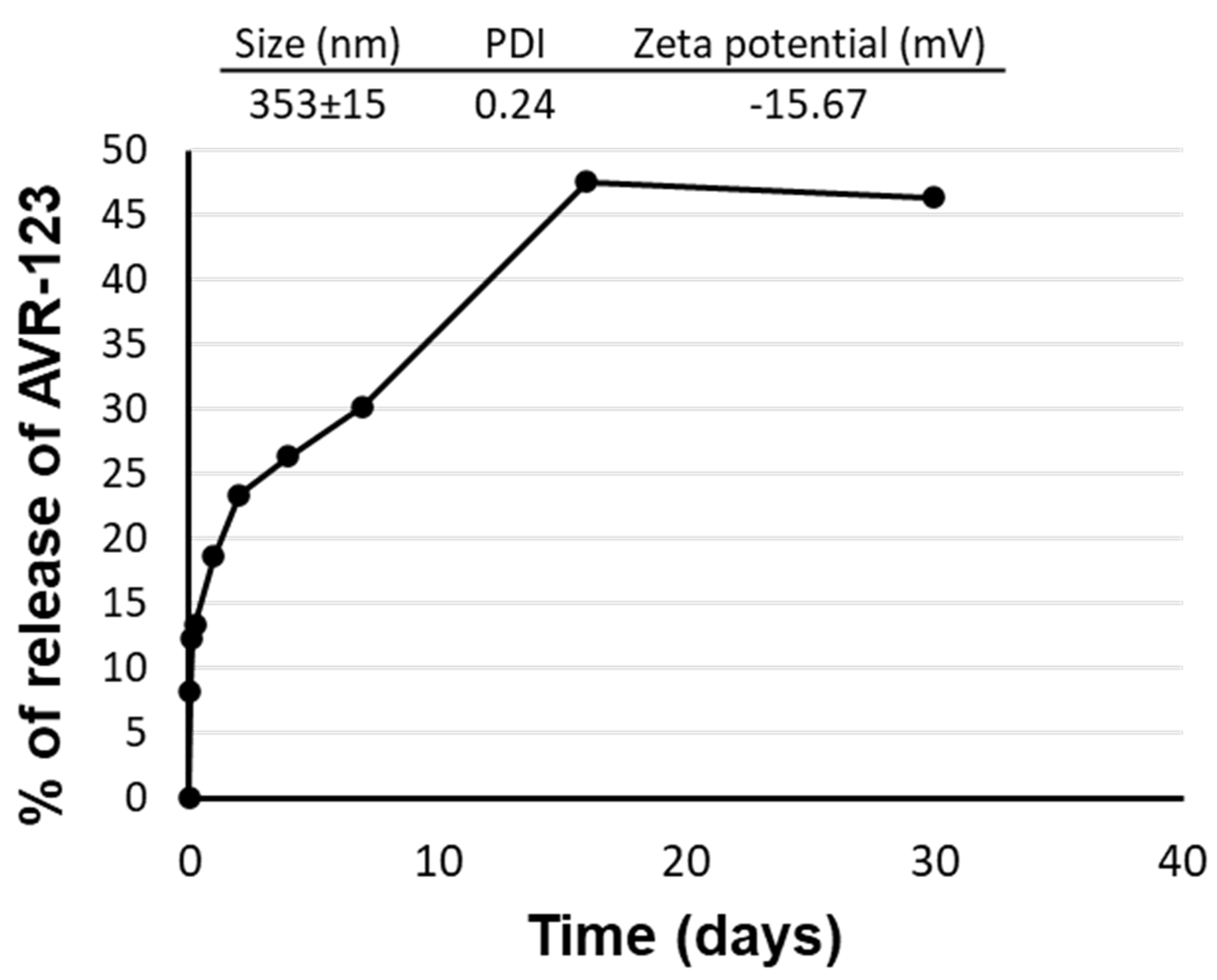
3.5. Formulation Development for AVR-123
3.6. AVR-123 Reduced Vaso-Obliteration and Angiogenesis in a Murine Oxygen-Induced Retinopathy (OIR) Model
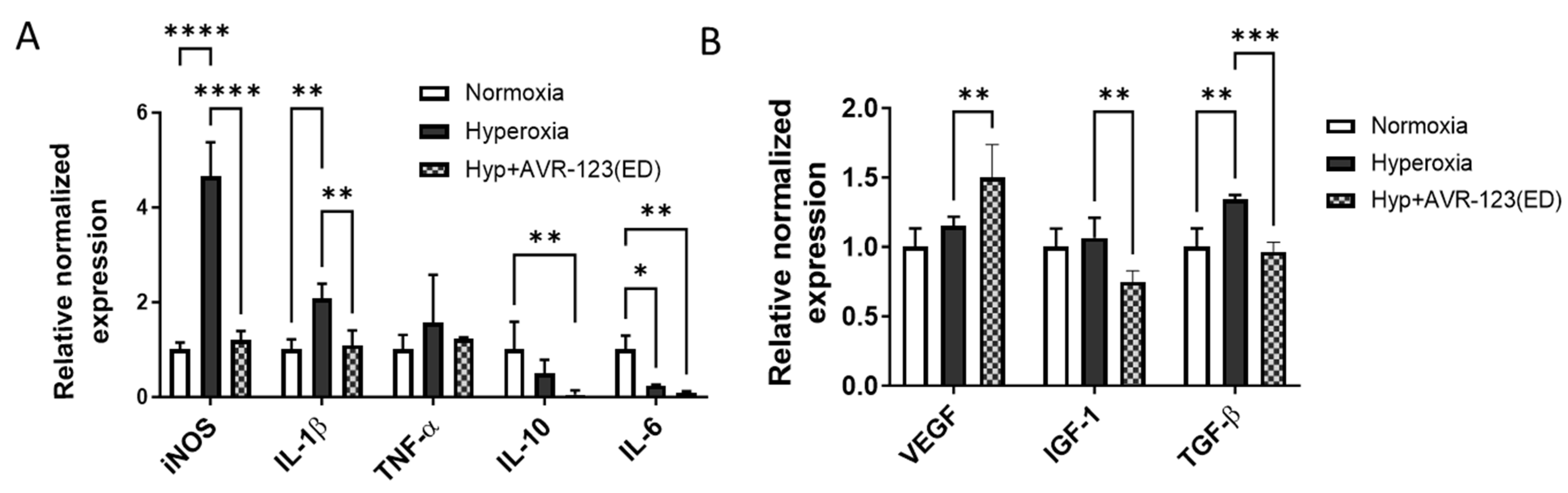
3.7. AVR-123 Treatment Reduced Pro-Inflammatory Cytokines While Not Negatively Affecting the Essential Growth Factors for Retinal Vascular Development
3.8. Treatment with AVR-123 Reduces Populations of Macrophages, CD8+ T Cells, and Neutrophils in Mouse Pup Spleens from the OIR Model
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Kim, S.J.; Port, A.D.; Swan, R.; Campbell, J.P.; Chan, R.V.P.; Chiang, M.F. Retinopathy of prematurity: A review of risk factors and their clinical significance. Surv. Ophthalmol. 2018, 63, 618–637. [Google Scholar] [CrossRef] [PubMed]
- Wu, P.Y.; Fu, Y.K.; Lien, R.I.; Chiang, M.C.; Lee, C.C.; Chen, H.C.; Hsueh, Y.J.; Chen, K.J.; Wang, N.K.; Liu, L.; et al. Systemic Cytokines in Retinopathy of Prematurity. J. Pers. Med. 2023, 13, 291. [Google Scholar] [CrossRef]
- Blencowe, H.; Lawn, J.E.; Vazquez, T.; Fielder, A.; Gilbert, C. Preterm-associated visual impairment and estimates of retinopathy of prematurity at regional and global levels for 2010. Pediatr. Res. 2013, 74 (Suppl. S1), 35–49. [Google Scholar] [CrossRef] [PubMed]
- Wood, E.H.; Chang, E.Y.; Beck, K.; Hadfield, B.R.; Quinn, A.R.; Harper, C.A. 80 Years of vision: Preventing blindness from retinopathy of prematurity. J. Perinatol. 2021, 41, 1216–1224. [Google Scholar] [CrossRef]
- Gyllensten, H.; Humayun, J.; Sjobom, U.; Hellstrom, A.; Lofqvist, C. Costs associated with retinopathy of prematurity: A systematic review and meta-analysis. BMJ Open 2022, 12, e057864. [Google Scholar] [CrossRef]
- Ozdemir, H.B.; Ozdek, S. Late sequelae of retinopathy of prematurity in adolescence and adulthood. Saudi J. Ophthalmol. 2022, 36, 270–277. [Google Scholar] [CrossRef]
- Smith, L.E. Pathogenesis of retinopathy of prematurity. Growth Horm. IGF Res. 2004, 14 (Suppl. A), S140–S144. [Google Scholar] [CrossRef]
- Erdol, H.; Hacioglu, D.; Kola, M.; Turk, A.; Aslan, Y. Investigation of the effect of hemoglobin F and A levels on development of retinopathy of prematurity. J. AAPOS 2017, 21, 136–140. [Google Scholar] [CrossRef] [PubMed]
- Hartnett, M.E. Discovering Mechanisms in the Changing and Diverse Pathology of Retinopathy of Prematurity: The Weisenfeld Award Lecture. Investig. Ophthalmol. Vis. Sci. 2019, 60, 1286–1297. [Google Scholar] [CrossRef]
- Stahl, A.; Brunder, M.C.; Lagreze, W.A.; Molnar, F.E.; Barth, T.; Eter, N.; Guthoff, R.; Krohne, T.U.; Pfeil, J.M.; Group, C.-R.S. Ranibizumab in retinopathy of prematurity—One-year follow-up of ophthalmic outcomes and two-year follow-up of neurodevelopmental outcomes from the CARE-ROP study. Acta Ophthalmol. 2022, 100, e91–e99. [Google Scholar] [CrossRef]
- Sankar, M.J.; Sankar, J.; Mehta, M.; Bhat, V.; Srinivasan, R. Anti-vascular endothelial growth factor (VEGF) drugs for treatment of retinopathy of prematurity. Cochrane Database Syst. Rev. 2016, 2, CD009734. [Google Scholar] [CrossRef]
- Stahl, A.; Sukgen, E.A.; Wu, W.C.; Lepore, D.; Nakanishi, H.; Mazela, J.; Moshfeghi, D.M.; Vitti, R.; Athanikar, A.; Chu, K.; et al. Effect of Intravitreal Aflibercept vs Laser Photocoagulation on Treatment Success of Retinopathy of Prematurity: The FIREFLEYE Randomized Clinical Trial. JAMA 2022, 328, 348–359. [Google Scholar] [CrossRef]
- Chang, Y.H.; Chien, L.N.; Chen, W.T.; Lin, I.C. Comparison of risks of arterial thromboembolic events and glaucoma with ranibizumab and aflibercept intravitreous injection: A nationwide population-based cohort study. PLoS ONE 2022, 17, e0267088. [Google Scholar] [CrossRef] [PubMed]
- Maloney, M.H.; Payne, S.R.; Herrin, J.; Sangaralingham, L.R.; Shah, N.D.; Barkmeier, A.J. Risk of Systemic Adverse Events after Intravitreal Bevacizumab, Ranibizumab, and Aflibercept in Routine Clinical Practice. Ophthalmology 2021, 128, 417–424. [Google Scholar] [CrossRef] [PubMed]
- Quinn, G.E.; Dobson, V.; Barr, C.C.; Davis, B.R.; Flynn, J.T.; Palmer, E.A.; Robertson, J.; Trese, M.T. Visual acuity in infants after vitrectomy for severe retinopathy of prematurity. Ophthalmology 1991, 98, 5–13. [Google Scholar] [CrossRef] [PubMed]
- Shah, P.K.; Narendran, V.; Kalpana, N.; Tawansy, K.A. Anatomical and visual outcome of stages 4 and 5 retinopathy of prematurity. Eye 2009, 23, 176–180. [Google Scholar] [CrossRef] [PubMed]
- Hansen, E.D.; Hartnett, M.E. A review of treatment for retinopathy of prematurity. Expert. Rev. Ophthalmol. 2019, 14, 73–87. [Google Scholar] [CrossRef]
- Sato, T.; Kusaka, S.; Shimojo, H.; Fujikado, T. Simultaneous analyses of vitreous levels of 27 cytokines in eyes with retinopathy of prematurity. Ophthalmology 2009, 116, 2165–2169. [Google Scholar] [CrossRef] [PubMed]
- Checchin, D.; Sennlaub, F.; Levavasseur, E.; Leduc, M.; Chemtob, S. Potential role of microglia in retinal blood vessel formation. Investig. Ophthalmol. Vis. Sci. 2006, 47, 3595–3602. [Google Scholar] [CrossRef]
- Ishida, S.; Yamashiro, K.; Usui, T.; Kaji, Y.; Ogura, Y.; Hida, T.; Honda, Y.; Oguchi, Y.; Adamis, A.P. Leukocytes mediate retinal vascular remodeling during development and vaso-obliteration in disease. Nat. Med. 2003, 9, 781–788. [Google Scholar] [CrossRef]
- Lee, J.; Dammann, O. Perinatal infection, inflammation, and retinopathy of prematurity. Semin. Fetal Neonatal Med. 2012, 17, 26–29. [Google Scholar] [CrossRef] [PubMed]
- Rivera, J.C.; Holm, M.; Austeng, D.; Morken, T.S.; Zhou, T.E.; Beaudry-Richard, A.; Sierra, E.M.; Dammann, O.; Chemtob, S. Retinopathy of prematurity: Inflammation, choroidal degeneration, and novel promising therapeutic strategies. J. Neuroinflammation 2017, 14, 165. [Google Scholar] [CrossRef] [PubMed]
- Langrish, C.L.; Buddle, J.C.; Thrasher, A.J.; Goldblatt, D. Neonatal dendritic cells are intrinsically biased against Th-1 immune responses. Clin. Exp. Immunol. 2002, 128, 118–123. [Google Scholar] [CrossRef] [PubMed]
- Corinti, S.; Albanesi, C.; la Sala, A.; Pastore, S.; Girolomoni, G. Regulatory activity of autocrine IL-10 on dendritic cell functions. J. Immunol. 2001, 166, 4312–4318. [Google Scholar] [CrossRef] [PubMed]
- Nguyen, M.; Leuridan, E.; Zhang, T.; De Wit, D.; Willems, F.; Van Damme, P.; Goldman, M.; Goriely, S. Acquisition of adult-like TLR4 and TLR9 responses during the first year of life. PLoS ONE 2010, 5, e10407. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.P.; Yu, S.L.; Huang, Z.J.; Huang, J.; Pan, J.; Feng, X.; Zhang, X.G.; Wang, J.H.; Wang, J. An impaired inflammatory cytokine response to gram-negative LPS in human neonates is associated with the defective TLR-mediated signaling pathway. J. Clin. Immunol. 2015, 35, 218–226. [Google Scholar] [CrossRef]
- Nussbaum, C.; Gloning, A.; Pruenster, M.; Frommhold, D.; Bierschenk, S.; Genzel-Boroviczeny, O.; von Andrian, U.H.; Quackenbush, E.; Sperandio, M. Neutrophil and endothelial adhesive function during human fetal ontogeny. J. Leukoc. Biol. 2013, 93, 175–184. [Google Scholar] [CrossRef]
- Weinberger, B.; Laskin, D.L.; Mariano, T.M.; Sunil, V.R.; DeCoste, C.J.; Heck, D.E.; Gardner, C.R.; Laskin, J.D. Mechanisms underlying reduced responsiveness of neonatal neutrophils to distinct chemoattractants. J. Leukoc. Biol. 2001, 70, 969–976. [Google Scholar] [CrossRef]
- Panda, S.K.; Kumar, S.; Tupperwar, N.C.; Vaidya, T.; George, A.; Rath, S.; Bal, V.; Ravindran, B. Chitohexaose activates macrophages by alternate pathway through TLR4 and blocks endotoxemia. PLoS Pathog. 2012, 8, e1002717. [Google Scholar] [CrossRef]
- Acharya, S.; Das, P.; Agarwaal, B. Novel Immunodulating Small. Molecules. Patent PCT/US2023/022481, 17 May 2020. [Google Scholar]
- Sun, Y.; Ni, Y.; Kong, N.; Huang, C. TLR2 signaling contributes to the angiogenesis of oxygen-induced retinopathy. Exp. Eye Res. 2021, 210, 108716. [Google Scholar] [CrossRef]
- Amankwa, C.E.; Gondi, S.R.; Dibas, A.; Weston, C.; Funk, A.; Nguyen, T.; Nguyen, K.T.; Ellis, D.Z.; Acharya, S. Novel Thiol Containing Hybrid Antioxidant-Nitric Oxide Donor Small Molecules for Treatment of Glaucoma. Antioxidants 2021, 10, 575. [Google Scholar] [CrossRef]
- Hinkle, L.; Le, D.; Nguyen, T.; Tran, V.; Amankwa, C.E.; Weston, C.; Shen, H.; Nguyen, K.T.; Rahimi, M.; Acharya, S. Nano encapsulated novel compound SA-10 with therapeutic activity in both acute and chronic murine hindlimb ischemia models. Nanomed. Nanotechnol. Biol. Med. 2021, 35, 102400. [Google Scholar] [CrossRef]
- Stankowska, D.L.; Millar, J.C.; Kodati, B.; Behera, S.; Chaphalkar, R.M.; Nguyen, T.; Nguyen, K.T.; Krishnamoorthy, R.R.; Ellis, D.Z.; Acharya, S. Nanoencapsulated hybrid compound SA-2 with long-lasting intraocular pressure-lowering activity in rodent eyes. Mol. Vis. 2021, 27, 37–49. [Google Scholar]
- Deliyanti, D.; Figgett, W.A.; Gebhardt, T.; Trapani, J.A.; Mackay, F.; Wilkinson-Berka, J.L. CD8+ T Cells Promote Pathological Angiogenesis in Ocular Neovascular Disease. Arterioscler. Thromb. Vasc. Biol. 2023, 43, 522–536. [Google Scholar] [CrossRef]
- Talia, D.; Zhu, T.; Agrotis, A.; Slattery, R.; Le Page, M.; Mackay-Fisson, F.; Wilkinson-Berka, J. The contribution of T cells to retinopathy of prematurity in mice. Investig. Ophthalmol. Vis. Sci. 2013, 54, 2027. [Google Scholar]
- Hu, Y.X.; Xu, X.X.; Shao, Y.; Yuan, G.L.; Mei, F.; Zhou, Q.; Cheng, Y.; Wang, J.; Wu, X.R. The prognostic value of lymphocyte-to-monocyte ratio in retinopathy of prematurity. Int. J. Ophthalmol. 2017, 10, 1716–1721. [Google Scholar] [CrossRef] [PubMed]
- Obata, S.; Matsumoto, R.; Kakinoki, M.; Sawada, O.; Sawada, T.; Saishin, Y.; Yanagi, T.; Maruo, Y.; Ohji, M. Blood neutrophil-to-lymphocyte ratio as a risk factor in treatment for retinopathy of prematurity. Graefe’s Arch. Clin. Exp. Ophthalmol. 2023, 261, 951–957. [Google Scholar] [CrossRef] [PubMed]
- Yoon, H.S. Neonatal innate immunity and Toll-like receptor. Korean J. Pediatr. 2010, 53, 985–988. [Google Scholar] [CrossRef]
- Kawai, T.; Akira, S. Signaling to NF-kappaB by Toll-like receptors. Trends Mol. Med. 2007, 13, 460–469. [Google Scholar] [CrossRef] [PubMed]
- Sanchez, M.C.; Chiabrando, G.A. Multitarget Activities of Muller Glial Cells and Low-Density Lipoprotein Receptor-Related Protein 1 in Proliferative Retinopathies. ASN Neuro 2022, 14, 17590914221136365. [Google Scholar] [CrossRef]
- Dvoriantchikova, G.; Barakat, D.J.; Hernandez, E.; Shestopalov, V.I.; Ivanov, D. Toll-like receptor 4 contributes to retinal ischemia/reperfusion injury. Mol. Vis. 2010, 16, 1907–1912. [Google Scholar]
- Tsioti, I.; Steiner, B.L.; Escher, P.; Zinkernagel, M.S.; Benz, P.M.; Kokona, D. Endothelial Toll-like receptor 4 is required for microglia activation in the murine retina after systemic lipopolysaccharide exposure. J. Neuroinflammation 2023, 20, 25. [Google Scholar] [CrossRef]
- Seidel, A.; Liu, L.; Jiang, Y.; Steinle, J.J. Loss of TLR4 in endothelial cells but not Muller cells protects the diabetic retina. Exp. Eye Res. 2021, 206, 108557. [Google Scholar] [CrossRef]
- Li, Y.; Deng, S.L.; Lian, Z.X.; Yu, K. Roles of Toll-Like Receptors in Nitroxidative Stress in Mammals. Cells 2019, 8, 576. [Google Scholar] [CrossRef]
- Li, P.; Chang, M. Roles of PRR-Mediated Signaling Pathways in the Regulation of Oxidative Stress and Inflammatory Diseases. Int. J. Mol. Sci. 2021, 22, 7688. [Google Scholar] [CrossRef] [PubMed]
- Canton, M.; Sanchez-Rodriguez, R.; Spera, I.; Venegas, F.C.; Favia, M.; Viola, A.; Castegna, A. Reactive Oxygen Species in Macrophages: Sources and Targets. Front. Immunol. 2021, 12, 734229. [Google Scholar] [CrossRef]
- Osiak, W.; Watroba, S.; Kapka-Skrzypczak, L.; Kurzepa, J. Two Faces of Heme Catabolic Pathway in Newborns: A Potential Role of Bilirubin and Carbon Monoxide in Neonatal Inflammatory Diseases. Oxid. Med. Cell Longev. 2020, 2020, 7140496. [Google Scholar] [CrossRef] [PubMed]
- Aranda, J.V.; Qu, J.; Valencia, G.B.; Beharry, K.D. Pharmacologic interventions for the prevention and treatment of retinopathy of prematurity. Semin. Perinatol. 2019, 43, 360–366. [Google Scholar] [CrossRef] [PubMed]
- Agnihotri, T.; Kim, N.; Cagnone, G.; Kim, J.S.; Heckel, E.; Pundir, S.; Gaub, P.; Wunnemann, F.; Szarek, W.; Schipper, H.; et al. AB038. Pathological neovascularization in retinopathy of prematurity is regulated by heme-derived iron trafficking. Ann. Eye Sci. 2020, 4, AB038. [Google Scholar] [CrossRef]
- Mezu-Ndubuisi, O.J.; Maheshwari, A. Role of the Endothelium in Neonatal Diseases. Newborn 2022, 1, 44–57. [Google Scholar] [CrossRef]
- Wang, Y.; Wang, Y.; Wang, X.; Ma, Y.; Li, Z.; Di, Y. LncRNA TUG1 Promotes Apoptosis, Invasion, and Angiogenesis of Retinal Endothelial Cells in Retinopathy of Prematurity via MiR-145-5p. Front. Med. 2022, 9, 803214. [Google Scholar] [CrossRef] [PubMed]
- Chen, S.; Sun, Q.; Sun, D.; Willette-Brown, J.; Anderson, M.J.; Gu, Q.; Lewandoski, M.; Hu, Y.; Zhu, F.; Wei, F.; et al. C-CBL is required for inhibition of angiogenesis through modulating JAK2/STAT3 activity in ROP development. Biomed. Pharmacother. 2020, 132, 110856. [Google Scholar] [CrossRef] [PubMed]
- Morabito, A.; De Maio, E.; Di Maio, M.; Normanno, N.; Perrone, F. Tyrosine kinase inhibitors of vascular endothelial growth factor receptors in clinical trials: Current status and future directions. Oncologist 2006, 11, 753–764. [Google Scholar] [CrossRef] [PubMed]
- Qin, S.; Li, A.; Yi, M.; Yu, S.; Zhang, M.; Wu, K. Recent advances on anti-angiogenesis receptor tyrosine kinase inhibitors in cancer therapy. J. Hematol. Oncol. 2019, 12, 27. [Google Scholar] [CrossRef] [PubMed]
- Kong, H.B.; Zheng, G.Y.; He, B.M.; Zhang, Y.; Zhou, Q. Clinical Efficacy and Safety of Propranolol in the Prevention and Treatment of Retinopathy of Prematurity: A Meta-Analysis of Randomized Controlled Trials. Front. Pediatr. 2021, 9, 631673. [Google Scholar] [CrossRef]
- Filippi, L.; Cavallaro, G.; Berti, E.; Padrini, L.; Araimo, G.; Regiroli, G.; Raffaeli, G.; Bozzetti, V.; Tagliabue, P.; Tomasini, B.; et al. Propranolol 0.2% Eye Micro-Drops for Retinopathy of Prematurity: A Prospective Phase IIB Study. Front. Pediatr. 2019, 7, 180. [Google Scholar] [CrossRef]
- Sehgal, P.; Narang, S.; Chawla, D.; Gupta, S.; Jain, S.; Sharma, U.; Katoch, D.; Kaur, J. Systemic biomarkers of retinopathy of prematurity in preterm babies. Int. Ophthalmol. 2023, 43, 1751–1759. [Google Scholar] [CrossRef]
- Hellgren, G.; Löfqvist, C.; Hansen-Pupp, I.; Gram, M.; Smith, L.E.; Ley, D.; Hellström, A. Increased postnatal concentrations of pro-inflammatory cytokines are associated with reduced IGF-I levels and retinopathy of prematurity. Growth Horm. IGF Res. 2018, 39, 19–24. [Google Scholar] [CrossRef]
- Zhang, Q.; Zhang, J.; Guan, Y.; Zhang, S.; Zhu, C.; Xu, G.T.; Wang, L. Suppression of retinal neovascularization by the iNOS inhibitor aminoguanidine in mice of oxygen-induced retinopathy. Graefes Arch. Clin. Exp. Ophthalmol. 2009, 247, 919–927. [Google Scholar] [CrossRef]
- Ninchoji, T.; Love, D.T.; Smith, R.O.; Hedlund, M.; Vestweber, D.; Sessa, W.C.; Claesson-Welsh, L. eNOS-induced vascular barrier disruption in retinopathy by c-Src activation and tyrosine phosphorylation of VE-cadherin. Elife 2021, 10, 64944. [Google Scholar] [CrossRef]
- Brooks, S.E.; Gu, X.; Samuel, S.; Marcus, D.M.; Bartoli, M.; Huang, P.L.; Caldwell, R.B. Reduced Severity of Oxygen-Induced Retinopathy in eNOS-Deficient Mice. Investig. Ophthalmol. Vis. Sci. 2001, 42, 222–228. [Google Scholar]
- Mueller-Buehl, A.M.; Tsai, T.; Hurst, J.; Theiss, C.; Peters, L.; Hofmann, L.; Herms, F.; Kuehn, S.; Schnichels, S.; Joachim, S.C. Reduced Retinal Degeneration in an Oxidative Stress Organ Culture Model through an iNOS-Inhibitor. Biology 2021, 10, 383. [Google Scholar] [CrossRef] [PubMed]
- Sennlaub, F.; Courtois, Y.; Goureau, O. Inducible nitric oxide synthase mediates retinal apoptosis in ischemic proliferative retinopathy. J. Neurosci. 2002, 22, 3987–3993. [Google Scholar] [CrossRef] [PubMed]
- Qiu, F.; Harris, D.L.; Kenyon, B.; Jamali, A.; Ortiz, G.; Hamrah, P. Normal Retina Contains Resident Plasmacytoid Dendritic Cells That Increase in a Mouse Model of Retinopathy of Prematurity. Investig. Ophthalmol. Vis. Sci. 2020, 61, 2793. [Google Scholar]
- Perry, V.H. The influence of systemic inflammation on inflammation in the brain: Implications for chronic neurodegenerative disease. Brain Behav. Immun. 2004, 18, 407–413. [Google Scholar] [CrossRef]
- Holm, M.; Morken, T.S.; Fichorova, R.N.; VanderVeen, D.K.; Allred, E.N.; Dammann, O.; Leviton, A.; Neonatology, E.S.; Ophthalmology, C. Systemic Inflammation-Associated Proteins and Retinopathy of Prematurity in Infants Born Before the 28th Week of Gestation. Investig. Ophthalmol. Vis. Sci. 2017, 58, 6419–6428. [Google Scholar] [CrossRef]
- Hu, K.; Jin, Y.; Chroneos, Z.; Han, X.; Liu, H.; Lin, L. Macrophage Functions and Regulation: Roles in Diseases and Implications in Therapeutics. J. Immunol. Res. 2018, 2018, 7590350. [Google Scholar] [CrossRef]
- Baban, B.; Lamoke, F.; Gutsaeva, D.; Bartoli, M. Dendritic cells-mediated polarization of retinal macrophages in human diabetic retina. Investig. Ophthalmol. Vis. Sci. 2015, 56, 4291. [Google Scholar]
- Chakravarthy, H.; Beli, E.; Navitskaya, S.; O’Reilly, S.; Wang, Q.; Kady, N.; Huang, C.; Grant, M.B.; Busik, J.V. Imbalances in Mobilization and Activation of Pro-Inflammatory and Vascular Reparative Bone Marrow-Derived Cells in Diabetic Retinopathy. PLoS ONE 2016, 11, e0146829. [Google Scholar] [CrossRef]



| Gene | Forward Sequence | Reverse Sequence |
|---|---|---|
| IL-1β | GCACTACAGGCTCCGAGATGAAC | TTGTCGTTGCTTGGTTCTCCTTGT |
| IL-6 | CAACGATGATGCACTTGCAGA | TGTGACTCCAGCTTATCTCTTGG |
| IL-10 | TGCTAACCGACTCCTTAATGCAGGAC | CCTTGATTTCTGGGCCATGCTTCTC |
| VEGF | ATCATGCGGATCAAACCTCACC | GGCTTTGTTCTGTCTTTCTTTGGTC |
| TGF-β2 | GCCCCTGCTGTACCTTCGT | TGCCATCAATACCTGCAAATCTC |
| IGF-1 | CAGTTCGTGTGTGGACCAAG | GAGTCTTGGGCATGTCAGTG |
| TNFα | 5′-GCCTCTTCTCATTCCTGCTTG-3 | 5′-CTGATGAGAGGGAGGCCATT-3 |
| iNOS | 5′-GTTCTCAGCCCAACAATACAAGA-3′ | 5′-GTGGACGGGTCGATGTCAC |
| β-actin | GGACTCCTATGTGGGTGACG | CTTCTCCATGTCGTCCCAGT |
| Antibody | Dilution | Catalog No. | Vendor |
|---|---|---|---|
| CD80-FITC | 1:200 | Cat# 557226 | BD Biosciences, San Jose, CA, USA |
| CD86-FITC | 1:200 | Cat# 555658 | BD Biosciences, San Jose, CA, USA |
| MHC II-APC-Cy7 | 1:400 | Cat# 56-5321-80 | eBioscience, Waltham, MA, USA |
| CD4-PE-Cy7 | 1:200 | Cat# 100526 | Biolegend, San Diego, CA, USA |
| CD8-PE-Cy7 | 1:200 | Cat# 100758 | Biolegend, San Diego, CA, USA |
| B220-FITC | 1:50 | Cat# 553088 | BD Biosciences, San Jose, CA, USA |
| CD3-PE | 1:200 | Cat# 555275 | eBiosceince, Waltham, MA, USA |
| F4/80-APC | 1:200 | Cat# 50-112-9524 | eBiosceince, Waltham, MA, USA |
| LY6G-PE-Texas Red | 1:400 | Cat# 50-112-4886 | eBioscience, Waltham, MA, USA |
| CD11b-FITC | 1:200 | Cat# 562793 | BD Biosciences, San Jose, CA, USA |
| CD11c-FITC | 1:100 | Cat# 561355 | BD Biosciences, San Jose, CA, USA |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Dayoub, A.S.; Acharya, E.; Dibas, A.; Jones, H.P.; Acharya, S. Novel Small Molecules with Anti-Inflammatory and Anti-Angiogenic Activity in a Mouse Model of Oxygen-Induced Retinopathy. Cells 2024, 13, 1371. https://doi.org/10.3390/cells13161371
Dayoub AS, Acharya E, Dibas A, Jones HP, Acharya S. Novel Small Molecules with Anti-Inflammatory and Anti-Angiogenic Activity in a Mouse Model of Oxygen-Induced Retinopathy. Cells. 2024; 13(16):1371. https://doi.org/10.3390/cells13161371
Chicago/Turabian StyleDayoub, Adam S., Eesha Acharya, Adnan Dibas, Harlan P. Jones, and Suchismita Acharya. 2024. "Novel Small Molecules with Anti-Inflammatory and Anti-Angiogenic Activity in a Mouse Model of Oxygen-Induced Retinopathy" Cells 13, no. 16: 1371. https://doi.org/10.3390/cells13161371





