Impact of Polyallylamine Hydrochloride on Gene Expression and Karyotypic Stability of Multidrug Resistant Transformed Cells
Abstract
:1. Introduction
2. Materials and Methods
2.1. Cells
2.2. Treatment Cells with Polymer Polyallylamine
2.3. G-banded Karyotyping
2.4. Cell Cycle Analysis
2.5. Viability Analysis
2.6. qRT-PCR Assay
2.7. MTT Assay
2.8. Colony Forming Assay
2.9. Statistical Analysis
3. Results
3.1. Characteristics of Cell Lines RJK and RJKEB
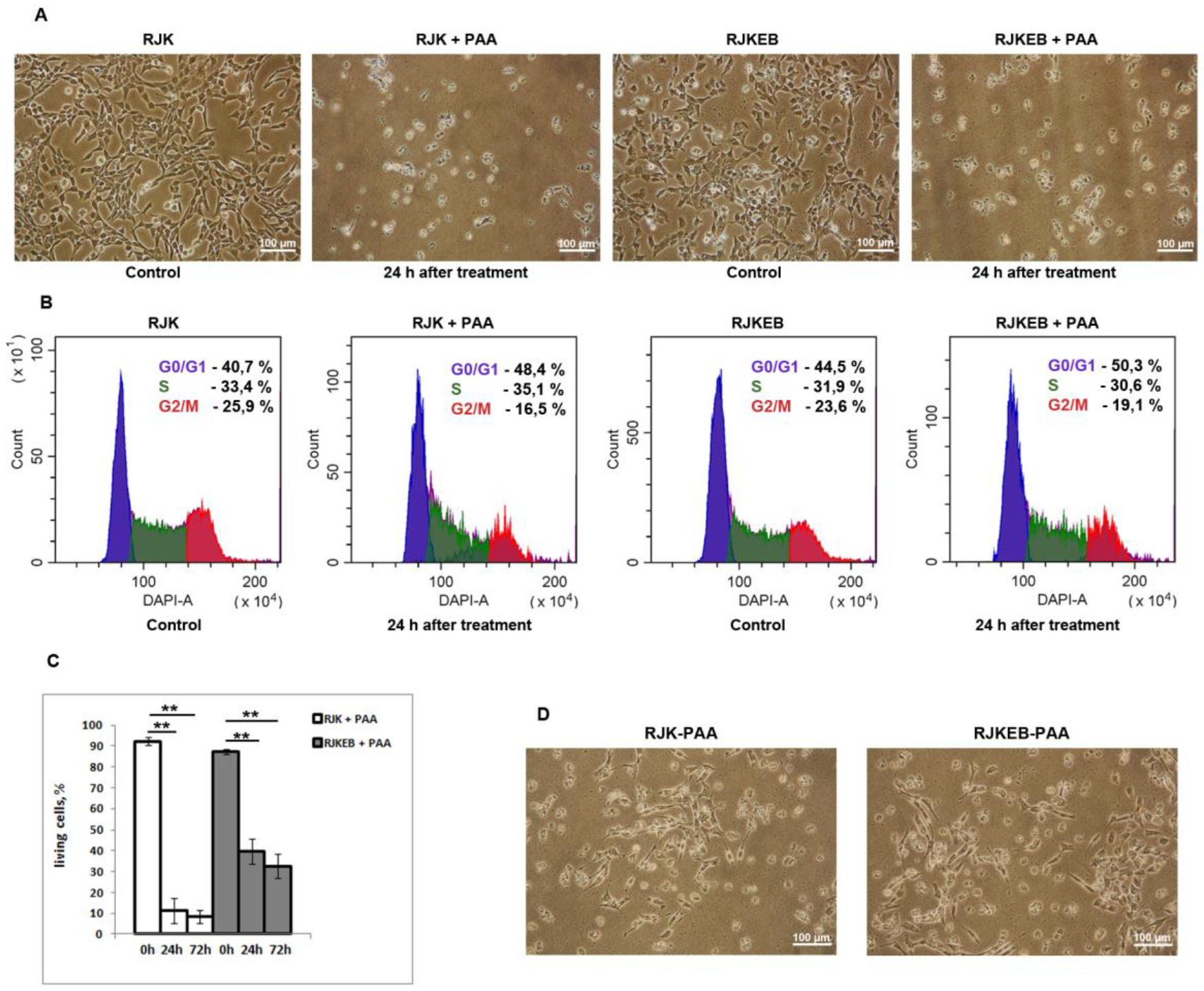
3.2. Morphology and Proliferative Features of RJK and RJKEB Cells after Exposure to PAA
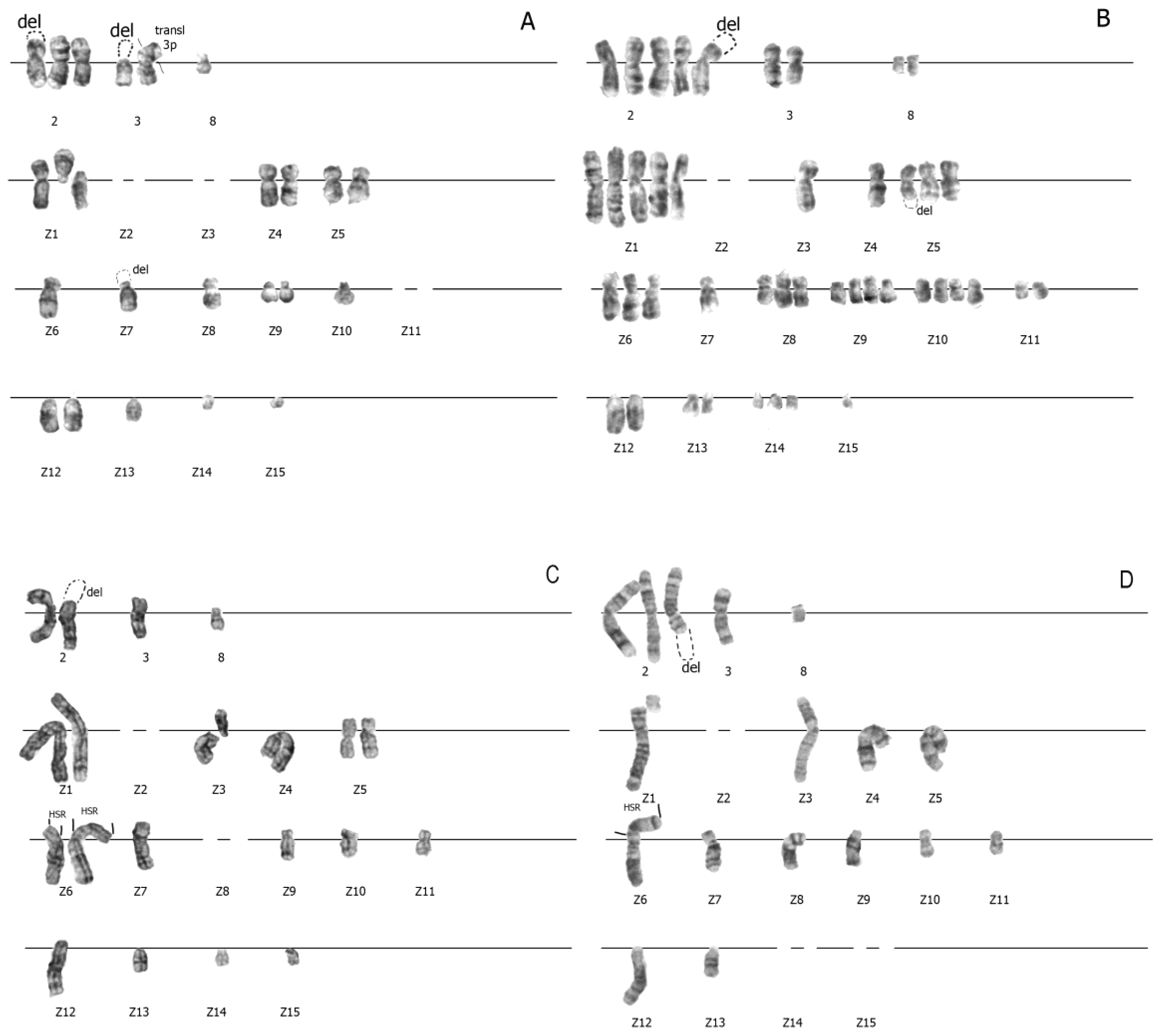
3.3. Increased Karyotypic Instability of RJK and RJKEB Cells Survived after Exposure to PAA
3.4. Expression of Genes Involved in Oncogenic Transformation in Progeny of RJK RJKEB Survived after Exposure to PAA
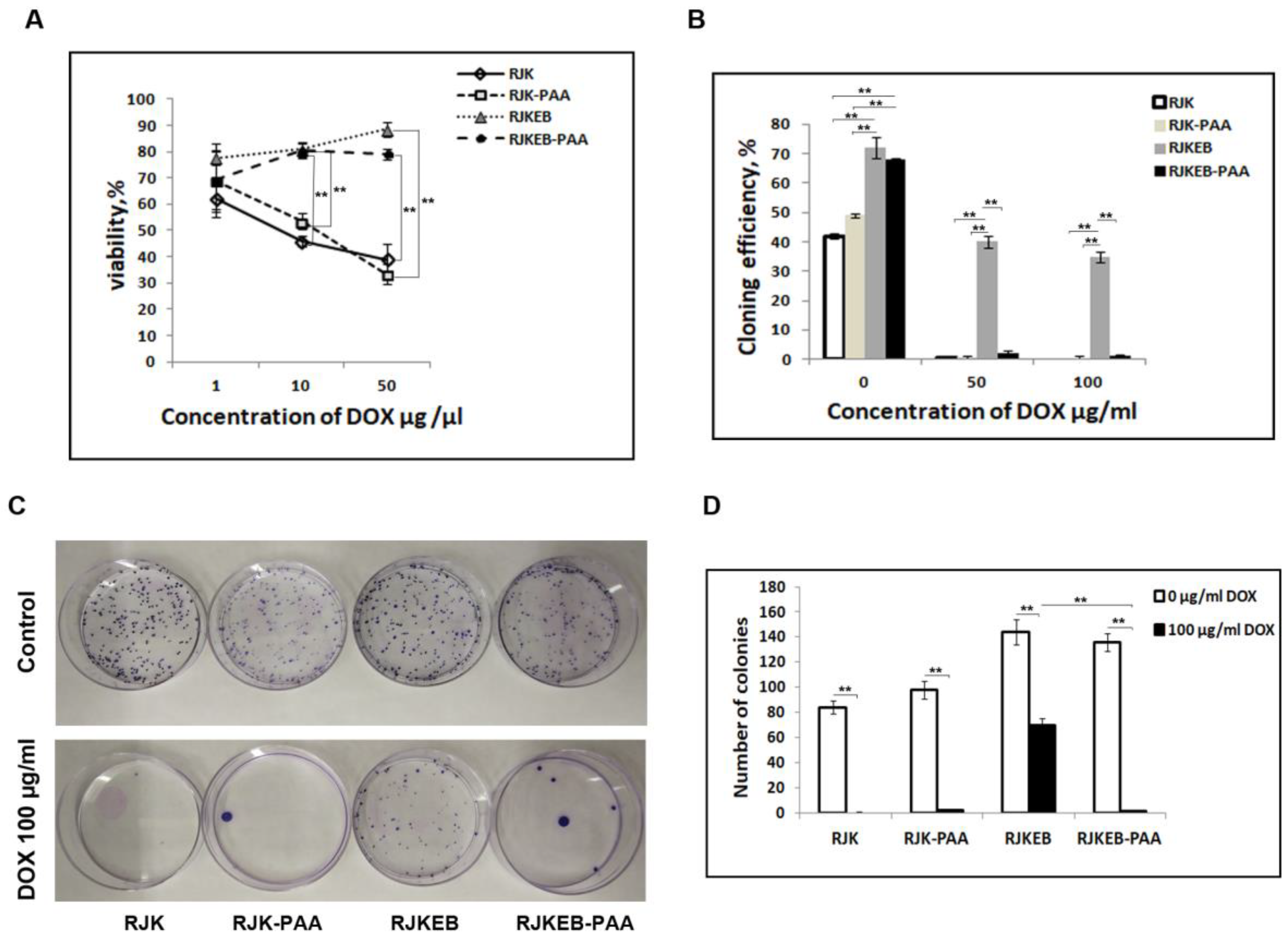
3.5. PAA Reduced RJKEB Cell Resistance to DOX
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Kim, H.; Kim, Y.; Park, J.; Hwang, N.S.; Lee, Y.K.; Hwang, Y. Recent Advances in Engineered Stem Cell-Derived Cell Sheets for Tissue Regeneration. Polymers 2019, 11, 209. [Google Scholar] [CrossRef] [Green Version]
- Avramović, N.; Mandić, B.; Savić-Radojević, A.; Simić, T. Polymeric Nanocarriers of Drug Delivery Systems in Cancer Therapy. Pharmaceutics 2020, 12, 298. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wang, B.; Zhang, Y.; Mao, Z.; Gao, C. Cellular Uptake of Covalent Poly(allylamine hydrochloride) Microcapsules and Its Influences on Cell Functions. Macromol. Biosci. 2012, 12, 1534–1545. [Google Scholar] [CrossRef] [PubMed]
- Martinez, J.S.; Schlenoff, J.B.; Keller, T.C.S. Collective epithelial cell sheet adhesion and migration on polyelectrolyte multilayers with uniform and gradients of compliance. Exp. Cell Res. 2016, 346, 17–29. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Di Silvio, D.; Martínez-Moro, M.; Salvador, C.; de Los Angeles Ramirez, M.; Cáceres-Vélez, P.R.; Ortore, M.G.; Dupin, D.; Andreozzi, P.; Moya, S.E. Self-assembly of poly(allylamine)/siRNA nanoparticles, their intracellular fate and siRNA delivery. J. Colloid Interface Sci. 2019, 557, 757–766. [Google Scholar] [CrossRef] [PubMed]
- Moby, V.; Kadi, A.; de Isla, N.; Stoltz, J.; Menu, P. Polyelectrolyte multilayer films: Effect of the initial anchoring layer on the cell growth. Bio-Med. Mater. Eng. 2008, 18, 199–204. [Google Scholar] [CrossRef]
- Jia, Z.; Xiu, P.; Roohani-Esfahani, S.-I.; Zreiqat, H.; Xiong, P.; Zhou, W.; Yan, J.; Cheng, Y.; Zheng, Y. Triple-Bioinspired Burying/Crosslinking Interfacial Coassembly Strategy for Layer-by-Layer Construction of Robust Functional Bioceramic Self-Coatings for Osteointegration Applications. ACS Appl. Mater. Interfaces 2019, 11, 4447–4469. [Google Scholar] [CrossRef]
- Tsai, H.-C.; Wang, J.-H.; Chen, Y.-A.; Tsai, L.-K.; Young, T.-H.; Li, Y.-C.E. A self-assembled layer-by-layer surface modification to fabricate the neuron-rich model from neural stem/precursor cells. J. Formos. Med. Assoc. 2020, 119, 430–438. [Google Scholar] [CrossRef]
- Janeesh, P.A.; Sami, H.; Dhanya, C.R.; Sivakumar, S.; Abraham, A. Biocompatibility and genotoxicity studies of polyallylamine hydrochloride nanocapsules in rats. RSC Adv. 2014, 4, 24484–24497. [Google Scholar] [CrossRef]
- Гpинчyк, T.M.; Шилинa, M.A.; Aлeкceeнкo, Л.Л.; Ивaнoвa, В.П. Кpaткoвpeмeннoe вoздeйcтвиe пoлиaллилaминa нa клeтки китaйcкoгo xoмячкa линии RJK c мнoжecтвeннoй лeкapcтвeннoй ycтoйчивocтью вeдeт к дecтaбилизaции cтpyктypы иx кapиoтипa. Tsitologiya 2020, 62, 446–454. [Google Scholar] [CrossRef]
- Ivanova, V.P.; Grinchuk, T.M.; Alekseenko, L.; Artsybasheva, I.V.; Gavrilova, I.I. Effect of synthetic polycation polyallylamine on adhesion and viability of CHL V-79 RJK Chinese hamster fibroblasts with various heat resistance. Cell Tissue Biol. 2010, 4, 520–528. [Google Scholar] [CrossRef]
- Berguetti, T.S.; Quintaes, L.S.P.; Pereira, T.H.; Robaina, M.; Cruz, A.L.S.; Maia, R.C.; De Souza, P.S.; Hancio, T. TNF-α Modulates P-Glycoprotein Expression and Contributes to Cellular Proliferation via Extracellular Vesicles. Cells 2019, 8, 500. [Google Scholar] [CrossRef] [Green Version]
- Meliksetian, M.B.; Berezkina, E.V.; A Pavlenko, M.; Grinchuk, T.M. Mechanisms of drug resistance of two cell lines of human chronic promyelocytic leukemia K562, resistant to DNA topoisomerase II inhibitors adriamycin and etoposide. Tsitologiya 1999, 41, 615–621. [Google Scholar]
- Hao, Q.; Chen, Y.; Zhou, X. The Janus Face of p53-Targeting Ubiquitin Ligases. Cells 2020, 9, 1656. [Google Scholar] [CrossRef] [PubMed]
- Bhushan, A.; Abramson, R.O.B.I.N.; Chiu, J.F.; Tritton, T.R. Expression of c-fos in human and murine multidrug-resistant cells. Mol. Pharmacol. 1992, 42, 69–74. [Google Scholar]
- Milani, A.; Basirnejad, M.; Bolhassani, A. Heat-shock proteins in diagnosis and treatment: An overview of different biochemical and immunological functions. Immunotherapy 2019, 11, 215–239. [Google Scholar] [CrossRef]
- Roninson, I.B. From amplification to function: The case of the MDR1 gene. Mutat. Res. Genet. Toxicol. 1992, 276, 151–161. [Google Scholar] [CrossRef]
- Grinchuk, T.M.; A Lipskaia, L.; Artsybasheva, I.V.; Sorokina, E.A.; Pan’Shina, I.T.; Ignatova, T.N. The karyotypic variability of Chinese hamster CHLV-79 RJK cells characterized by multiple drug resistance resulting from the amplification of the mdr gene family. Tsitologiya 1996, 38, 161. (In Russian) [Google Scholar]
- Pavlov, G.M.; Panarin, E.F.; Korneeva, E.; Gavrilova, I.; Tarasova, N. Molecular Properties and Electrostatic Interactions of Linear Poly(allylamine hydrochloride) Chains. Prog. Colloid. Polym. 2006, 131, 134–140. [Google Scholar] [CrossRef]
- Ray, M.; Mohandas, T.; Hamerton, J. Proposed banding nomenclature for the Chinese hamster chromosomes (Cricetulus griseus). Cytogenet Cell Genet. 1976, 16, 83–91. [Google Scholar] [CrossRef]
- Wang, X.; Zhang, H.; Chen, X. Drug resistance and combating drug resistance in cancer. Cancer Drug Resist. 2019, 2, 141–160. [Google Scholar] [CrossRef] [Green Version]
- Pan, J.; Rostamizadeh, K.; Filipczak, N.; Torchilin, V.P. Polymeric Co-Delivery Systems in Cancer Treatment: An Overview on Component Drugs’ Dosage Ratio Effect. Molecules 2019, 24, 1035. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Borgonovo, T.; Vaz, I.M.; Senegaglia, A.C.; Rebelatto, C.L.K.; Brofman, P.R.S. Genetic evaluation of mesenchymal stem cells by G-banded karyotyping in a Cell Technology Center. Rev. Bras. Hematol. Hemoter. 2014, 36, 202–207. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Durkin, S.G.; Glover, T.W. Chromosome Fragile Sites. Annu. Rev. Genet. 2007, 41, 169–192. [Google Scholar] [CrossRef] [PubMed]
- Bjerregaard, V.A.; Özer, Ö.; Hickson, I.D.; Liu, Y. The Detection and Analysis of Chromosome Fragile Sites. Methods Mol. Biol. 2017, 1672, 471–482. [Google Scholar] [CrossRef]
- Grinchuk, T.M.; Pavlenko, M.A.; Artsybasheva, I.V.; Meliksetyan, M.B.; Alekseenko, L.L.; Pugovkina, N.A. Hamster cells CHL V-79 RJK selected for resistance to etoposide exhibit multiple changes of karyotype. Tsitologiia 1999, 41, 190–199. [Google Scholar] [PubMed]
- Grinchuk, T.M.; Shilina, M.A.; Alekseenko, L. Long-term cultivation of Chinese hamster fibroblasts V-79 RJK under elevated temperature results in karyotype destabilization. Cell Tissue Biol. 2015, 9, 119–126. [Google Scholar] [CrossRef]
- Ferguson, L.R.; Chen, H.; Collins, A.R.; Connell, M.; Damia, G.; Dasgupta, S.; Malhotra, M.; Meeker, A.K.; Amedei, A.; Amin, A.; et al. Genomic instability in human cancer: Molecular insights and opportunities for therapeutic attack and prevention through diet and nutrition. Semin. Cancer Biol. 2015, 35, S5–S24. [Google Scholar] [CrossRef]
- Andor, N.; Maley, C.C.; Ji, H.P. Genomic Instability in Cancer: Teetering on the Limit of Tolerance. Cancer Res. 2017, 77, 2179–2185. [Google Scholar] [CrossRef] [Green Version]
- Storchova, Z.; Pellman, D. From polyploidy to aneuploidy, genome instability and cancer. Nat. Rev. Mol. Cell Biol. 2004, 5, 45–54. [Google Scholar] [CrossRef]
- Grinchuk, T.M.; Ignatova, T.N.; Sorokina, E.A.; Artsybasheva, I.V.; Pan’Shina, I.T. Chromosomal polymorphism of mammalian cells resistant to drugs in multiple passages. I. Karyotype analysis of Chinese hamster cells resistant to ethidium bromide in the early passages of the first steps of selection. Tsitologiya 1988, 30, 312–320. [Google Scholar]
- Kuznetsova, A.Y.; Seget, K.; Moeller, G.K.; de Pagter, M.S.; de Roos, J.A.D.M.; Dürrbaum, M.; Kuffer, C.; Müller, S.; Zaman, G.J.R.; Kloosterman, W.P.; et al. Chromosomal instability, tolerance of mitotic errors and multidrug resistance are promoted by tetraploidization in human cells. Cell Cycle 2015, 14, 2810–2820. [Google Scholar] [CrossRef] [PubMed]
- Sheltzer, J.M.; Ko, J.H.; Replogle, J.M.; Burgos, N.C.H.; Chung, E.S.; Meehl, C.M.; Sayles, N.M.; Passerini, V.; Storchova, Z.; Amon, A. Single-chromosome gains commonly function as tumor suppressors. Cancer Cell 2017, 31, 240–255. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Dabas, N.; Byrnes, D.M.; Rosa, A.M.; Eller, M.S.; Grichnik, J.M. Diagnostic role of chromosomal instability in melanoma. J. Ski. Cancer 2012, 2012, 1–7. [Google Scholar] [CrossRef] [Green Version]
- Shilina, M.A.; Grinchuk, T.M.; Anatskaya, O.; Vinogradov, A.; Alekseenko, L.; Elmuratov, A.; Nikolsky, N.N. Cytogenetic and transcriptomic analysis of human endometrial msc retaining proliferative activity after sublethal heat shock. Cells 2018, 7, 184. [Google Scholar] [CrossRef] [Green Version]
- Lee, J.H.; Berger, J.M. Cell Cycle-Dependent Control and Roles of DNA Topoisomerase II. Genes 2019, 10, 859. [Google Scholar] [CrossRef] [Green Version]
- Feng, Y.; Zhang, H.; Gao, W.; Wen, S.; Huangfu, H.; Sun, R.; Bai, W.; Wang, B. Expression of DNA topoisomerase II-α: Clinical significance in laryngeal carcinoma. Oncol. Lett. 2014, 8, 1575–1580. [Google Scholar] [CrossRef] [Green Version]
- Sakurai, R.; Kaira, K.; Miura, Y.; Sunaga, N.; Saito, R.; Oyama, T.; Hisada, T.; Yamada, M. Clinical significance of topoisomerase-II expression in patients with advanced non-small cell lung cancer treated with amrubicin. Thorac. Cancer 2020, 11, 426–435. [Google Scholar] [CrossRef]
- Shaulian, E.; Karin, M. AP-1 as a regulator of cell life and death. Nat. Cell Biol. 2002, 4, E131–E136. [Google Scholar] [CrossRef]
- Yang, W.; Zhao, S.; Wu, B.; Xu, J.; Wu, Z.; Guo, J.; Zeng, R. BATF2 inhibits chemotherapy resistance by suppressing AP-1 in vincristine-resistant gastric cancer cells. Cancer Chemother. Pharmacol. 2019, 84, 1279–1288. [Google Scholar] [CrossRef]
- Muhammad, N.; Bhattacharya, S.; Steele, R.; Phillips, N.; Ray, R.B. Involvement of c-Fos in the promotion of cancer stem-like cell properties in head and neck squamous cell carcinoma. Clin. Cancer Res. 2016, 23, 3120–3128. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Durchdewald, M.; Angel, P.; Hess, J. The transcription factor Fos: A Janus-type regulator in health and disease. Histol. Histopathol. 2009, 24, 1451–1461. [Google Scholar] [PubMed]
- Mikula, M.; Gotzmann, J.; Fischer, A.N.M.; Wolschek, M.F.; Thallinger, C.; Schulte-Hermann, R.; Beug, H.; Mikulits, W. The proto-oncoprotein c-Fos negatively regulates hepatocellular tumorigenesis. Oncogene 2003, 22, 6725–6738. [Google Scholar] [CrossRef] [Green Version]
- Meinhold-Heerlein, I.; Bauerschlag, D.; Hilpert, F.; Dimitrov, P.; Sapinoso, L.M.; Orlowska-Volk, M.; Bauknecht, T.; Park, T.-W.; Jonat, W.; Jacobsen, A.; et al. Molecular and prognostic distinction between serous ovarian carcinomas of varying grade and malignant potential. Oncogene 2004, 24, 1053–1065. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hein, S.; Kanowski, C.; Mahner, S.; Löning, T.; Jänicke, F.; Milde-Langosch, K. Expression of Jun and Fos proteins in ovarian tumors of different malignant potential and in ovarian cancer cell lines. Oncol. Rep. 2009, 22, 177–183. [Google Scholar] [CrossRef] [Green Version]
- Oliveira-Ferrer, L.; Rößler, K.; Haustein, V.; Schröder, C.; Wicklein, D.; Maltseva, D.; Khaustova, N.; Samatov, T.; Tonevitsky, A.; Mahner, S.; et al. c-FOS suppresses ovarian cancer progression by changing adhesion. Br. J. Cancer 2013, 110, 753–763. [Google Scholar] [CrossRef] [Green Version]
- Anuj; Arivazhagan, L.; Venkatraman, G.; Rayala, S.K. Increased Expression of MicroRNA 551a by c-Fos Reduces Focal Adhesion Kinase Levels and Blocks Tumorigenesis. Mol. Cell. Biol. 2019, 39. [Google Scholar] [CrossRef] [Green Version]
- Vydra, N.; Toma-Jonik, A.; Głowala-Kosińska, M.; Gogler-Piglowska, A.; Widlak, W. Overexpression of heat shock transcription factor 1 enhances the resistance of melanoma cells to doxorubicin and paclitaxel. BMC Cancer 2013, 13, 504. [Google Scholar] [CrossRef] [Green Version]
- Kim, S.-H.; Yeo, G.-S.; Lim, Y.-S.; Kang, C.-D.; Kim, C.-M.; Chung, B.-S. Suppression of multidrug resistance via inhibition of heat shock factor by quercetin in MDR cells. Exp. Mol. Med. 1998, 30, 87–92. [Google Scholar] [CrossRef]
- Shevtsov, M.; Multhoff, G.; Mikhaylova, E.; Shibata, A.; Guzhova, I.; Margulis, B. Combination of Anti-Cancer Drugs with Molecular Chaperone Inhibitors. Int. J. Mol. Sci. 2019, 20, 5284. [Google Scholar] [CrossRef] [Green Version]
- Shevtsov, M.; Balogi, Z.; Khachatryan, W.; Gao, H.; Vígh, L.; Multhoff, G. Membrane-Associated Heat Shock Proteins in Oncology: From Basic Research to New Theranostic Targets. Cells 2020, 9, 1263. [Google Scholar] [CrossRef]
- Sampson, J.; O’Regan, L.; Dyer, M.J.S.; Bayliss, R.; Fry, A.M. Hsp72 and Nek6 Cooperate to Cluster Amplified Centrosomes in Cancer Cells. Cancer Res. 2017, 77, 4785–4796. [Google Scholar] [CrossRef] [Green Version]
- Jagadish, N.; Agarwal, S.; Gupta, N.; Fatima, R.; Devi, S.; Kumar, V.; Suri, A.; Kumar, R.; Suri, V.; Sadasukhi, T.C.; et al. Heat shock protein 70-2 (HSP70-2) overexpression in breast cancer. J. Exp. Clin. Cancer Res. 2016, 35, 1–14. [Google Scholar] [CrossRef] [Green Version]
- Sun, G.; Cao, Y.; Xu, Y.; Huai, D.; Chen, P.; Guo, J.; Li, M.; Dai, Y. Overexpression of Hsc70 promotes proliferation, migration, and invasion of human glioma cells. J. Cell. Biochem. 2019, 120, 10707–10714. [Google Scholar] [CrossRef]
- Wu, J.; Liu, T.; Rios, Z.; Mei, Q.; Lin, X.; Cao, S. Heat Shock Proteins and Cancer. Trends Pharmacol. Sci. 2017, 38, 226–256. [Google Scholar] [CrossRef] [PubMed]
- Jego, G.; Hazoumé, A.; Eseigneuric, R.; Garrido, C. Targeting heat shock proteins in cancer. Cancer Lett. 2013, 332, 275–285. [Google Scholar] [CrossRef]
- Yun, C.W.; Kim, H.J.; Lim, J.H.; Lee, S.H. Heat Shock Proteins: Agents of Cancer Development and Therapeutic Targets in Anti-Cancer Therapy. Cells 2019, 9, 60. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- He, N.; Zhang, L.; Cui, J.; Li, Z. Bone marrow vascular niche: Home for hematopoietic stem cells. Bone Marrow Res. 2014, 2014, 128436. [Google Scholar] [CrossRef] [Green Version]
- Kim, R.H.; Chen, W.; Hu, S.; Shin, K.-H.; Park, N.-H.; Kang, M.K.; Kim, R.H.; Kim, R. Association of hsp90 to the hTERT promoter is necessary for hTERT expression in human oral cancer cells. J. Carcinog. 2008, 29, 2425–2431. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bai, J.; Zhou, G.; Qiu, Y.; Hu, Y.; Liu, J.; Zhao, J.; Zhang, S.; Zhang, J. HSP90 inhibitor AUY922 can reverse Fulvestrant induced feedback reaction in human breast cancer cells. Cancer Sci. 2017, 108, 1177–1184. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Tien, A.H.; Sadar, M.D. Keys to unlock androgen receptor translocation. J. Biol. Chem. 2019, 294, 8711–8712. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kostrzewa-Nowak, D.; Paine, M.J.I.; Wolf, C.R.; Tarasiuk, J. The role of bioreductive activation of doxorubicin in cytotoxic activity against leukaemia HL60-sensitive cell line and its multidrug-resistant sublines. Br. J. Cancer 2005, 93, 89–97. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Moghimi, S.M.; Symonds, P.; Murray, J.C.; Hunter, A.C.; Debska, G.; Szewczyk, A. A two-stage poly(ethylenimine)-mediated cytotoxicity: Implications for gene transfer/therapy. Mol. Ther. 2005, 11, 990–995. [Google Scholar] [CrossRef] [PubMed]
- Khansarizadeh, M.; Mokhtarzadeh, A.; Rashedinia, M.; Taghdisi, S.M.; Lari, P.; Abnous, K.H.; Ramezani, M. Identification of possible cytotoxicity mechanism of polyethylenimine by proteomics analysis. Hum. Exp. Toxicol. 2015, 35, 377–387. [Google Scholar] [CrossRef] [PubMed]
- Roy, S.; Sarkar, A.; Jaiswal, A. Poly(allylamine hydrochloride)-functionalized reduced graphene oxide for synergistic chemo-photothermal therapy. Nanomedicine 2019, 14, 255–274. [Google Scholar] [CrossRef] [PubMed]


| Symbol | Primer Sequence | Amplification Conditions | PCR Product Size (bp) | NCBI Reference Sequence |
|---|---|---|---|---|
| p53 | F 5′ GTTGGCTCTGACTGTACCAC 3′ R 5′ AGGGTGAAATATTCTCCATC 3′ | 93 °C, 20 s, 57 °C, 20 s, 72 °C 30 s | 317 | NM_001243976.1 |
| topo2-α | F 5′ GGGAGACTCAGCCAAAACAC 3′ R 5′ CAGCATCATCTTCAGGTCCA 3′ | 93 °C, 20 s, 58 °C, 20 s, 72 °C 30 s | 560 | NM_001246738.1 |
| mdr1 | F 5′ ATCGACGGTCAGGACATCAG 3′ R 5′ TTCAGCGATAGTGGTGGCAA 3′ | 93 °C, 20 s, 60 °C, 20 s, 72 °C 30 s | 102 | XM_027439202.1 |
| c-fos | F 5′ GCAGCCAAATGCTGGAATCG 3′ R 5′ CCAGTGATGTCTTGGGCTCA 3′ | 93 °C, 20 s, 60 °C, 20 s, 72 °C 30 s | 310 | NM_001246683.1 |
| grp78 | F 5′ GATGCGGCCAAGAACCAGCT 3′ R 5′ CGCATGACATTCAGTCCAGC 3′ | 93 °C, 20 s, 63 °C, 20 s, 72 °C 30 s | 359 | NM_001246739.2 |
| hsp90 | F 5′ AATCGGAAGAAGCTTTCAGA 3′ R 5′ GTGCTTGTGACAATACAGCA 3′ | 93 °C, 20 s,56 °C, 20 s, 72 °C 30 s | 257 | NM_001246821.1 |
| hsc70 | F 5′ ATCCCCAAGATTCAGAAGCT 3′ R 5′ TTGATGAGGACAGTCATGAC 3′ | 93 °C, 20 s, 56 °C, 20 s, 72 °C 30 s | 218 | NM_001246729.1 |
| actin | F 5′ GCTGAGAGGGAAATTGTGCGTG 3′ R 5′ CGGTGGACGATGGAGGGGCCG 3′ | 93 °C, 20 s, 68 °C, 20 s, 72 °C 30 s | 506 | XM_007648665.3 |
| Variability of Chromosome Copy Number | Cell Number with Modified Chromosome Structure, % | ||||||
|---|---|---|---|---|---|---|---|
| Cells | Chromosome Variability | Modal Class | Breaks with Preservation of Genet Material | Deletions | Translocations | HSRs Presence | |
| RJK | 16-23 | 17-19 | 0-2 | 16 | 22 | 11 | 0 |
| RJK-PAA | 15-32 | Not evident | 0-5 | 11 | 50 | 17 | 0 |
| RJKEB | 18-32 | 18 | 0-3 | 5 | 20 | 10 | 100 |
| RJKEB-PAA | 15-21 | 18-20 | 0-3 | 33 | 55 | 0 | 100 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Alekseenko, L.; Shilina, M.; Kozhukharova, I.; Lyublinskaya, O.; Fridlyanskaya, I.; Nikolsky, N.; Grinchuk, T. Impact of Polyallylamine Hydrochloride on Gene Expression and Karyotypic Stability of Multidrug Resistant Transformed Cells. Cells 2020, 9, 2332. https://doi.org/10.3390/cells9102332
Alekseenko L, Shilina M, Kozhukharova I, Lyublinskaya O, Fridlyanskaya I, Nikolsky N, Grinchuk T. Impact of Polyallylamine Hydrochloride on Gene Expression and Karyotypic Stability of Multidrug Resistant Transformed Cells. Cells. 2020; 9(10):2332. https://doi.org/10.3390/cells9102332
Chicago/Turabian StyleAlekseenko, Larisa, Mariia Shilina, Irina Kozhukharova, Olga Lyublinskaya, Irina Fridlyanskaya, Nikolay Nikolsky, and Tatiana Grinchuk. 2020. "Impact of Polyallylamine Hydrochloride on Gene Expression and Karyotypic Stability of Multidrug Resistant Transformed Cells" Cells 9, no. 10: 2332. https://doi.org/10.3390/cells9102332





