Rebuilding Tendons: A Concise Review on the Potential of Dermal Fibroblasts
Abstract
1. Introduction: Tendon Injuries and Repair Strategies
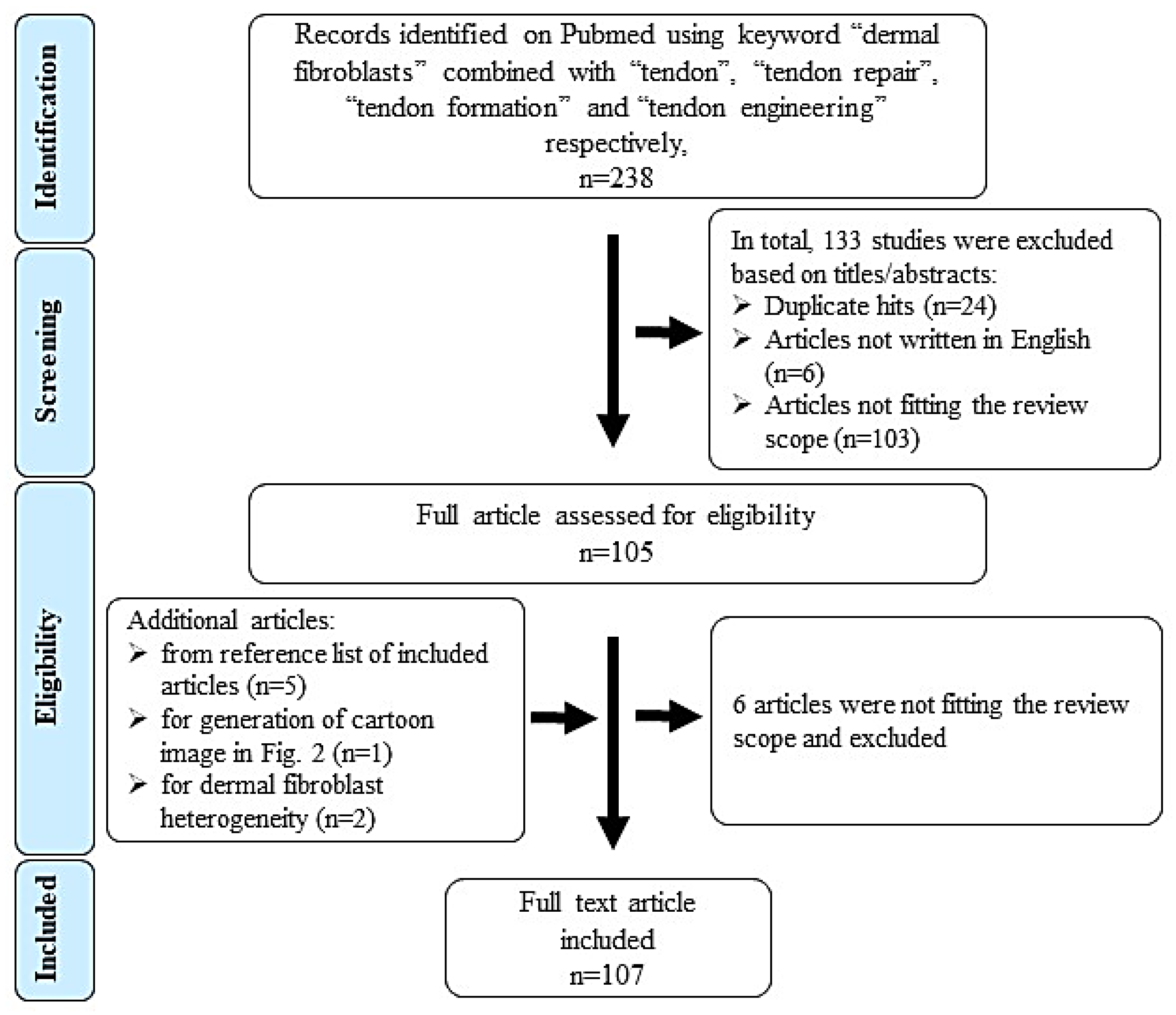
2. Review Scope and Literature Search Method
3. Comparison of Tissue Structure and Composition of Tendon and Dermis
4. Molecular Similarities between DFs and TCs: Analysis Based on Open-Source Databank
5. Differentiation Potential of DFs
5.1. Differentiation Potential of DFs Towards Other Cell Types
5.2. Tenogenic Potential of DFs
6. In Vitro Studies
6.1. Molecular Factors
6.2. Mechanical Factors
6.3. Cultivation Environment
7. In Vivo Studies
8. Clinical Applications of Autologous DFs in Tendon Repair
9. Conclusions and Outlook
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Lemme, N.J.; Li, N.Y.; DeFroda, S.F.; Kleiner, J.; Owens, B.D. Epidemiology of Achilles Tendon Ruptures in the United States: Athletic and Nonathletic Injuries From 2012 to 2016. Orthop. J. Sports Med. 2018, 6, 2325967118808238. [Google Scholar] [CrossRef] [PubMed]
- de Jong, J.P.; Nguyen, J.T.; Sonnema, A.J.; Nguyen, E.C.; Amadio, P.C.; Moran, S.L. The incidence of acute traumatic tendon injuries in the hand and wrist: A 10-year population-based study. Clin. Orthop. Surg. 2014, 6, 196–202. [Google Scholar] [CrossRef] [PubMed]
- Sanders, T.L.; Maradit Kremers, H.M.; Bryan, A.J.; Larson, D.R.; Dahm, D.L.; Levy, B.A.; Stuart, M.J.; Krych, A.J. Incidence of Anterior Cruciate Ligament Tears and Reconstruction: A 21-Year Population-Based Study. Am. J. Sports Med. 2016, 44, 1502–1507. [Google Scholar] [CrossRef] [PubMed]
- Holm, C.; Kjaer, M.; Eliasson, P. Achilles tendon rupture-treatment and complications: A systematic review. Scand. J. Med. Sci. Sports 2015, 25, e1–e10. [Google Scholar] [CrossRef]
- Deng, S.; Sun, Z.; Zhang, C.; Chen, G.; Li, J. Surgical Treatment Versus Conservative Management for Acute Achilles Tendon Rupture: A Systematic Review and Meta-Analysis of Randomized Controlled Trials. J. Foot Ankle Surg. 2017, 56, 1236–1243. [Google Scholar] [CrossRef]
- Bitar, A.C.; Demange, M.K.; D’Elia, C.O.; Camanho, G.L. Traumatic patellar dislocation: Nonoperative treatment compared with MPFL reconstruction using patellar tendon. Am. J. Sports Med. 2012, 40, 114–122. [Google Scholar] [CrossRef]
- Chechik, O.; Dolkart, O.; Mozes, G.; Rak, O.; Alhajajra, F.; Maman, E. Timing matters: NSAIDs interfere with the late proliferation stage of a repaired rotator cuff tendon healing in rats. Arch. Orthop. Trauma Surg. 2014, 134, 515–520. [Google Scholar] [CrossRef]
- Doral, M.N.; Bozkurt, M.; Turhan, E.; Dönmez, G.; Demirel, M.; Kaya, D.; Ateşok, K.; Atay, O.A.; Maffulli, N. Achilles tendon rupture: Physiotherapy and endoscopy-assisted surgical treatment of a common sports injury. Open Access J. Sports Med. 2010, 1, 233–240. [Google Scholar] [CrossRef]
- Wu, Y.; Lin, L.; Li, H.; Zhao, Y.; Liu, L.; Jia, Z.; Wang, D.; He, Q.; Ruan, D. Is surgical intervention more effective than non-surgical treatment for acute Achilles tendon rupture? A systematic review of overlapping meta-analyses. Int. J. Surg. 2016, 36, 305–311. [Google Scholar] [CrossRef] [PubMed]
- Ochen, Y.; Beks, R.B.; van Heijl, M.; Hietbrink, F.; Leenen, L.; van der Velde, D.; Heng, M.; van der Meijden, O.; Groenwold, R.; Houwert, R.M. Operative treatment versus nonoperative treatment of Achilles tendon ruptures: Systematic review and meta-analysis. BMJ 2019, 364, k5120. [Google Scholar] [CrossRef] [PubMed]
- Rensing, N.; Waterman, B.R.; Frank, R.M.; Heida, K.A.; Orr, J.D. Low Risk for Local and Systemic Complications After Primary Repair of 1626 Achilles Tendon Ruptures. Foot Ankle Spec. 2017, 10, 216–226. [Google Scholar] [CrossRef]
- Glazebrook, M.; Rubinger, D. Functional Rehabilitation for Nonsurgical Treatment of Acute Achilles Tendon Rupture. Foot Ankle Clin. 2019, 24, 387–398. [Google Scholar] [CrossRef] [PubMed]
- Kauwe, M. Acute Achilles Tendon Rupture: Clinical Evaluation, Conservative Management, and Early Active Rehabilitation. Clin. Podiatr. Med. Surg. 2017, 34, 229–243. [Google Scholar] [CrossRef] [PubMed]
- Gaspar, D.; Spanoudes, K.; Holladay, C.; Pandit, A.; Zeugolis, D. Progress in cell-based therapies for tendon repair. Adv. Drug Deliv. Rev. 2015, 84, 240–256. [Google Scholar] [CrossRef] [PubMed]
- De Deyne, P.; Haut, R.C. Some effects of gamma irradiation on patellar tendon allografts. Connect. Tissue Res. 1991, 27, 51–62. [Google Scholar] [CrossRef]
- Roe, S.C.; Milthorpe, B.K.; True, K.; Rogers, G.J.; Schindhelm, K. The effect of gamma irradiation on a xenograft tendon bioprosthesis. Clin. Mater. 1992, 9, 149–154. [Google Scholar] [CrossRef]
- Milthorpe, B.K. Xenografts for tendon and ligament repair. Biomaterials 1994, 15, 745–752. [Google Scholar] [CrossRef]
- Nakamura, S.; Katsuki, M. Tendon grafting for multiple extensor tendon ruptures of fingers in rheumatoid hands. J. Hand Surg. Br. 2002, 27, 326–328. [Google Scholar] [CrossRef]
- Schneider, M.; Angele, P.; Järvinen, T.; Docheva, D. Rescue plan for Achilles: Therapeutics steering the fate and functions of stem cells in tendon wound healing. Adv. Drug Deliv. Rev. 2018, 129, 352–375. [Google Scholar] [CrossRef]
- Docheva, D.; Müller, S.A.; Majewski, M.; Evans, C.H. Biologics for tendon repair. Adv. Drug Deliv. Rev. 2015, 84, 222–239. [Google Scholar] [CrossRef]
- Yan, Z.; Yin, H.; Nerlich, M.; Pfeifer, C.G.; Docheva, D. Boosting tendon repair: Interplay of cells, growth factors and scaffold-free and gel-based carriers. J. Exp. Orthop. 2018, 5, 1. [Google Scholar] [CrossRef] [PubMed]
- Chen, Z.; Wang, Y.; Shi, C. Therapeutic Implications of Newly Identified Stem Cell Populations from the Skin Dermis. Cell Transplant. 2015, 24, 1405–1422. [Google Scholar] [CrossRef] [PubMed]
- Arda, O.; Göksügür, N.; Tüzün, Y. Basic histological structure and functions of facial skin. Clin. Dermatol. 2014, 32, 3–13. [Google Scholar] [CrossRef] [PubMed]
- Tempfer, H.; Traweger, A. Tendon Vasculature in Health and Disease. Front. Physiol. 2015, 6, 330. [Google Scholar] [CrossRef]
- Sharma, P.; Maffulli, N. Tendinopathy and tendon injury: The future. Disabil. Rehabil. 2008, 30, 1733–1745. [Google Scholar] [CrossRef]
- James, R.; Kesturu, G.; Balian, G.; Chhabra, A.B. Tendon: Biology, biomechanics, repair, growth factors, and evolving treatment options. J. Hand Surg. Am. 2008, 33, 102–112. [Google Scholar] [CrossRef]
- Hulmes, D.J. Building collagen molecules, fibrils, and suprafibrillar structures. J. Struct. Biol. 2002, 137, 2–10. [Google Scholar] [CrossRef]
- Nourissat, G.; Berenbaum, F.; Duprez, D. Tendon injury: From biology to tendon repair. Nat. Rev. Rheumatol 2015, 11, 223–233. [Google Scholar] [CrossRef]
- Losquadro, W.D. Anatomy of the Skin and the Pathogenesis of Nonmelanoma Skin Cancer. Facial Plast. Surg. Clin. North. Am. 2017, 25, 283–289. [Google Scholar] [CrossRef]
- Bi, Y.; Ehirchiou, D.; Kilts, T.M.; Inkson, C.A.; Embree, M.C.; Sonoyama, W.; Li, L.; Leet, A.I.; Seo, B.M.; Zhang, L.; et al. Identification of tendon stem/progenitor cells and the role of the extracellular matrix in their niche. Nat. Med. 2007, 13, 1219–1227. [Google Scholar] [CrossRef]
- Kohler, J.; Popov, C.; Klotz, B.; Alberton, P.; Prall, W.C.; Haasters, F.; Müller-Deubert, S.; Ebert, R.; Klein-Hitpass, L.; Jakob, F.; et al. Uncovering the cellular and molecular changes in tendon stem/progenitor cells attributed to tendon aging and degeneration. Aging Cell 2013, 12, 988–999. [Google Scholar] [CrossRef]
- Tempfer, H.; Wagner, A.; Gehwolf, R.; Lehner, C.; Tauber, M.; Resch, H.; Bauer, H.C. Perivascular cells of the supraspinatus tendon express both tendon- and stem cell-related markers. Histochem. Cell Biol. 2009, 131, 733–741. [Google Scholar] [CrossRef] [PubMed]
- Woodley, D.T. Distinct Fibroblasts in the Papillary and Reticular Dermis: Implications for Wound Healing. Derm. Clin. 2017, 35, 95–100. [Google Scholar] [CrossRef]
- Sassolas, B. [Anatomy of normal skin]. Soins Rev. Ref. Infirm. 2010, 28. [Google Scholar]
- Korosec, A.; Frech, S.; Lichtenberger, B.M. Isolation of Papillary and Reticular Fibroblasts from Human Skin by Fluorescence-activated Cell Sorting. J. Vis. Exp. 2019, 147, e59372. [Google Scholar] [CrossRef]
- Rinn, J.L.; Bondre, C.; Gladstone, H.B.; Brown, P.O.; Chang, H.Y. Anatomic demarcation by positional variation in fibroblast gene expression programs. PLoS Genet. 2006, 2, e119. [Google Scholar] [CrossRef]
- Mackley, J.R.; Ando, J.; Herzyk, P.; Winder, S.J. Phenotypic responses to mechanical stress in fibroblasts from tendon, cornea and skin. Biochem. J. 2006, 396, 307–316. [Google Scholar] [CrossRef]
- Novak, D.; Weina, K.; Utikal, J. From skin to other cell types of the body. J. Dtsch. Derm. Ges. 2014, 12, 789–792. [Google Scholar] [CrossRef]
- Vierbuchen, T.; Wernig, M. Direct lineage conversions: Unnatural but useful. Nat. Biotechnol. 2011, 29, 892–907. [Google Scholar] [CrossRef]
- Vierbuchen, T.; Ostermeier, A.; Pang, Z.P.; Kokubu, Y.; Südhof, T.C.; Wernig, M. Direct conversion of fibroblasts to functional neurons by defined factors. Nature 2010, 463, 1035–1041. [Google Scholar] [CrossRef]
- Pang, Z.P.; Yang, N.; Vierbuchen, T.; Ostermeier, A.; Fuentes, D.R.; Yang, T.Q.; Citri, A.; Sebastiano, V.; Marro, S.; Südhof, T.C.; et al. Induction of human neuronal cells by defined transcription factors. Nature 2011, 476, 220–223. [Google Scholar] [CrossRef]
- Avaliani, N.; Pfisterer, U.; Heuer, A.; Parmar, M.; Kokaia, M.; Andersson, M. Directly Converted Human Fibroblasts Mature to Neurons and Show Long-Term Survival in Adult Rodent Hippocampus. Stem Cells Int. 2017, 2017, 5718608. [Google Scholar] [CrossRef]
- Pfisterer, U.; Kirkeby, A.; Torper, O.; Wood, J.; Nelander, J.; Dufour, A.; Björklund, A.; Lindvall, O.; Jakobsson, J.; Parmar, M. Direct conversion of human fibroblasts to dopaminergic neurons. Proc. Natl. Acad. Sci. USA 2011, 108, 10343–10348. [Google Scholar] [CrossRef]
- Nam, Y.J.; Song, K.; Luo, X.; Daniel, E.; Lambeth, K.; West, K.; Hill, J.A.; DiMaio, J.M.; Baker, L.A.; Bassel-Duby, R.; et al. Reprogramming of human fibroblasts toward a cardiac fate. Proc. Natl. Acad. Sci. USA 2013, 110, 5588–5593. [Google Scholar] [CrossRef]
- Steens, J.; Unger, K.; Klar, L.; Neureiter, A.; Wieber, K.; Hess, J.; Jakob, H.G.; Klump, H.; Klein, D. Direct conversion of human fibroblasts into therapeutically active vascular wall-typical mesenchymal stem cells. Cell. Mol. Life Sci. 2020, 77, 3401–3422. [Google Scholar] [CrossRef]
- Kitada, M.; Murakami, T.; Wakao, S.; Li, G.; Dezawa, M. Direct conversion of adult human skin fibroblasts into functional Schwann cells that achieve robust recovery of the severed peripheral nerve in rats. Glia 2019, 67, 950–966. [Google Scholar] [CrossRef]
- Szabo, E.; Rampalli, S.; Risueño, R.M.; Schnerch, A.; Mitchell, R.; Fiebig-Comyn, A.; Levadoux-Martin, M.; Bhatia, M. Direct conversion of human fibroblasts to multilineage blood progenitors. Nature 2010, 468, 521–526. [Google Scholar] [CrossRef]
- Hou, Y.P.; Zhang, Z.Y.; Xing, X.Y.; Zhou, J.; Sun, J. Direct conversion of human fibroblasts into functional Leydig-like cells by SF-1, GATA4 and NGFI-B. Am. J. Transl. Res. 2018, 10, 175–183. [Google Scholar]
- Zhang, K.; Liu, G.H.; Yi, F.; Montserrat, N.; Hishida, T.; Esteban, C.R.; Belmonte, J.I. Direct conversion of human fibroblasts into retinal pigment epithelium-like cells by defined factors. Protein Cell 2014, 5, 48–58. [Google Scholar] [CrossRef][Green Version]
- Wang, W.; He, A.; Zhang, Z.; Zhang, W.; Zhou, G.; Cao, Y.; Liu, W. Induction of transient tenogenic phenotype of high-density cultured human dermal fibroblasts. Connect. Tissue Res. 2015, 56, 288–299. [Google Scholar] [CrossRef]
- Lin, D.; Alberton, P.; Caceres, M.D.; Volkmer, E.; Schieker, M.; Docheva, D. Tenomodulin is essential for prevention of adipocyte accumulation and fibrovascular scar formation during early tendon healing. Cell Death Dis. 2017, 8, e3116. [Google Scholar] [CrossRef] [PubMed]
- Shukunami, C.; Oshima, Y.; Hiraki, Y. Molecular cloning of tenomodulin, a novel chondromodulin-I related gene. Biochem. Biophys. Res. Commun. 2001, 280, 1323–1327. [Google Scholar] [CrossRef]
- Brandau, O.; Meindl, A.; Fässler, R.; Aszódi, A. A novel gene, tendin, is strongly expressed in tendons and ligaments and shows high homology with chondromodulin-I. Dev. Dyn. 2001, 221, 72–80. [Google Scholar] [CrossRef]
- Docheva, D.; Hunziker, E.B.; Fässler, R.; Brandau, O. Tenomodulin is necessary for tenocyte proliferation and tendon maturation. Mol. Cell. Biol. 2005, 25, 699–705. [Google Scholar] [CrossRef]
- Jiang, Y.; Shi, Y.; He, J.; Zhang, Z.; Zhou, G.; Zhang, W.; Cao, Y.; Liu, W. Enhanced tenogenic differentiation and tendon-like tissue formation by tenomodulin overexpression in murine mesenchymal stem cells. J. Tissue Eng. Regen. Med. 2017, 11, 2525–2536. [Google Scholar] [CrossRef]
- Shi, Y.; Xiong, Y.; Jiang, Y.; Zhang, Z.; Zhou, G.; Zhang, W.; Cao, Y.; He, J.; Liu, W. Conditional tenomodulin overexpression favors tenogenic lineage differentiation of transgenic mouse derived cells. Gene 2017, 598, 9–19. [Google Scholar] [CrossRef]
- Tischer, T.; Vogt, S.; Aryee, S.; Steinhauser, E.; Adamczyk, C.; Milz, S.; Martinek, V.; Imhoff, A.B. Tissue engineering of the anterior cruciate ligament: A new method using acellularized tendon allografts and autologous fibroblasts. Arch. Orthop. Trauma Surg. 2007, 127, 735–741. [Google Scholar] [CrossRef]
- Deng, D.; Liu, W.; Xu, F.; Yang, Y.; Zhou, G.; Zhang, W.J.; Cui, L.; Cao, Y. Engineering human neo-tendon tissue in vitro with human dermal fibroblasts under static mechanical strain. Biomaterials 2009, 30, 6724–6730. [Google Scholar] [CrossRef]
- Brink, H.E.; Bernstein, J.; Nicoll, S.B. Fetal dermal fibroblasts exhibit enhanced growth and collagen production in two- and three-dimensional culture in comparison to adult fibroblasts. J. Tissue Eng. Regen. Med. 2009, 3, 623–633. [Google Scholar] [CrossRef]
- Jiang, D.; Xu, B.; Yang, M.; Zhao, Z.; Zhang, Y.; Li, Z. Efficacy of tendon stem cells in fibroblast-derived matrix for tendon tissue engineering. Cytotherapy 2014, 16, 662–673. [Google Scholar] [CrossRef]
- Wang, W.; Li, J.; Wang, K.; Zhang, Z.; Zhang, W.; Zhou, G.; Cao, Y.; Ye, M.; Zou, H.; Liu, W. Induction of predominant tenogenic phenotype in human dermal fibroblasts via synergistic effect of TGF-β and elongated cell shape. Am. J. Physiol., Cell Physiol. 2016, 310, 357–372. [Google Scholar] [CrossRef]
- Chattopadhyay, A.; Galvez, M.G.; Bachmann, M.; Legrand, A.; McGoldrick, R.; Lovell, A.; Jacobs, M.; Crowe, C.; Umansky, E.; Chang, J. Tendon Regeneration with Tendon Hydrogel-Based Cell Delivery: A Comparison of Fibroblasts and Adipose-Derived Stem Cells. Plast. Reconstr. Surg. 2016, 138, 617–626. [Google Scholar] [CrossRef]
- Wang, W.; He, J.; Feng, B.; Zhang, Z.; Zhang, W.; Zhou, G.; Cao, Y.; Fu, W.; Liu, W. Aligned nanofibers direct human dermal fibroblasts to tenogenic phenotype in vitro and enhance tendon regeneration in vivo. Nanomedicine (Lond.) 2016, 11, 1055–1072. [Google Scholar] [CrossRef]
- Di Giancamillo, A.; Deponti, D.; Raimondi, M.T.; Boschetti, F.; Gervaso, F.; Modina, S.; Mangiavini, L.; Peretti, G.M. Comparison between different cell sources and culture strategies for tendon tissue engineering. J. Biol. Regul. Homeost. Agents 2017, 31, 61–66. [Google Scholar]
- Chen, C.H.; Chen, S.H.; Kuo, C.Y.; Li, M.L.; Chen, J.P. Response of Dermal Fibroblasts to Biochemical and Physical Cues in Aligned Polycaprolactone/Silk Fibroin Nanofiber Scaffolds for Application in Tendon Tissue Engineering. Nanomaterials (Basel) 2017, 7, 219. [Google Scholar] [CrossRef]
- Nakanishi, Y.; Okada, T.; Takeuchi, N.; Kozono, N.; Senju, T.; Nakayama, K.; Nakashima, Y. Histological evaluation of tendon formation using a scaffold-free three-dimensional-bioprinted construct of human dermal fibroblasts under in vitro static tensile culture. Regen. Ther. 2019, 11, 47–55. [Google Scholar] [CrossRef]
- Gaspar, D.; Ryan, C.; Zeugolis, D.I. Multifactorial bottom-up bioengineering approaches for the development of living tissue substitutes. FASEB J. 2019, 33, 5741–5754. [Google Scholar] [CrossRef]
- Pascher, A.; Steinert, A.F.; Palmer, G.D.; Betz, O.; Gouze, J.N.; Gouze, E.; Pilapil, C.; Ghivizzani, S.C.; Evans, C.H.; Murray, M.M. Enhanced repair of the anterior cruciate ligament by in situ gene transfer: Evaluation in an in vitro model. Mol. Ther. 2004, 10, 327–336. [Google Scholar] [CrossRef]
- Juneja, S.C.; Schwarz, E.M.; O’Keefe, R.J.; Awad, H.A. Cellular and molecular factors in flexor tendon repair and adhesions: A histological and gene expression analysis. Connect. Tissue Res. 2013, 54, 218–226. [Google Scholar] [CrossRef]
- Jørgensen, H.G.; McLellan, S.D.; Crossan, J.F.; Curtis, A.S. Neutralisation of TGF beta or binding of VLA-4 to fibronectin prevents rat tendon adhesion following transection. Cytokine 2005, 30, 195–202. [Google Scholar] [CrossRef]
- Katzel, E.B.; Wolenski, M.; Loiselle, A.E.; Basile, P.; Flick, L.M.; Langstein, H.N.; Hilton, M.J.; Awad, H.A.; Hammert, W.C.; O’Keefe, R.J. Impact of Smad3 loss of function on scarring and adhesion formation during tendon healing. J. Orthop. Res. 2011, 29, 684–693. [Google Scholar] [CrossRef] [PubMed]
- Lichtman, M.K.; Otero-Vinas, M.; Falanga, V. Transforming growth factor beta (TGF-β) isoforms in wound healing and fibrosis. Wound Repair Regen. 2016, 24, 215–222. [Google Scholar] [CrossRef] [PubMed]
- Fang, X.; Hu, X.; Zheng, Z.; Tao, K.; Wang, H.; Guan, H.; Shi, J.; Ji, P.; Cai, W.; Bai, X.; et al. Smad interacting protein 1 influences transforming growth factor-β1/Smad signaling in extracellular matrix protein production and hypertrophic scar formation. J. Mol. Histol. 2019, 50, 503–514. [Google Scholar] [CrossRef] [PubMed]
- Koyanagi, M.; Takahashi, J.; Arakawa, Y.; Doi, D.; Fukuda, H.; Hayashi, H.; Narumiya, S.; Hashimoto, N. Inhibition of the Rho/ROCK pathway reduces apoptosis during transplantation of embryonic stem cell-derived neural precursors. J. Neurosci. Res. 2008, 86, 270–280. [Google Scholar] [CrossRef]
- Deng, Z.; Jia, Y.; Liu, H.; He, M.; Yang, Y.; Xiao, W.; Li, Y. RhoA/ROCK pathway: Implication in osteoarthritis and therapeutic targets. Am. J. Transl. Res. 2019, 11, 5324–5331. [Google Scholar]
- Xu, B.; Song, G.; Ju, Y.; Li, X.; Song, Y.; Watanabe, S. RhoA/ROCK, cytoskeletal dynamics, and focal adhesion kinase are required for mechanical stretch-induced tenogenic differentiation of human mesenchymal stem cells. J. Cell. Physiol. 2012, 227, 2722–2729. [Google Scholar] [CrossRef]
- Maharam, E.; Yaport, M.; Villanueva, N.L.; Akinyibi, T.; Laudier, D.; He, Z.; Leong, D.J.; Sun, H.B. Rho/Rock signal transduction pathway is required for MSC tenogenic differentiation. Bone Res. 2015, 3, 15015. [Google Scholar] [CrossRef]
- Harb, N.; Archer, T.K.; Sato, N. The Rho-Rock-Myosin signaling axis determines cell-cell integrity of self-renewing pluripotent stem cells. PLoS ONE 2008, 3, e3001. [Google Scholar] [CrossRef]
- Thomas, G.P.; el Haj, A.J. Bone marrow stromal cells are load responsive in vitro. Calcif. Tissue Int. 1996, 58, 101–108. [Google Scholar] [CrossRef]
- Bayer, M.L.; Schjerling, P.; Herchenhan, A.; Zeltz, C.; Heinemeier, K.M.; Christensen, L.; Krogsgaard, M.; Gullberg, D.; Kjaer, M. Release of tensile strain on engineered human tendon tissue disturbs cell adhesions, changes matrix architecture, and induces an inflammatory phenotype. PLoS ONE 2014, 9, e86078. [Google Scholar] [CrossRef]
- Youngstrom, D.W.; Barrett, J.G. Engineering Tendon: Scaffolds, Bioreactors, and Models of Regeneration. Stem Cells Int. 2016, 2016, 3919030. [Google Scholar] [CrossRef]
- Lomas, A.J.; Ryan, C.N.; Sorushanova, A.; Shologu, N.; Sideri, A.I.; Tsioli, V.; Fthenakis, G.C.; Tzora, A.; Skoufos, I.; Quinlan, L.R.; et al. The past, present and future in scaffold-based tendon treatments. Adv. Drug Deliv. Rev. 2015, 84, 257–277. [Google Scholar] [CrossRef] [PubMed]
- Friess, W. Collagen--biomaterial for drug delivery. Eur. J. Pharm. Biopharm. 1998, 45, 113–136. [Google Scholar] [CrossRef]
- Kew, S.J.; Gwynne, J.H.; Enea, D.; Abu-Rub, M.; Pandit, A.; Zeugolis, D.; Brooks, R.A.; Rushton, N.; Best, S.M.; Cameron, R.E. Regeneration and repair of tendon and ligament tissue using collagen fibre biomaterials. Acta Biomater. 2011, 7, 3237–3247. [Google Scholar] [CrossRef] [PubMed]
- Juncosa-Melvin, N.; Matlin, K.S.; Holdcraft, R.W.; Nirmalanandhan, V.S.; Butler, D.L. Mechanical stimulation increases collagen type I and collagen type III gene expression of stem cell—Collagen sponge constructs for patellar tendon repair. Tissue Eng. 2007, 13, 1219–1226. [Google Scholar] [CrossRef]
- Caliari, S.R.; Harley, B.A. The effect of anisotropic collagen-GAG scaffolds and growth factor supplementation on tendon cell recruitment, alignment, and metabolic activity. Biomaterials 2011, 32, 5330–5340. [Google Scholar] [CrossRef]
- Caliari, S.R.; Weisgerber, D.W.; Ramirez, M.A.; Kelkhoff, D.O.; Harley, B.A. The influence of collagen-glycosaminoglycan scaffold relative density and microstructural anisotropy on tenocyte bioactivity and transcriptomic stability. J. Mech. Behav. Biomed. Mater. 2012, 11, 27–40. [Google Scholar] [CrossRef]
- Zeugolis, D.I.; Paul, G.R.; Attenburrow, G. Cross-linking of extruded collagen fibers--a biomimetic three-dimensional scaffold for tissue engineering applications. J. Biomed. Mater. Res. A. 2009, 89, 895–908. [Google Scholar] [CrossRef]
- Zeugolis, D.I.; Paul, R.G.; Attenburrow, G. Extruded collagen-polyethylene glycol fibers for tissue engineering applications. J. Biomed. Mater. Res. Part B Appl. Biomater. 2008, 85, 343–352. [Google Scholar] [CrossRef]
- Wang, Q.; Han, G.; Yan, S.; Zhang, Q. 3D Printing of Silk Fibroin for Biomedical Applications. Materials (Basel) 2019, 12, 504. [Google Scholar] [CrossRef]
- Yang, M. Silk-based biomaterials. Microsc. Res. Tech. 2017, 80, 321–330. [Google Scholar] [CrossRef] [PubMed]
- Altman, G.H.; Horan, R.L.; Lu, H.H.; Moreau, J.; Martin, I.; Richmond, J.C.; Kaplan, D.L. Silk matrix for tissue engineered anterior cruciate ligaments. Biomaterials 2002, 23, 4131–4141. [Google Scholar] [CrossRef]
- Tabata, I.I. The importance of drug delivery systems in tissue engineering. Pharm. Sci. Technol. Today 2000, 3, 80–89. [Google Scholar] [CrossRef]
- Wang, W.; Deng, D.; Wang, B.; Zhou, G.; Zhang, W.; Cao, Y.; Zhang, P.; Liu, W. Comparison of Autologous, Allogeneic, and Cell-Free Scaffold Approaches for Engineered Tendon Repair in a Rabbit Model-A Pilot Study. Tissue Eng. Part A 2017, 23, 750–761. [Google Scholar] [CrossRef] [PubMed]
- Hsieh, C.F.; Yan, Z.; Schumann, R.G.; Milz, S.; Pfeifer, C.G.; Schieker, M.; Docheva, D. In Vitro Comparison of 2D-Cell Culture and 3D-Cell Sheets of Scleraxis-Programmed Bone Marrow Derived Mesenchymal Stem Cells to Primary Tendon Stem/Progenitor Cells for Tendon Repair. Int. J. Mol. Sci. 2018, 19, 2272. [Google Scholar] [CrossRef]
- Yan, Z.; Yin, H.; Brochhausen, C.; Pfeifer, C.G.; Alt, V.; Docheva, D. Aged Tendon Stem/Progenitor Cells Are Less Competent to Form 3D Tendon Organoids Due to Cell Autonomous and Matrix Production Deficits. Front. Bioeng. Biotechnol. 2020, 8, 406. [Google Scholar] [CrossRef]
- Sun, H.; Liu, W.; Zhou, G.; Zhang, W.; Cui, L.; Cao, Y. Tissue engineering of cartilage, tendon and bone. Front. Med. 2011, 5, 61–69. [Google Scholar] [CrossRef]
- Bottagisio, M.; Lovati, A.B. A review on animal models and treatments for the reconstruction of Achilles and flexor tendons. J. Mater. Sci. Mater. Med. 2017, 28, 45. [Google Scholar] [CrossRef]
- Hast, M.W.; Zuskov, A.; Soslowsky, L.J. The role of animal models in tendon research. Bone Jt. Res. 2014, 3, 193–202. [Google Scholar] [CrossRef]
- Loiselle, A.E.; Kelly, M.; Hammert, W.C. Biological Augmentation of Flexor Tendon Repair: A Challenging Cellular Landscape. J. Hand Surg. Am. 2016, 41, 144–149. [Google Scholar] [CrossRef]
- He, Q.; Li, Q.; Chen, B.; Wang, Z. Repair of flexor tendon defects of rabbit with tissue engineering method. Chin. J. Traumatol. 2002, 5, 200–208. [Google Scholar]
- Liu, W.; Chen, B.; Deng, D.; Xu, F.; Cui, L.; Cao, Y. Repair of tendon defect with dermal fibroblast engineered tendon in a porcine model. Tissue Eng. 2006, 12, 775–788. [Google Scholar] [CrossRef] [PubMed]
- Chen, B.; Ding, J.; Zhang, W.; Zhou, G.; Cao, Y.; Liu, W.; Wang, B. Tissue Engineering of Tendons: A Comparison of Muscle-Derived Cells, Tenocytes, and Dermal Fibroblasts as Cell Sources. Plast. Reconstr. Surg. 2016, 137, 536–544. [Google Scholar] [CrossRef] [PubMed]
- Kwon, J.; Kim, Y.H.; Rhee, S.M.; Kim, T.I.; Lee, J.; Jeon, S.; Oh, J.H. Effects of Allogenic Dermal Fibroblasts on Rotator Cuff Healing in a Rabbit Model of Chronic Tear. Am. J. Sports Med. 2018, 46, 1901–1908. [Google Scholar] [CrossRef] [PubMed]
- Clarke, A.W.; Alyas, F.; Morris, T.; Robertson, C.J.; Bell, J.; Connell, D.A. Skin-derived tenocyte-like cells for the treatment of patellar tendinopathy. Am. J. Sports Med. 2011, 39, 614–623. [Google Scholar] [CrossRef] [PubMed]
- Connell, D.; Datir, A.; Alyas, F.; Curtis, M. Treatment of lateral epicondylitis using skin-derived tenocyte-like cells. Br. J. Sports Med. 2009, 43, 293–298. [Google Scholar] [CrossRef]
- Young, M. Stem cell applications in tendon disorders: A clinical perspective. Stem Cells Int. 2012, 2012, 637836. [Google Scholar] [CrossRef]


| Inclusion Criteria | Exclusion Criteria |
|---|---|
| Title/abstract | |
|
|
| Full text | |
|
|
| |
| |
| Species | Material of Scaffold | Stimulation Factors | Results | Ref. |
|---|---|---|---|---|
| Mice | - | Static mechanical strain |
| [37] |
| Rabbit | Acellularized tendon | - |
| [57] |
| Human | PGA | Static mechanical strain |
| [58] |
| Rat | PGA | - |
| [59] |
| Rat | dFM | - |
| [60] |
| Human | - | High density culture |
| [50] |
| Human | Grooved silicone membrane | Morphology, exogenous TGF-β1 and Rock inhibitor |
| [61] |
| Human | Native hydrogel | Cell density |
| [62] |
| Human | PCL/gelatin nanofiber | Pattern of nanofibers |
| [63] |
| Pig | Collagen I | Static and dynamic mechanical strain |
| [64] |
| Mice | - | TNMD overexpression |
| [56] |
| Rabbit | PCL/Silk Fibroin Nanofiber | Aligned vs. not-aligned nanofibers |
| [65] |
| Human | BIO-3D printed tissue ring | Static mechanical strain |
| [66] |
| Human | - | MMC and mechanical stimulation |
| [67] |
| Model | Cell Type | Scaffold Type | Implants Classification | Results | Ref. |
|---|---|---|---|---|---|
| Rabbit Achilles tendon defect | Rabbit DFs | HA-ECM | Scaffold with DFs Scaffold alone Just suture |
| [101] |
| Pig flexor tendon defect | Pig DFs | PGA | Scaffold with DFs Scaffold with TCs Scaffold alone |
| [102] |
| Rat patellar tendon injury | TSCs | dFM | TSCs with dFM TSCs alone |
| [60] |
| Rat subcutaneous pocket | DFs, TCs and MDCs | PGA | Scaffold with DFs Scaffold with TCs Scaffold with MDCs |
| [103] |
| Rat subcutaneous pocket | DFs, ADSCs | Tendon tissue derived hydrogel | Scaffold with DFs Scaffold with ADSCs |
| [62] |
| Rabbit Achilles tendon defect | DFs | PGA and PLA composed scaffold | Scaffold with autologous DFs Scaffold with allogeneic DFs Scaffold alone |
| [94] |
| Rabbit Achilles tendon partial defect | Rabbit DFs | PLC/Silk Fibroin nanofiber scaffolds | Acellular RPSF Acellular APSF Cells/RPSF Cells/APSF |
| [65] |
| Rabbit rotator cuff tear | DFs | - | DFs suspension with fibrin Fibrin only Saline only |
| [104] |
| Clinical Diagnosis | Study Design | Interventions | Results | Ref. |
|---|---|---|---|---|
| Refractory lateral epicondylitis | Prospective clinical pilot study. | Autologous cells suspended in autologous plasma. |
| [106] |
| Refractory patellar tendinopathy | Randomizedcontrol trial | Autologous cells suspended in autologous plasma. Autologous plasma alone. |
| [105] |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Chu, J.; Lu, M.; Pfeifer, C.G.; Alt, V.; Docheva, D. Rebuilding Tendons: A Concise Review on the Potential of Dermal Fibroblasts. Cells 2020, 9, 2047. https://doi.org/10.3390/cells9092047
Chu J, Lu M, Pfeifer CG, Alt V, Docheva D. Rebuilding Tendons: A Concise Review on the Potential of Dermal Fibroblasts. Cells. 2020; 9(9):2047. https://doi.org/10.3390/cells9092047
Chicago/Turabian StyleChu, Jin, Ming Lu, Christian G. Pfeifer, Volker Alt, and Denitsa Docheva. 2020. "Rebuilding Tendons: A Concise Review on the Potential of Dermal Fibroblasts" Cells 9, no. 9: 2047. https://doi.org/10.3390/cells9092047
APA StyleChu, J., Lu, M., Pfeifer, C. G., Alt, V., & Docheva, D. (2020). Rebuilding Tendons: A Concise Review on the Potential of Dermal Fibroblasts. Cells, 9(9), 2047. https://doi.org/10.3390/cells9092047








