Absence of W Chromosome in Psychidae Moths and Implications for the Theory of Sex Chromosome Evolution in Lepidoptera
Abstract
:1. Introduction
2. Materials and Methods
2.1. Chromosome and Polyploid Nuclei Preparations
2.2. DNA Extraction
2.3. Comparative Genomic Hybridization
2.4. Microscopy and Image Processing
2.5. Flow Cytometry
3. Results
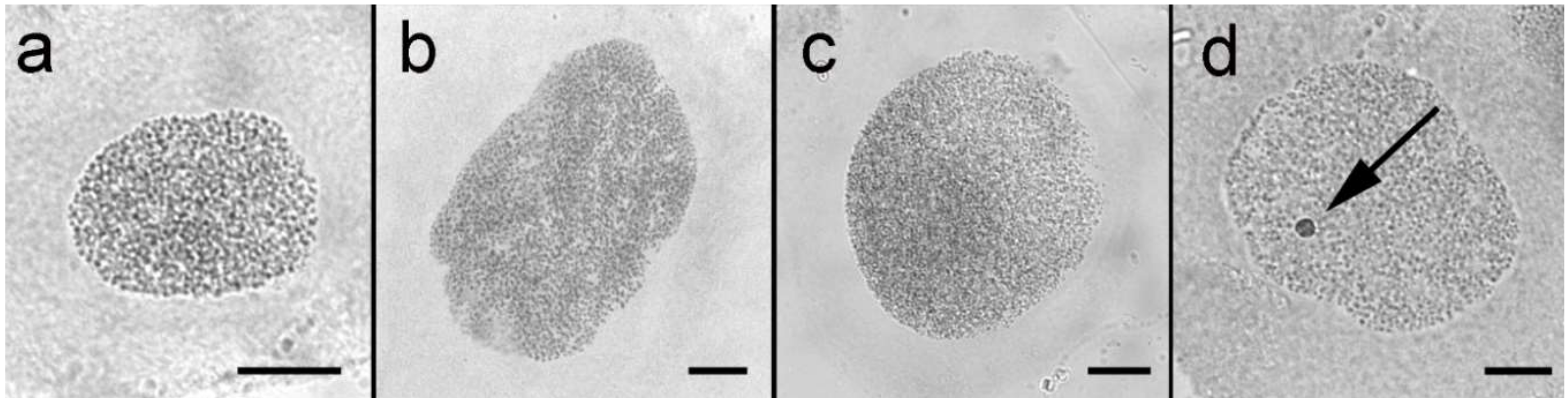
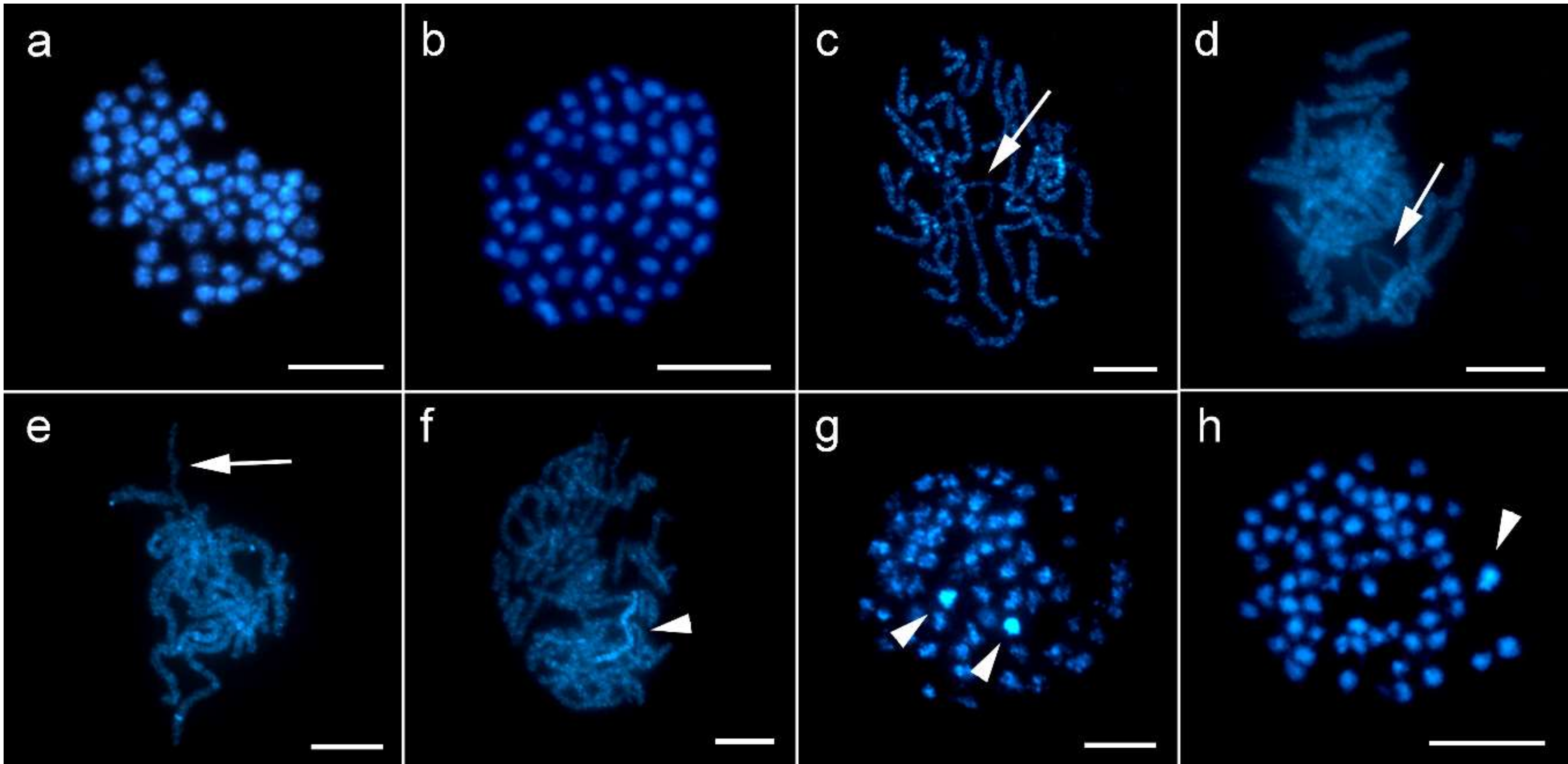
3.1. Sex Chromatin and Chromosome Number
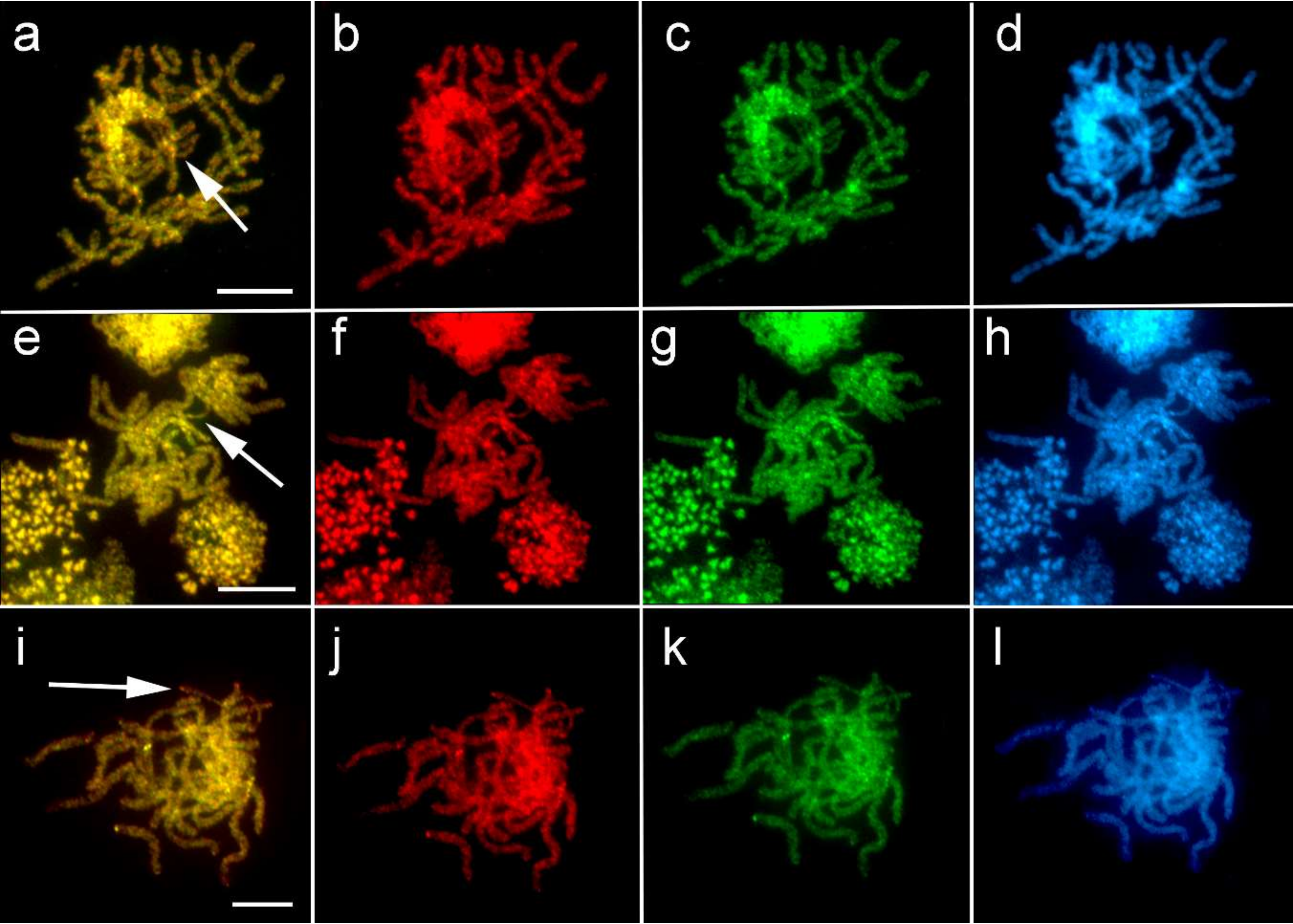
3.2. Comparative Genomic Hybridization (CGH)
3.3. Flow Cytometry
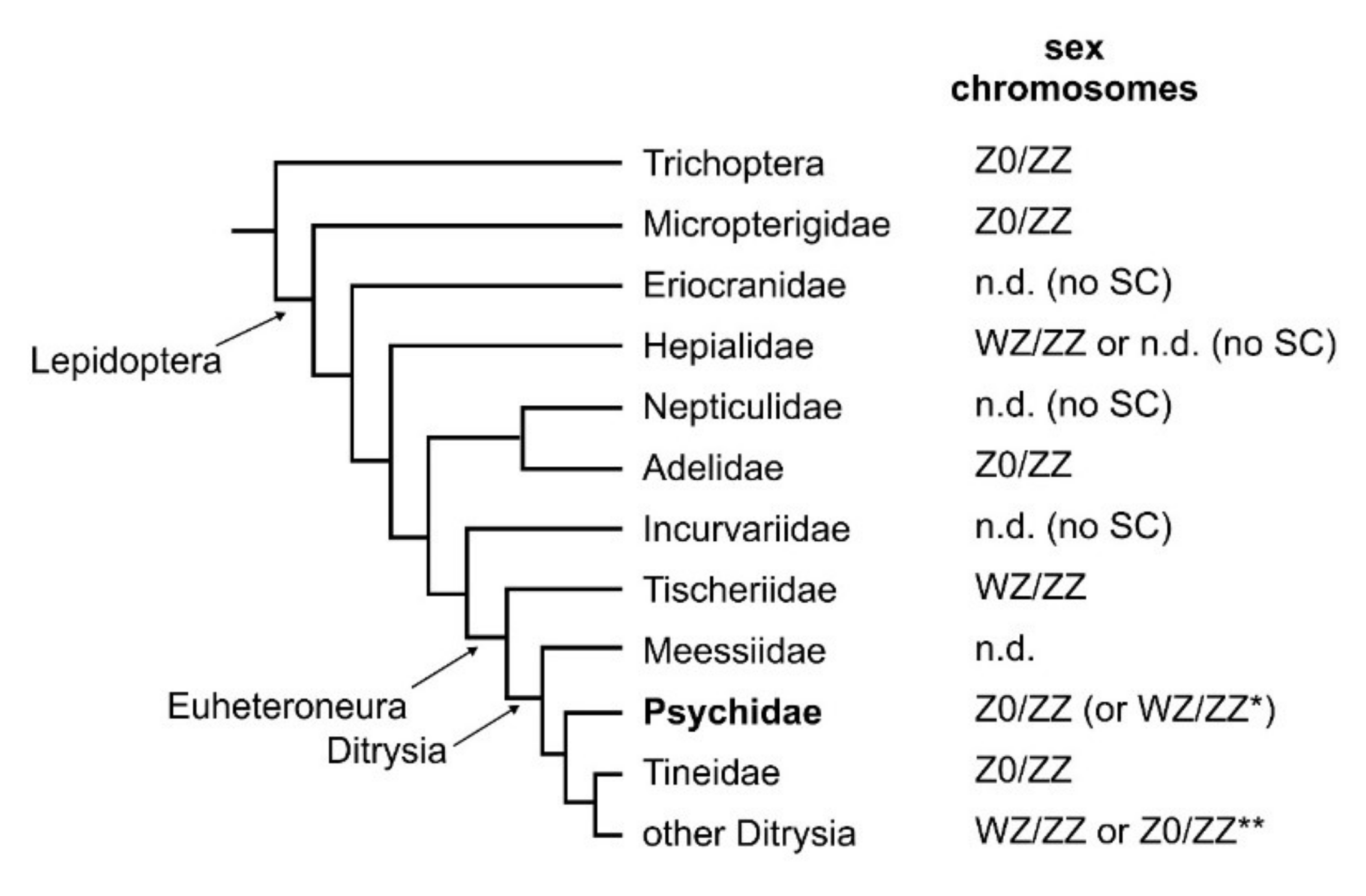
4. Discussion
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Traut, W.; Sahara, K.; Marec, F. Sex chromosomes and sex determination in Lepidoptera. Sex. Dev. 2007, 1, 332–346. [Google Scholar] [CrossRef] [PubMed]
- Beldade, P.; Saenko, S.V.; Pul, N.; Long, A.D. A Gene-based linkage map for Bicyclus anynana butterflies allows for a comprehensive analysis of synteny with the lepidopteran reference genome. PLoS Genet. 2009, 5, e1000366. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Van’t Hof, A.E.; Nguyen, P.; Dalíková, M.; Edmonds, N.; Marec, F.; Saccheri, I.J. Linkage map of the peppered moth, Biston betularia (Lepidoptera, Geometridae): A model of industrial melanism. Heredity 2013, 110, 283–295. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Fraïsse, C.; Picard, M.A.L.; Vicoso, B. The deep conservation of the Lepidoptera Z chromosome suggests a non-canonical origin of the W. Nat. Commun. 2017, 8, 1486. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Dalíková, M.; Zrzavá, M.; Hladová, I.; Nguyen, P.; Šonský, I.; Flegrová, M.; Kubíčková, S.; Voleníková, A.; Kawahara, A.Y.; Peters, R.S.; et al. New insights into the evolution of the W chromosome in Lepidoptera. J. Hered. 2017, 108, 709–719. [Google Scholar] [CrossRef]
- Marec, F.; Sahara, K.; Traut, W. Rise and fall of the W chromosome in Lepidoptera. In Molecular Biology and Genetics of the Lepidoptera, 1st ed.; Goldsmith, M.R., Marec, F., Eds.; CRC Press: Boca Raton, FL, USA, 2009; ISBN 978-113-811-172-1. [Google Scholar]
- Traut, W.; Mosbacher, G.C. Geschlechtschromatin bei Lepidopteren. Chromosoma 1968, 25, 343–356. [Google Scholar] [CrossRef]
- Ennis, T.J. Sex chromatin and chromosome numbers in Lepidoptera. Can. J. Genet. Cytol. 1976, 18, 119–130. [Google Scholar] [CrossRef]
- Traut, W.; Marec, F. Sex chromatin in Lepidoptera. Q. Rev. Biol. 1996, 71, 239–256. [Google Scholar] [CrossRef]
- Vlašánek, P.; Bartoňová, A.; Marec, F.; Konvička, M. Elusive Parnassius mnemosyne (Linnaeus, 1758) larvae: Habitat selection, sex determination and sex ratio (Lepidoptera: Papilionidae). SHILAP Rev. Lepidopterol. 2017, 45, 561–569. [Google Scholar]
- Traut, W.; Marec, F. Sex chromosome differentiation in some species of Lepidoptera (Insecta). Chromosome Res. 1997, 5, 283–291. [Google Scholar] [CrossRef]
- Lukhtanov, V.A. Sex chromatin and sex chromosome systems in nonditrysian Lepidoptera (Insecta). J. Zool. Syst. Evol. Res. 2000, 38, 73–79. [Google Scholar] [CrossRef] [Green Version]
- Sahara, K.; Yoshido, A.; Traut, W. Sex chromosome evolution in moths and butterflies. Chromosome Res. 2012, 20, 83–94. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Seiler, J. Researches on the sex-chromosomes of Psychidæ (Lepidoptera). Biol. Bull. 1919, 36, 399–404. [Google Scholar] [CrossRef]
- Seiler, J. Geschlechtschromosomen-Untersuchungen an Psychiden. Z. Indukt. Abstamm. Vererbungsl. 1922, 31, 1–99. (In German) [Google Scholar] [CrossRef]
- Seiler, J. Untersuchungen über die Entstehung der Parthenogenese bei Solenobia triquetrella F.R. II. Analyse der diploid parthenogenetischen Solenobia triquetrella F.R. (Lepidoptera, Psychidae). Chromosoma 1960, 11, 29–102. (In German) [Google Scholar] [CrossRef] [PubMed]
- Narbel-Hofstetter, M. Cytologie comparée de l’espèce parthénogénétique Luffia ferchaultella Steph. et de l’espèce bisexuée L. Lapidella goeze (Lepidoptera, Psychidae). Chromosoma 1961, 15, 505–552. (In French) [Google Scholar] [CrossRef] [PubMed]
- Narbel, M. La Cytologie de la parthenogénèse chez Apterona helix Sieb. Rev. Suisse Zool. 1946, 53, 625–681. (In French) [Google Scholar] [CrossRef]
- Nilsson, N.; ’Löfstedt, C.; Dävring, L. Unusual sex chromosome inheritance in six species of small ermine moths (Yponomeuta, Yponomeutidae, Lepidoptera). Hereditas 2008, 108, 259–265. [Google Scholar] [CrossRef]
- Rishi, S.; Sahni, G.; Rishi, K.K. Inheritance of unusual sex chromosome evidenced by AAWZ sex trivalent in Trabala vishnu (Lasiocampidae, Lepidoptera). Cytobios 1999, 394, 85–94. [Google Scholar]
- Yoshido, A.; Marec, F.; Sahara, K. Resolution of sex chromosome constitution by genomic in situ hybridization and fluorescence in situ hybridization with (TTAGG)n telomeric probe in some species of Lepidoptera. Chromosoma 2005, 114, 193–202. [Google Scholar] [CrossRef] [Green Version]
- Marec, F.; Traut, W. Sex chromosome pairing and sex chromatin bodies in W-Z translocation strains of Ephestia kuehniella (Lepidoptera). Genome 1994, 37, 426–435. [Google Scholar] [CrossRef] [PubMed]
- Lindsley, D.L.; Zimm, G.G. The Genome of Drosophila Melanogaster; Academic Press: San Diego, CA, USA, 1992; ISBN 978-012-450-990-0. [Google Scholar]
- Glaser, R.W. The growth of insect blood cells in vitro. Psyche 1917, 24, 1–7. [Google Scholar] [CrossRef] [Green Version]
- Dalíková, M.; Zrzavá, M.; Kubíčková, S.; Marec, F. W-enriched satellite sequence in the Indian meal moth, Plodia interpunctella (Lepidoptera, Pyralidae). Chromosome Res. 2017, 25, 241–252. [Google Scholar] [CrossRef] [PubMed]
- Mediouni, J.; Fuková, I.; Frydrychová, R.; Dhouibi, M.H.; Marec, F. Karyotype, sex chromatin and sex chromosome differentiation in the carob moth, Ectomyelois ceratoniae (Lepidoptera: Pyralidae). Caryologia 2004, 57, 184–194. [Google Scholar] [CrossRef]
- Traut, W.; Eickhof, U.; Schorch, J.C. Identification and analysis of sex chromosomes by comparative genomic hybridization (CGH). Methods Cell Sci. 2001, 23, 155–161. [Google Scholar] [CrossRef] [PubMed]
- Kato, A.; Albert, P.S.; Vega, J.M.; Birchler, J.A. Sensitive fluorescence in situ hybridization signal detection in maize using directly labeled probes produced by high concentration DNA polymerase nick translation. Biotech. Histochem. 2006, 81, 71–78. [Google Scholar] [CrossRef]
- Buntrock, L.; Marec, F.; Krueger, S.; Traut, W. Organ growth without cell division: Somatic polyploidy in a moth, Ephestia kuehniella. Genome 2012, 55, 755–763. [Google Scholar] [CrossRef]
- Gregory, T.R.; Johnston, J.S. Genome size diversity in the family Drosophilidae. Heredity 2008, 101, 228–238. [Google Scholar] [CrossRef] [Green Version]
- Seiler, J. Studies on the origin of parthenogenesis in Solenobia triquetrella F.R. (Lepidoptera, Psychidae). Chromosoma 1965, 16, 463–476. [Google Scholar] [CrossRef]
- Seiler, J. Vergleich des Fuhlers der reinen Geschlechter und der triploiden Intersexe von Solenobia triquetrella. Mol. Gen. Genet. 1972, 115, 146–194. [Google Scholar] [CrossRef]
- Suomalainen, E. On the sex chromosome trivalent in some Lepidoptera females. Chromosoma 1969, 28, 298–308. [Google Scholar] [CrossRef]
- Traut, W.; Weith, A.; Traut, G. Structural mutants of the W chromosome in Ephestia (Insecta, Lepidoptera). Genetica 1986, 70, 69–79. [Google Scholar] [CrossRef]
- Šíchová, J.; Voleníková, A.; Dincă, V.; Nguyen, P.; Vila, R.; Sahara, K.; Marec, F. Dynamic karyotype evolution and unique sex determination systems in Leptidea wood white butterflies. BMC Evol. Biol. 2015, 15, 89. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Robinson, R. Lepidoptera genetics. In International Series of Monographs in Pure and Applied Biology, 1st ed.; Kerkut, G.A., Ed.; Pergamon Press: Oxford, UK, 1970; ISBN 978-148-315-470-1. [Google Scholar]
- Seiler, J. Untersuchungen über die Entstehung der Parthenogenese bei Solenobia triquetrella F.R. Chromosoma 1959, 10, 73–114. [Google Scholar] [CrossRef] [PubMed]
- Nguyen, P.; Sýkorová, M.; Šíchová, J.; Kůta, V.; Dalíková, M.; Čapková Frydrychová, R.; Neven, L.G.; Sahara, K.; Marec, F. Neo-sex chromosomes and adaptive potential in tortricid pests. Proc. Natl. Acad. Sci. USA 2013, 110, 6931–6936. [Google Scholar] [CrossRef] [Green Version]
- Nguyen, P.; Carabajal Paladino, L. On the neo-sex chromosomes of Lepidoptera. In Evolutionary Biology: Convergent Evolution, Evolution of Complex Traits, Concepts and Methods, 1st ed.; Pontarotti, P., Ed.; Springer International Publishing: Cham, Switzerland, 2016; ISBN 978-331-941-324-2. [Google Scholar]
- Mongue, A.J.; Nguyen, P.; Voleníková, A.; Walters, J.R. Neo-sex chromosomes in the monarch butterfly, Danaus plexippus. G3 Genes Genomes Genet. 2017, 7, 3281–3294. [Google Scholar] [CrossRef] [Green Version]
- Regier, J.C.; Mitter, C.; Kristensen, N.P.; Davis, D.R.; van Nieukerken, E.J.; Rota, J.; Simonsen, T.J.; Mitter, K.T.; Kawahara, A.Y.; Yen, S.; et al. A molecular phylogeny for the oldest (nonditrysian) lineages of extant Lepidoptera, with implications for classification, comparative morphology and life-history evolution. Syst. Entomol. 2015, 40, 671–704. [Google Scholar] [CrossRef]
- Mutanen, M.; Wahlberg, N.; Kaila, L. Comprehensive gene and taxon coverage elucidates radiation patterns in moths and butterflies. Proc. R. Soc. B 2010, 277, 2839–2848. [Google Scholar] [CrossRef] [Green Version]
- Regier, J.C.; Mitter, C.; Zwick, A.; Bazinet, A.L.; Cummings, M.P.; Kawahara, A.Y.; Sohn, J.-C.; Zwickl, D.J.; Cho, S.; Davis, D.R.; et al. A large-scale, higher-level, molecular phylogenetic study of the insect order Lepidoptera (moths and butterflies). PLoS ONE 2013, 8, e58568. [Google Scholar] [CrossRef]
- Bazinet, A.; Mitter, K.; Davis, D.; Nieukerken, E.; Cummings, M.; Mitter, C. Phylotranscriptomics resolves ancient divergences in the Lepidoptera. Syst. Entomol. 2017, 42, 305–316. [Google Scholar] [CrossRef]
- Seiler, J. Sexuality as a developmental process. Proc. 11th Inter. Congr. Genet. 1964, 2, 199–207. [Google Scholar]
- Narbel-Hofstetter, M. La cytologie de la parthénogénèse chez Luffia ferchaultella Stph. (Lépid. Psychide). Communication préliminaire. Rev. Suisse Zool. 1954, 61, 416–419. [Google Scholar] [CrossRef]
- Narbel-Hofstetter, M. Cytologie de la pseudogamie chez Luffia lapidella GOEZE (Lepidoptera, Psychidae). Chromosoma 1963, 13, 623–645. [Google Scholar] [CrossRef]
- Narbel, M. La Cytologie de la parthenogénèse chez une Psychide: Apterona helix. Arch. Klaus. Stift. Vererb. Forsch. 1946, 20, 473–475. (In French) [Google Scholar]

© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hejníčková, M.; Koutecký, P.; Potocký, P.; Provazníková, I.; Voleníková, A.; Dalíková, M.; Visser, S.; Marec, F.; Zrzavá, M. Absence of W Chromosome in Psychidae Moths and Implications for the Theory of Sex Chromosome Evolution in Lepidoptera. Genes 2019, 10, 1016. https://doi.org/10.3390/genes10121016
Hejníčková M, Koutecký P, Potocký P, Provazníková I, Voleníková A, Dalíková M, Visser S, Marec F, Zrzavá M. Absence of W Chromosome in Psychidae Moths and Implications for the Theory of Sex Chromosome Evolution in Lepidoptera. Genes. 2019; 10(12):1016. https://doi.org/10.3390/genes10121016
Chicago/Turabian StyleHejníčková, Martina, Petr Koutecký, Pavel Potocký, Irena Provazníková, Anna Voleníková, Martina Dalíková, Sander Visser, František Marec, and Magda Zrzavá. 2019. "Absence of W Chromosome in Psychidae Moths and Implications for the Theory of Sex Chromosome Evolution in Lepidoptera" Genes 10, no. 12: 1016. https://doi.org/10.3390/genes10121016
APA StyleHejníčková, M., Koutecký, P., Potocký, P., Provazníková, I., Voleníková, A., Dalíková, M., Visser, S., Marec, F., & Zrzavá, M. (2019). Absence of W Chromosome in Psychidae Moths and Implications for the Theory of Sex Chromosome Evolution in Lepidoptera. Genes, 10(12), 1016. https://doi.org/10.3390/genes10121016





