Abstract
Suicide in Bipolar Disorder (BD) is a relevant clinical concern. Genetics may shape the individual risk for suicide behavior in BD, together with known clinical factors. The lack of consistent replication in BD may be associated with its multigenetic component. In the present contribution we analyzed a sample of BD individuals (from STEP-BD database) to identify the genetic variants potentially associated with three different suicide-related phenotypes: (1) a feeling that the life was not worth living; (2) fantasies about committing a violent suicide; (3) previous attempted suicide. The sample under analysis included 1115 BD individuals. None of the SNPs reached genome-wide significance. However, a trend of association was evidenced for rs2767403, an intron variant of AOPEP gene, in association with phenotype #1 (p = 5.977 × 10−6). The molecular pathway analysis showed a significant enrichment in all the investigated phenotypes on pathways related to post synaptic signaling, neurotransmission and neurodevelopment. Further, NOTCH signaling or the γ-aminobutyric acid (GABA)-ergic signaling were found to be associated with specific suicide-related phenotypes. The present investigation contributes to the hypothesis that the genetic architecture of suicide behaviors in BD is related to alteration of entire pathways rather than single genes. In particular, our molecular pathway analysis points on some specific molecular events that could be the focus of further research in this field.
1. Introduction
Bipolar disorder (BD) is a group of recurrent affective disorders characterized by episodes of mania (a pathological elation of mood sometimes with psychotic symptoms), hypomania (a lesser form of mania) and/or depression (a pathological depressed mood sometimes with psychotic symptoms). BD definition has undergone major changes over the last decades. In this evolving classification system, one clinical phenotype remains of unchanged and vital relevance: suicide behavior. Suicide behavior is a prime concern in BD as BD patients hold the highest risk of suicide when compared with all other psychiatric subjects [1]: the expected lifespan in this disease is reduced by 8.5–16.7 years compared to the general population [2], and the higher suicide rates in BD are strictly implicated with this decrease [3]. Further, the high prevalence of BD, 0.3 to 1.5% in the general population, makes suicide risk an important concern in modern society [4,5] (Please refer to Appendix A for definitions of suicide related phenotypes and Appendix B for clinical risk factor along with preventive strategies for suicide behavior).
One-third up-to one-half of BD patients will attempt suicide at least once and 15–20% of BD individuals die from suicide. Suicide attempts in BD have a higher rate of success: 1 out 3–4 completers compared to the 1 out of 30 completers observed in the general population [6]. Genetics may be one of the factors contributing to the risk of suicide in BD, as reported by numerous adoption studies, twin studies and family studies. Data obtained evidenced that the rates of suicide attempts and completions are higher in individuals with a family history of attempters and/or completers (please refer to Table 1). Further, the genetic component for suicide seems to be partly shared with the inheritance of psychiatric disorders [7,8]: Indeed, recent GWAS analyses showed interesting correlation between suicide attempts and depressive symptoms, neuroticism, Schizophrenia, insomnia and major depressive disorder [9,10]. However, the genetic background behind suicide behaviors remains elusive: GWAS studies with polygenic risk scores have detected numerous SNPs potentially correlated with suicide [11,12,13]. The heritability calculated from these common variations only ranges between 4 and 4.6% [10,14], suggesting a complex biological background and a potential multifactorial origin behind this disease.

Table 1.
Main Previous Findings about the Genetics of Suicide Behavior in Affective Disorders.
The high rates of suicide attempts and committed suicides with BD call for further research aiming at predict and hopefully reduce this behavior and ameliorate the devastating consequences for both relatives and the patients themselves. In this study we tried to investigate associations between suicidality and genetic variations in genotyped participants from the STEP-BD program (The Treatment Enhancement Program for Bipolar Disorder) [15] with a particular focus on biological pathways, which have a higher capacity of explaining complex phenotypes.
2. Materials and Methods
2.1. Clinical Sample
The sample under analysis was obtained from the NIMH genetics available Treatment Enhancement Program for Bipolar Disorder (STEP-BD). The STEP-BP remains one of the largest public investigations conducted so far for BD. It was located in the USA and initially enrolled 4361 participants distributed in 21 sites. Of them, only half gave consent for genetic analyses (please refer to Section 2.3). The trial included both naturalistic and randomized nested studies. Subjects had the opportunity to enter the randomized studies within the general trial once and re-enter the naturalistic design at exit. The study is described in details here [15].
2.2. Participants
Individuals included in the study were 18 years or older subjects with BD I or II. Diagnoses were confirmed by the Mini International Neuropsychiatric Interview (MINI) [35]. Moreover, a set of information was retrieved from the Affective Disorder Evaluation (ADE) set of questions prepared and focused on that specific study [15]. At the time of randomization all the subjects met criteria for acute depression at the MINI. Every patient received a standard care and follow-up, until accepting and meeting the criteria for being offered a randomized clinical treatment according to STEP-BD clinical states (SCSs) [15,36]. SCSs comprised: (1) the “acute depression SCS”; (2) the “refractory depression SCS”, which included patients that failed to respond to 12 months treatment or at least two trials in the past; and (3) the “relapse prevention pathway” in which the index episode (mania, hypomania or mixed) occurred in patients under either lithium or valproate treatment and with normal levels of Thyroid-stimulating hormone and creatinine [15,36].
2.3. Original Genetic Sample
About half of the original participants in the STEP-BD gave consent for the genetic analysis. From the original data (from the NIMH genetics database) there were 2453 people (1218 males, 1235 females), with a total genotype rate of 0.99 before quality control. Genotyping was performed using the Affymetrix GeneChip Human Mapping 500K Array Set (Affymetrix, part of Thermo Fisher Scientific, Waltham, MA, USA) by the Genetic Analysis Platform at the Broad Institute of Harvard and Massachusetts Institute of Technology. The link to the original study can be found at [15]. 372,193 variants were available in the original file before quality control. The Hapmap genome database b23 was instrumental for the imputation process. The sample was further filtered to exclude individuals with no information on the phenotypes under analysis. The final sample comprised 670 males and 485 females.
2.4. Outcomes
Three phenotypes were under analysis. Subjects that experienced (1) a feeling that the life was not worth living, AND/OR (2) fantasies about committing a violent suicide AND/OR (3) previous attempted suicide; were classified as “cases” or “controls” for main analysis: Individuals who had a score > 0 in one specific phenotype were considered cases for that phenotype. Individuals who had a score = 0 for a specific phenotype were considered controls for that specific phenotype. This analysis was performed with the aim of evidencing potential differences in the genetic background between the three phenotypes. These variables were chosen because they cover some relevant psychopathological aspects of suicide behavior, rather than the dichotomic attempted/completed suicide VS non suicide behavior. Additionally, an exploratory analysis which evaluated individuals with none of the described phenotypes as “controls” versus individuals with at least one of the described phenotypes as “cases” was performed. The aim of the exploratory analysis was to evidence the main genetic/biological differences between suicidal and non-suicidal bipolar patients. Only subjects with bipolar disorder were included in both analyses.
2.5. Clinical Covariates
Age, gender, ethnical background, marital status, living alone, education, kind of job and drug or alcohol abuse at the entry of the study, were the covariates included in the analysis. Variables are detailed in Table 1. Age, gender, ethnical background, marital status, living alone, kind of job and employment status and drug or alcohol abuse at the entry of the study were retrieved from the DF database and downloaded after permission from the NIMH genetics database. As for the ethnical background, the questionnaire included two questions, the first being “What is your primary race” and including answers as for example, “White or Caucasian” or “Black of African American”. The second was “Are you Hispanic or Latino?” BD classification type was excluded from covariates since the phenotype under analysis is associated with the depressive phase, which is similar in type I and type II and this classification is based mainly on manic phase.
2.6. Statistical Model and Flow of Analysis
All analyses were conducted in R [37], or in bash environment. Plink [38], gtool (https://www.well.ox.ac.uk/, (accessed on 1 April 2021)) and impute [39] were instrumental to perform the genetic analyses. The bash environment provided the basis for the use of plink, impute and gtool. It also allowed for an effective manipulation of phenotype data, in order to adapt them to the input requirements for the above-mentioned programs. The single nucleotide association analyses were conducted in Plink after the genotype was imputed (with gtool and impute), pruned and checked for quality control to standard defaults for this kind of analysis. Molecular pathway analyses were performed on R environment, using the result of the single nucleotide association analysis as input, and providing as output the molecular pathways found to be enriched in mutations associated with the phenotypes under analysis. Figures and tables were also created in R. R script is available on request.
2.7. Analysis of Clinical Data
The clinical phenotypes were created and their association with the clinical covariates tested with the appropriate statistical test (for example ANOVA or Chi2, or the correspondent nonparametric test) in order to protect the genetic analysis from clinical stratification factors. Covariates that were significantly associated with the outcomes under analysis were included in files containing binary variables. For example, if “living alone” was found to be associated with the phenotypes under analysis, the variable was commuted to a binary 1 or 2 variable where 1 was for example “married or living as married” and 2 contained every other clinical variation of the original variable. This process was instrumental to reduce the degree of freedom of the analysis, to avoid the risk of a NA (not assessable) result from the plink analysis (genetic association analysis).
2.8. Analysis of Genetic Data
The standard quality thresholds were applied to the original sample before pruning and imputing. Minor allele frequency was set at 0.05, genotype rate was set at 0.95, Hardy Weinberg Equilibrium was set at 0.00001. Pruning was set at the standard --indep 50, 5, 2, where 50 is the number of SNPs considered at every step, 5 is the number of SNPs to be shift at every step and 2 is the VIF threshold (1/ (1 − R2) where R2 is the multiple correlation coefficient). An R2 equals to 10 implies that two SNPs carry the same signal. An R2 equals to 1 implies that two SNPs are completely independent. The enrichment analysis was conducted using R software suite, through Bioconductor [40] and the package ReactomePA [41]. The ReactomePA (https://bioconductor.org/packages/release/bioc/html/ReactomePA.html, accessed on 1 April 2021) is a manually curated database that includes chemical reactions, biological processes and molecular pathways. To test for possible stratification factors associated with the phenotypes under analysis at plink test was performed under standard parameters for test the non-genetic identity of the samples (cases and controls, Permutation test for between group IBS differences where IBS stands for identical by state). Please refer to Figure 1 and Table 2. Single tests for association were generated for every SNP under a regression model. SNPs associated with the investigated phenotype were ranked according to the pValue of association. SNPs showing a significant (p < 0.05) association with the phenotypes under analysis were selected. It is of note, that this level of significance is not be used in a classic GWAS study because of the risk of false positives. This—or similar—classic level of significance was nevertheless chosen in the present as in previous studies [42,43,44,45,46] to identify all the possible significant associations throughout the genome. Moreover, the same level of significance for this kind of analysis is standard for the package in use. The risk for false positives was then controlled by correcting for multiple testing, a function that is embedded in the R package in use. In particular, Bonferroni correction (p.adjust) and False Discovery Rate correction (q value) were provided. The genes that harbored such variations were identified and investigated for enrichment. Enrichment in this context means, that the number of SNPs that are significantly associated with the phenotypes under analysis is larger than expected by chance. As a consequence, the genetic variations distributed in specific molecular pathways, those enriched, are shown to have a potential role in differentiating cases from controls. This approach takes into consideration the likely multigenetic nature of such complex phenotypes and provides more power for this kind of analysis. The analysis was conducted in a Linux system in Bash language; the computations were conducted through access at the Aalborg University superPC.
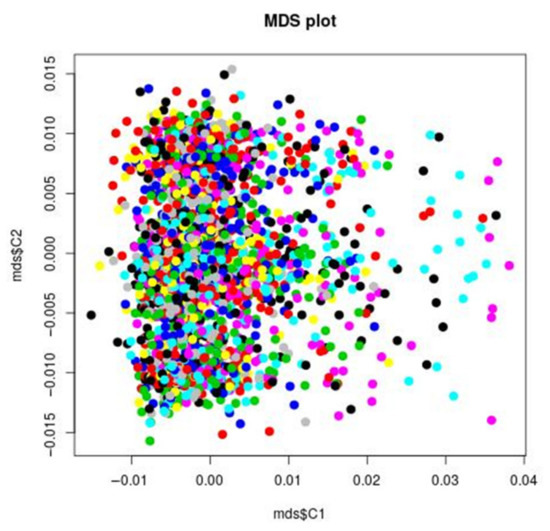
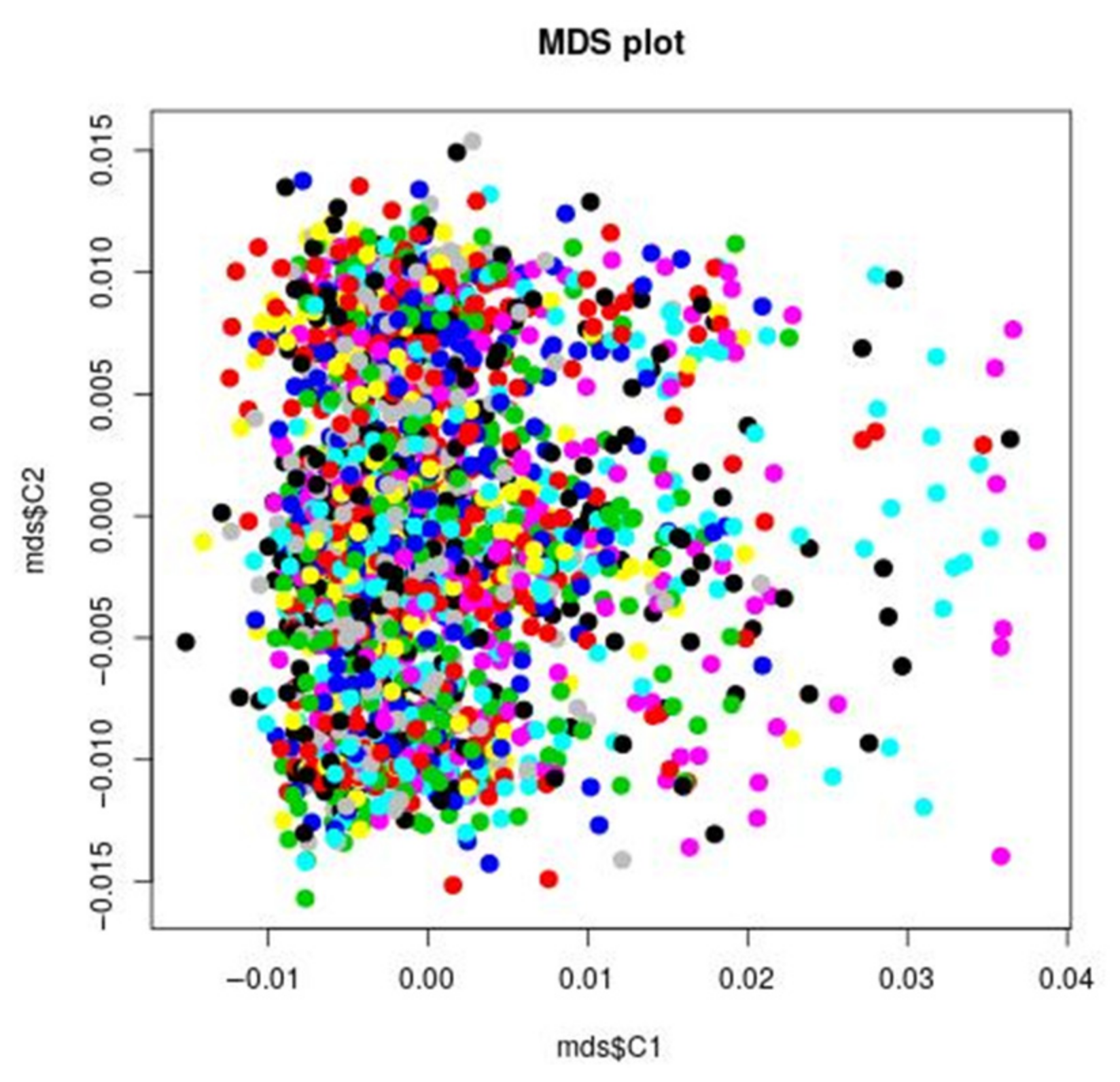
Figure 1.
MDS plot: IBS clustering analysis of the genetic stratification factors. Increasing values of the first component do not correspond to visual significant increasing or decreasing values of the second component, suggesting that the covariance between the two components in the genetic is minimal. For example, to small and increasing values of the first component correspond both small and higher values of the second component. The points are concentrated in the left end of the figure, indicating a larger variance of the first component. In order to further test this visual impression, the plink permutation test for between group IBS differences confirmed that there was no significant group genetic differences (stratification factors) with respect to all the phenotypes under analysis. That does not necessarily mean, that there is no genetic stratification in the STEP-BD sample, but that this stratification is not of main significant interest when considering the phenotypes under analysis. The pairwise clustering based on IBS (identity by state) is useful for detecting pairs of individuals who look more different from each other than what is expected in a random, homogeneous sample. This method allows for identification of clusters of patients, that are more genetically similar to each other than they are similar to the rest of the sample. Such groups are identified by different colors in the figure.

Table 2.
Test between group IBS (identical by state) differences for stratification factors.
3. Results
The sample under analysis and the covariate analysis are described in Table 3. Gender was not included in the analysis because not associated with the phenotypes under analysis and because there was no evidence for major genetic stratification factors for the phenotypes under analysis.

Table 3.
Sample characteristics and clinical covariate analysis.
3.1. Main Analysis
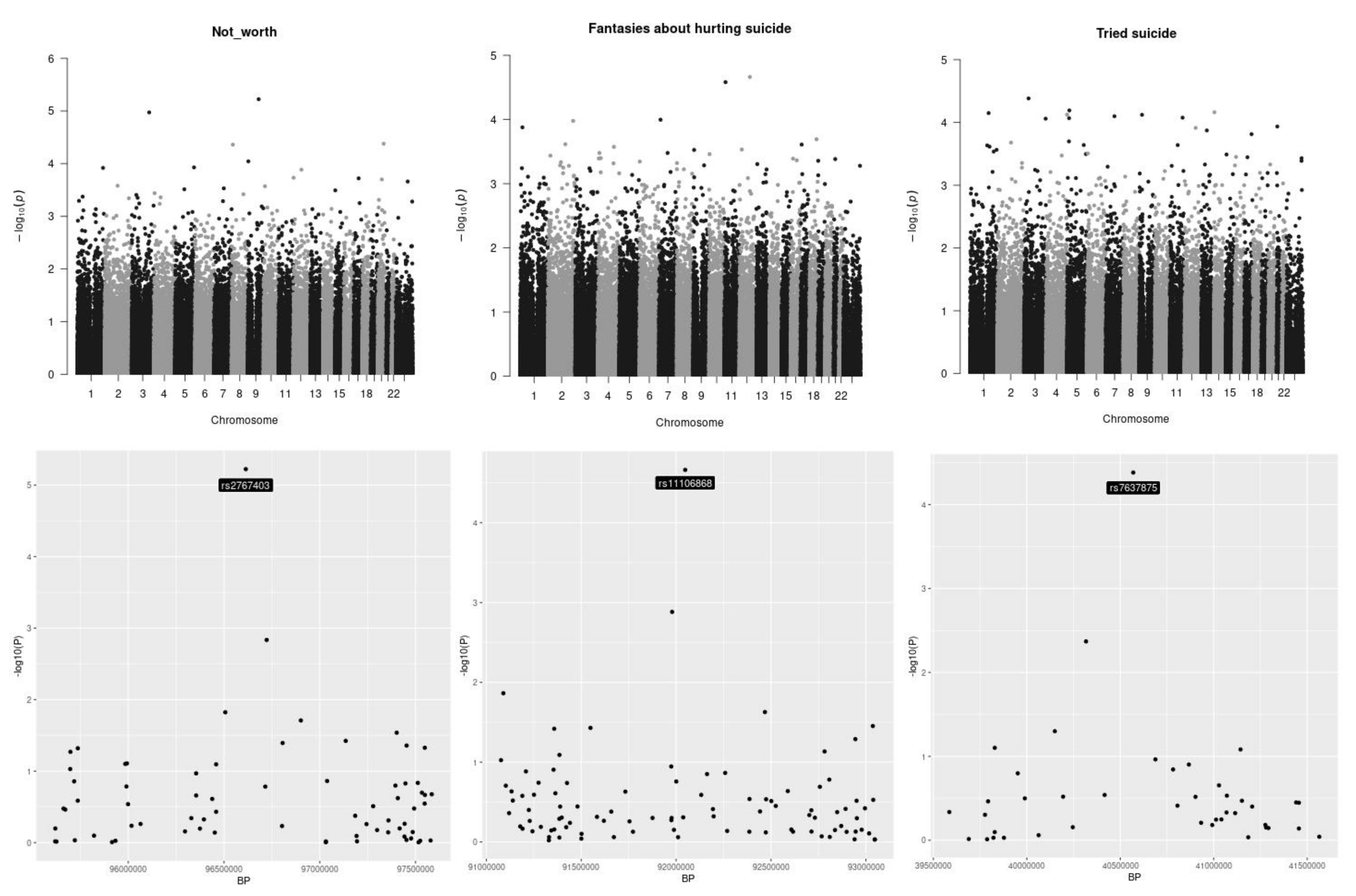
Figure 1 reports the analysis of the genetic stratification factors. Figure 2 reports the result of the genome wide analysis. Figure 3 reports the result of the molecular pathway analysis. 957418 SNPs were available for the analysis after standard quality control and imputation. Briefly, no single variation reached genome wide significance in the GWAS analysis. The strongest association resulted for rs2767403 (C > G) (p = 5.977 × 10−6) in association with phenotype #1 (Not worth). Rs2767403 is an intron variant of the AOPEP (aminopeptidase O), a gene implicated in the angiotensin IV pathway.
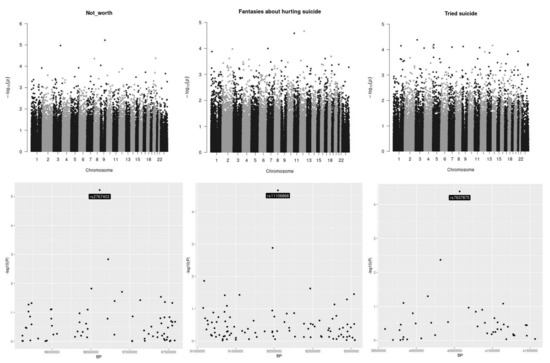
Figure 2.
Manhattan plots. Manhattan plots for the three outcomes under analysis are presented in the top of the picture. At the bottom of the picture the genomic areas with the strongest associations are shown. The outcomes under analysis are, respectively, from left to right, “having a feeling of not worth”, “fantasies about a hurting suicide” and “tried suicide”. None of the single investigated SNP reached the genome-wide threshold.
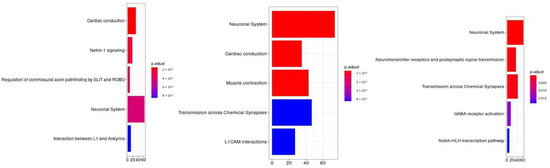
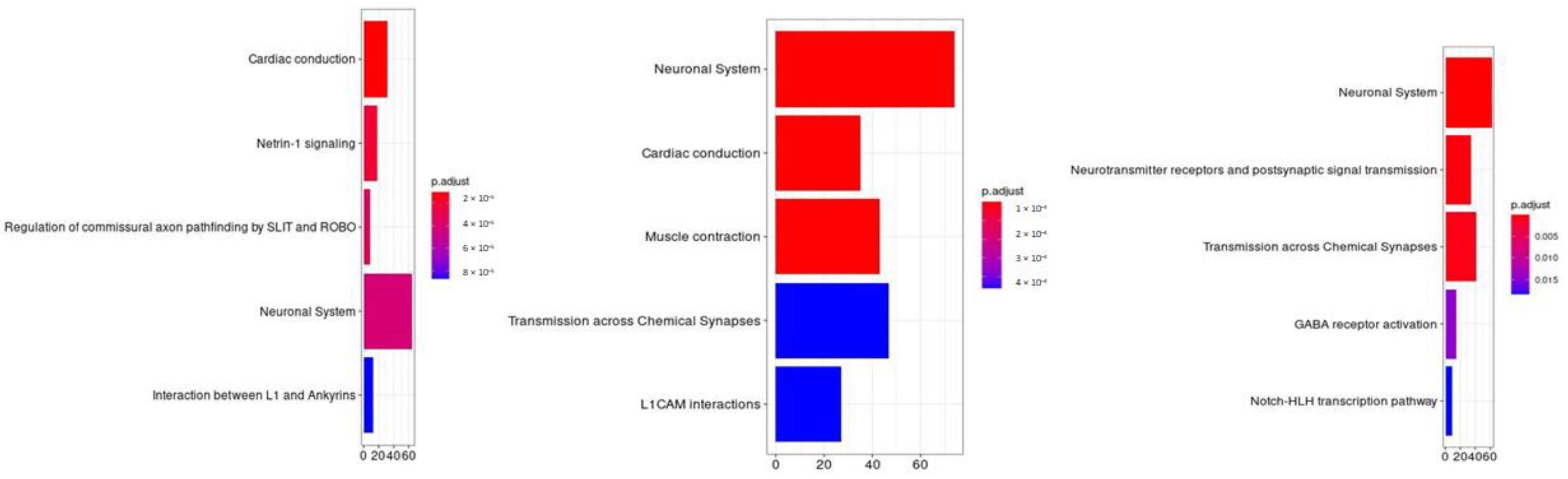
Figure 3.
Molecular pathway analysis. The molecular pathways are listed in the Y axis. X axis describes the number of genes found to be enriched in association with, respectively, from left to right, “having a feeling of not worth”, “fantasies about a hurting suicide” and “tried suicide”.
According to in silico analyses of the effects of rs2767403 on splicing, this mutation introduces a donor splice site within the AOPEP sequence, which can potentially alter the splicing pattern of AOPEP mRNA. Table 4 reports the results of the in silico analysis that was performed using Genomnis HSF software (https://www.genomnis.com/, accessed on 21 April 2021).

Table 4.
Splicing Analysis of rs2767403.
The molecular pathways analysis reported a list of molecular pathways enriched in genetic variations associated with the phenotypes under analysis. Some shared molecular pathways enriched in variations associated with all the phenotypes under investigation were the pathways involved in post synaptic signaling, neurotransmission in general and neurodevelopment (axon). Finally, NOTCH signaling and the GABAergic signaling were found to be associated with specific suicidal behaviors (please refer to Table 5, Figure 4 and Figure 5).

Table 5.
Result from the molecular pathway analysis (main analysis).
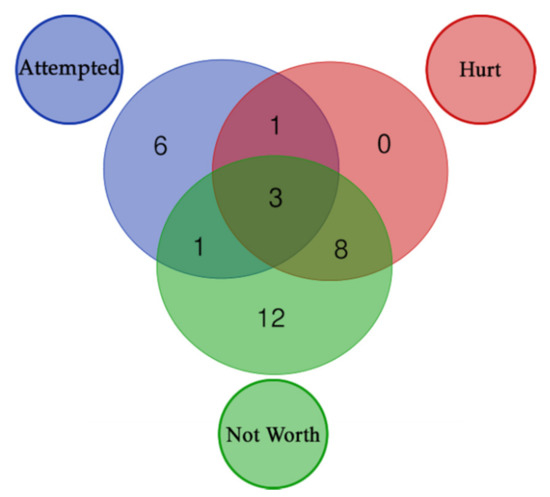
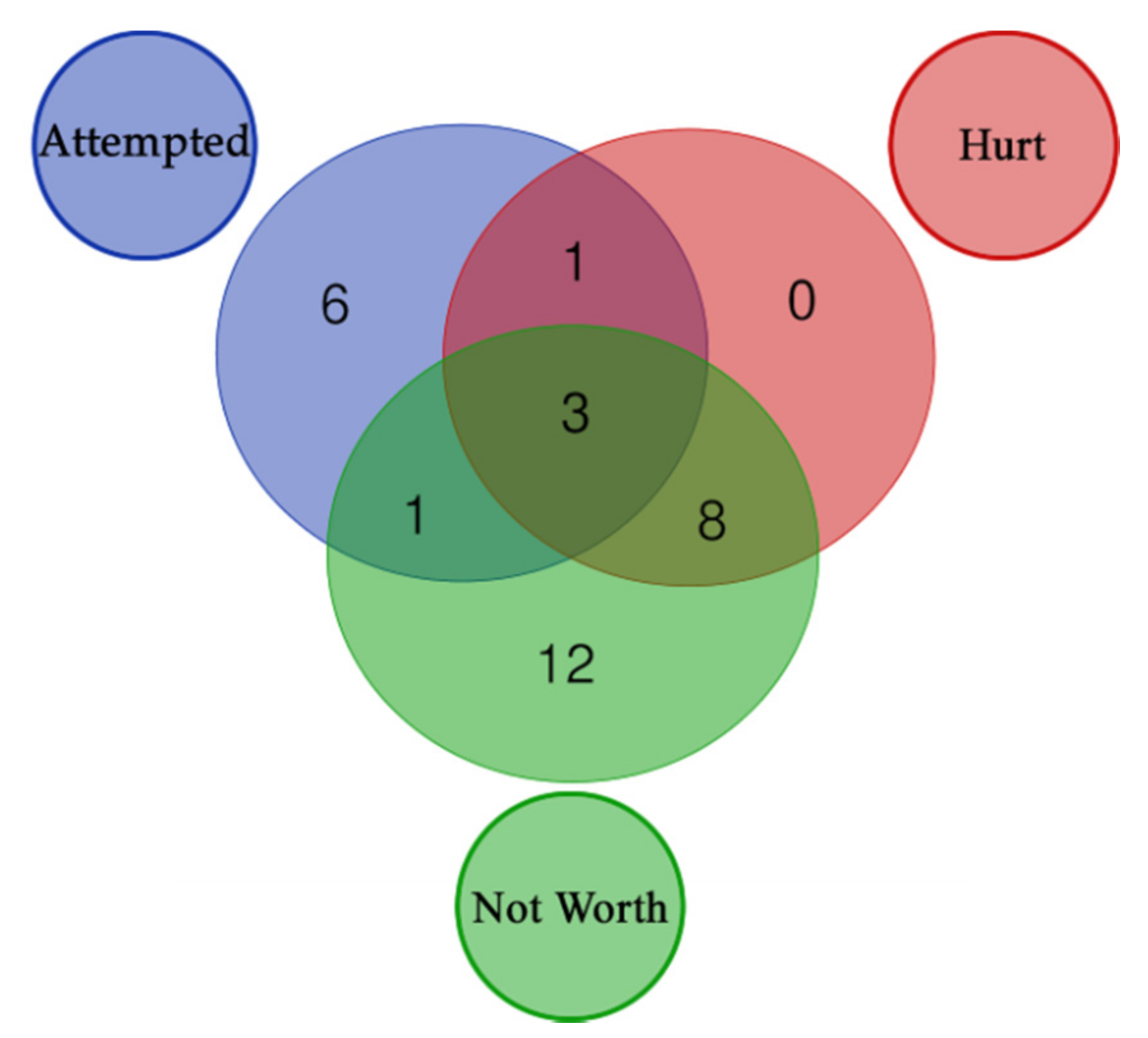
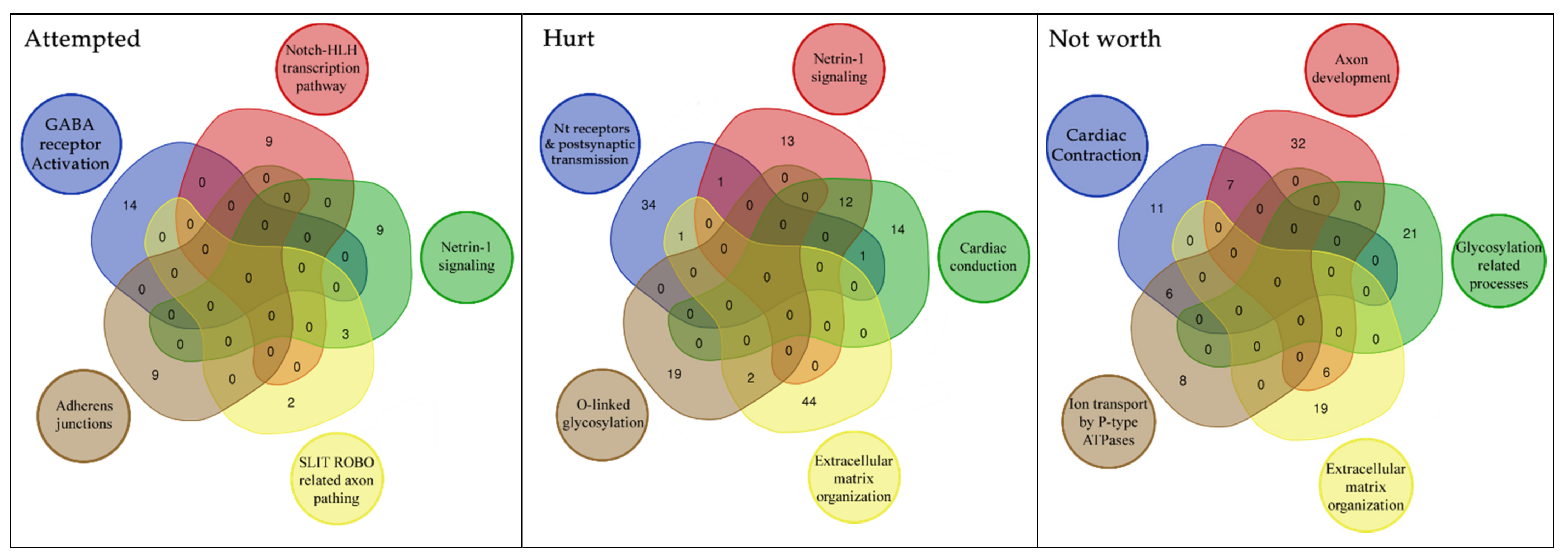
Figure 4.
Distribution of enriched pathways for the 3 suicide classes. The figure reports the Venn Diagram of enriched pathways distribution for the 3 suicide classes. Attempted + Hurt + Not Worth (3): R-HSA-373752, R-HSA-112316, R-HSA-112315; Attempted + Hurt (1): R-HSA-112314; Attempted + Not Worth (1): R-HSA-428542; Hurt + Not Worth (8): R-HSA-397014, R-HSA-5173105, R-HSA-1474244, R-HSA-445095, R-HSA-5578775, R-HSA-5576891, R-HSA-373760, R-HSA-5576892; Attempted (6): R-HSA-421270, R-HSA-350054, R-HSA-446728, R-HSA-1500931, R-HSA-977443, R-HSA-418990; Not Worth (12): R-HSA-1650814, R-HSA-5083635, R-HSA-5173214, R-HSA-1474290, R-HSA-8948216, R-HSA-3906995, R-HSA-2022928, R-HSA-375165, R-HSA-3000178, R-HSA-983712, R-HSA-419037, R-HSA-936837.
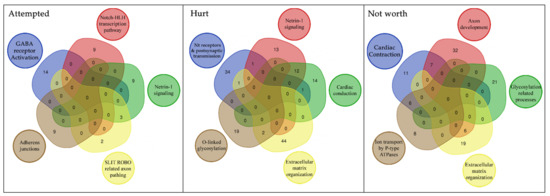
Figure 5.
Overlap of the genetic structure associated with specific suicide related phenotypes in BD. This figure reports the unique pathways we found involved in the three suicide classes. Further we reported through a venn diagram the number of genes shared by each of these pathways.
3.2. Exploratory Analysis
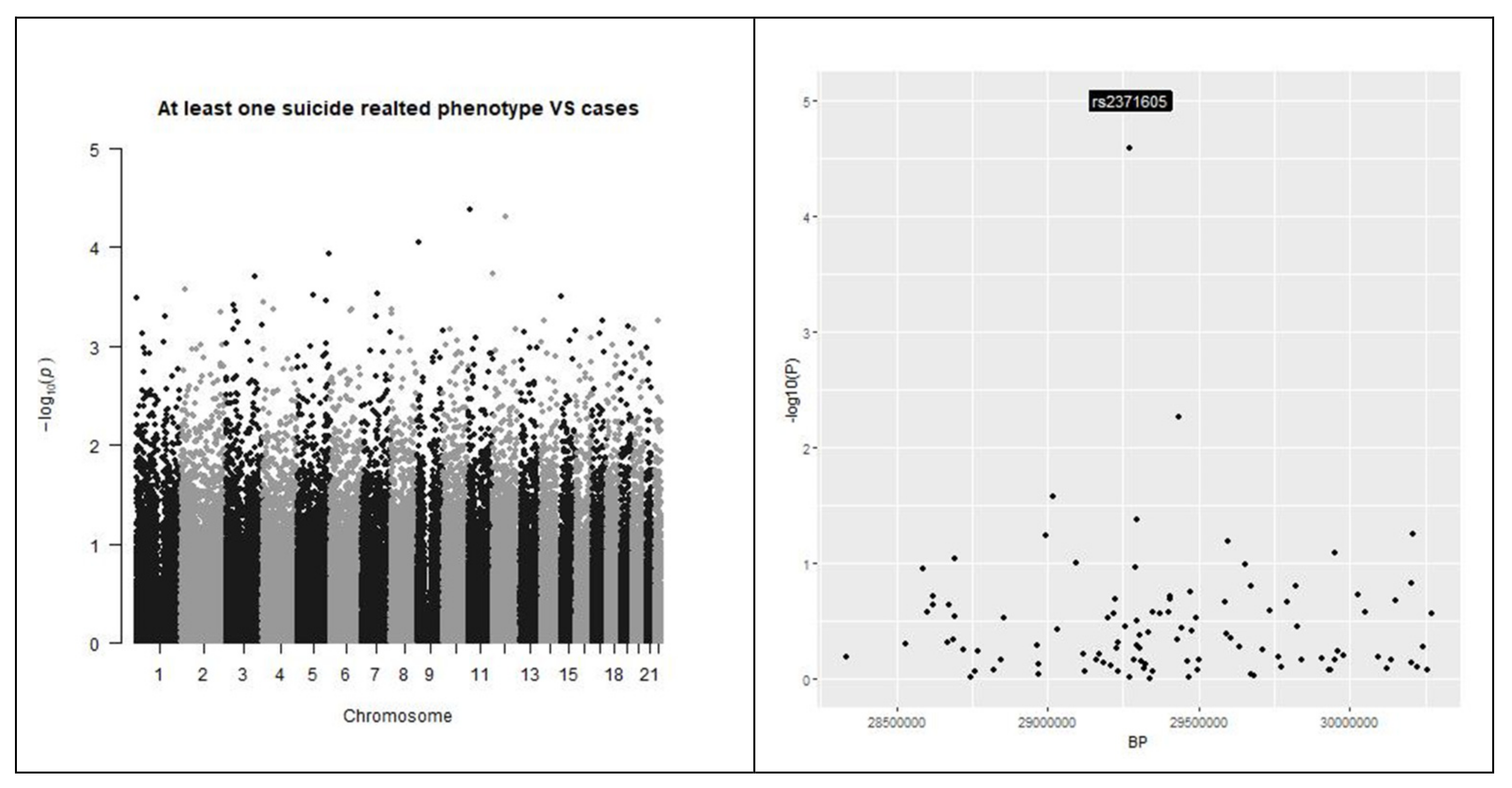
The result of the analyses on individuals reporting at least one of the investigated phenotypes (cases) against individuals reporting none (controls) showed no significant data in single SNPs analysis (please refer to Figure 6). The molecular pathways analysis reported a list of molecular pathways enriched in genetic variations associated with the phenotypes under analysis. Results are reported in Table 6 and Figure 7.
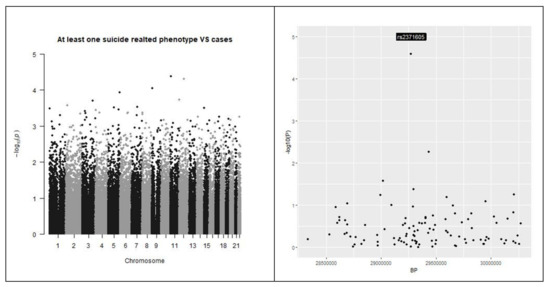
Figure 6.
Manhattan plots. Left: Manhattan plots for the outcome under analysis were reported. Right: the genomic areas with the strongest associations with outcome are shown. The outcome under analysis is suicidal behavior versus controls. None of the single investigated SNP reached the genome-wide threshold.

Table 6.
Result from the molecular pathway analysis (exploratory analysis).
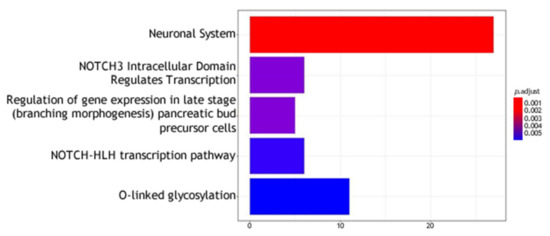
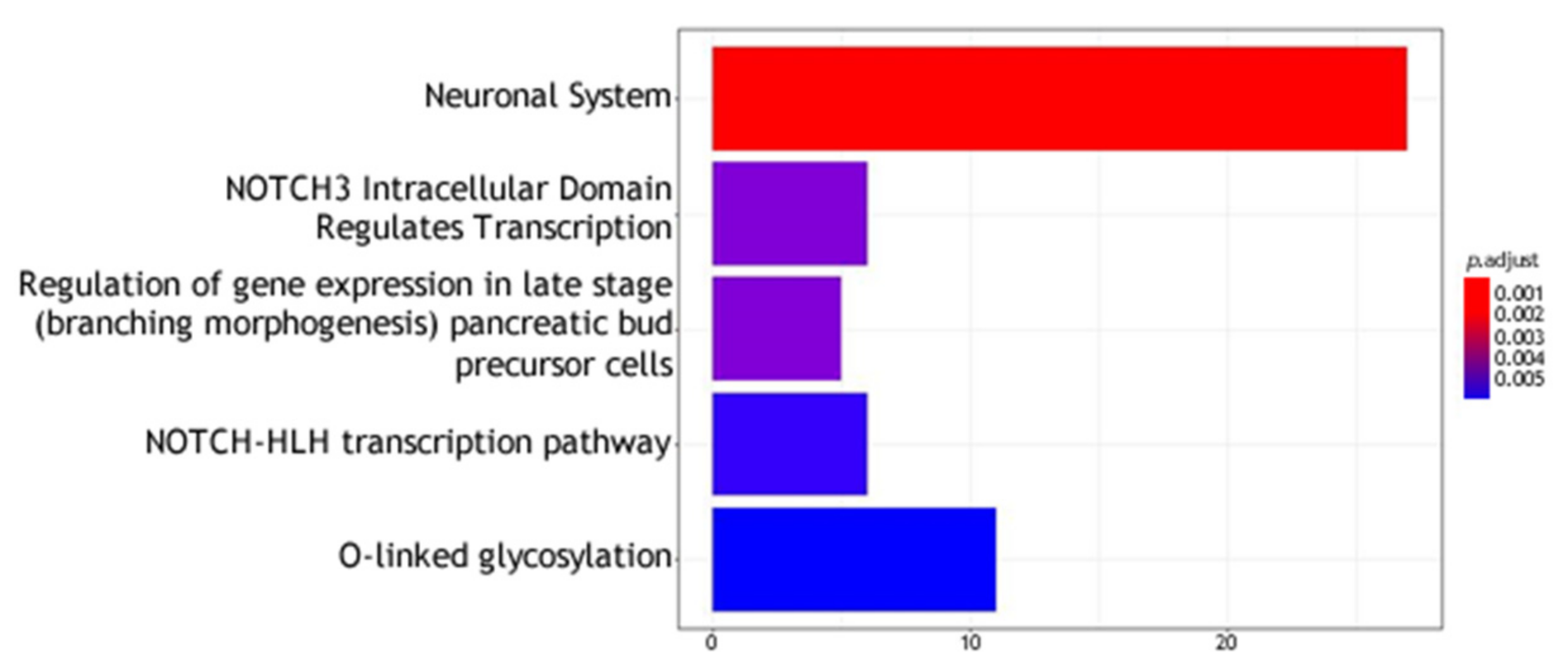
Figure 7.
Molecular pathway analysis. The molecular pathways are listed in the Y axis. X axis describes the number of genes found to be enriched in the suicidal behavior versus controls analysis.
4. Discussion
Suicidal attempts are dramatic, frequent events in psychiatric clinical practice. Even though several studies have been carried out, there are no data on the biological factors influencing the risk of this event that can be used in the current clinical practice. In this paper we have focused on the potential genetic backbone which may be indicative of risk in BD.
4.1. Analysis of Single SNPs
In our study, no single DNA variation reached genome-wide significance, emphasiz ing that no gene or SNP, alone, has a high enough impact on suicide risk. Still, some insights could be obtained from the observed trend. The most significant association, with a p = 5.977 × 10−6, was related to an intronic variant (rs2767403) of the AOPEP gene. The other two SNPs with a low (but not significant) p-value (rs11106868 and rs7637875) were localized in intergenic regions as such no further analysis was conducted. Regarding the exploratory analysis, no SNPs reached the significant threshold and the most significant trend (rs2371605) was localized in an intergenic region. According to in silico analyses, rs2767403 introduces a donor splice site within the AOPEP sequence (Table 4).
Even though this prediction highlights the potential variability caused by rs2767403, the AOPEP gene encodes a metallo protease linked to the renin-angiotensin system [47]. In particular, it is able to cleave angiotensin III (but not I and II) to generate Angiotensin IV. It is mainly expressed in heart, but it can also be found, with lower concentrations, in brain. Overall, AOPEP RNA has poor tissue specificity and can be found in almost all tissue of the body. Unfortunately, except for its correlation with the renin-angiotensin system, little is known about its function. Of note, several studies consistently associated AOPEP with polycystic ovary syndrome (PCOS) [48,49,50,51,52,53].
Of interest, the AOPEP region also holds the miR-23b/27b/24 cluster [54]. This cluster is composed of three miRNA genes located within 14 intron of C9orf3 (AOPEP) [54]. The precise mechanism of regulation of the miR-23b/27b/24 cluster expression is still not clear [54]. According to current knowledge on intronic miRNAs biogenesis, the pri-miR-23b/27b/24 cluster is potentially transcribed as part of the transcript of the AOPEP gene. Therefore, potential alterations of AOPEP maturation processes by rs2767403 can potentially interfere with the cluster expression.
The role of these miRNAs has not been fully elucidated, however studies examining the roles of miR-23b, miR-27b and miR-24-1 have demonstrated their multiple functions, ranging from metabolic disorders to proliferation and development disorders [55,56,57,58,59,60]. These miRNAs are highly expressed in vascularized tissues [60]. In addition, miR-27b was shown to target the NOTCH ligand Delta-like ligand 4 (Dll4) [55]. To note, the NOTCH pathway was enriched in our pathway analysis. Within their functions, this cluster, and in particular miR-23b and miR-27b, seem to exert an important control on neuronal apoptosis, modulating the expression of Apaf-1 gene (in a murine model) [61]. Apaf-1 is a key apoptotic protein associated with neuronal apoptosis [62]. This protein levels are extremely low in adult brains. Conversely, the expression of miR-23-27 cluster is significantly higher in adults than in embryos. Even though its function in vivo remains elusive, this cluster likely regulates Apaf-1 expression, thus affecting the sensitivity of neurons to apoptosis during development. A potential alteration of this tight regulation system can lead to impaired neural nets (brain) development. Even though there is currently no evidence in literature that correlates this cluster and suicide, miRNAs expression can be closely related to neurophysiology and suicidal behavior [63,64].
4.2. Molecular Pathway Analysis
Regarding our pathway analysis, our data evidenced that specific molecular cascades are enriched in the above defined suicide classes (please refer to Table 5). The three classes of suicide (Attempted, Hurt and Not worth) shared some of these pathways, as also evidenced by exploratory analysis, while others were unique. In particular, the biological processes involved in post synaptic signaling, neurotransmission in general and neurodevelopment (axon) were enriched in all these classes and in the exploratory analysis (Neuronal System), although GABA signaling was only enriched in the Attempted class. In addition, NOTCH signaling and cell-cell communication pathway were found to be significantly enriched only in this class. The Not worth class was related to the biological processes involved in ion equilibrium, with extracellular matrix and glycosylation processes. Figure 4 reports a Venn diagram showing pathways distribution in the 3 classes.
A further focus on the genes within each enriched pathway explains the significance of some apparently unrelated pathways. Indeed, as can be seen in the Venn diagrams (Figure 5), cardiac conduction related pathway shares 12 genes with Netrin-1 signaling (axon development related process). Common genes are the SCN genes, which encode for sodium voltage-gated channel subunits. They are important in both cardiac and brain function. In particular, the pathway associated with SCN genes seems to be involved in the biological processes of pain, especially in the development of inflammatory pain [65,66,67,68]. This could explain the link between these genes (and related pathways) and suicide risk, as pain is a robust predictor of suicidal desire [69].
In the Not Worth class, the pathway of cardiac function also shared its genetic background with the ion transport by P-type ATPases pathway. The genes shared by the two play a role in both cardiac function and neurotransmission related processes. Transmembrane ion transport by ATPases is closely related to the membrane potential. Alterations within this process likely alter neurotransmission processes. Interestingly, literature data associate ATPase activity coupled to the transport of ions across the cell membrane with suicide risk [70,71].
The associated pathways related to the neurotransmission processes (common in the three classes) and to GABA neurotransmission, specific for suicide attempters were deeply investigated in the literature. The data obtained showed that the perturbation of glutamatergic and GABAergic neurotransmission systems, which play roles in excitatory and inhibitory responses, respectively, contribute to the neurobiology of psychiatric disorders and have been associated with suicide [70,71,72,73,74,75,76,77,78,79]. The mechanisms by which such complex behaviors are not well understood, but likely involve the function of both ionotropic GABAA and metabotropic GABAB receptors (GABAAR and GABABR) [79,80].
In addition, the Attempted class was associated with processes related to adherent junctions. The molecules involved in this cascade have been linked to anxiety and mood disorders [81], which represent substantial risk factors contributing to suicidal behavior [81].
Finally, NOTCH signaling was also associated with the class of suicide attempters. Recent studies have associated alterations of neuronal plasticity in specific brain areas with suicidal behavior [82]. In this context, the NOTCH signaling pathway plays a relevant role in neuronal plasticity as well as cell survival and migration [83], which are biological processes proven to be altered in suicide victims [84]. It also has been identified as critical regulator of neurogenesis and gliogenesis in the adult brain [85,86]. During the last decade, alterations in neurogenesis processes were identified in suicide victims [87]. The alteration of these processes is supported by the structural anomalies that can be observed in different brain areas of suicide attempters such as PFC and HIP [87,88,89]. The role of the NOTCH signaling pathway in neurogenesis strongly suggests its potential involvement in suicidal behavior.
The Hurt and Not Worth classes were found to be linked to less neuron-specific processes: Glycosylation and ExtraCellular Matrix (ECM) Organization. Even though, these biological cascades may not seem to be important for brain functioning, literature data has demonstrated their essential role for the physiological function of the brain [90]. Indeed, a large portion of the brain volume is made up of ECM and its interaction with local cells is essential for functions such as memory and learning [91]. ECM also has a prominent role in brain development, maturation of neural circuits and adult neuroplasticity (cell migration, axonal outgrowth and synaptogenesis) [92]. It also has important roles in neurotransmission and signal transduction since it influences the exchange of ions and neurotransmitters through extracellular space [92,93]. Proteins used to anchor cells to ECM, such as CD44, have been found to be expressed by several brain cells including neurons [94], astrocytes [95] and microglia [96]. Interestingly, these proteins required to organize ECM have been found to be related to suicidal behavior [97,98], likely influencing brain homeostasis through their immune control [90,99] and regulation of the blood-brain barrier permeability [90,100]. This multifactorial role of the ECM suggests that alterations in its organization could potentially lead to impaired brain function and, consequently, increasing the risk for neuropsychiatric and/or neurodegenerative diseases [90,101,102]. The role of ECM is strongly related to glycosylation. In the brain, the formation and structure of the ECM contains the hyaluronic polysaccharide, and a large variety of glycoproteins and proteoglycans [103]. The glycosylation process is tightly regulated as it is an enzymatic modification that is site and substrate specific [104]. Further glycosylation plays an important role in various cellular processes, from cell adhesion and pattern recognition [105] to more specific brain-related roles in controlling neurite sprouting and development [104,106,107]. This process may alter the synthesis of gangliosides, impairing the physiological function of the brain [108]. Even though no literature data exposed a precise causal correlation between altered glycosylation processes and suicidal behavior, this pathway is implicated in several disease states including psychiatric and neurodegenerative disorders [109,110,111,112,113,114,115,116].
5. Conclusions
Bipolar Disorder (BD) is a recurrent, frequent and devastating affective disorder. The current treatments mitigate the exacerbations of BD symptoms at the cost of side effects, including weight gain, together with a non-optimal efficacy. Moreover, suicide rates are higher among individuals with BD. Clinical interviews and treatment alliance are the current strategies employed to decrease suicide rate in BD. There are no current biologic variables that can help identify the BD individuals at risk for suicide. The identification of such variables would prompt a Copernical revolution in the treatment of these individuals. Suicide behavior is a complex phenotype. It is unreasonable to look for the “suicide gene” or “suicide genetic variation”. It is more likely that several genetic variations distributed in different genes concur in shaping the general risk for suicide behavior. Our paper enforces the complex, multigenetic background behind suicidal behavior, since the lack of significant association on single gene analysis, although we should also report some suggestive trends on AOPEP gene. In addition, environmental factors related to distal and proximal history, e.g., early-life adversity and psychiatric disorder also need to be taken in consideration when trying to evaluate the risk of suicide. In particular, including epigenetic and epigenomic modifications in individuals could represent an important step for future studies to include the effects of environment on genes’ expression. Despite this, we have highlighted how some specific biological processes, when altered can contribute to the risk of suicide more than others. We also evidenced slight differences between Suicide Attempters, Not Worth and Hurt classes, with the first more related to neuro-like processes: Neurotransmission, and Development (in particular GABA and NOTCH which were unique for this class). While others interestingly correlated with processes such as ECM organization and glycosylation processes. Growing evidence is available correlating these cascades to brain functions [91,92], suggesting that they may underlie the development and progression of neurologic alterations which can affect suicide risk. In the present research the combined effect of several genetic variations is analyzed in order to identify BD individuals at risk for suicide. A hypothesis—free analysis is conducted throughout the whole genome of each individual. Genes and their embedded variations are grouped in consistent molecular pathways across the whole genome and tested for “enrichment”. That means that the molecular pathways that contain more genetic variations significantly associated with suicide behavior than expected by chance are identified. The result of the present investigation helps in prompting further analysis of specific molecular pathways. This knowledge can be used both for the identification of BD that have a genetic predisposition to suicide behavior, and to the engineering of specific drugs able to tackle the molecular pathways at risk.
Author Contributions
L.K.M.L. and M.C. written the first draft; A.D. retrieved the sample data and performed data analyses; S.B. cured the revision of the first draft and performed further literature search; A.D. and C.C. supervised the study. All authors contributed to write the final version of the manuscript.
Funding
This research received no external funding.
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
The following study was based on STEP-BD public database. Ethics approval and patients’ informed consent to analyses were already included in the dataset.
Data Availability Statement
The Data and biomaterials analyzed in this study were obtained from the STEP-BD study (2N01MH080001-001), available on NIMH Repository with permission of NIMH (https://www.nimhgenetics.org/, accessed on 21 April 2021).
Acknowledgments
Data and biomaterials were collected for the Systematic Treatment Enhancement Program for Bipolar Disorder (STEP-BD), a multi-center, longitudinal (5–8 years) project selected from responses to RFP #NIMH-98-DS-0001, “Treatment for Bipolar Disorder.” The project was led by Gary Sachs, and coordinated by Massachusetts General Hospital in Boston, MA. The NIMH grant number was 2N01MH080001-001. Given the major public health implications of identifying genes responsible for severe neuropsychiatric disorders, the National Institute of Mental Health (NIMH) has funded a Human Genetics Initiative. The goal of this Initiative is to establish a national resource of clinical data and biomaterials that are collected from individuals with Alzheimer disease, schizophrenia or bipolar I disorder (BP), in order to aid researchers in understanding the genetic bases of these disorders. The NIMH Bipolar Disorder Genetics Initiative is supported by the Office of Human Genetics and Genomic Resources in NIMH’s Division of Neuroscience and Basic Behavioral Science (DNBBS). Since 1996, data and biomaterials (cell lines and DNA samples) have been available to qualified investigators who study the genetics of BP and may be accessed by following a set of instructions. We thank Rasmus Licht and René Ernst Nielsen for suggestions and guide in the writing process.
Conflicts of Interest
The authors declare no conflict of interest.
Appendix A. Definitions of Suicide—Related Phenotypes
The term suicidal ideation, i.e., suicidal thoughts or ideas, describes a broad range of contemplations, wishes and preoccupations with death and suicide. There is no established definition of suicidal ideation, making the clinical assessment difficult, and also research is hindered by the need of operational definitions leading to inability to compare findings [117].
Suicide attempt is defined by WHO as “any non-fatal suicidal behavior”, further described as any “intentional self-inflicted poisoning, injury or self-harm which may or may not have a fatal intent or outcome”, while suicide is defined as “the act of deliberately killing oneself”. Worldwide approximately 800,000 individuals commit suicide each year, it is the 10th leading cause of death overall and the number is probably underestimated due to the sensitivity of the circumstance and countries where suicide is illegal.
Appendix B. Clinical Risk Factors and Prevention Measures for Suicide Behavior in BD
Risk factors—Some risk factors are seen as general risk factors for suicide, especially regarding socio-demographic factors; gender (male), marital status (single, widowed, divorced), living alone, age under 35 years or over 75 years, no children and unemployment. In addition, some clinical risk factors are overall risk factors for committing suicide, e.g., history of suicide attempt and family history of completed suicide. Other clinical risk factors are related to the characteristics and phase of the illness: predominant depressive episode, major depressive episode, rapid cycling subtype, earlier age of onset, longer duration of untreated illness, previous hospitalization, concurrent medical comorbidity and mood-incongruent psychotic symptoms. Psychiatric comorbidity in the form of personality disorders (borderline, antisocial, histrionic and narcissistic) also higher the risk.
Prevention strategies—When it comes to the prevention of suicide in BD, several studies have shown the anti-suicidal effect of lithium treatment, including a large Swedish study with over 50,000 patients. The register-based longitudinal study found a decreased incidence of suicide-related events of 14% compared to those that received valproate [118]. Another study found a 5-fold greater risk of suicide attempts among patients with poor adherence to long-term lithium maintenance treatment compared to those with high adherence [119]. Electroconvulsive therapy (ECT) has proven to be effective in treating acute suicidal danger in severely depressed patients; furthermore ECT also prevents subsequent suicidal behavior [120]. Medical treatment should never stand alone; a general national guideline for preventing suicide could consist of education of the patient as well as friends and family, a 24-h telephone hotline, the development of a personalized safety planning and for some psychotherapy.
References
- Miller, J.; Black, D.W. Bipolar disorder and suicide: A review. Curr. Psychiatry Rep. 2020, 22, 6. [Google Scholar] [CrossRef]
- Hansen, P.S.; Laursen, M.F.; Grøntved, S.; Straszek, S.P.V.; Licht, R.W.; Nielsen, R.E. Increasing mortality gap for patients diagnosed with bipolar disorder-A nationwide study with 20 years of follow-up. Bipolar Disord. 2019, 21, 270–275. [Google Scholar] [CrossRef]
- Crump, C.; Sundquist, K.; Winkleby, M.A.; Sundquist, J. Comorbidities and mortality in bipolar disorder: A Swedish national cohort study. JAMA Psychiatry 2013, 70, 931–939. [Google Scholar] [CrossRef]
- Pinto, J.V.; Saraf, G.; Kozicky, J.; Beaulieu, S.; Sharma, V.; Parikh, S.V.; Yatham, L.N. Remission and recurrence in bipolar disorder: The data from health outcomes and patient eval-uations in bipolar disorder (HOPE-BD) study. J Affect. Disord. 2020, 268, 150–157. [Google Scholar] [CrossRef] [PubMed]
- Bauer, M.; Pfennig, A. Epidemiology of Bipolar Disorders. Epilepsia 2005, 46, 8–13. [Google Scholar] [CrossRef]
- Gonda, X.; Pompili, M.; Serafini, G.; Montebovi, F.; Campi, S.; Dome, P.; Duleba, T.; Girardi, P.; Rihmer, Z. Suicidal behavior in bipolar disorder: Epidemiology, characteristics and major risk factors. J. Affect. Disord. 2012, 143, 16–26. [Google Scholar] [CrossRef] [PubMed]
- Voracek, M.; Loibl, L.M. Genetics of suicide: A systematic review of twin studies. Wien. Klin. Wochenschr. 2007, 119, 463–475. [Google Scholar] [CrossRef]
- Brent, D.A.; Mann, J.J. Family genetic studies, suicide, and suicidal behavior. Am. J. Med. Genet. Part C Semin. Med. Genet. 2005, 133C, 13–24. [Google Scholar] [CrossRef]
- Mullins, N.; Bigdeli, T.B.; Børglum, A.; Coleman, J.; Demontis, D.; Mehta, D.D.; Power, R.A.; Ripke, S.; Stahl, E.A.; Starnawska, A.; et al. GWAS of suicide attempt in psychiatric disorders and association with major depression polygenic risk scores. Am. J. Psychiatry 2019, 176, 651–660. [Google Scholar] [CrossRef]
- Ruderfer, D.M.; Walsh, C.G.; Aguirre, M.W.; Tanigawa, Y.; Ribeiro, J.D.; Franklin, J.C.; Rivas, M.A. Significant shared heritability underlies suicide attempt and clinically predicted probability of attempting suicide. Mol. Psychiatry 2020, 25, 2422–2430. [Google Scholar] [CrossRef] [PubMed]
- Stein, M.B.; Ware, E.B.; Mitchell, C.; Chen, C.-Y.; Borja, S.; Cai, T.; Dempsey, C.L.; Fullerton, C.S.; Gelernter, J.; Heeringa, S.G.; et al. Genomewide association studies of suicide attempts in US soldiers. Am. J. Med. Genet. Part B Neuropsychiatr. Genet. 2017, 174, 786–797. [Google Scholar] [CrossRef]
- Mullins, N.; Perroud, N.; Uher, R.; Butler, A.W.; Cohen-Woods, S.; Rivera, M.; Malki, K.; Euesden, J.; Power, R.; Tansey, K.; et al. Genetic relationships between suicide attempts, suicidal ideation and major psychiatric disorders: A genome-wide association and polygenic scoring study. Am. J. Med. Genet. Part B Neuropsychiatr. Genet. 2014, 165, 428–437. [Google Scholar] [CrossRef]
- Galfalvy, H.; Haghighi, F.; Hodgkinson, C.; Goldman, D.; Oquendo, M.A.; Burke, A.; Huang, Y.-Y.; Giegling, I.; Rujescu, D.; Bureau, A.; et al. A genome-wide association study of suicidal behavior. Am. J. Med. Genet. Part B Neuropsychiatr. Genet. 2015, 168, 557–563. [Google Scholar] [CrossRef] [PubMed]
- Erlangsen, A.; Appadurai, V.; Wang, Y.; Turecki, G.; Mors, O.; Werge, T.; Mortensen, P.B.; Starnawska, A.; Børglum, A.; Schork, A.; et al. Genetics of suicide attempts in individuals with and without mental disorders: A population-based genome-wide association study. Mol. Psychiatry 2020, 25, 2410–2421. [Google Scholar] [CrossRef] [PubMed]
- Sachs, G.S.; Thase, M.E.; Otto, M.; Bauer, M.; Miklowitz, D.; Wisniewski, S.; Lavori, P.; Lebowitz, B.; Rudorfer, M.; Frank, E.; et al. Rationale, design, and methods of the systematic treatment enhancement program for bipolar disorder (STEP-BD). Biol. Psychiatry 2003, 53, 1028–1042. [Google Scholar] [CrossRef]
- Bellivier, F.; Laplanche, J.-L.; Leboyer, M.; Feingold, J.; Bottos, C.; Allilaire, J.-F.; Launay, J.-M. Serotonin transporter gene and manic depressive illness: An association study. Biol. Psychiatry 1997, 41, 750–752. [Google Scholar] [CrossRef]
- Ohara, K.; Nagai, M.; Tsukamoto, T.; Tani, K.; Suzuki, Y.; Ohara, K. 5-HT2A receptor gene promoter polymor-phism--1438G/A and mood disorders. Neuroreport 1998, 9, 1139–1141. [Google Scholar] [CrossRef]
- Ohara, K.; Nagai, M.; Tsukamoto, T.; Tani, K.; Suzuki, Y.; Ohara, K. Functional polymorphism in the serotonin transporter promoter at the SLC6A4 locus and mood disorders. Biol. Psychiatry 1998, 44, 550–554. [Google Scholar] [CrossRef]
- Rujescu, D.; Giegling, I.; Dahmen, N.; Szegedi, A.; Anghelescu, I.; Gietl, A.; Schäfer, M.; Müller-Siecheneder, F.; Bondy, B.; Möller, H.J. Association study of suicidal behavior and affective disorders with a genetic polymorphism in ABCG1, a positional candidate on chromosome 21q22.3. Neuropsychobiology 2000, 42, 22–25. [Google Scholar] [CrossRef]
- Zubenko, G.S.; Maher, B.S.; Hughes HB 3rd Zubenko, W.N.; Scott Stiffler, J.; Marazita, M.L. Genome-wide linkage survey for genetic loci that affect the risk of suicide attempts in families with recurrent, early-onset, major depression. Am. J. Med. Genet. B Neuropsychiatr. Genet. 2004, 129B, 47–54. [Google Scholar] [CrossRef]
- Hesselbrock, V.; Dick, D.; Hesselbrock, M.; Foroud, T.; Schuckit, M.; Edenberg, H.; Bucholz, K.; Kramer, J.; Reich, T.; Goate, A.; et al. The search for genetic risk factors associated with suicidal behavior. Alcohol. Clin. Exp. Res. 2004, 28, 70S–76S. [Google Scholar] [CrossRef]
- De Lara, C.L.; Dumais, A.; Rouleau, G.; Lesage, A.; Dumont, M.; Chawky, N.; Alda, M.; Benkelfat, C.; Turecki, G. STin2 Variant and Family History of Suicide as Significant Predictors of Suicide Completion in Major Depression. Biol. Psychiatry 2006, 59, 114–120. [Google Scholar] [CrossRef]
- Cheng, R.; Juo, S.H.; Loth, J.E.; Nee, J.; Iossifov, I.; Blumenthal, R.; Sharpe, L.; Kanyas, K.; Lerer, B.; Lilliston, B.; et al. Genome-wide linkage scan in a large bipolar disorder sample from the National Institute of Mental Health genetics initiative suggests putative loci for bipolar disorder, psychosis, suicide, and panic disorder. Mol. Psychiatry 2006, 11, 252–260. [Google Scholar] [CrossRef]
- Willour, V.L.; Zandi, P.P.; Badner, J.A.; Steele, J.; Miao, K.; Lopez, V.; MacKinnon, D.F.; Mondimore, F.M.; Schweizer, B.; McInnis, M.G.; et al. Attempted Suicide in Bipolar Disorder Pedigrees: Evidence for Linkage to 2p12. Biol. Psychiatry 2007, 61, 725–727. [Google Scholar] [CrossRef] [PubMed]
- Iga, J.-I.; Ueno, S.-I.; Yamauchi, K.; Numata, S.; Tayoshi-Shibuya, S.; Kinouchi, S.; Nakataki, M.; Song, H.; Hokoishi, K.; Tanabe, H.; et al. The Val66Met polymorphism of the brain-derived neurotrophic factor gene is associated with psychotic feature and suicidal behavior in Japanese major depressive patients. Am. J. Med. Genet. B Neuropsychiatr. Genet. 2007, 144B, 1003–1006. [Google Scholar] [CrossRef] [PubMed]
- Perlis, R.H.; Huang, J.; Purcell, S.; Fava, M.; Rush, A.J.; Sullivan, P.F.; Hamilton, S.P.; McMahon, F.J.; Schulze, T.; Potash, J.B.; et al. Genome-wide association study of suicide attempts in mood disorder patients. Am. J. Psychiatry 2010, 167, 1499–1507. [Google Scholar] [CrossRef]
- Schosser, A.; Butler, A.W.; Ising, M.; Perroud, N.; Uher, R.; Ng, M.Y.; Cohen-Woods, S.; Craddock, N.; Owen, M.J.; Korszun, A.; et al. Genomewide Association Scan of Suicidal Thoughts and Behaviour in Major Depression. PLoS ONE 2011, 6, e20690. [Google Scholar] [CrossRef] [PubMed]
- Willour, V.L.; Seifuddin, F.; Mahon, P.B.; Jancic, D.; Pirooznia, M.; Steele, J.; Schweizer, B.; Goes, F.S.; Mondimore, F.M.; Mackinnon, D.F.; et al. A genome-wide association study of attempted suicide. Mol. Psychiatry 2011, 17, 433–444. [Google Scholar] [CrossRef]
- Schosser, A.; Calati, R.; Serretti, A.; Massat, I.; Kocabas, N.A.; Papageorgiou, K.; Linotte, S.; Mendlewicz, J.; Souery, D.; Zohar, J.; et al. The impact of COMT gene polymorphisms on suicidality in treatment resistant major depressive disorder—A European Multicenter Study. Eur. Neuropsychopharmacol. 2012, 22, 259–266. [Google Scholar] [CrossRef]
- Zai, C.C.; Gonçalves, V.F.; Tiwari, A.K.; Gagliano, S.A.; Hosang, G.; de Luca, V.; Shaikh, S.A.; King, N.; Chen, Q.; Xu, W.; et al. A genome-wide association study of suicide severity scores in bipolar disorder. J. Psychiatr. Res. 2015, 65, 23–29. [Google Scholar] [CrossRef]
- Gross, J.A.; Bureau, A.; Croteau, J.; Galfalvy, H.; Oquendo, M.A.; Haghighi, F.; Turecki, G. A genome-wide copy number variant study of suicidal behavior. PLoS ONE 2015, 10, e0128369. [Google Scholar] [CrossRef]
- Sokolowski, M.; Wasserman, J. Polygenic associations of neurodevelopmental genes in suicide attempt. Mol. Psychiatry 2016, 21, 1381–1390. [Google Scholar] [CrossRef] [PubMed]
- Laursen, T.M.; Trabjerg, B.B.; Mors, O.; Børglum, A.; Hougaard, D.M.; Mattheisen, M.; Meier, S.M.; Byrne, E.; Mortensen, P.B.; Munk-Olsen, T.; et al. Association of the polygenic risk score for schizophrenia with mortality and suicidal behavior—A Danish population-based study. Schizophr. Res. 2017, 184, 122–127. [Google Scholar] [CrossRef] [PubMed]
- Levey, D.F.; Polimanti, R.; Cheng, Z.; Zhou, H.; Nuñez, Y.Z.; Jain, S.; He, F.; Sun, X.; Ursano, R.J.; Kessler, R.C.; et al. Genetic associations with suicide attempt severity and genetic overlap with major depression. Transl. Psychiatry 2019, 9, 22. [Google Scholar] [CrossRef]
- Sheehan, D.V.; Lecrubier, Y.; Sheehan, K.H.; Amorim, P.; Janavs, J.; Weiller, E.; Hergueta, T.; Baker, R.; Dunbar, G.C. The Mini-International Neuropsychiatric Interview (M.I.N.I.): The development and validation of a structured diagnostic psychiatric interview for DSM-IV and ICD-10. J. Clin. Psychiatry 1998, 59, 22–33. [Google Scholar] [PubMed]
- Bowden, C.; Perlis, R.; Thase, M.; Ketter, T.; Ostacher, M.; Calabrese, J.; Reilly-Harrington, N.; Gonzalez, J.; Singh, V.; Nierenberg, A.; et al. Aims and Results of the NIMH Systematic Treatment Enhancement Program for Bipolar Disorder (STEP-BD). CNS Neurosci. Ther. 2011, 18, 243–249. [Google Scholar] [CrossRef]
- Team R Core. R: A Language and Environment for Statistical Computing; R Foundation for Statistical Computing: Vienna, Austria, 2020. [Google Scholar]
- Purcell, S.; Neale, B.; Todd-Brown, K.; Thomas, L.; Ferreira, M.A.R.; Bender, D.; Maller, J.; Sklar, P.; de Bakker, P.I.W.D.; Daly, M.J.; et al. PLINK: A Tool Set for Whole-genome association and population-based linkage analyses. Am. J. Hum. Genet. 2007, 81, 559–575. [Google Scholar] [CrossRef]
- Howie, B.N.; Donnelly, P.; Marchini, J. A flexible and accurate genotype imputation method for the next generation of genome-wide association studies. PLoS Genet. 2009, 5, e1000529. [Google Scholar] [CrossRef]
- Huber, W.; Carey, V.J.; Gentleman, R.; Anders, S.; Carlson, M.; Carvalho, B.S.; Bravo, H.C.; Davis, S.; Gatto, L.; Girke, T.; et al. Orchestrating high-throughput genomic analysis with Bioconductor. Nat. Methods 2015, 12, 115–121. [Google Scholar] [CrossRef] [PubMed]
- Yu, G.; He, Q.-Y. ReactomePA: An R/Bioconductor package for reactome pathway analysis and visualization. Mol. BioSyst. 2016, 12, 477–479. [Google Scholar] [CrossRef] [PubMed]
- Corfitsen, H.T.; Drago, A. Enriched developmental biology molecular pathways impact on antipsychotics-induced weight gain. Pharm. Genom. 2020, 30, 9–20. [Google Scholar] [CrossRef] [PubMed]
- Corfitsen, H.T.; Krantz, B.; Larsen, A.; Drago, A. Molecular pathway analysis associates alterations in obesity-related genes and antipsychotic-induced weight gain. Acta Neuropsychiatr. 2020, 32, 72–83. [Google Scholar] [CrossRef] [PubMed]
- Drago, A.; Fischer, E.K. A molecular pathway analysis informs the genetic risk for arrhythmias during antipsychotic treatment. Int. Clin. Psychopharmacol. 2018, 33, 1–14. [Google Scholar] [CrossRef] [PubMed]
- Fischer, E.K.; Drago, A. A molecular pathway analysis stresses the role of inflammation and oxidative stress towards cognition in schizophrenia. J. Neural Transm. 2017, 124, 765–774. [Google Scholar] [CrossRef]
- O’Dushlaine, C.; Kenny, E.; Heron, E.; Donohoe, G.; Gill, M.; Morris, D.; International Schizophrenia Consortium; Corvin, A. Molecular pathways involved in neuronal cell adhesion and membrane scaffolding contribute to schizophrenia and bipolar disorder susceptibility. Mol. Psychiatry 2011, 16, 286–292. [Google Scholar] [CrossRef]
- Ichikawa, M.; Konoshita, T.; Makino, Y.; Suzuki, J.; Ishizuka, T.; Nakamura, H. An association study of C9orf3, a novel component of the renin-angiotensin system, and hypertension in diabetes. Sci. Rep. 2020, 10, 1–7. [Google Scholar] [CrossRef]
- Zeber-Lubecka, N.; Hennig, E.E. Genetic susceptibility to joint occurrence of polycystic ovary syndrome and hashimoto’s thyroiditis: How far is our understanding? Front. Immunol. 2021, 12, 606620. [Google Scholar] [CrossRef] [PubMed]
- Hayes, M.G.; Urbanek, M.; Ehrmann, D.A.; Armstrong, L.L.; Lee, J.Y.; Sisk, R.; Karaderi, T.; Barber, T.M.; McCarthy, M.I.; Franks, S.; et al. Genome-wide association of polycystic ovary syndrome implicates alterations in gonadotropin secretion in European ancestry populations. Nat. Commun. 2015, 6, 7502. [Google Scholar] [CrossRef]
- Louwers, Y.V.; Stolk, L.; Uitterlinden, A.G.; Laven, J.S. Cross-ethnic meta-analysis of genetic variants for polycystic ovary syndrome. J. Clin. Endocrinol. Metab. 2013, 98, E2006–E2012. [Google Scholar] [CrossRef] [PubMed]
- Day, F.R.; Hinds, D.A.; Tung, J.Y.; Stolk, L.; Styrkarsdottir, U.; Saxena, R.; Bjonnes, A.; Broer, L.; Dunger, D.B.; Halldórsson, B.V.; et al. Causal mechanisms and balancing selection inferred from genetic associations with polycystic ovary syndrome. Nat. Commun. 2015, 6, 8464. [Google Scholar] [CrossRef]
- Brower, M.A.; Jones, M.R.; Rotter, J.I.; Krauss, R.M.; Legro, R.S.; Azziz, R.; Goodarzi, M.O. Further investigation in europeans of susceptibility variants for polycystic ovary syndrome discovered in genome-wide association studies of Chinese individuals. J. Clin. Endocrinol. Metab. 2015, 100, E182–E186. [Google Scholar] [CrossRef] [PubMed]
- Day, F.; Karaderi, T.; Jones, M.R.; Meun, C.; He, C.; Drong, A.; Kraft, P.; Lin, N.; Huang, H.; Broer, L.; et al. Large-scale genome-wide meta-analysis of polycystic ovary syndrome suggests shared genetic architecture for different diagnosis criteria. PLoS Genet. 2018, 14, e1007813. [Google Scholar] [CrossRef]
- Nishida, K.; Kuwano, Y.; Rokutan, K. The MicroRNA-23b/27b/24 Cluster Facilitates Colon Cancer Cell Migration by Targeting FOXP2. Cancers 2020, 12, 174. [Google Scholar] [CrossRef]
- Jiang, Y.-H.; Man, Y.-Y.; Liu, Y.; Yin, C.-J.; Li, J.-L.; Shi, H.-C.; Zhao, H.; Zhao, S.-G. Loss of miR-23b/27b/24-1 Cluster Impairs Glucose Tolerance via Glycolysis Pathway in Mice. Int. J. Mol. Sci. 2021, 22, 550. [Google Scholar] [CrossRef] [PubMed]
- McAlinden, A.; Varghese, N.; Wirthlin, L.; Chang, L.-W. Differentially Expressed MicroRNAs in Chondrocytes from Distinct Regions of Developing Human Cartilage. PLoS ONE 2013, 8, e75012. [Google Scholar] [CrossRef] [PubMed]
- He, H.-C.; Zhu, J.-G.; Chen, X.-B.; Chen, S.-M.; Han, Z.-D.; Dai, Q.-S.; Ling, X.-H.; Fu, X.; Lin, Z.-Y.; Deng, Y.-H.; et al. MicroRNA-23b downregulates peroxiredoxin III in human prostate cancer. FEBS Lett. 2012, 586, 2451–2458. [Google Scholar] [CrossRef]
- Bang, C.; Fiedler, J.; Thum, T. Cardiovascular importance of the microRNA-23/27/24 family. Microcirculation 2012, 19, 208–214. [Google Scholar] [CrossRef] [PubMed]
- Zhu, S.; Pan, W.; Song, X.; Liu, Y.; Shao, X.; Tang, Y.; Liang, D.; He, D.; Wang, H.; Liu, W.; et al. The microRNA miR-23b suppresses IL-17-associated autoimmune inflammation by targeting TAB2, TAB3 and IKK-α. Nat. Med. 2012, 18, 1077–1086. [Google Scholar] [CrossRef]
- Boon, R.A.; Hergenreider, E.; Dimmeler, S. Atheroprotective mechanisms of shear stress-regulated microRNAs. Thromb. Haemost. 2012, 108, 616–620. [Google Scholar] [CrossRef] [PubMed]
- Chen, Q.; Xu, J.; Li, L.; Li, H.; Mao, S.; Zhang, F.; Zen, K.; Zhang, C.-Y.; Zhang, Q. MicroRNA-23a/b and microRNA-27a/b suppress Apaf-1 protein and alleviate hypoxia-induced neuronal apoptosis. Cell Death Dis. 2014, 5, e1132. [Google Scholar] [CrossRef]
- Johnson, C.E.; Huang, Y.Y.; Parrish, A.B.; Smith, M.I.; Vaughn, A.E.; Zhang, Q.; Wright, K.; Van Dyke, T.; Wechsler-Reya, R.J.; Kornbluth, S.; et al. Differential Apaf-1 levels allow cytochrome c to induce apoptosis in brain tumors but not in normal neural tissues. Proc. Natl. Acad. Sci. USA 2007, 104, 20820–20825. [Google Scholar] [CrossRef]
- Lopez, J.P.; Fiori, L.M.; Gross, J.A.; LaBonte, B.; Yerko, V.; Mechawar, N.; Turecki, G. Regulatory role of miRNAs in polyamine gene expression in the prefrontal cortex of depressed suicide completers. Int. J. Neuropsychopharmacol. 2013, 17, 23–32. [Google Scholar] [CrossRef]
- Kouter, K.; Paska, A.V. Biomarkers for suicidal behavior: miRNAs and their potential for diagnostics through liquid biopsy—A systematic review. Epigenomics 2020, 12, 2219–2235. [Google Scholar]
- Cox, J.J.; Reimann, F.; Nicholas, A.K.; Thornton, G.; Roberts, E.; Springell, K.; Karbani, G.; Jafri, H.; Mannan, J.; Raashid, Y.; et al. An SCN9A channelopathy causes congenital inability to experience pain. Nat. Cell Biol. 2006, 444, 894–898. [Google Scholar] [CrossRef] [PubMed]
- Fertleman, C.R.; Baker, M.D.; Parker, K.A.; Moffatt, S.; Elmslie, F.V.; Abrahamsen, B.; Ostman, J.; Klugbauer, N.; Wood, J.N.; Gardiner, R.M.; et al. SCN9A mutations in paroxysmal extreme pain disorder: Allelic variants underlie distinct channel defects and phenotypes. Neuron 2006, 52, 767–774. [Google Scholar] [CrossRef] [PubMed]
- Han, C.; Dib-Hajj, S.D.; Lin, Z.; Li, Y.; Eastman, E.M.; Tyrrell, L.; Cao, X.; Yang, Y.; Waxman, S.G. Early- and late-onset inherited erythromelalgia: Genotype–phenotype correlation. Brain 2009, 132, 1711–1722. [Google Scholar] [CrossRef] [PubMed]
- Eberhardt, M.; Nakajima, J.; Klinger, A.B.; Neacsu, C.; Hühne, K.; O’Reilly, A.O.; Kist, A.M.; Lampe, A.K.; Fischer, K.; Gibson, J. Inherited pain: Sodium channel Nav1.7 A1632T mutation causes erythromelalgia due to a shift of fast inactivation. J. Biol. Chem. 2014, 289, 1971–1980. [Google Scholar] [CrossRef]
- Tsai, M.; Lari, H.; Saffy, S.; Klonsky, E.D. Examining the Three-Step Theory (3ST) of Suicide in a Prospective Study of Adult Psychiatric Inpatients. Behav. Ther. 2021, 52, 673–685. [Google Scholar] [CrossRef]
- Klempan, T.A.; Sequeira, A.; Canetti, L.; Lalovic, A.; Ernst, C.; Ffrench-Mullen, J.; Turecki, G. Altered expression of genes involved in ATP biosynthesis and GABAergic neurotransmission in the ventral prefrontal cortex of suicides with and without major depression. Mol. Psychiatry 2007, 14, 175–189. [Google Scholar] [CrossRef] [PubMed]
- Pantazatos, S.P.; Huang, Y.-Y.; Rosoklija, G.B.; Dwork, A.J.; Arango, V.; Mann, J.J. Whole-transcriptome brain expression and exon-usage profiling in major depression and suicide: Evidence for altered glial, endothelial and ATPase activity. Mol. Psychiatry 2017, 22, 760–773. [Google Scholar] [CrossRef] [PubMed]
- Duman, R.S.; Sanacora, G.; Krystal, J.H. Altered connectivity in depression: GABA and glutamate neurotransmitter deficits and reversal by novel treatments. Neuron 2019, 102, 75–90. [Google Scholar] [CrossRef]
- Levinson, A.J.; Fitzgerald, P.; Favalli, G.; Blumberger, D.M.; Daigle, M.; Daskalakis, Z.J. Evidence of cortical inhibitory deficits in major depressive disorder. Biol. Psychiatry 2010, 67, 458–464. [Google Scholar] [CrossRef] [PubMed]
- Zhao, J.; Verwer, R.; Gao, S.-F.; Qi, X.-R.; Lucassen, P.J.; Kessels, H.; Swaab, D. Prefrontal alterations in GABAergic and glutamatergic gene expression in relation to depression and suicide. J. Psychiatr. Res. 2018, 102, 261–274. [Google Scholar] [CrossRef]
- Ghosal, S.; Bang, E.; Yue, W.; Hare, B.D.; Lepack, A.E.; Girgenti, M.J.; Duman, R.S. Activity-dependent brain-derived neu-rotrophic factor release is required for the rapid antidepressant actions of scopolamine. Biol. Psychiatry 2018, 83, 29–37. [Google Scholar] [CrossRef] [PubMed]
- Lener, M.S.; Niciu, M.J.; Ballard, E.D.; Park, M.; Park, L.T.; Nugent, A.C.; Zarate, C.A., Jr. Glutamate and γ-aminobutyric acid systems in the pathophysiology of major depression and antidepressant response to ketamine. Biol. Psychiatry 2016, 81, 886–897. [Google Scholar] [CrossRef]
- Lin, L.C.; Sibille, E. Somatostatin, neuronal vulnerability and behavioral emotionality. Mol. Psychiatry 2015, 20, 377–387. [Google Scholar] [CrossRef] [PubMed]
- Godfrey, K.E.M.; Gardner, A.C.; Kwon, S.; Chea, W.; Muthukumaraswamy, S.D. Differences in excitatory and inhibitory neu-rotransmitter levels between depressed patients and healthy controls: A systematic review and meta-analysis. J. Psychiatr. Res. 2018, 105, 33–44. [Google Scholar] [CrossRef]
- Fee, C.; Banasr, M.; Sibille, E. Somatostatin-positive γ-aminobutyric acid interneuron deficits in depression: Cortical microcircuit and therapeutic perspectives. Biol. Psychiatry 2017, 82, 549–559. [Google Scholar] [CrossRef]
- Radhu, N.; de Jesus, D.R.; Ravindran, L.N.; Zanjani, A.; Fitzgerald, P.; Daskalakis, Z.J. A meta-analysis of cortical inhibition and excitability using transcranial magnetic stimulation in psychiatric disorders. Clin. Neurophysiol. 2013, 124, 1309–1320. [Google Scholar] [CrossRef] [PubMed]
- Tanti, A.; Lutz, P.-E.; Kim, J.; O’Leary, L.; Théroux, J.-F.; Turecki, G.; Mechawar, N. Evidence of decreased gap junction coupling between astrocytes and oligodendrocytes in the anterior cingulate cortex of depressed suicides. Neuropsychopharmacology 2019, 44, 2099–2111. [Google Scholar] [CrossRef]
- Monsalve, E.M.; García-Gutiérrez, M.S.; Navarrete, F.; Giner, S.; Laborda, J.; Manzanares, J. Abnormal expression pattern of notch receptors, ligands, and downstream effectors in the dorsolateral prefrontal cortex and amygdala of suicidal victims. Mol. Neurobiol. 2013, 49, 957–965. [Google Scholar] [CrossRef]
- Alberi, L.; Liu, S.; Wang, Y.; Badie, R.; Smith-Hicks, C.; Wu, J.; Pierfelice, T.J.; Abazyan, B.; Mattson, M.P.; Kuhl, D.; et al. Activity-induced notch signaling in neurons requires Arc/Arg3.1 and is essential for synaptic plasticity in hippocampal networks. Neuron 2011, 69, 437–444. [Google Scholar] [CrossRef]
- Torres-Platas, S.G.; Hercher, C.; Davoli, M.A.; Maussion, G.; LaBonte, B.; Turecki, G.; Mechawar, N. Astrocytic hypertrophy in anterior cingulate white matter of depressed suicides. Neuropsychopharmacology 2011, 36, 2650–2658. [Google Scholar] [CrossRef]
- Bagheri-Mohammadi, S. Adult neurogenesis and the molecular signalling pathways in brain: The role of stem cells in adult hippocampal neurogenesis. Int. J. Neurosci. 2021, 1–13. [Google Scholar] [CrossRef]
- Snyder, J.L.; Kearns, C.A.; Appel, B. Fbxw7 regulates Notch to control specification of neural precursors for oligodendrocyte fate. Neural Dev. 2012, 7, 15. [Google Scholar] [CrossRef] [PubMed]
- Dwivedi, Y. Brain-derived neurotrophic factor in suicide pathophysiology. In The Primate Visual System; CRC Press: Boca Raton, FL, USA, 2012. [Google Scholar]
- Soloff, P.H.; Pruitt, P.; Sharma, M.; Radwan, J.; White, R.; Diwadkar, V.A. Structural brain abnormalities and suicidal behavior in borderline personality disorder. J. Psychiatr. Res. 2012, 46, 516–525. [Google Scholar] [CrossRef] [PubMed]
- Hwang, J.-P.; Lee, T.-W.; Tsai, S.-J.; Chen, T.-J.; Yang, C.-H.; Lirng, J.-F.; Tsai, C.-F. Cortical and subcortical abnormalities in late-onset depression with history of suicide attempts investigated with MRI and Voxel-based morphometry. J. Geriatr. Psychiatry Neurol. 2010, 23, 171–184. [Google Scholar] [CrossRef]
- Ventorp, F.; Barzilay, R.; Erhardt, S.; Samuelsson, M.; Träskman-Bendz, L.; Janelidze, S.; Weizman, A.; Offen, D.; Brundin, L. The CD44 ligand hyaluronic acid is elevated in the cerebrospinal fluid of suicide attempters and is associated with increased blood–brain barrier permeability. J. Affect. Disord. 2016, 193, 349–354. [Google Scholar] [CrossRef] [PubMed]
- Dityatev, A.; Schachner, M.; Sonderegger, P. The dual role of the extracellular matrix in synaptic plasticity and homeostasis. Nat. Rev. Neurosci. 2010, 11, 735–746. [Google Scholar] [CrossRef] [PubMed]
- Gundelfinger, E.D.; Frischknecht, R.; Choquet, D.; Heine, M. Converting juvenile into adult plasticity: A role for the brain’s extracellular matrix. Eur. J. Neurosci. 2010, 31, 2156–2165. [Google Scholar] [CrossRef] [PubMed]
- Tamura, H.; Ishikawa, Y.; Shiosaka, S. Does extracellular proteolysis control mammalian cognition? Rev. Neurosci. 2013, 24, 365–374. [Google Scholar] [CrossRef]
- Glezer, I.; Bittencourt, J.C.; Rivest, S. Neuronal expression of Cd36, Cd44, and Cd83 antigen transcripts maps to distinct and specific murine brain circuits. J. Comp. Neurol. 2009, 517, 906–924. [Google Scholar] [CrossRef] [PubMed]
- Kaaijk, P.; Pals, S.T.; Morsink, F.; Bosch, D.; Troost, D. Differential expression of CD44 splice variants in the normal human central nervous system. J. Neuroimmunol. 1997, 73, 70–77. [Google Scholar] [CrossRef]
- Jones, L.L.; Liu, Z.; Shen, J.; Werner, A.; Kreutzberg, G.W.; Raivich, G. Regulation of the cell adhesion molecule CD44 after nerve transection and direct trauma to the mouse brain. J. Comp. Neurol. 2000, 426, 468–492. [Google Scholar] [CrossRef]
- Thalmeier, A.; Dickmann, M.; Giegling, I.; Schneider, B.; Hartmann, A.M.; Maurer, K.; Schnabel, A.; Kauert, G.; Möller, H.-J.; Rujescu, D. Gene expression profiling of post-mortem orbitofrontal cortex in violent suicide victims. Int. J. Neuropsychopharmacol. 2007, 11, 217–228. [Google Scholar] [CrossRef] [PubMed]
- Galfalvy, H.; Zalsman, G.; Huang, Y.-Y.; Murphy, L.; Rosoklija, G.; Dwork, A.J.; Haghighi, F.; Arango, V.; Mann, J.J. A pilot genome wide association and gene expression array study of suicide with and without major depression. World J. Biol. Psychiatry 2011, 14, 574–582. [Google Scholar] [CrossRef]
- Serafini, G.; Parisi, V.M.; Aguglia, A.; Amerio, A.; Sampogna, G.; Fiorillo, A.; Pompili, M.; Amore, M. A Specific inflammatory profile underlying suicide risk? Systematic review of the main literature findings. Int. J. Environ. Res. Public Health 2020, 17, 2393. [Google Scholar] [CrossRef] [PubMed]
- Zetterberg, H.; Jakobsson, J.; Redsäter, M.; Andreasson, U.; Pålsson, E.; Ekman, C.J.; Sellgren, C.; Johansson, A.G.; Blennow, K.; Landén, M. Blood–cerebrospinal fluid barrier dysfunction in patients with bipolar disorder in relation to antipsychotic treatment. Psychiatry Res. 2014, 217, 143–146. [Google Scholar] [CrossRef] [PubMed]
- Lubbers, B.R.; Smit, A.B.; Spijker, S.; Oever, M.C.V.D. Neural ECM in addiction, schizophrenia, and mood disorder. Prog. Brain Res. 2014, 214, 263–284. [Google Scholar] [PubMed]
- Robinson, A. Living history: An autobiography of Arthur Robinson. Am. J. Med. Genet. 1990, 35, 475–480. [Google Scholar] [CrossRef]
- Zimmermann, D.R.; Dours-Zimmermann, M.T. Extracellular matrix of the central nervous system: From neglect to challenge. Histochem. Cell Biol. 2008, 130, 635–653. [Google Scholar] [CrossRef]
- Abou-Abbass, H.; Abou-El-Hassan, H.; Bahmad, H.; Zibara, K.; Zebian, A.; Youssef, R.; Ismail, J.; Zhu, R.; Zhou, S.; Dong, X.; et al. Glycosylation and other PTMs alterations in neurodegenerative diseases: Current status and future role in neurotrauma. Electrophoresis 2016, 37, 1549–1561. [Google Scholar] [CrossRef]
- Wei, X.; Li, L. Comparative glycoproteomics: Approaches and applications. Briefings Funct. Genom. Proteom. 2009, 8, 104–113. [Google Scholar] [CrossRef] [PubMed]
- Yi, J.-H.; Katagiri, Y.; Susarla, B.; Figge, D.; Symes, A.; Geller, H.M. Alterations in sulfated chondroitin glycosaminoglycans following controlled cortical impact injury in mice. J. Comp. Neurol. 2012, 520, 3295–3313. [Google Scholar] [CrossRef] [PubMed]
- Myer, D.J.; Gurkoff, G.G.; Lee, S.M.; Hovda, D.A.; Sofroniew, M.V. Essential protective roles of reactive astrocytes in traumatic brain injury. Brain 2006, 129, 2761–2772. [Google Scholar] [CrossRef]
- Desplats, P.A.; Denny, C.A.; Kass, K.E.; Gilmartin, T.; Head, S.R.; Sutcliffe, J.G.; Seyfried, T.N.; Thomas, E.A. Glycolipid and ganglioside metabolism imbalances in Huntington’s disease. Neurobiol. Dis. 2007, 27, 265–277. [Google Scholar] [CrossRef]
- Chauhan, N.B. Chronic neurodegenerative consequences of traumatic brain injury. Restor. Neurol. Neurosci. 2014, 32, 337–365. [Google Scholar] [CrossRef]
- Silveyra, M.-X.; Cuadrado-Corrales, N.; Marcos, A.; Barquero, M.-S.; Rabano, A.; Calero, M.; Saez-Valero, J. Altered glycosylation of acetylcholinesterase in Creutzfeldt-Jakob disease. J. Neurochem. 2006, 96, 97–104. [Google Scholar] [CrossRef] [PubMed]
- Willison, H.J.; Goodyear, C. Glycolipid antigens and autoantibodies in autoimmune neuropathies. Trends Immunol. 2013, 34, 453–459. [Google Scholar] [CrossRef]
- Karis, K.; Eskla, K.-L.; Kaare, M.; Täht, K.; Tuusov, J.; Visnapuu, T.; Innos, J.; Jayaram, M.; Timmusk, T.; Weickert, C.S.; et al. Altered expression profile of IgLON family of neural cell adhesion molecules in the dorsolateral prefrontal cortex of schizophrenic patients. Front. Mol. Neurosci. 2018, 11, 8. [Google Scholar] [CrossRef] [PubMed]
- An, H.; Zhou, L.; Yu, Y.; Fan, H.; Fan, F.; Tan, S.; Wang, Z.; Boz, Z.; Shi, J.; Yang, F.; et al. Serum NCAM levels and cognitive deficits in first episode schizophrenia patients versus health controls. Schizophr. Res. 2018, 192, 457–458. [Google Scholar] [CrossRef]
- Pivac, N.; Knežević, A.; Gornik, O.; Pučić, M.; Igl, W.; Peeters, H.; Crepel, A.; Steyaert, J.; Novokmet, M.; Redžić, I.; et al. Human plasma glycome in attention-deficit hyperactivity disorder and autism spectrum disorders. Mol. Cell. Proteom. 2011, 10, 110–004200. [Google Scholar] [CrossRef] [PubMed]
- Yamagata, H.; Uchida, S.; Matsuo, K.; Harada, K.; Kobayashi, A.; Nakashima, M.; Higuchi, F.; Watanuki, T.; Matsubara, T.; Watanabe, Y. Altered plasma protein glycosylation in a mouse model of depression and in patients with major depression. J. Affect. Disord. 2018, 233, 79–85. [Google Scholar] [CrossRef] [PubMed]
- Huang, Y.; Xu, Y.; Liu, A. Increased levels of serum glycosylated hemoglobin are associated with depressive symptoms in a population with cancer (>/=49 Years): An antidepressant-stratified analysis. Clin. Interv. Aging 2021, 16, 205–212. [Google Scholar] [CrossRef] [PubMed]
- Harmer, B.; Lee, S.; Duong, T.V.H.; Saadabadi, A. Suicidal Ideation; StatPearls: Treasure Island, FL, USA, 2021.
- Song, J.; Sjölander, A.; Joas, E.; Bergen, S.; Runeson, B.; Larsson, H.; Landén, M.; Lichtenstein, P. suicidal behavior during lithium and valproate treatment: A within-individual 8-year prospective study of 50,000 patients with bipolar disorder. Am. J. Psychiatry 2017, 174, 795–802. [Google Scholar] [CrossRef]
- Gonzalez-Pinto, A.; Mosquera, F.; Alonso, M.; López, P.; Ramírez, F.; Vieta, E.; Baldessarini, R.J. Suicidal risk in bipolar I disorder patients and adherence to long-term lithium treatment. Bipolar Disord. 2006, 8, 618–624. [Google Scholar] [CrossRef]
- Wasserman, D.; Rihmer, Z.; Rujescu, D.; Sarchiapone, M.; Sokolowski, M.; Titelman, D.; Zalsman, G.; Zemishlany, Z.; Carli, V. The European Psychiatric Association (EPA) guidance on suicide treatment and prevention. Eur. Psychiatry 2012, 27, 129–141. [Google Scholar] [CrossRef] [PubMed]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).