TUBB3 M323V Syndrome Presents with Infantile Nystagmus
Abstract
1. Introduction
2. Case Report
3. Discussion
Author Contributions
Funding
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Rim, J.H.; Lee, S.-T.; Gee, H.Y.; Lee, B.J.; Choi, J.R.; Park, H.W.; Han, S.-H.; Han, J. Accuracy of next-generation sequencing for molecular diagnosis in patients with infantile nystagmus syndrome. JAMA Ophthalmol. 2017, 135, 1376–1385. [Google Scholar] [CrossRef] [PubMed]
- Khanna, S.; Dell’Osso, L.F. The diagnosis and treatment of infantile nystagmus syndrome (INS). Sci. World J. 2006, 6, 1385–1397. [Google Scholar] [CrossRef]
- Forssman, B.; Ringner, B. Prevalence and inheritance of congenital nystagmus in a swedish population. Ann. Hum. Genet. 1971, 35, 139–147. [Google Scholar] [CrossRef]
- Stewart-Brown, S.; Haslum, M. Partial sight and blindness in children of the 1970 birth cohort at 10 years of age. J. Epidemiol. Community Health 1988, 42, 17–23. [Google Scholar] [CrossRef]
- Papageorgiou, E.; McLean, R.J.; Gottlob, I. Nystagmus in childhood. Pediatr. Neonatol. 2014, 55, 341–351. [Google Scholar] [CrossRef]
- Kuht, H.J.; Han, J.; Maconachie, G.D.E.; Park, S.E.; Lee, S.T.; McLean, R.; Sheth, V.; Hisaund, M.; Dawar, B.; Sylvius, N.; et al. Slc38a8 mutations result in arrested retinal development with loss of cone photoreceptor specialization. Hum. Mol. Genet. 2020, 29, 2989–3002. [Google Scholar] [CrossRef]
- Han, J.; Lee, T.; Lee, J.B.; Han, S.H. Retinal microstructures are altered in patients with idiopathic infantile nystagmus. Graefes Arch. Clin. Exp. Ophthalmol. 2017, 255, 1661–1668. [Google Scholar] [CrossRef] [PubMed]
- Aamir, A.; Kuht, H.J.; Grønskov, K.; Brooks, B.P.; Thomas, M.G. Clinical utility gene card for oculocutaneous (oca) and ocular albinism (oa)-an update. Eur. J. Hum. Genet. 2021. [Google Scholar] [CrossRef]
- Lee, J.; Lee, H.; Lee, Y.M.; Kuht, H.J.; Thomas, M.G.; Kim, S.J.; Lee, S.T.; Han, J. Dync2h1 variants cause leber congenital amaurosis without syndromic features. Clin. Genet. 2021. [Google Scholar] [CrossRef] [PubMed]
- Yoo, T.K.; Han, S.H.; Han, J. Rp2 rod-cone dystrophy causes spasmus nutans-like nystagmus. J. Neuroophthalmol. 2021, 41, e91–e93. [Google Scholar] [CrossRef] [PubMed]
- Surl, D.; Shin, S.; Lee, S.T.; Choi, J.R.; Lee, J.; Byeon, S.H.; Han, S.H.; Lim, H.T.; Han, J. Copy number variations and multiallelic variants in korean patients with leber congenital amaurosis. Mol. Vis. 2020, 26, 26–35. [Google Scholar]
- Dawar, B.; Kuht, H.J.; Han, J.; Maconachie, G.D.E.; Thomas, M.G. Clinical utility gene card for frmd7-related infantile nystagmus. Eur J. Hum. Genet. 2021. [Google Scholar] [CrossRef] [PubMed]
- Pilling, R.; Thompson, J.; Gottlob, I. Social and visual function in nystagmus. Br. J. Ophthalmol. 2005, 89, 1278–1281. [Google Scholar] [CrossRef] [PubMed]
- Richards, M.D.; Wong, A. Infantile nystagmus syndrome: Clinical characteristics, current theories of pathogenesis, diagnosis, and management. Can. J. Ophthalmol. 2015, 50, 400–408. [Google Scholar] [CrossRef] [PubMed]
- Thomas, M.G.; Maconachie, G.D.; Sheth, V.; McLean, R.J.; Gottlob, I. Development and clinical utility of a novel diagnostic nystagmus gene panel using targeted next-generation sequencing. Eur. J. Hum. Genet. 2017, 25, 725–734. [Google Scholar] [CrossRef] [PubMed]
- Lasser, M.; Tiber, J.; Lowery, L.A. The role of the microtubule cytoskeleton in neurodevelopmental disorders. Front. Cell. Neurosci. 2018, 12, 165. [Google Scholar] [CrossRef]
- Parker, A.L.; Kavallaris, M.; McCarroll, J.A. Microtubules and their role in cellular stress in cancer. Front. Oncol. 2014, 4, 153. [Google Scholar] [CrossRef]
- Arrigoni, F.; Romaniello, R.; Peruzzo, D.; Poretti, A.; Bassi, M.T.; Pierpaoli, C.; Valente, E.M.; Nuovo, S.; Boltshauser, E.; Huisman, T.A.G.M. The spectrum of brainstem malformations associated to mutations of the tubulin genes family: MRI and DTI analysis. Eur. Radiol. 2019, 29, 770–782. [Google Scholar] [CrossRef]
- Whitman, M.C.; Andrews, C.; Chan, W.M.; Tischfield, M.A.; Stasheff, S.F.; Brancati, F.; Ortiz-Gonzalez, X.; Nuovo, S.; Garaci, F.; MacKinnon, S.E. Two unique tubb3 mutations cause both cfeom3 and malformations of cortical development. Am. J. Med. Genet. Part A 2016, 170, 297–305. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.H.; Hwang, J.M. Imaging of cranial nerves iii, iv, vi in congenital cranial dysinnervation disorders. Korean J. Ophthalmol. 2017, 31, 183–193. [Google Scholar] [CrossRef]
- Tischfield, M.A.; Baris, H.N.; Wu, C.; Rudolph, G.; Van Maldergem, L.; He, W.; Chan, W.-M.; Andrews, C.; Demer, J.L.; Robertson, R.L. Human tubb3 mutations perturb microtubule dynamics, kinesin interactions, and axon guidance. Cell 2010, 140, 74–87. [Google Scholar] [CrossRef] [PubMed]
- Thomas, M.G.; Maconachie, G.D.; Constantinescu, C.S.; Chan, W.-M.; Barry, B.; Hisaund, M.; Sheth, V.; Kuht, H.J.; Dineen, R.A.; Harieaswar, S. Congenital monocular elevation deficiency associated with a novel tubb3 gene variant. Br. J. Ophthalmol. 2020, 104, 547–550. [Google Scholar] [CrossRef] [PubMed]
- Richards, S.; Aziz, N.; Bale, S.; Bick, D.; Das, S.; Gastier-Foster, J.; Grody, W.W.; Hegde, M.; Lyon, E.; Spector, E. Standards and guidelines for the interpretation of sequence variants: A joint consensus recommendation of the american college of medical genetics and genomics and the association for molecular pathology. Genet. Med. 2015, 17, 405–423. [Google Scholar] [CrossRef]
- Poirier, K.; Saillour, Y.; Bahi-Buisson, N.; Jaglin, X.H.; Fallet-Bianco, C.; Nabbout, R.; Castelnau-Ptakhine, L.; Roubertie, A.; Attie-Bitach, T.; Desguerre, I. Mutations in the neuronal β-tubulin subunit tubb3 result in malformation of cortical development and neuronal migration defects. Hum. Mol. Genet. 2010, 19, 4462–4473. [Google Scholar] [CrossRef] [PubMed]
- Tischfield, M.A.; Cederquist, G.Y.; Gupta Jr, M.L.; Engle, E.C. Phenotypic spectrum of the tubulin-related disorders and functional implications of disease-causing mutations. Curr. Opin. Genet. Dev. 2011, 21, 286–294. [Google Scholar] [CrossRef]
- Pettersen, E.F.; Goddard, T.D.; Huang, C.C.; Meng, E.C.; Couch, G.S.; Croll, T.I.; Morris, J.H.; Ferrin, T.E. Ucsf chimerax: Structure visualization for researchers, educators, and developers. Protein Sci. 2021, 30, 70–82. [Google Scholar] [CrossRef]
- Hebebrand, M.; Hüffmeier, U.; Trollmann, R.; Hehr, U.; Uebe, S.; Ekici, A.B.; Kraus, C.; Krumbiegel, M.; Reis, A.; Thiel, C.T. The mutational and phenotypic spectrum of tuba1a-associated tubulinopathy. Orphanet. J. Rare Dis. 2019, 14, 1–13. [Google Scholar] [CrossRef]
- Abdollahi, M.R.; Morrison, E.; Sirey, T.; Molnar, Z.; Hayward, B.E.; Carr, I.M.; Springell, K.; Woods, C.G.; Ahmed, M.; Hattingh, L.; et al. Mutation of the variant alpha-tubulin tuba8 results in polymicrogyria with optic nerve hypoplasia. Am. J. Hum. Genet. 2009, 85, 737–744. [Google Scholar] [CrossRef]
- Aiken, J.; Buscaglia, G.; Bates, E.A.; Moore, J.K. The α-tubulin gene tuba1a in brain development: A key ingredient in the neuronal isotype blend. J. Dev. Biol 2017, 5, 8. [Google Scholar] [CrossRef] [PubMed]
- Thomas, M.G.; Maconachie, G.D.E.; Kuht, H.J.; Chan, W.M.; Sheth, V.; Hisaund, M.; McLean, R.J.; Barry, B.; Al-Diri, B.; Proudlock, F.A.; et al. Optic nerve head and retinal abnormalities associated with congenital fibrosis of the extraocular muscles. Int. J. Mol. Sci. 2021, 22, 2575. [Google Scholar] [CrossRef] [PubMed]
- Sharma, R.K.; Netland, P.A. Early born lineage of retinal neurons express class III beta-tubulin isotype. Brain Res. 2007, 1176, 11–17. [Google Scholar] [CrossRef] [PubMed]
- Yonehara, K.; Fiscella, M.; Drinnenberg, A.; Esposti, F.; Trenholm, S.; Krol, J.; Franke, F.; Scherf, B.G.; Kusnyerik, A.; Müller, J.; et al. Congenital nystagmus gene frmd7 is necessary for establishing a neuronal circuit asymmetry for direction selectivity. Neuron 2016, 89, 177–193. [Google Scholar] [CrossRef] [PubMed]
- Konus, I.; Ozsoy, E.; Turkcuoglu, P.; Emre, S.; Duman, F. Evaluation of metabolite changes in the occipital cortex of patients with idiopathic infantile nystagmus or bilateral ametropic amblyopia by magnetic resonance spectroscopy. Korean J. Ophthalmol. 2019, 33, 406–413. [Google Scholar] [CrossRef] [PubMed]
- Bertsch, M.; Floyd, M.; Kehoe, T.; Pfeifer, W.; Drack, A.V. The clinical evaluation of infantile nystagmus: What to do first and why. Ophthalmic Genet. 2017, 38, 22–33. [Google Scholar] [CrossRef]

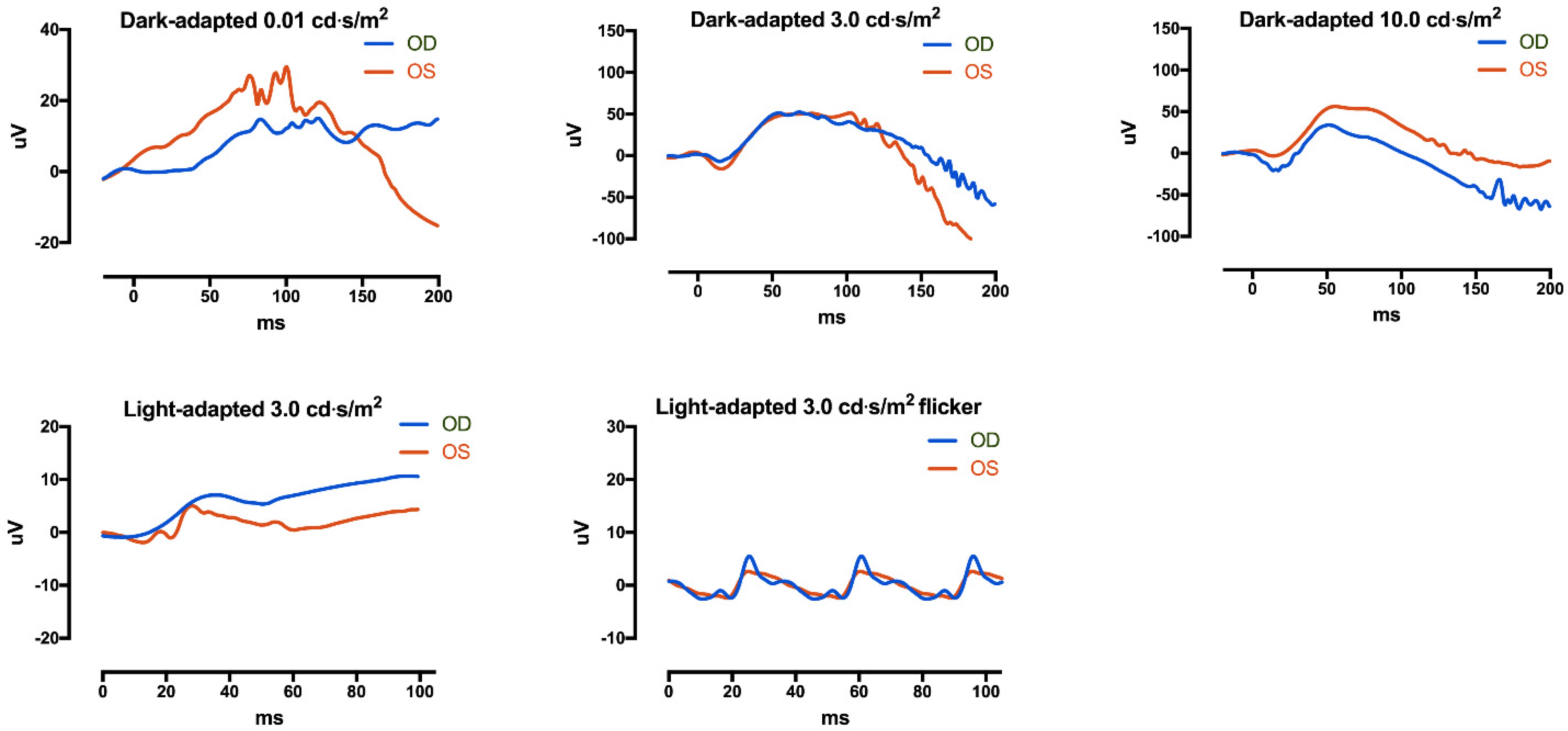
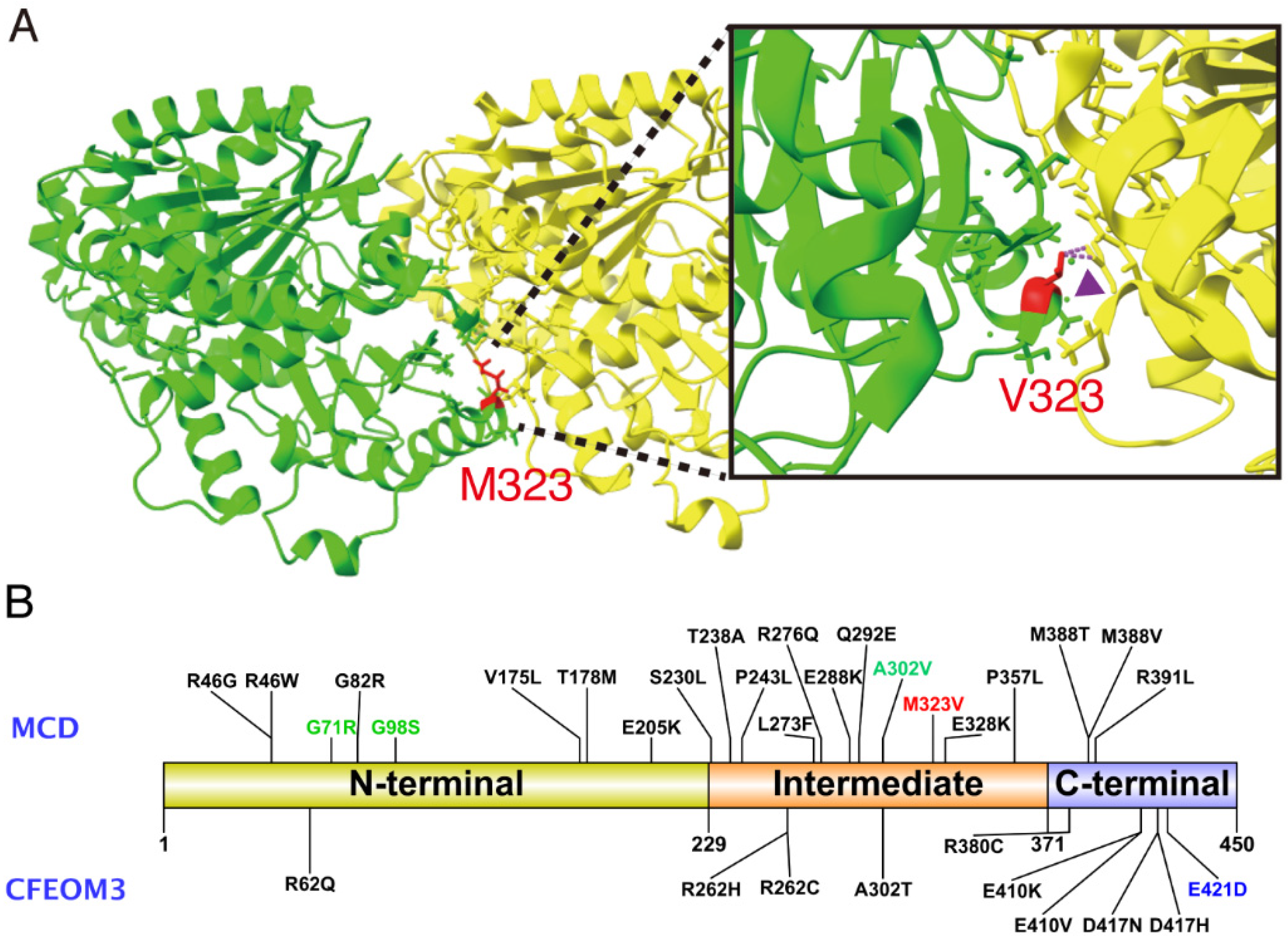
| Clinical Characteristics | Patient 1 | Patient 2 | Patient 3 | Patient 4 | Patient 5 | Patient 6 | Patient 7 | |
|---|---|---|---|---|---|---|---|---|
| TUBB3 variant | p.M323V | p.M323V | p.A302V (homo) | p.G71R | p.G71R | p.G98S | p.M323V | |
| Inheritance pattern | AD | AD | Isolated (homo) | Isolated | Isolated | Isolated | Isolated | |
| Age | 36 years | 2 years | 1 year | 5 years | 9 years | 2 years | 6 months | |
| Gender | Male | Male | Female | Female | Male | Female | Male | |
| Ethnicity | NA | NA | NA | European | European | European | Korean | |
| OFC | 3rd p | 25th p | 3rd p | NA | NA | NA | 3-50th p | |
| Motor delay | Hypotonia | Hypotonia | Hypotonia | Hypotonia | Hypotonia | Hypotonia | Absent | |
| Cognitive function | Severe ID | LD | NA | ID | ID | ID | ID, LD | |
| Epilepsy | Absent | Absent | Absent | NA | NA | NA | Absent | |
| CFEOM | No | No | No | Yes | Yes | Yes | No | |
| Nystagmus | Horizontal nystagmus | Horizontal nystagmus | Multidirectional nystagmus | Rotary nystagmus | Horizontal nystagmus | Horizontal nystagmus | Horizontal nystagmus | |
| Cortical dysgenesis | Gyral disorganization | Gyral disorganization | Gyral disorganization | Gyral disorganization | Gyral disorganization | Gyral disorganization | Gyral disorganization | |
| Cerebellum | vermis | Dysplastic | Dysplastic | Dysplastic | Dysplastic | Dysplastic | Dysplastic | Dysplastic |
| Hemisphere | Dysplastic | Normal | Normal | Normal | Normal | Normal | Normal | |
| Brainstem | Hypoplastic | Hypoplastic | Hypoplastic | Hypoplastic | Hypoplastic | Hypoplastic | Normal | |
| Corpus callosum | Thin | Thin | Thin | Thin | Thin | Thin | Asymmetric | |
| Basal ganglia | Hypertrophic/mild fusion | Hypertrophic/mild fusion | Fusion caudate/ putamen | Hypertrophic/ fusion | Hypertrophic/ fusion | Hypertrophic/ fusion | Asymmetric | |
| Literatures | Poirier et al. Hum Mol Genet (2010) | Poirier et al. Hum Mol Genet (2010) | Poirier et al. Hum Mol Genet (2010) | Whitman et al. Am J Med Genet A. (2016) | Whitman et al. Am J Med Genet A. (2016) | Whitman et al. Am J Med Genet A. (2016) | This study | |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Jin, S.; Park, S.-E.; Won, D.; Lee, S.-T.; Han, S.-H.; Han, J. TUBB3 M323V Syndrome Presents with Infantile Nystagmus. Genes 2021, 12, 575. https://doi.org/10.3390/genes12040575
Jin S, Park S-E, Won D, Lee S-T, Han S-H, Han J. TUBB3 M323V Syndrome Presents with Infantile Nystagmus. Genes. 2021; 12(4):575. https://doi.org/10.3390/genes12040575
Chicago/Turabian StyleJin, Soohwa, Sung-Eun Park, Dongju Won, Seung-Tae Lee, Sueng-Han Han, and Jinu Han. 2021. "TUBB3 M323V Syndrome Presents with Infantile Nystagmus" Genes 12, no. 4: 575. https://doi.org/10.3390/genes12040575
APA StyleJin, S., Park, S.-E., Won, D., Lee, S.-T., Han, S.-H., & Han, J. (2021). TUBB3 M323V Syndrome Presents with Infantile Nystagmus. Genes, 12(4), 575. https://doi.org/10.3390/genes12040575






