Population Genetics of the European Roma—A Review
Abstract
:1. Introduction
2. Origins of the Roma from Genetic Evidence
3. The Romani Diaspora out of India
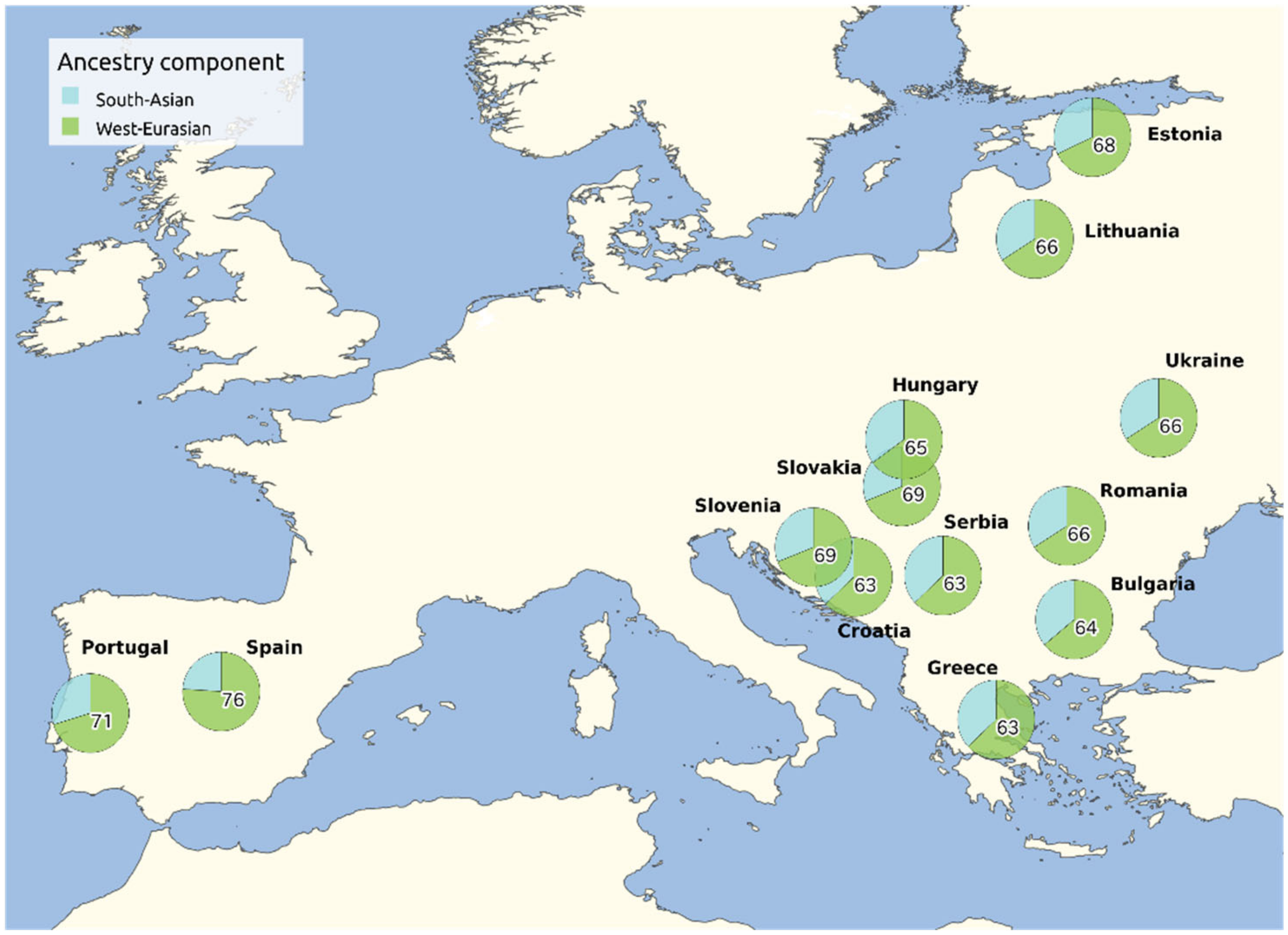
4. The Genetic Differentiation and Heterogeneity among Different European Romani Subpopulations
5. The Genetic Diversity of the Roma: Current Insights in the Bottleneck and Founder Events
6. Gene Flow Sex Bias in the Roma Population
7. Biomedical Implications of the Complex Romani Population Demography
8. Future Perspectives
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Hancock, I. On Romani Origins and Identity. The Romani Archives and Documentation Center, University of Texas at Austin, USA. 2006. Available online: https://www.oocities.org/romani_life_society/indexpics/HistoryOrigins.pdf (accessed on 1 June 2022).
- Fraser, A.M. The Gypsies (The Peoples of Europe); Blackwell Pub.: Hoboken, NJ, USA, 1992; ISBN 978-0-631-19605-1. [Google Scholar]
- Gresham, D.; Morar, B.; Underhill, P.A.; Passarino, G.; Lin, A.A.; Wise, C.; Angelicheva, D.; Calafell, F.; Oefner, P.J.; Shen, P.; et al. Origins and Divergence of the Roma (Gypsies). Am. Soc. Hum. Genet. 2001, 69, 1314–1331. [Google Scholar] [CrossRef] [Green Version]
- Moorjani, P.; Patterson, N.; Loh, P.-R.; Lipson, M.; Kisfali, P.; Melegh, B.I.; Bonin, M.; Kádaši, L.; Rieß, O.; Berger, B.; et al. Reconstructing Roma History from Genome-Wide Data. PLoS ONE 2013, 8, e58633. [Google Scholar] [CrossRef]
- Mendizabal, I.; Lao, O.; Marigorta, U.M.; Wollstein, A.; Gusmão, L.; Ferak, V.; Ioana, M.; Jordanova, A.; Kaneva, R.; Kouvatsi, A.; et al. Report Reconstructing the Population History of European Romani from Genome-Wide Data. Curr. Biol. 2012, 22, 2342–2349. [Google Scholar] [CrossRef] [Green Version]
- Martínez-Cruz, B.; Mendizabal, I.; Harmant, C.; Pablo, R.D.; Ioana, M.; Angelicheva, D.; Kouvatsi, A.; Makukh, H.; Netea, M.G.; Pamjav, H.; et al. Origins, Admixture and Founder Lineages in European Roma. Eur. J. Hum. Genet. 2016, 24, 937–943. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Font-Porterias, N.; Arauna, L.R.; Poveda, A.; Bianco, E.; Rebato, E.; Prata, M.J.; Calafell, F.; Comas, D. European Roma Groups Show Complex West Eurasian Admixture Footprints and a Common South Asian Genetic Origin. PLOS Genet. 2019, 15, e1008417. [Google Scholar] [CrossRef] [PubMed]
- Kenrick, D. Historical Dictionary of the Gypsies (Romanies); Scarecrow Press: Lanham, MD, USA, 2007; ISBN 978-0-8108-6440-5. [Google Scholar]
- Covrig, A. Why Roma Do Not Declare Their Identity—Careful Decision or Unpremeditated Refusal? J. Study Relig. Ideol. 2010, 3, 90–101. [Google Scholar]
- Roma Equality, Inclusion and Participation in the EU. Available online: https://ec.europa.eu/info/policies/justice-and-fundamental-rights/combatting-discrimination/roma-eu/roma-equality-inclusion-and-participation-eu_en (accessed on 8 June 2022).
- Matras, Y. Romani A Linguistic Introduction; UK Cambridge University Press: Cambridge, UK, 2002; ISBN 0-511-03580-2. [Google Scholar]
- Zatreanu, M.; Halwachs, D.W. Romani in Europe. Ministry of Education and Research–Bucharest Romania, Department of Linguistics at the University of Graz, Austria. 2003. Available online: https://www.coe.int/t/dg4/education/roma/Source/RomaniEurope_EN.pdf (accessed on 8 June 2022).
- Iovita, R.P.; Schurr, T.G. Reconstructing the origins and migrations of diasporic populations: The case of the European Gypsies. Am. Anthropol. 2004, 106, 267–281. [Google Scholar]
- Marushiakova, E.; Popov, V. Gypsies (Roma) in Bulgaria (Volume 18); Peter Lang Pub Incorporated: New York, NY, USA, 1997. [Google Scholar]
- Grierson, G.A. Doms, Jâts, and the Origin of the Gypsies. J. Gypsy Lore Soc. 1888, 1, 71. [Google Scholar]
- Frossard, E.B.D. The Gypsies in France. Saturday Mag. 1835, 6, 39–40. [Google Scholar]
- Crawfurd, J. On the Origin of the Gypsies. Trans. Ethnol. Soc. Lond. 1865, 3, 25–36. [Google Scholar] [CrossRef]
- Colocci, A. The Gypsies: History of an Errant People; Arnaldo Forni Editore: Bologna, Italy, 1889. [Google Scholar]
- Pamjav, H.; Zalán, A.; Béres, J.; Nagy, M.; Chang, Y.M. Genetic Structure of the Paternal Lineage of the Roma People. Am. J. Phys. Anthropol. 2011, 145, 21–29. [Google Scholar] [CrossRef] [PubMed]
- Regueiro, M.; Rivera, L.; Chennakrishnaiah, S.; Popovic, B.; Andjus, S.; Milasin, J.; Herrera, R.J. Ancestral Modal Y-STR Haplotype Shared among Romani and South Indian Populations. Gene 2012, 504, 296–302. [Google Scholar] [CrossRef]
- Mendizabal, I.; Valente, C.; Gusmão, A.; Alves, C.; Gomes, V.; Goios, A.; Parson, W.; Calafell, F.; Alvarez, L.; Amorim, A.; et al. Reconstructing the Indian Origin and Dispersal of the European Roma: A Maternal Genetic Perspective. PLoS ONE 2011, 6, e15988. [Google Scholar] [CrossRef] [Green Version]
- Kalaydjieva, L.; Gresham, D.; Calafell, F. Genetic Studies of the Roma (Gypsies): A Review. BMC Med. Genet. 2001, 2, 5. [Google Scholar] [CrossRef]
- Morar, B.; Gresham, D.; Angelicheva, D.; Tournev, I.; Gooding, R.; Guergueltcheva, V.; Schmidt, C.; Abicht, A.; Lochmüller, H.; Tordai, A.; et al. Mutation History of the Roma/Gypsies. Am. J. Hum. Genet. 2004, 75, 596–609. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kalaydjieva, L.; Morar, B.; Chaix, R.; Tang, H. A Newly Discovered Founder Population: The Roma/Gypsies. BioEssays 2005, 27, 1084–1094. [Google Scholar] [CrossRef]
- Bloch, J.; David, H.; Foletier, F.D.V. The Gypsies; FeniXX: Singapore, 1969; ISBN 978-2-7059-1051-8. [Google Scholar]
- Brearley, M. The Persecution of Gypsies in Europe. Am. Behav. Sci. 2001, 45, 588–599. [Google Scholar] [CrossRef] [Green Version]
- Taylor, B. Another Darkness, Another Dawn: A History of Gypsies, Roma and Travellers; Reaktion Books: London, UK, 2014; ISBN 978-1-78023-297-3. [Google Scholar]
- Barany, Z. The East European Gypsies in the Imperial Age. Ethn. Racial Stud. 2001, 24, 50–63. [Google Scholar] [CrossRef]
- Crowe, D.; Kolsti, J.; Hancock, I. The Gypsies of Eastern Europe; Routledge: London, UK, 2016; ISBN 978-1-315-49024-3. [Google Scholar]
- Kamusella, T.; Jaskułowski, K. Nationalisms Today; Peter Lang: New York, NY, USA, 2009; ISBN 978-3-03911-883-0. [Google Scholar]
- Marushiakova, E.; Popov, V.Z.; Popov, V. Gypsies in the Ottoman Empire: A Contribution to the History of the Balkans; University of Hertfordshire Press: Hatfield, UK, 2001; Volume 22, ISBN 978-1-902806-02-0. [Google Scholar]
- Lewy, G. The Nazi Persecution of the Gypsies; Oxford University Press: Oxford, UK, 2000; ISBN 978-0-19-802904-5. [Google Scholar]
- Ioanid, R. The Holocaust in Romania: The Destruction of Jews and Gypsies Under the Antonescu Regime, 1940–1944; Ivan R. Dee: Chicago, IL, USA, 2008; ISBN 978-1-4616-9490-8. [Google Scholar]
- Kenrick, D.; Puxon, G. Gypsies Under the Swastika; University of Hertfordshire Press: Hatfield, UK, 2009; ISBN 978-1-902806-80-8. [Google Scholar]
- Trevisan, P. ‘Gypsies’ in Fascist Italy: From Expelled Foreigners to Dangerous Italians. Soc. Hist. 2017, 42, 342–364. [Google Scholar] [CrossRef]
- Kotljarchuk, A. World War II and the Registration of Roma in Sweden: The Role of Experts and Census-Takers. Holocaust Genocide Stud. 2017, 31, 457–479. [Google Scholar] [CrossRef]
- Palmer, M.D.; Burgess, S.M. The Wiley-Blackwell Companion to Religion and Social Justice; John Wiley & Sons: Hoboken, NJ, USA, 2020; ISBN 978-1-119-57210-7. [Google Scholar]
- A Persisting Concern: Anti-Gypsyism as a Barrier to Roma Inclusion. Available online: https://fra.europa.eu/en/publication/2018/persisting-concern-anti-gypsyism-barrier-roma-inclusion (accessed on 5 September 2022).
- Council of Europe Strategic Action Plan for Roma and Traveller Inclusion (2020–2025). Available online: https://edoc.coe.int/en/roma-and-travellers/8508-council-of-europe-strategic-action-plan-for-roma-and-traveller-inclusion-2020-2025.html (accessed on 14 September 2022).
- ECRI General Policy Recommendation No. 13. Available online: https://www.coe.int/en/web/european-commission-against-racism-and-intolerance/recommendation-no.13 (accessed on 5 September 2022).
- Bernasovský, I.; Suchý, J.; Bernasovská, K.; Vargová, T. Blood Groups of Roms (Gypsies) in Czechoslovakia. Am. J. Phys. Anthropol. 1976, 45, 277–279. [Google Scholar] [CrossRef] [PubMed]
- Bernasovský, I.; Jurícková, J.; Ferák, V. Population Genetic Study in Gypsies (Roms) from Slovakia: Distribution of Blood Group Genetic Markers. Anthropol. Sci. 1994, 102, 409–419. [Google Scholar] [CrossRef] [Green Version]
- Gáliková, J.; Vilímová, M.; Ferák, V.; Mayerová, A. Haptoglobin Types in Gypsies from Slovakia (Czechoslovakia). Hum. Hered. 1969, 19, 480–485. [Google Scholar] [CrossRef]
- Piccolo, F.; Jeanpierre, M.; Leturcq, F.; Dodé, C.; Azibi, K.; Toutain, A.; Merlini, L.; Jarre, L.; Navarro, C.; Krishnamoorthy, R.; et al. A Founder Mutation in the γ-Sarcoglycan Gene of Gypsies Possibly Predating Their Migration Out of India. Hum. Mol. Genet. 1996, 5, 2019–2022. [Google Scholar] [CrossRef] [PubMed]
- Lasa, A.; Piccolo, F.; de Diego, C.; Jeanpierre, M.; Colomer, J.; Rodríguez, M.J.; Urtizberea, J.A.; Baiget, M.; Kaplan, J.C.; Gallano, P. Severe Limb Girdle Muscular Dystrophy in Spanish Gypsies: Further Evidence for a Founder Mutation in the γ-Sarcoglycan Gene. Eur. J. Hum. Genet. 1998, 6, 396–399. [Google Scholar] [CrossRef] [Green Version]
- Abicht, A.; Stucka, R.; Karcagi, V.; Herczegfalvi, A.; Horváth, R.; Mortier, W.; Schara, U.; Ramaekers, V.; Jost, W.; Brunner, J.; et al. A Common Mutation (Ε1267delG) in Congenital Myasthenic Patients of Gypsy Ethnic Origin. Neurology 1999, 53, 1564. [Google Scholar] [CrossRef] [PubMed]
- Álvarez, A.; del Castillo, I.; Villamar, M.; Aguirre, L.A.; González-Neira, A.; López-Nevot, A.; Moreno-Pelayo, M.A.; Moreno, F. High Prevalence of the W24X Mutation in the Gene Encoding Connexin-26 (GJB2) in Spanish Romani (Gypsies) with Autosomal Recessive Non-Syndromic Hearing Loss. Am. J. Med. Genet. 2005, 137A, 255–258. [Google Scholar] [CrossRef]
- Bouwer, S.; Angelicheva, D.; Chandler, D.; Seeman, P.; Tournev, I.; Kalaydjieva, L. Carrier Rates of the Ancestral Indian W24X Mutation in GJB2 in the General Gypsy Population and Individual Subisolates. Genet. Test. 2007, 11, 455–458. [Google Scholar] [CrossRef]
- Azmanov, D.N.; Dimitrova, S.; Florez, L.; Cherninkova, S.; Draganov, D.; Morar, B.; Saat, R.; Juan, M.; Arostegui, J.I.; Ganguly, S.; et al. LTBP2 and CYP1B1 Mutations and Associated Ocular Phenotypes in the Roma/Gypsy Founder Population. Eur. J. Hum. Genet. 2011, 19, 326–333. [Google Scholar] [CrossRef]
- Maheshwari, M.; Vijaya, R.; Ghosh, M.; Shastri, S.; Kabra, M.; Menon, P.S.N. Screening of Families with Autosomal Recessive Non-Syndromic Hearing Impairment (ARNSHI) for Mutations in GJB2 Gene: Indian Scenario. Am. J. Med. Genet. 2003, 120A, 180–184. [Google Scholar] [CrossRef]
- Gómez-Carballa, A.; Pardo-seco, J.; Fachal, L.; Vega, A.; Cebey, M.; Salas, A. Indian Signatures in the Westernmost Edge of the European Romani Diaspora: New Insight from Mitogenomes. PLoS ONE 2013, 8, e75397. [Google Scholar] [CrossRef] [Green Version]
- Zalán, A.; Béres, J.; Pamjav, H. Paternal Genetic History of the Vlax Roma. Forensic Sci. Int. Genet. 2011, 5, 109–113. [Google Scholar] [CrossRef] [PubMed]
- Rai, N.; Chaubey, G.; Tamang, R.; Pathak, A.K.; Singh, V.K.; Karmin, M.; Singh, M.; Rani, D.S.; Anugula, S.; Yadav, B.K.; et al. The Phylogeography of Y-Chromosome Haplogroup H1a1a-M82 Reveals the Likely Indian Origin of the European Romani Populations. PLoS ONE 2012, 7, e48477. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Melegh, B.I.; Banfai, Z.; Hadzsiev, K.; Miseta, A.; Melegh, B. Refining the South Asian Origin of the Romani People. BMC Genet. 2017, 18, 82. [Google Scholar] [CrossRef]
- Reich, D.; Thangaraj, K.; Patterson, N.; Price, A.L.; Singh, L. Reconstructing Indian Population History. Nature 2009, 461, 489–494. [Google Scholar] [CrossRef] [Green Version]
- Narasimhan, V.M.; Patterson, N.; Moorjani, P.; Rohland, N.; Bernardos, R.; Mallick, S.; Lazaridis, I.; Nakatsuka, N.; Olalde, I.; Lipson, M. The Formation of Human Populations in South and Central Asia. Science 2019, 365, eaat7487. [Google Scholar] [CrossRef]
- Bianco, E.; Laval, G.; Font-Porterias, N.; García-Fernández, C.; Dobon, B.; Sabido-Vera, R.; Stefanovska, E.S.; Kučinskas, V.; Makukh, H.; Pamjav, H.; et al. Recent Common Origin, Reduced Population Size, and Marked Admixture Have Shaped European Roma Genomes. Mol. Biol. Evol. 2020, 37, 3175–3187. [Google Scholar] [CrossRef]
- Matras, Y.; Bakker, P.; Kyuchukov, H. The Typology and Dialectology of Romani; John Benjamins B.V.: Amsterdam, The Netherlands, 1997. [Google Scholar]
- Bánfai, Z.; Ádám, V.; Pöstyéni, E.; Büki, G.; Czakó, M.; Miseta, A.; Melegh, B. Revealing the Impact of the Caucasus Region on the Genetic Legacy of Romani People from Genome-Wide Data. PLoS ONE 2018, 13, e0202890. [Google Scholar] [CrossRef]
- Derenko, M.; Malyarchuk, B.; Bahmanimehr, A.; Denisova, G.; Perkova, M.; Farjadian, S.; Yepiskoposyan, L. Complete Mitochondrial DNA Diversity in Iranians. PLoS ONE 2013, 8, e80673. [Google Scholar] [CrossRef] [Green Version]
- Tarkhnishvili, D.; Gavashelishvili, A.; Murtskhvaladze, M.; Gabelaia, M.; Tevzadze, G. Human Paternal Lineages, Languages, and Environment in the Caucasus. Hum. Biol. 2014, 86, 113–130. [Google Scholar] [CrossRef] [Green Version]
- Malyarchuk, B.A.; Perkova, M.A.; Derenko, M.V.; Vanecek, T.; Lazur, J.; Gomolcak, P. Mitochondrial DNA Variability in Slovaks, with Application to the Roma Origin. Ann. Hum. Genet. 2008, 72, 228–240. [Google Scholar] [CrossRef] [PubMed]
- Gusmão, A.; Gusmão, L.; Gomes, V.; Alves, C.; Calafell, F.; Amorim, A.; Prata, M.J. A Perspective on the History of the Iberian Gypsies Provided by Phylogeographic Analysis of Y-Chromosome Lineages. Ann. Hum. Genet. 2008, 72, 215–227. [Google Scholar] [CrossRef] [PubMed]
- Klarić, I.M.; Salihović, M.P.; Lauc, L.B.; Zhivotovsky, L.A.; Rootsi, S.; Janićijević, B. Dissecting the Molecular Architecture and Origin of Bayash Romani Patrilineages: Genetic Influences from South-Asia and the Balkans. Am. J. Phys. Anthropol. 2009, 138, 333–342. [Google Scholar] [CrossRef] [PubMed]
- Malyarchuk, B.A.; Grzybowski, T.; Derenko, M.V.; Czarny, J.; Miścicka-Śliwka, D. Mitochondrial DNA Diversity in the Polish Roma. Ann. Hum. Genet. 2006, 70, 195–206. [Google Scholar] [CrossRef]
- Kalaydjieva, L.; Calafell, F.; Jobling, M.A.; Angelicheva, D.; Knijff, P.D.; Rosser, Z.H.; Hurles, M.E.; Underhill, P.; Tournev, I.; Marushiakova, E.; et al. Patterns of Inter- and Intra-Group Genetic Diversity in the Vlax Roma as Revealed by Y Chromosome and Mitochondrial DNA Lineages. Eur. J. Hum. Genet. 2001, 9, 97–104. [Google Scholar] [CrossRef] [Green Version]
- Pokupčić, K.; Cukrov, S.; Klarić, I.M.; Salihović, M.P.; Lauc, L.B.; Blažanović, A.; Janićijević, B. Y-STR Genetic Diversity of Croatian (Bayash) Roma. Forensic Sci. Int. Genet. 2008, 2, e11–e13. [Google Scholar] [CrossRef]
- Ahmic, A.; Kalamujic, B.; Ismailovic, A.; Lasic, L.; Halilovic, E.; Mujkic, I.; Pojskic, N. Mitochondrial DNA Diversity of the Roma from Northeastern Bosnia, Bosnia and Herzegovina. HOMO 2018, 69, 347–356. [Google Scholar] [CrossRef]
- Font-Porterias, N.; Caro-Consuegra, R.; Lucas-Sánchez, M.; Lopez, M.; Giménez, A.; Carballo-Mesa, A.; Bosch, E.; Calafell, F.; Quintana-Murci, L.; Comas, D. The Counteracting Effects of Demography on Functional Genomic Variation: The Roma Paradigm. Mol. Biol. Evol. 2021, 38, 2804–2817. [Google Scholar] [CrossRef]
- Bánfai, Z.; Melegh, B.I.; Sümegi, K.; Hadzsiev, K.; Miseta, A.; Kásler, M.; Melegh, B. Revealing the Genetic Impact of the Ottoman Occupation on Ethnic Groups of East-Central Europe and on the Roma Population of the Area. Front. Genet. 2019, 10, 558. [Google Scholar] [CrossRef] [Green Version]
- Morar, B.; Azmanov, D.N.; Kalaydjieva, L. Roma (Gypsies): Genetic Studies. In eLS; John Wiley & Sons: Hoboken, NJ, USA, 2013; ISBN 978-0-470-01590-2. [Google Scholar]
- Pereira, V.; Gusmão, L.; Valente, C.; Pereira, R.; Carneiro, J.; Gomes, I.; Morling, N.; Amorim, A.; João Prata, M. Refining the Genetic Portrait of Portuguese Roma through X-Chromosomal Markers. Am. J. Phys. Anthropol. 2012, 148, 389–394. [Google Scholar] [CrossRef]
- Reidla, M.; Kivisild, T.; Metspalu, E.; Kaldma, K.; Tambets, K.; Tolk, H.-V.; Parik, J.; Loogväli, E.-L.; Derenko, M.; Malyarchuk, B.; et al. Origin and Diffusion of MtDNA Haplogroup X. Am. J. Hum. Genet. 2003, 73, 1178–1190. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Chapman, N.H.; Thompson, E.A. The Effect of Population History on the Lengths of Ancestral Chromosome Segments. Genetics 2002, 162, 449–458. [Google Scholar] [CrossRef]
- Chaix, R.; Austerlitz, F.; Morar, B.; Kalaydjieva, L.; Heyer, E. Vlax Roma History: What Do Coalescent-Based Methods Tell Us? Eur. J. Hum. Genet. 2004, 12, 285–292. [Google Scholar] [CrossRef] [Green Version]
- Irwin, J.; Egyed, B.; Saunier, J.; Szamosi, G.; O’Callaghan, J.; Padar, Z.; Parsons, T.J. Hungarian MtDNA Population Databases from Budapest and the Baranya County Roma. Int. J. Legal Med. 2007, 121, 377–383. [Google Scholar] [CrossRef]
- Salihović, M.P.; Barešić, A.; Klarić, I.M.; Cukrov, S.; Lauc, L.B.; Janićijević, B. The Role of the Vlax Roma in Shaping the European Romani Maternal Genetic History. Am. J. Phys. Anthropol. 2011, 146, 262–270. [Google Scholar] [CrossRef] [PubMed]
- Peričić, M.; Klarić, I.M.; Lauc, L.B.; Janićijević, B.; Đorđević, D.; Efremovska, L.; Rudan, P. Population Genetics of 8 Y Chromosome STR Loci in Macedonians and Macedonian Romani (Gypsy). Forensic Sci. Int. 2005, 154, 257–261. [Google Scholar] [CrossRef] [PubMed]
- Juyal, G.; Mondal, M.; Luisi, P.; Laayouni, H.; Sood, A.; Midha, V.; Heutink, P.; Bertranpetit, J.; Thelma, B.K.; Casals, F. Population and Genomic Lessons from Genetic Analysis of Two Indian Populations. Hum. Genet. 2014, 133, 1273–1287. [Google Scholar] [CrossRef]
- Mondal, M.; Bergström, A.; Xue, Y.; Calafell, F.; Laayouni, H.; Casals, F.; Majumder, P.P.; Tyler-Smith, C.; Bertranpetit, J. Y-Chromosomal Sequences of Diverse Indian Populations and the Ancestry of the Andamanese. Hum. Genet. 2017, 136, 499–510. [Google Scholar] [CrossRef] [Green Version]
- Maria Theresia and Joseph II Policies of Assimilation in the Age of Enlightened Absolutism [Rombase]. Available online: http://rombase.uni-graz.at/cgi-bin/art.cgi?src=data/hist/modern/maria.en.xml (accessed on 9 June 2022).
- Gusmão, A.; Valente, C.; Gomes, V.; Alves, C.; Amorim, A.; Prata, M.J.; Gusmão, L. A Genetic Historical Sketch of European Gypsies: The Perspective from Autosomal Markers. Am. J. Phys. Anthropol. 2010, 141, 507–514. [Google Scholar] [CrossRef]
- Inotai, D.; Szilvasi, A.; Benko, S.; Boros-Major, A.; Illes, Z.; Bors, A.; Kiss, K.P.; Rajczy, K.; Gelle-Hossó, A.; Buhler, S.; et al. HLA Genetic Diversity in Hungarians and Hungarian Gypsies: Complementary Differentiation Patterns and Demographic Signals Revealed by HLA-A, -B and -DRB1 in Central Europe. Tissue Antigens 2015, 86, 115–121. [Google Scholar] [CrossRef]
- Burton, M.L.; Moore, C.C.; Whiting, J.W.M.; Romney, A.K.; Aberle, D.F.; Barcelo, J.A.; Dow, M.M.; Guyer, J.I.; Kronenfeld, D.B.; Levy, J.E.; et al. Regions Based on Social Structure. Curr. Anthropol. 1996, 37, 87–123. [Google Scholar]
- Gamella, J.F. Marriage, Gender and Transnational Migrations in Fertility Transitions of Romanian Roma Women. Intersect. East Eur. J. Soc. Polit. 2018, 4, 57–85. [Google Scholar] [CrossRef]
- Weyrauch, W.O. Gypsy Law: Romani Legal Traditions and Culture; University of California Press: Berkeley, CA, USA, 2001. [Google Scholar]
- Heyer, E.; Chaix, R.; Pavard, S.; Austerlitz, F. Sex-Specific Demographic Behaviours That Shape Human Genomic Variation. Mol. Ecol. 2012, 21, 597–612. [Google Scholar] [CrossRef] [PubMed]
- Goldberg, A.; Rosenberg, N.A. Beyond 2/3 and 1/3: The Complex Signatures of Sex-Biased Admixture on the X Chromosome. Genetics 2015, 201, 263. [Google Scholar] [CrossRef]
- García-Fernández, C.; Font-Porterias, N.; Kučinskas, V.; Sukarova-Stefanovska, E.; Pamjav, H.; Makukh, H.; Dobon, B.; Bertranpetit, J.; Netea, M.G.; Calafell, F.; et al. Sex-Biased Patterns Shaped the Genetic History of Roma. Sci. Rep. 2020, 10, 14464. [Google Scholar] [CrossRef]
- Hancock, I.F. We Are the Romani People; University of Hertfordshire Press: Hatfield, UK, 2002; ISBN 978-1-902806-19-8. [Google Scholar]
- Mendizabal, I.; Lao, O.; Marigorta, U.M.; Kayser, M.; Comas, D. Implications of Population History of European Romani on Genetic Susceptibility to Disease. Hum. Hered. 2013, 76, 194–200. [Google Scholar] [CrossRef]
- Font-Porterias, N.; Giménez, A.; Carballo-Mesa, A.; Calafell, F.; Comas, D. Admixture Has Shaped Romani Genetic Diversity in Clinically Relevant Variants. Front. Genet. 2021, 12, 683880. [Google Scholar] [CrossRef]
- Dobon, B.; Ter Horst, R.; Laayouni, H.; Mondal, M.; Bianco, E.; Comas, D.; Ioana, M.; Bosch, E.; Bertranpetit, J.; Netea, M.G. The shaping of immunological responses through natural selection after the Roma Diaspora. Sci. Rep. 2020, 10, 1–12. [Google Scholar] [CrossRef]
- Simko, V.; Ginter, E. Short Life Expectancy and Metabolic Syndrome in Romanies (Gypsies) in Slovakia. Cent. Eur. J. Public Health 2010, 18, 16. [Google Scholar] [CrossRef] [Green Version]
- Parekh, N.; Rose, T. Health Inequalities of the Roma in Europe: A Literature Review. Cent. Eur. J. Public Health 2011, 19, 139–142. [Google Scholar]
- Dobranici, M.; Buzea, A.; Popescu, R. The Cardiovascular Risk Factors of the Roma (Gypsies) People in Central-Eastern Europe: A Review of the Published Literature. J. Med. Life 2012, 5, 382–389. [Google Scholar] [PubMed]
- Nunes, M.A.; Kučerová, K.; Lukáč, O.; Kvapil, M.; Brož, J. Prevalence of Diabetes Mellitus among Roma Populations—A Systematic Review. Int. J. Environ. Res. Public. Health 2018, 15, 2607. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sarantaus, L.; Huusko, P.; Eerola, H.; Launonen, V.; Vehmanen, P.; Rapakko, K.; Gillanders, E.; Syrjäkoski, K.; Kainu, T.; Vahteristo, P.; et al. Multiple Founder Effects and Geographical Clustering of BRCA1 and BRCA2 Families in Finland. Eur. J. Hum. Genet. 2000, 8, 757–763. [Google Scholar] [CrossRef] [PubMed]
- Loudianos, G.; Dessi, V.; Lovicu, M.; Angius, A.; Figus, A.; Lilliu, F.; de Virgiliis, S.; Nurchi, A.M.; Deplano, A.; Moi, P.; et al. Molecular Characterization of Wilson Disease in the Sardinian Population—Evidence of a Founder Effect. Hum. Mutat. 1999, 14, 294–303. [Google Scholar] [PubMed]
- Gosselin, I.; Thiffault, I.; Tétreault, M.; Chau, V.; Dicaire, M.-J.; Loisel, L.; Emond, M.; Senderek, J.; Mathieu, J.; Dupré, N.; et al. Founder SH3TC2 Mutations Are Responsible for a CMT4C French-Canadians Cluster. Neuromuscul. Disord. 2008, 18, 483–492. [Google Scholar] [CrossRef] [PubMed]
- Martínez-Frías, M.L.; Bermejo, E. Prevalence of Congenital Anomaly Syndromes in a Spanish Gypsy Population. J. Med. Genet. 1992, 29, 483–486. [Google Scholar]
- Werissa, N.A.; Piko, P.; Fiatal, S.; Kosa, Z.; Sandor, J.; Adany, R. SNP-Based Genetic Risk Score Modeling Suggests No Increased Genetic Susceptibility of the Roma Population to Type 2 Diabetes Mellitus. Genes 2019, 10, E942. [Google Scholar] [CrossRef]
- Zeljko, H.; Škarić-Jurić, T.; Smolej Narančić, N.; Peričić Salihović, M.; Martinović Klarić, I.; Barbalić, M.; Starčević, B.; Barać Lauc, L.; Janićijević, B. Traditional CVD Risk Factors and Socio-Economic Deprivation in Roma Minority Population of Croatia. Coll. Antropol. 2008, 32, 667–676. [Google Scholar]
- De Courten, B.V.; de Courten, M.; Hanson, R.L.; Zahorakova, A.; Egyenes, H.P.; Tataranni, P.A.; Bennett, P.H.; Vozar, J. Higher Prevalence of Type 2 Diabetes, Metabolic Syndrome and Cardiovascular Diseases in Gypsies than in Non-Gypsies in Slovakia. Diabetes Res. Clin. Pract. 2003, 62, 95–103. [Google Scholar]
- Škarić-Jurić, T.; Zeljko, H.M.; Tomas, Ž.; Peričić Salihović, M.; Smolej Narančić, N.; Janićijević, B. Genetic Risk Factors for Cardiovascular Diseases in the Roma Minority Population of Croatia. Eur. J. Hum. Genet. 2009, 17 (Suppl. S2), 262. [Google Scholar]
- Soltesz, B.; Piko, P.; Sandor, J.; Kosa, Z.; Adany, R.; Fiatal, S. The Genetic Risk for Hypertension Is Lower among the Hungarian Roma Population Compared to the General Population. PLoS ONE 2020, 15, e0234547. [Google Scholar] [CrossRef]
- Ginter, E.; Simko, V.; Wsolova, L. Fall of the Iron Curtain: Male Life Expectancy in Slovakia, in the Czech Republic and in Europe. Cent. Eur. J. Public Health 2009, 17, 171. [Google Scholar] [PubMed]
- Popejoy, A.B.; Fullerton, S.M. Genomics Is Failing on Diversity. Nature 2016, 538, 161–164. [Google Scholar] [PubMed] [Green Version]
- Sirugo, G.; Williams, S.M.; Tishkoff, S.A. The Missing Diversity in Human Genetic Studies. Cell 2019, 177, 26–31. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Alfonso-Sánchez, M.A.; Espinosa, I.; Gómez-Pérez, L.; Poveda, A.; Rebato, E.; Peña, J.A. Tau haplotypes support the Asian ancestry of the Roma population settled in the Basque Country. Heredity 2018, 120, 91–99. [Google Scholar] [PubMed] [Green Version]
- Patel, K.; Patel, M.; Mukhi, H. Despite Advances in Precision Medicine, Disparities Persist. Target. Ther. Oncol. 2020, 9, 16–112. [Google Scholar]
- Ingelman-Sundberg, M.; Mkrtchian, S.; Zhou, Y.; Lauschke, V.M. Integrating Rare Genetic Variants into Pharmacogenetic Drug Response Predictions. Hum. Genomics 2018, 12, 26. [Google Scholar] [CrossRef]
- Knoppers, B.M. Framework for Responsible Sharing of Genomic and Health-Related Data. HUGO J. 2014, 8, 1–6. [Google Scholar] [CrossRef] [Green Version]
- Claw, K.G.; Anderson, M.Z.; Begay, R.L.; Tsosie, K.S.; Fox, K.; Garrison, N. A Framework for Enhancing Ethical Genomic Research with Indigenous Communities. Nat. Commun. 2018, 9, 1–7. [Google Scholar] [CrossRef] [Green Version]
- Lipphardt, V.; Rappold, G.A.; Surdu, M. Representing Vulnerable Populations in Genetic Studies: The Case of the Roma. Sci. Context 2021, 34, 69–100. [Google Scholar] [CrossRef]
- Hudson, M.; Garrison, N.; Sterling, R.; Caron, N.R.; Fox, K.; Yracheta, J.; Anderson, J.; Wilcox, P.; Arbour, L.; Brown, A. Rights, Interests and Expectations: Indigenous Perspectives on Unrestricted Access to Genomic Data. Nat. Rev. Genet. 2020, 21, 377–384. [Google Scholar] [CrossRef] [PubMed]
- Lemke, A.A.; Esplin, E.D.; Goldenberg, A.J.; Gonzaga-Jauregui, C.; Hanchard, N.A.; Harris-Wai, J.; Ideozu, J.E.; Isasi, R.; Landstrom, A.P.; Prince, A.E. Addressing Underrepresentation in Genomics Research through Community Engagement. Am. J. Hum. Genet. 2022, 109, 1563–1571. [Google Scholar] [CrossRef] [PubMed]

Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ena, G.F.; Aizpurua-Iraola, J.; Font-Porterias, N.; Calafell, F.; Comas, D. Population Genetics of the European Roma—A Review. Genes 2022, 13, 2068. https://doi.org/10.3390/genes13112068
Ena GF, Aizpurua-Iraola J, Font-Porterias N, Calafell F, Comas D. Population Genetics of the European Roma—A Review. Genes. 2022; 13(11):2068. https://doi.org/10.3390/genes13112068
Chicago/Turabian StyleEna, Giacomo Francesco, Julen Aizpurua-Iraola, Neus Font-Porterias, Francesc Calafell, and David Comas. 2022. "Population Genetics of the European Roma—A Review" Genes 13, no. 11: 2068. https://doi.org/10.3390/genes13112068




