PLOD Family: A Novel Biomarker for Prognosis and Personalized Treatment in Soft Tissue Sarcoma
Abstract
:1. Introduction
2. Materials and Methods
2.1. ONCOMINE Analysis
2.2. Datasets
2.3. Kaplan–Meier Plotter Analysis
2.4. Gene Enrichment Analysis
2.5. TIMER Analysis
2.6. Immune Infiltration Analysis
3. Results
3.1. The Expression of PLOD Family Members in STS
3.2. Survival Analysis Based on the Expression of PLOD Family Members
3.3. The Expression of PLOD Family Members in Context of Age, Gender, and Grading
3.4. KEGG Pathway and GO Enrichment Analyses for PLOD Family Members
3.5. The Expression of PLOD Family Members with Tumor Immune Infiltration Cells
3.6. Cellular and Molecular Characteristics of PLOD Family Members
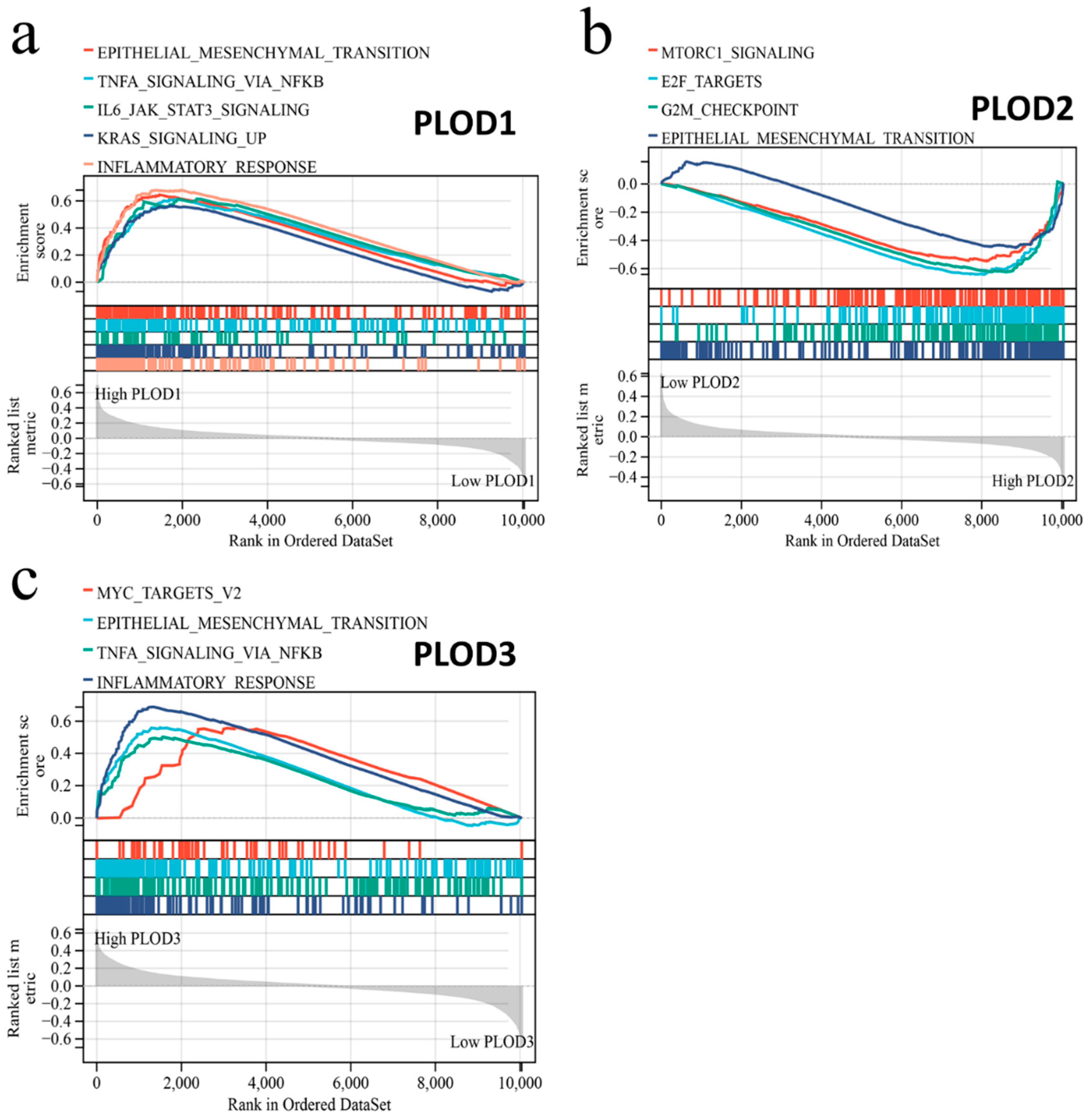
3.7. Gene Set Enrichment Analysis of PLOD Family Members
4. Discussion
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Ayodele, O.; Abdul Razak, A.R. Immunotherapy in soft-tissue sarcoma. Curr. Oncol. 2019, 26, 17–23. [Google Scholar] [CrossRef]
- Dancsok, A.R.; Asleh-Aburaya, K.; Nielsen, T.O. Advances in sarcoma diagnostics and treatment. Oncotarget 2017, 8, 7068–7093. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bourcier, K.; Le Cesne, A.; Tselikas, L.; Adam, J.; Mir, O.; Honore, C.; de Baere, T. Basic Knowledge in Soft Tissue Sarcoma. Cardiovasc. Intervent. Radiol. 2019, 42, 1255–1261. [Google Scholar] [CrossRef] [PubMed]
- Tawbi, H.A.; Burgess, M.; Bolejack, V.; Van Tine, B.A.; Schuetze, S.M.; Hu, J.; D’Angelo, S.; Attia, S.; Riedel, R.F.; Priebat, D.A.; et al. Pembrolizumab in advanced soft-tissue sarcoma and bone sarcoma (SARC028): A multicentre, two-cohort, single-arm, open-label, phase 2 trial. Lancet Oncol. 2017, 18, 1493–1501. [Google Scholar] [CrossRef]
- O’Sullivan, B.; Davis, A.M.; Turcotte, R.; Bell, R.; Catton, C.; Chabot, P.; Wunder, J.; Kandel, R.; Goddard, K.; Sadura, A.; et al. Preoperative versus postoperative radiotherapy in soft-tissue sarcoma of the limbs: A randomised trial. Lancet 2002, 359, 2235–2241. [Google Scholar] [CrossRef]
- Ratan, R.; Patel, S.R. Chemotherapy for soft tissue sarcoma. Cancer 2016, 122, 2952–2960. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Correa, R.; Gómez-Millán, J.; Lobato, M.; Fernández, A.; Ordoñez, R.; Castro, C.; Lupiañez, Y.; Medina, J.A. Radiotherapy in soft-tissue sarcoma of the extremities. Clin. Transl. Oncol. 2018, 20, 1127–1135. [Google Scholar] [CrossRef]
- Miwa, S.; Yamamoto, N.; Hayashi, K.; Takeuchi, A.; Igarashi, K.; Tsuchiya, H. Therapeutic targets for bone and soft-tissue sarcomas. Int. J. Mol. Sci. 2019, 20, 170. [Google Scholar] [CrossRef] [Green Version]
- Morales, E.; Olson, M.; Iglesias, F.; Dahiya, S.; Luetkens, T.; Atanackovic, D. Role of immunotherapy in Ewing sarcoma. J. Immunother. Cancer 2020, 8, e000653. [Google Scholar] [CrossRef]
- Von Mehren, M.; Randall, R.L.; Benjamin, R.S.; Boles, S.; Bui, M.M.; Ganjoo, K.N.; George, S.; Gonzalez, R.J.; Heslin, M.J.; Kane, J.M.; et al. Soft Tissue Sarcoma, Version 2.2018, NCCN Clinical Practice Guidelines in Oncology. J. Natl. Compr. Cancer Netw. 2018, 16, 536–563. [Google Scholar] [CrossRef]
- Casali, P.G.; Abecassis, N.; Bauer, S.; Biagini, R.; Bielack, S.; Bonvalot, S.; Boukovinas, I.; Bovee, J.V.M.G.; Brodowicz, T.; Broto, J.M.; et al. Soft tissue and visceral sarcomas: ESMO-EURACAN Clinical Practice Guidelines for diagnosis, treatment and follow-up. Ann. Oncol. Off. J. Eur. Soc. Med. Oncol. 2018, 29, iv51–iv67. [Google Scholar] [CrossRef] [PubMed]
- Gronchi, A.; Maki, R.G.; Jones, R.L. Treatment of soft tissue sarcoma: A focus on earlier stages. Future Oncol. 2017, 13, 13–21. [Google Scholar] [CrossRef] [PubMed]
- Katz, D.; Palmerini, E.; Pollack, S.M. More Than 50 Subtypes of Soft Tissue Sarcoma: Paving the Path for Histology-Driven Treatments. Am. Soc. Clin. Oncol. Educ. B 2018, 38, 925–938. [Google Scholar] [CrossRef] [PubMed]
- Ray-Coquard, I.; Serre, D.; Reichardt, P.; Martín-Broto, J.; Bauer, S. Options for treating different soft tissue sarcoma subtypes. Future Oncol. 2018, 14, 25–49. [Google Scholar] [CrossRef] [PubMed]
- Gamboa, A.C.; Gronchi, A.; Cardona, K. Soft-tissue sarcoma in adults: An update on the current state of histiotype-specific management in an era of personalized medicine. CA Cancer J. Clin. 2020, 70, 200–229. [Google Scholar] [CrossRef] [Green Version]
- Haas, R.L. Preoperative radiotherapy in soft tissue sarcoma: From general guidelines to personalized medicine. Chin. Clin. Oncol. 2018, 7, 41. [Google Scholar] [CrossRef] [Green Version]
- Reed, D.; Altiok, S. Metastatic soft tissue sarcoma chemotherapy: An opportunity for personalized medicine. Cancer Control 2011, 18, 188–195. [Google Scholar] [CrossRef] [Green Version]
- Desar, I.M.E.; Ottevanger, P.B.; Benson, C.; van der Graaf, W.T.A. Systemic treatment in adult uterine sarcomas. Crit. Rev. Oncol. Hematol. 2018, 122, 10–20. [Google Scholar] [CrossRef]
- Chamberlain, F.; Engelmann, B.; Al-Muderis, O.; Messiou, C.; Thway, K.; Miah, A.; Zaidi, S.; Constantinidou, A.; Benson, C.; Gennatas, S.; et al. Low-grade Fibromyxoid Sarcoma: Treatment outcomes and efficacy of chemotherapy. In Vivo 2020, 34, 239–245. [Google Scholar] [CrossRef]
- Skapek, S.X.; Ferrari, A.; Gupta, A.A.; Lupo, P.J.; Butler, E.; Shipley, J.; Barr, F.G.; Hawkins, D.S. Rhabdomyosarcoma. Nat. Rev. Dis. Prim. 2019, 5, 1. [Google Scholar] [CrossRef]
- Bonnans, C.; Chou, J.; Werb, Z. Remodelling the extracellular matrix in development and disease. Nat. Rev. Mol. Cell Biol. 2014, 15, 786–801. [Google Scholar] [CrossRef] [PubMed]
- Raghav, P.K.; Verma, Y.K.; Gangenahalli, G.U. Peptide screening to knockdown Bcl-2’s anti-apoptotic activity: Implications in cancer treatment. Int. J. Biol. Macromol. 2012, 50, 796–814. [Google Scholar] [CrossRef] [PubMed]
- Raghav, P.K.; Kumar, R.; Kumar, V.; Raghava, G.P.S. Docking-based approach for identification of mutations that disrupt binding between Bcl-2 and Bax proteins: Inducing apoptosis in cancer cells. Mol. Genet. Genom. Med. 2019, 7, e910. [Google Scholar] [CrossRef] [PubMed]
- De Graaff, M.A.; De Rooij, M.A.J.; Van Den Akker, B.E.W.M.; Gelderblom, H.; Chibon, F.; Coindre, J.M.; Marino-Enriquez, A.; Fletcher, J.A.; Cleton-Jansen, A.M.; Bovée, J.V.M.G. Inhibition of Bcl-2 family members sensitises soft tissue leiomyosarcomas to chemotherapy. Br. J. Cancer 2016, 114, 1219–1226. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Raghav, P.K.; Singh, A.K.; Gangenahalli, G. Stem cell factor and NSC87877 combine to enhance c-Kit mediated proliferation of human megakaryoblastic cells. PLoS ONE 2018, 13, e0206364. [Google Scholar] [CrossRef]
- Raghav, P.K.; Singh, A.K.; Gangenahalli, G. Stem cell factor and NSC87877 synergism enhances c-Kit mediated proliferation of human erythroid cells. Life Sci. 2018, 214, 84–97. [Google Scholar] [CrossRef]
- Ding, X.; Lu, D.; Fan, J. A natural product phillygenin suppresses osteosarcoma growth and metastasis by regulating the SHP-1/JAK2/STAT3 signaling. Biosci. Biotechnol. Biochem. 2021, 85, 307–314. [Google Scholar] [CrossRef]
- Qi, Y.; Xu, R. Roles of PLODs in collagen synthesis and cancer progression. Front. Cell Dev. Biol. 2018, 6, 66. [Google Scholar] [CrossRef]
- Vahidnezhad, H.; Youssefian, L.; Saeidian, A.H.; Touati, A.; Pajouhanfar, S.; Baghdadi, T.; Shadmehri, A.A.; Giunta, C.; Kraenzlin, M.; Syx, D.; et al. Mutations in PLOD3, encoding lysyl hydroxylase 3, cause a complex connective tissue disorder including recessive dystrophic epidermolysis bullosa-like blistering phenotype with abnormal anchoring fibrils and type VII collagen deficiency. Matrix Biol. 2019, 81, 91–106. [Google Scholar] [CrossRef] [Green Version]
- Li, S.S.; Lian, Y.F.; Huang, Y.L.; Huang, Y.H.; Xiao, J. Overexpressing PLOD family genes predict poor prognosis in gastric cancer. J. Cancer 2020, 11, 121–131. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Heikkinen, J.; Hautala, T.; Kivirikko, K.I.; Myllylä, R. Structure and expression of the human lysyl hydroxylase gene (PLOD): Introns 9 and 16 contain Alu sequences at the sites of recombination in Ehlers-Danlos syndrome type VI patients. Genomics 1994, 24, 464–471. [Google Scholar] [CrossRef] [PubMed]
- Eisinger-Mathason, T.S.K.; Zhang, M.; Qiu, Q.; Skuli, N.; Nakazawa, M.S.; Karakasheva, T.; Mucaj, V.; Shay, J.E.S.; Stangenberg, L.; Sadri, N.; et al. Hypoxia-dependent modification of collagen networks promotes sarcoma metastasis. Cancer Dis. 2013, 3, 1190–1205. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Valtavaara, M.; Szpirer, C.; Szpirer, J.; Myllylä, R. Primary structure, tissue distribution, and chromosomal localization of a novel isoform of lysyl hydroxylase (lysyl hydroxylase 3). J. Biol. Chem. 1998, 273, 12881–12886. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Valtavaara, M.; Papponen, H.; Pirttilä, A.M.; Hiltunen, K.; Helander, H.; Myllylä, R. Cloning and characterization of a novel human lysyl hydroxylase isoform highly expressed in pancreas and muscle. J. Biol. Chem. 1997, 272, 6831–6834. [Google Scholar] [CrossRef] [Green Version]
- Wang, Z.; Shi, Y.; Ying, C.; Jiang, Y.; Hu, J. Hypoxia-induced PLOD1 overexpression contributes to the malignant phenotype of glioblastoma via NF-κB signaling. Oncogene 2021, 40, 1458–1475. [Google Scholar] [CrossRef]
- Wang, H.; Luo, W.; Dai, L. Expression and prognostic role of PLOD1 in malignant glioma. Oncol. Targets. Ther. 2020, 13, 13285–13297. [Google Scholar] [CrossRef]
- Zhang, Y.; Wu, Y.; Su, X. PLOD1 promotes cell growth and aerobic glycolysis by regulating the SOX9/PI3K/Akt/mTOR signaling pathway in gastric cancer. Front. Biosci.-Landmark 2021, 26, 322–334. [Google Scholar] [CrossRef]
- Jiang, H.; Guo, W.; Yuan, S.; Song, L. PLOD1 is a prognostic biomarker and mediator of proliferation and invasion in osteosarcoma. Biomed Res. Int. 2020, 2020, 3418398. [Google Scholar] [CrossRef]
- Li, B.; Yang, H.; Shen, B.; Huang, J.; Qin, Z. Procollagen-lysine, 2-oxoglutarate 5-dioxygenase 1 increases cellular proliferation and colony formation capacity in lung cancer via activation of E2F transcription factor 1. Oncol. Lett. 2021, 22. [Google Scholar] [CrossRef]
- Xu, M.; Fang, S.; Xie, A. Posttranscriptional control of PLOD1 in adipose-derived stem cells regulates scar formation through altering macrophage polarization. Ann. Transl. Med. 2021, 9, 1573. [Google Scholar] [CrossRef]
- Yamada, Y.; Kato, M.; Arai, T.; Sanada, H.; Uchida, A.; Misono, S.; Sakamoto, S.; Komiya, A.; Ichikawa, T.; Seki, N. Aberrantly expressed PLOD1 promotes cancer aggressiveness in bladder cancer: A potential prognostic marker and therapeutic target. Mol. Oncol. 2019, 13, 1898–1912. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Koenig, S.N.; Cavus, O.; Williams, J.; Bernier, M.; Tonniges, J.; Sucharski, H.; Dew, T.; Akel, M.; Baker, P.; Madiai, F.; et al. New mechanistic insights to PLOD1-mediated human vascular disease. Transl. Res. 2022, 239, 1–17. [Google Scholar] [CrossRef] [PubMed]
- Qi, Q.; Huang, W.; Zhang, H.; Zhang, B.; Sun, X.; Ma, J.; Zhu, C.; Wang, C. Bioinformatic analysis of PLOD family member expression and prognostic value in non-small cell lung cancer. Transl. Cancer Res. 2021, 10, 2707–2724. [Google Scholar] [CrossRef] [PubMed]
- Gilkes, D.M.; Bajpai, S.; Chaturvedi, P.; Wirtz, D.; Semenza, G.L. Hypoxia-inducible f0actor 1 (HIF-1) promotes extracellular matrix remodeling under hypoxic conditions by inducing P4HA1, P4HA2, and PLOD2 expression in fibroblasts. J. Biol. Chem. 2013, 288, 10819–10829. [Google Scholar] [CrossRef] [Green Version]
- Van der Slot, A.J.; Zuurmond, A.M.; Bardoel, A.F.J.; Wijmenga, C.; Pruijs, H.E.H.; Sillence, D.O.; Brinckmann, J.; Abraham, D.J.; Black, C.M.; Verzijl, N.; et al. Identification of PLOD2 as Telopeptide Lysyl Hydroxylase, an Important Enzyme in Fibrosis. J. Biol. Chem. 2003, 278, 40967–40972. [Google Scholar] [CrossRef] [Green Version]
- Gistelinck, C.; Weis, M.A.; Rai, J.; Schwarze, U.; Niyazov, D.; Song, K.M.; Byers, P.H.; Eyre, D.R. Abnormal Bone Collagen Cross-Linking in Osteogenesis Imperfecta/Bruck Syndrome Caused by Compound Heterozygous PLOD2 Mutations. JBMR Plus 2021, 5, e10454. [Google Scholar] [CrossRef]
- Sun, Y.; Wang, S.; Zhang, X.; Wu, Z.; Li, Z.; Ding, Z.; Huang, X.; Chen, S.; Jing, Y.; Zhang, X.; et al. Identification and validation of plod2 as an adverse prognostic biomarker for oral squamous cell carcinoma. Biomolecules 2021, 11, 1842. [Google Scholar] [CrossRef]
- Cheriyamundath, S.; Kumar, A.; Gavert, N.; Brabletz, T.; Ben-ze’ev, A. The collagen-modifying enzyme plod2 is induced and required during l1-mediated colon cancer progression. Int. J. Mol. Sci. 2021, 22, 3552. [Google Scholar] [CrossRef]
- Du, H.; Pang, M.; Hou, X.; Yuan, S.; Sun, L. PLOD2 in cancer research. Biomed. Pharmacother. 2017, 90, 670–676. [Google Scholar] [CrossRef]
- Cao, F.; Kang, X.H.; Cui, Y.H.; Wang, Y.; Zhao, K.L.; Wang, Y.N.; Kou, W.Z.; Miao, Z.H.; Cao, X.J. Upregulation of PLOD2 promotes invasion and metastasis of osteosarcoma cells. Zhonghua Zhong Liu Za Zhi 2019, 41, 435–440. [Google Scholar] [CrossRef]
- Hu, H.-L.; Wang, C.-F.; Wei, X.-H.; Lv, J.-X.; Cao, X.-H.; Shi, Y.-Y.; Han, L.-F.; Zhang, Y.-N. Correlation between procollagen-lysine, 2-oxoglutarate 5-dioxygenase 2 and breast cancer. Int. J. Clin. Exp. Pathol. 2019, 12, 1015–1021. [Google Scholar] [PubMed]
- Li, G.; Wang, X.; Liu, G. PLOD2 Is a Potent Prognostic Marker and Associates with Immune Infiltration in Cervical Cancer. Biomed Res. Int. 2021, 2021, 5512340. [Google Scholar] [CrossRef] [PubMed]
- Du, W.; Liu, N.; Zhang, Y.; Liu, X.; Yang, Y.; Chen, W.; He, Y. PLOD2 promotes aerobic glycolysis and cell progression in colorectal cancer by upregulating HK2. Biochem. Cell Biol. 2020, 98, 386–395. [Google Scholar] [CrossRef] [PubMed]
- Cao, C.; Ma, Q.; Huang, X.; Li, A.; Liu, J.; Ye, J.; Gui, Y. Targeted Demethylation of the PLOD2 mRNA Inhibits the Proliferation and Migration of Renal Cell Carcinoma. Front. Mol. Biosci. 2021, 8, 521. [Google Scholar] [CrossRef] [PubMed]
- Saito, K.; Mitsui, A.; Sumardika, I.W.; Yokoyama, Y.; Sakaguchi, M.; Kondo, E. PLOD2-driven IL-6/STAT3 signaling promotes the invasion and metastasis of oral squamous cell carcinoma via activation of integrin β1. Int. J. Oncol. 2021, 58, 29. [Google Scholar] [CrossRef]
- Du, H.; Chen, Y.; Hou, X.; Huang, Y.; Wei, X.; Yu, X.; Feng, S.; Wu, Y.; Zhan, M.; Shi, X.; et al. Plod2 regulated by transcription factor foxa1 promotes metastasis in nsclc. Cell Death Dis. 2017, 8, e3143. [Google Scholar] [CrossRef]
- Sheng, X.; Li, Y.; Li, Y.; Liu, W.; Lu, Z.; Zhan, J.; Xu, M.; Chen, L.; Luo, X.; Cai, G.; et al. PLOD2 contributes to drug resistance in laryngeal cancer by promoting cancer stem cell-like characteristics. BMC Cancer 2019, 19, 840. [Google Scholar] [CrossRef] [Green Version]
- Okumura, Y.; Noda, T.; Eguchi, H.; Sakamoto, T.; Iwagami, Y.; Yamada, D.; Asaoka, T.; Wada, H.; Kawamoto, K.; Gotoh, K.; et al. Hypoxia-Induced PLOD2 is a Key Regulator in Epithelial-Mesenchymal Transition and Chemoresistance in Biliary Tract Cancer. Ann. Surg. Oncol. 2018, 25, 3728–3737. [Google Scholar] [CrossRef]
- Gong, S.; Duan, Y.; Wu, C.; Osterhoff, G.; Schopow, N.; Kallendrusch, S. A human pan-cancer system analysis of procollagen-lysine, 2-oxoglutarate 5-dioxygenase 3 (Plod3). Int. J. Mol. Sci. 2021, 22, 9903. [Google Scholar] [CrossRef]
- Rautavuoma, K.; Takaluoma, K.; Sormunen, R.; Myllyharju, J.; Kivirikko, K.I.; Soininen, R. Premature aggregation of type IV collagen and early lethality in lysyl hydroxylase 3 null mice. Proc. Natl. Acad. Sci. USA 2004, 101, 14120–14125. [Google Scholar] [CrossRef] [Green Version]
- Sipilä, L.; Ruotsalainen, H.; Sormunen, R.; Baker, N.L.; Lamande, S.R.; Vapola, M.; Wang, C.; Sado, Y.; Aszodi, A.; Myllyla, R. Secretion and assembly of type IV and VI collagens depend on glycosylation of hydroxylysines. J. Biol. Chem. 2007, 282, 33381–33388. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wang, B.; Xu, L.; Ge, Y.; Cai, X.; Li, Q.; Yu, Z.; Wang, J.; Wang, Y.; Lu, C.; Wang, D.; et al. PLOD3 is upregulated in gastric cancer and correlated with clinicopathologic characteristics. Clin. Lab. 2019, 65, 25–30. [Google Scholar] [CrossRef] [PubMed]
- Deng, X.; Pan, Y.; Yang, M.; Liu, Y.; Li, J. PLOD3 Is Associated with Immune Cell Infiltration and Genomic Instability in Colon Adenocarcinoma. Biomed Res. Int. 2021, 2021, 4714526. [Google Scholar] [CrossRef] [PubMed]
- Shi, J.; Bao, M.; Wang, W.; Wu, X.; Li, Y.; Zhao, C.; Liu, W. Integrated Profiling Identifies PLOD3 as a Potential Prognostic and Immunotherapy Relevant Biomarker in Colorectal Cancer. Front. Immunol. 2021, 12, 3941. [Google Scholar] [CrossRef]
- Tsai, C.K.; Huang, L.C.; Tsai, W.C.; Huang, S.M.; Lee, J.T.; Hueng, D.Y. Overexpression of PLOD3 promotes tumor progression and poor prognosis in gliomas. Oncotarget 2018, 9, 15705–15720. [Google Scholar] [CrossRef] [Green Version]
- Zhang, J.; Tian, Y.; Mo, S.; Fu, X. Overexpressing PLOD Family Genes Predict Poor Prognosis in Pancreatic Cancer. Int. J. Gen. Med. 2022, 15, 3077–3096. [Google Scholar] [CrossRef]
- Yang, B.; Zhao, Y.; Wang, L.; Zhao, Y.; Wei, L.; Chen, D.; Chen, Z. Identification of PLOD Family Genes as Novel Prognostic Biomarkers for Hepatocellular Carcinoma. Front. Oncol. 2020, 10, 1695. [Google Scholar] [CrossRef]
- Meng, Y.; Sun, J.; Zhang, G.; Yu, T.; Piao, H. Clinical Prognostic Value of the PLOD Gene Family in Lung Adenocarcinoma. Front. Mol. Biosci. 2022, 8, 770729. [Google Scholar] [CrossRef]
- Gong, S.; Wu, C.; Köhler, F.; Meixensberger, J.; Schopow, N.; Kallendrusch, S. Procollagen-Lysine, 2-Oxoglutarate 5-Dioxygenase Family: Novel Prognostic Biomarkers and Tumor Microenvironment Regulators for Lower-Grade Glioma. Front. Cell. Neurosci. 2022, 16, 838548. [Google Scholar] [CrossRef]
- Charoentong, P.; Finotello, F.; Angelova, M.; Mayer, C.; Efremova, M.; Rieder, D.; Hackl, H.; Trajanoski, Z. Pan-cancer Immunogenomic Analyses Reveal Genotype-Immunophenotype Relationships and Predictors of Response to Checkpoint Blockade. Cell Rep. 2017, 18, 248–262. [Google Scholar] [CrossRef] [Green Version]
- Yoshihara, K.; Shahmoradgoli, M.; Martínez, E.; Vegesna, R.; Kim, H.; Torres-Garcia, W.; Treviño, V.; Shen, H.; Laird, P.W.; Levine, D.A.; et al. Inferring tumour purity and stromal and immune cell admixture from expression data. Nat. Commun. 2013, 4, 2612. [Google Scholar] [CrossRef] [PubMed]
- Subramanian, A.; Tamayo, P.; Mootha, V.K.; Mukherjee, S.; Ebert, B.L.; Gillette, M.A.; Paulovich, A.; Pomeroy, S.L.; Golub, T.R.; Lander, E.S.; et al. Gene set enrichment analysis: A knowledge-based approach for interpreting genome-wide expression profiles. Proc. Natl. Acad. Sci. USA 2005, 102, 15545–15550. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hjalt, T.A.; Amendt, B.A.; Murray, J.C. PITX2 regulates procollagen lysyl hydroxylase (PLOD) gene expression: Implications for the pathology of Rieger syndrome. J. Cell Biol. 2001, 153, 545–552. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Pastushenko, I.; Blanpain, C. EMT Transition States during Tumor Progression and Metastasis. Trends Cell Biol. 2019, 29, 212–226. [Google Scholar] [CrossRef] [Green Version]
- Dongre, A.; Weinberg, R.A. New insights into the mechanisms of epithelial–mesenchymal transition and implications for cancer. Nat. Rev. Mol. Cell Biol. 2019, 20, 69–84. [Google Scholar] [CrossRef] [PubMed]
- Yue, X.Y.; Sun, X.F.; Che, K.; Hu, J.X.; Lv, W.S.; Sun, X.L.; Geng, Z.; Chi, J.W.; Wang, Y.G. Lpl-C310R mutation is associated with impaired glucose tolerance and endoplasmic reticulum stress in skeletal muscle. Biochem. Biophys. Res. Commun. 2020, 529, 480–486. [Google Scholar] [CrossRef] [PubMed]
- Oakes, S.A.; Papa, F.R. The role of endoplasmic reticulum stress in human pathology. Annu. Rev. Pathol. Mech. Dis. 2015, 10, 173–194. [Google Scholar] [CrossRef] [Green Version]
- Lim, P.J.; Lindert, U.; Opitz, L.; Hausser, I.; Rohrbach, M.; Giunta, C. Transcriptome profiling of primary skin fibroblasts reveal distinct molecular features between PLOD1-and FKBP14-kyphoscoliotic Ehlers–Danlos syndrome. Genes 2019, 10, 517. [Google Scholar] [CrossRef] [Green Version]
- Baek, J.H.; Yun, H.S.; Kwon, G.T.; Lee, J.; Kim, J.Y.; Jo, Y.; Cho, J.M.; Lee, C.W.; Song, J.Y.; Ahn, J.; et al. PLOD3 suppression exerts an anti-tumor effect on human lung cancer cells by modulating the PKC-delta signaling pathway. Cell Death Dis. 2019, 10, 156. [Google Scholar] [CrossRef]
- Amodio, G.; Sasso, E.; D’Ambrosio, C.; Scaloni, A.; Moltedo, O.; Franceschelli, S.; Zambrano, N.; Remondelli, P. Identification of a microRNA (miR-663a) induced by ER stress and its target gene PLOD3 by a combined microRNome and proteome approach. Cell Biol. Toxicol. 2016, 32, 285–303. [Google Scholar] [CrossRef]
- Zhang, W.G.; Chen, L.; Dong, Q.; He, J.; Zhao, H.D.; Li, F.l.; Li, H. Mmu-miR-702 functions as an anti-apoptotic mirtron by mediating ATF6 inhibition in mice. Gene 2013, 531, 235–242. [Google Scholar] [CrossRef] [PubMed]
- Zheng, X.; Jin, X.; Liu, X.; Liu, B.; Li, P.; Ye, F.; Zhao, T.; Chen, W.; Li, Q. Inhibition of endoplasmic reticulum stress-induced autophagy promotes the killing effect of X-rays on sarcoma in mice. Biochem. Biophys. Res. Commun. 2020, 522, 612–617. [Google Scholar] [CrossRef] [PubMed]
- Kraus, M.; Malenke, E.; Gogel, J.; Müller, H.; Rückrich, T.; Overkleeft, H.; Ovaa, H.; Koscielniak, E.; Hartmann, J.T.; Driessen, C. Ritonavir induces endoplasmic reticulum stress and sensitizes sarcoma cells toward bortezomib-induced apoptosis. Mol. Cancer Ther. 2008, 7, 1940–1948. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wu, T.; Dai, Y. Tumor microenvironment and therapeutic response. Cancer Lett. 2017, 387, 61–68. [Google Scholar] [CrossRef]
- Denton, A.E.; Roberts, E.W.; Fearon, D.T. Stromal cells in the tumor microenvironment. Adv. Exp. Med. Biol. 2018, 1060, 99–114. [Google Scholar] [CrossRef]
- Jin, M.Z.; Jin, W.L. The updated landscape of tumor microenvironment and drug repurposing. Signal Transduct. Target. Ther. 2020, 5, 166. [Google Scholar] [CrossRef]
- Alahari, S.; Farrell, A.; Ermini, L.; Park, C.; Sallais, J.; Roberts, S.; Gillmore, T.; Litvack, M.; Post, M.; Caniggia, I. JMJD6 Dysfunction Due to Iron Deficiency in Preeclampsia Disrupts Fibronectin Homeostasis Resulting in Diminished Trophoblast Migration. Front. Cell Dev. Biol. 2021, 9, 987. [Google Scholar] [CrossRef]
- Raghav, P.K.; Mann, Z. Cancer stem cells targets and combined therapies to prevent cancer recurrence. Life Sci. 2021, 277, 119465. [Google Scholar] [CrossRef]
- Qing, L.; Chen, H.; Tang, J.; Jia, X. Exosomes and Their MicroRNA Cargo: New Players in Peripheral Nerve Regeneration. Neurorehabil. Neural Repair 2018, 32, 765–776. [Google Scholar] [CrossRef] [Green Version]
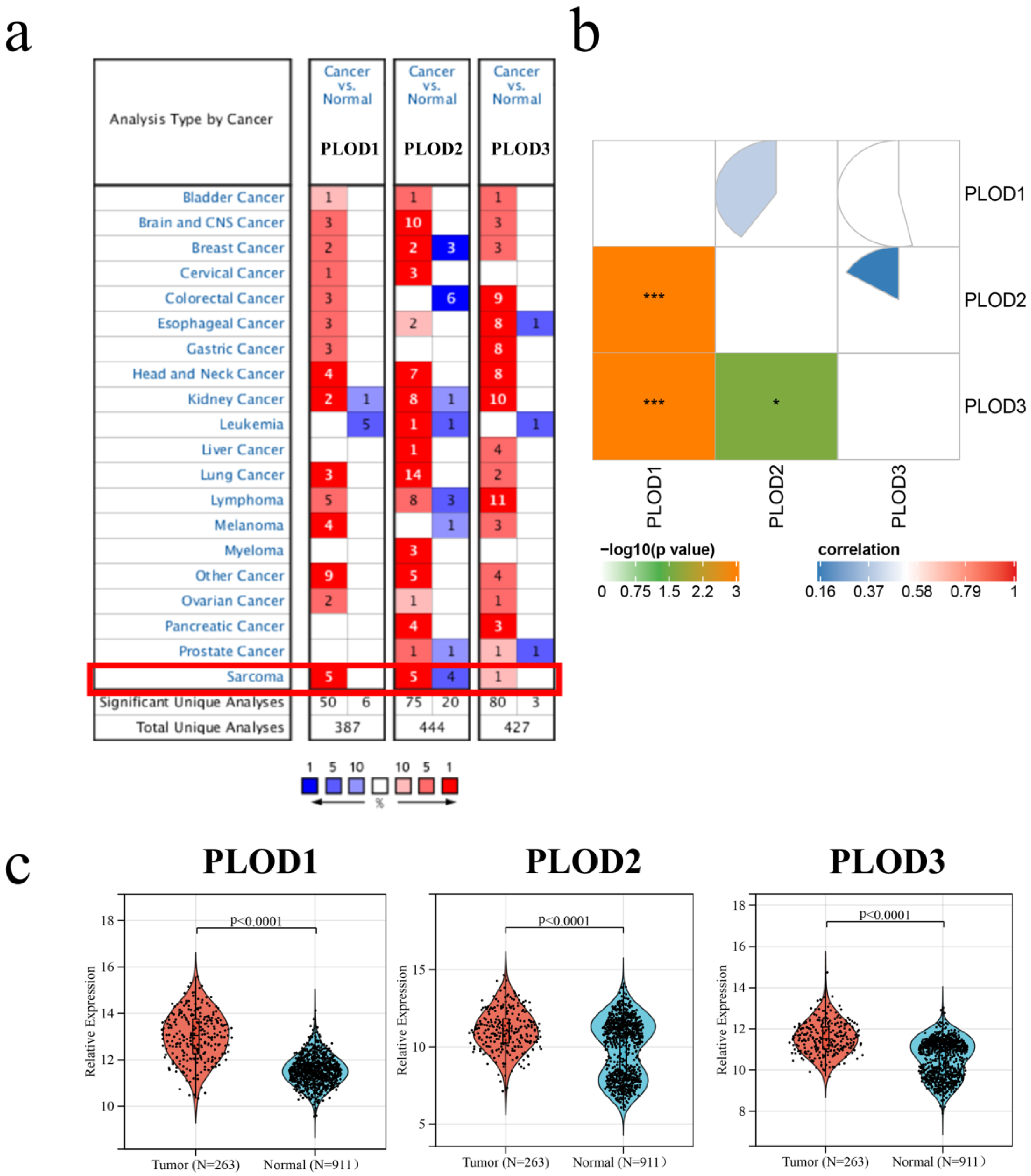






| Characteristics | All (N = 189) |
|---|---|
| Age group (Median) | |
| Younger (≤60 years) | 96 (50.79%) |
| Older (>60 years) | 93 (49.21%) |
| Gender | |
| Male | 87 (46.03%) |
| Female | 102 (53.97%) |
| Pathologic tumor size | |
| ≤10.5 cm | 95 (50.26%) |
| >10.5 cm | 94 (49.74%) |
| Radiotherapy | |
| Yes | 54 (28.57%) |
| No | 135 (71.43%) |
| Pharmaceutical therapy | |
| Yes | 45 (23.81%) |
| No | 144 (76.19%) |
| FNCLCC grade | |
| 1/2 | 115 (60.85%) |
| 3 | 74 (39.15%) |
| Vital status | |
| Alive | 117 (61.90%) |
| Dead | 72 (38.10%) |
| Histological type | |
| DDLPS | 49 (25.93%) |
| LMS | 68 (35.98%) |
| UPS | 41 (21.69%) |
| MFS | 17 (8.99%) |
| SS | 10 (5.29%) |
| MPNST | 4 (2.12%) |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Gong, S.; Schopow, N.; Duan, Y.; Wu, C.; Kallendrusch, S.; Osterhoff, G. PLOD Family: A Novel Biomarker for Prognosis and Personalized Treatment in Soft Tissue Sarcoma. Genes 2022, 13, 787. https://doi.org/10.3390/genes13050787
Gong S, Schopow N, Duan Y, Wu C, Kallendrusch S, Osterhoff G. PLOD Family: A Novel Biomarker for Prognosis and Personalized Treatment in Soft Tissue Sarcoma. Genes. 2022; 13(5):787. https://doi.org/10.3390/genes13050787
Chicago/Turabian StyleGong, Siming, Nikolas Schopow, Yingjuan Duan, Changwu Wu, Sonja Kallendrusch, and Georg Osterhoff. 2022. "PLOD Family: A Novel Biomarker for Prognosis and Personalized Treatment in Soft Tissue Sarcoma" Genes 13, no. 5: 787. https://doi.org/10.3390/genes13050787
APA StyleGong, S., Schopow, N., Duan, Y., Wu, C., Kallendrusch, S., & Osterhoff, G. (2022). PLOD Family: A Novel Biomarker for Prognosis and Personalized Treatment in Soft Tissue Sarcoma. Genes, 13(5), 787. https://doi.org/10.3390/genes13050787







