Prenatal Diagnosis of Talipes Equinovarus by Ultrasound and Chromosomal Microarray Analysis: A Chinese Single-Center Retrospective Study
Abstract
:1. Introduction
2. Materials and Methods
2.1. Study Cohort
2.2. Chromosomal Microarray Analysis
2.3. Statistical Analysis
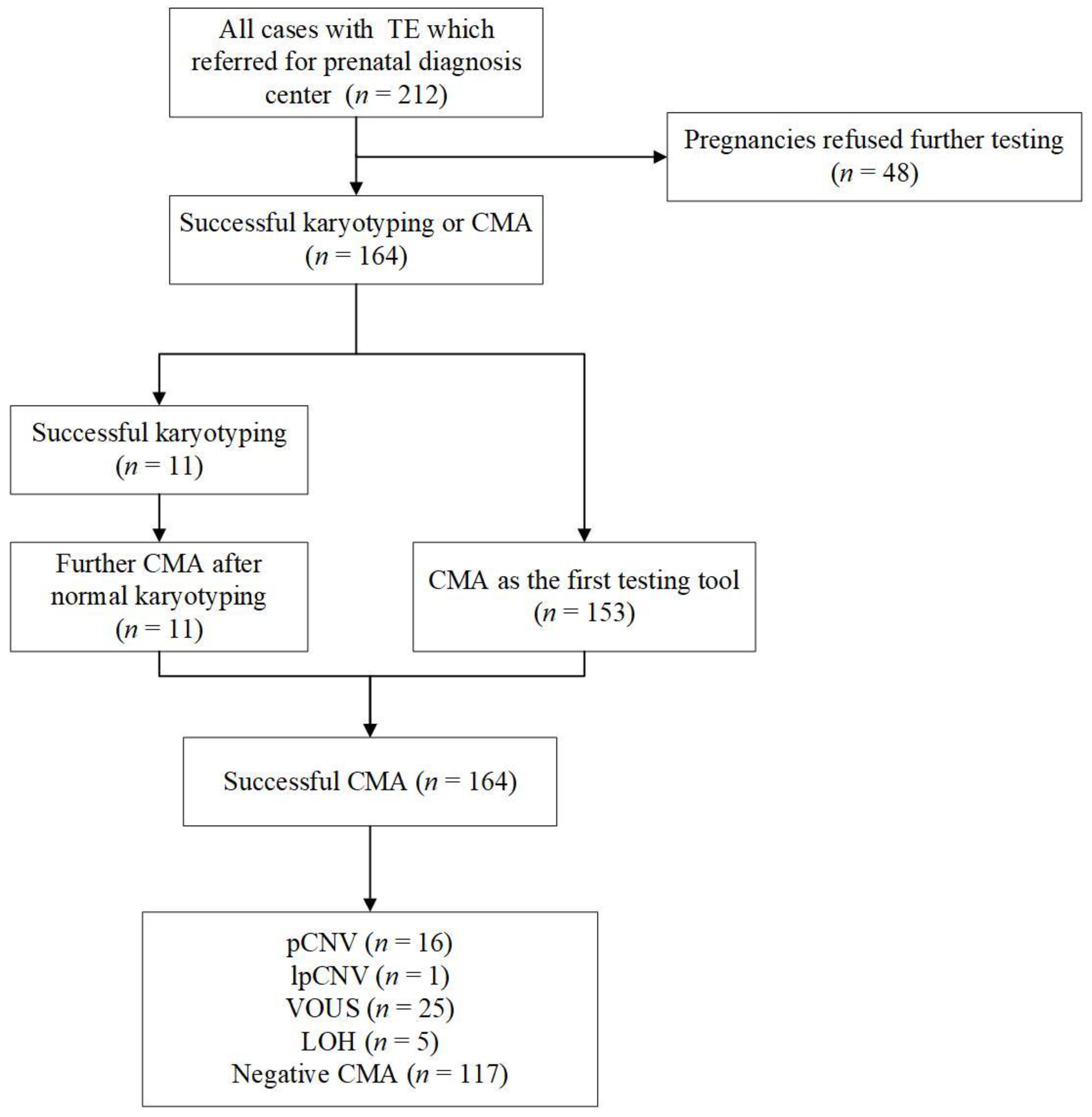
3. Results
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
References
- Sucu, M.; Demir, S.C. The relationship between isolated pes equinovarus and aneuploidies and perinatal outcomes: Results of a tertiary center. Turk. J. Obstet. Gynecol. 2020, 17, 270–277. [Google Scholar] [CrossRef]
- Faldini, C.; Fenga, D.; Sanzarello, I.; Nanni, M.; Traina, F.; Rosa, M.A.A. Prenatal Diagnosis of Clubfoot: A Review of Current Available Methodology. Folia Med. 2017, 59, 247–253. [Google Scholar] [CrossRef] [PubMed]
- Ruzzini, L.; De Salvatore, S.; Longo, U.G.; Marino, M.; Greco, A.; Piergentili, I.; Costici, P.F.; Denaro, V. Prenatal Diagnosis of Clubfoot: Where Are We Now? Systematic Review and Meta-Analysis. Diagnostics 2021, 11, 2235. [Google Scholar] [CrossRef] [PubMed]
- McKinney, J.; Rac, M.W.F.; Gandhi, M. Congenital talipes equinovarus (clubfoot). Am. J. Obstet. Gynecol. 2019, 221, B10–B12. [Google Scholar] [CrossRef]
- Ester, A.R.; Weymouth, K.S.; Burt, A.; Wise, C.A.; Scott, A.; Gurnett, C.A.; Dobbs, M.B.; Blanton, S.H.; Hecht, J.T. Altered transmission of HOX and apoptotic SNPs identify a potential common pathway for clubfoot. Am. J. Med. Genet. A 2009, 149A, 2745–2752. [Google Scholar] [CrossRef]
- Menke, D.B.; Guenther, C.; Kingsley, D.M. Dual hindlimb control elements in the Tbx4 gene and region-specific control of bone size in vertebrate limbs. Development 2008, 135, 2543–2553. [Google Scholar] [CrossRef] [PubMed]
- Yang, H.; Zheng, Z.; Cai, H.; Li, H.; Ye, X.; Zhang, X.; Wang, Z.; Fu, Q. Three novel missense mutations in the filamin B gene are associated with isolated congenital talipes equinovarus. Hum. Genet. 2016, 135, 1181–1189. [Google Scholar] [CrossRef]
- Sanzarello, I.; Nanni, M.; Faldini, C. The clubfoot over the centuries. J. Pediatr. Orthop. B 2017, 26, 143–151. [Google Scholar] [CrossRef]
- Viaris de le Segno, B.; Gruchy, N.; Bronfen, C.; Dolley, P.; Leporrier, N.; Creveuil, C.; Benoist, G. Prenatal diagnosis of clubfoot: Chromosomal abnormalities associated with fetal defects and outcome in a tertiary center. J. Clin. Ultrasound. 2016, 44, 100–105. [Google Scholar] [CrossRef]
- Singer, A.; Maya, I.; Banne, E.; Feldman, H.B.; Vinkler, C.; Ben Shachar, S.; Sagi-Dain, L. Prenatal clubfoot increases the risk for clinically significant chromosomal microarray results—Analysis of 269 singleton pregnancies. Early Hum. Dev. 2020, 145, 105047. [Google Scholar] [CrossRef]
- Alvarado, D.M.; Buchan, J.G.; Frick, S.L.; Herzenberg, J.E.; Dobbs, M.B.; Gurnett, C.A. Copy number analysis of 413 isolated talipes equinovarus patients suggests role for transcriptional regulators of early limb development. Eur. J. Hum. Genet. 2013, 21, 373–380. [Google Scholar] [CrossRef] [PubMed]
- Liao, C.; Fu, F.; Li, R.; Xie, G.-E.; Zhang, Y.-L.; Li, J.; Li, D.-Z. Implementation of high-resolution SNP arrays in the investigation of fetuses with ultrasound malformations: 5 years of clinical experience. Clin. Genet. 2014, 86, 264–269. [Google Scholar] [CrossRef] [PubMed]
- Riggs, E.R.; Andersen, E.F.; Cherry, A.M.; Kantarci, S.; Kearney, H.; Patel, A.; Raca, G.; Ritter, D.I.; South, S.T.; Thorland, E.C. Technical standards for the interpretation and reporting of constitutional copy-number variants: A joint consensus recommendation of the American College of Medical Genetics and Genomics (ACMG) and the Clinical Genome Resource (ClinGen). Genet. Med. 2020, 22, 245–257. [Google Scholar] [CrossRef]
- Li, M.; Li, J.; Fu, H.; Xu, J.; Li, Q.; Lin, L.; Geng, G.; Ouyang, L. Genetics analysis of fetal foot varus during the second and third trimester of pregnancy by single nucleotide polymorphisms array. Chin. J. Fam. Plann. 2021, 29, 1757–1760. [Google Scholar]
- Fantasia, I.; Dibello, D.; Di Carlo, V.; Colin, G.; Barbieri, M.; Belcaro, C.; Magni, E.; Faletra, F.; Laura, T.; Stampalija, T. Prenatal diagnosis of isolated clubfoot: Diagnostic accuracy and long-term postnatal outcomes. Eur. J. Obstet. Gynecol. Reprod. Biol. 2021, 264, 60–64. [Google Scholar] [CrossRef] [PubMed]
- Basit, S.; Khoshhal, K.I. Genetics of clubfoot; recent progress and future perspectives. Eur. J. Med. Genet. 2018, 61, 107–113. [Google Scholar] [CrossRef]
- Homans, J.F.; Tromp, I.N.; Colo, D.; Schlösser, T.P.C.; Kruyt, M.C.; Deeney, V.F.X.; Crowley, T.B.; McDonald-McGinn, D.M.; Castelein, R.M. Orthopaedic manifestations within the 22q11.2 Deletion syndrome: A systematic review. Am. J. Med. Genet. A 2018, 176, 2104–2120. [Google Scholar] [CrossRef]
- Campbell, H.; Carothers, A.D.; Rudan, I.; Hayward, C.; Biloglav, Z.; Barac, L.; Pericic, M.; Janicijevic, B.; Smolej-Narancic, N.; Polasek, O.; et al. Effects of genome-wide heterozygosity on a range of biomedically relevant human quantitative traits. Hum. Mol. Genet. 2007, 16, 233–241. [Google Scholar] [CrossRef]
- Ku, C.S.; Naidoo, N.; Teo, S.M.; Pawitan, Y. Regions of homozygosity and their impact on complex diseases and traits. Hum. Genet. 2011, 129, 1–15. [Google Scholar] [CrossRef]
- Robinson, W.P. Mechanisms leading to uniparental disomy and their clinical consequences. Bioessays 2000, 22, 452–459. [Google Scholar] [CrossRef]
- Yamazawa, K.; Ogata, T.; Ferguson-Smith, A.C. Uniparental disomy and human disease: An overview. Am. J. Med. Genet. C Semin. Med. Genet. 2010, 154C, 329–334. [Google Scholar] [CrossRef] [PubMed]
- Lapunzina, P.; Monk, D. The consequences of uniparental disomy and copy number neutral loss-of-heterozygosity during human development and cancer. Biol. Cell 2011, 103, 303–317. [Google Scholar] [CrossRef] [PubMed]
- Liu, J.; He, Z.; Lin, S.; Wang, Y.; Huang, L.; Huang, X.; Luo, Y. Absence of heterozygosity detected by single-nucleotide polymorphism array in prenatal diagnosis. Ultrasound. Obstet. Gynecol. 2021, 57, 314–323. [Google Scholar] [CrossRef] [PubMed]
- Hordyjewska-Kowalczyk, E.; Nowosad, K.; Jamsheer, A.; Tylzanowski, P. Genotype-phenotype correlation in clubfoot (talipes equinovarus). J. Med. Genet. 2022, 59, 209–219. [Google Scholar] [CrossRef] [PubMed]
- Liehr, T.; Rautenstrauss, B.; Grehl, H.; Bathke, K.D.; Ekici, A.; Rauch, A.; Rott, H.-D. Mosaicism for the Charcot-Marie-Tooth disease type 1A duplication suggests somatic reversion. Hum. Genet. 1996, 98, 22–28. [Google Scholar] [CrossRef]
- Cervera, M.; Sánchez, S.; Molina, B.; Alcántara-Ortigoza, M.A.; Del Castillo, V.; Carnevale, A.; Angel, A.G.-D. Trisomy of the short arm of chromosome 5 due to a de novo inversion and duplication (5)(p15.3 p13.3). Am. J. Med. Genet. A 2005, 136A, 381–385. [Google Scholar] [CrossRef]
- Collinson, J.M.; Lindström, N.O.; Neves, C.; Wallace, K.; Meharg, C.; Charles, R.H.; Ross, Z.K.; Fraser, A.M.; Mbogo, I.; Oras, K.; et al. The developmental and genetic basis of ‘clubfoot’ in the peroneal muscular atrophy mutant mouse. Development 2018, 145, dev160093. [Google Scholar] [CrossRef]
- Bill, P.L.; Versfeld, G.A. Congenital clubfoot: An electromyographic study. J. Pediatr. Orthop. 1982, 2, 139–142. [Google Scholar] [CrossRef]
- Feldbrin, Z.; Gilai, A.N.; Ezra, E.; Khermosh, O.; Kramer, U.; Wientroub, S. Muscle imbalance in the aetiology of idiopathic club foot. An electromyographic study. J. Bone Jt. Surg. Br. 1995, 77, 596–601. [Google Scholar] [CrossRef]
- Herceg, M.B.; Weiner, D.S.; Agamanolis, D.P.; Hawk, D. Histologic and histochemical analysis of muscle specimens in idiopathic talipes equinovarus. J. Pediatr. Orthop. 2006, 26, 91–93. [Google Scholar] [CrossRef]

| Case | Maternal Age (years) | GA (weeks) | Ultrasound Findings | CMA Results | Type of CNV | Size (Mb) | Outcome |
|---|---|---|---|---|---|---|---|
| 1 * | 29.6 | 30.6 | Isolated TE | arrXp22.31(6,449,752–8,143,319) × 1 | Deletion | 1.69 | Live birth |
| 2 | 39.9 | 18.9 | Isolated TE | arr22q11.21(18,636,749−21,800,471) × 1 | Deletion | 3.16 | TOP |
| 3 | 20.1 | 28 | Isolated TE | arr17p12(14,087,918−15,503,234) × 3 | Duplication | 1.42 | TOP |
| 4 | 30.3 | 26 | Isolated TE | arr22q11.21(18,916,842−21,800,471) × 1 | Deletion | 2.88 | TOP |
| 5 | 34.9 | 28.6 | Isolated TE | arr18q21.32q23(57,600,965−78,014,123) × 1 | Deletion | 20.41 | TOP |
| 6 | 28.2 | 28.4 | Isolated TE | arr22q11.21(18,916,842−21,465,662) × 1 | Deletion | 2.55 | TOP |
| 7 | 26.4 | 24 | Isolated TE | arr(21) × 2~3 | Duplication | 33.09 | TOP |
| 8 | 27.9 | 26.4 | TE; VSD | arr4p16.3p15.33(68,345−14,195,870) × 1 | Deletion | 14.13 | TOP |
| 9 | 39.3 | 20.7 | TE; CPCs | arr(18) × 3 | Duplication | 77.88 | TOP |
| 10 | 31.1 | 19.6 | TE; VSD | arr22q11.21(18,916,842−21,465,662) × 1 | Deletion | 2.55 | TOP |
| 11 | 26.0 | 19 | TE; CLP; HPE; SGA | arr6p25.3p24.3(156,975−9,116,357) × 3 arr21q22.13q22.3(38,242,327−48,093,361) × 1 | Duplication Deletion | 8.96 9.85 | TOP |
| 12 # | 29.2 | 33 | TE; oligohydramnios | arr22q11.21(20,717,654−21,465,659) × 1 | Deletion | 0.75 | Live birth |
| 13 | 28.9 | 24.6 | TE; cholecystomegaly | arr16p13.11(14,896,385−16,328,840) × 3 | Duplication | 1.43 | Live birth |
| 14 | 28.6 | 25.7 | TE; iCTR | arr5p15.33p13.2(2,103,059−37,483,088) × 3 | Duplication | 25.38 | TOP |
| 15 | 35.9 | 25 | TE; VSD | arr(18) × 3 | Duplication | 77.88 | TOP |
| 16 | 32.9 | 23.4 | TE; FGR; porencephaly | arr13q22.1q33.1(74,307,209−102,461,029) × 1 | Deletion | 28.15 | TOP |
| 17 | 31.5 | 12.7 | TE; CH; omphalocele | arr(18) × 3 | Duplication | 77.88 | TOP |
| Groups | CSVs | VOUS | Live Birth | TOP |
|---|---|---|---|---|
| Singleton vs. Twins | ||||
| Singleton (n = 148) | 16 (10.8%) | 24 (16.2%) | 95 (64.2%) | 48 (32.4%) |
| Twins (n = 16) | 1 (6.3%) | 1 (6.3%) | 8 (50.0%) | 7 (43.8%) |
| p-value | 0.891 | 0.492 | 0.265 | 0.362 |
| Isolated vs. Non-isolated * | ||||
| Isolated (n = 111) | 6 (5.4%) | 19 (17.1%) | 85 (76.6%) | 22 (19.8%) |
| Non-isolated (n = 37) | 10 (27.0%) | 5 (13.5%) | 10 (27.0%) | 26 (70.3%) |
| p-value | 0.000 | 0.607 | 0.000 | 0.000 |
| Unilateral vs. Bilateral * | ||||
| Unilateral (n = 69) | 8 (11.6%) | 14 (20.3%) | 38 (55.1%) | 28 (40.6%) |
| Bilateral (n = 79) | 8 (10.1%) | 10 (12.7%) | 57 (72.2%) | 20 (25.3%) |
| p-value | 0.774 | 0.209 | 0.031 | 0.048 |
| Left foot vs. Right foot * | ||||
| Left foot (n = 32) | 2 (6.3%) | 5 (15.6%) | 20 (62,5%) | 12 (37.5%) |
| Right foot (n = 37) | 6 (16.2%) | 9 (24.3%) | 18 (48.6%) | 16 (43.2%) |
| p-value | 0.362 | 0.370 | 0.249 | 0.752 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Huang, R.; Yang, X.; Zhou, H.; Fu, F.; Cheng, K.; Wang, Y.; Ma, C.; Li, R.; Jing, X.; Han, J.; et al. Prenatal Diagnosis of Talipes Equinovarus by Ultrasound and Chromosomal Microarray Analysis: A Chinese Single-Center Retrospective Study. Genes 2022, 13, 1573. https://doi.org/10.3390/genes13091573
Huang R, Yang X, Zhou H, Fu F, Cheng K, Wang Y, Ma C, Li R, Jing X, Han J, et al. Prenatal Diagnosis of Talipes Equinovarus by Ultrasound and Chromosomal Microarray Analysis: A Chinese Single-Center Retrospective Study. Genes. 2022; 13(9):1573. https://doi.org/10.3390/genes13091573
Chicago/Turabian StyleHuang, Ruibin, Xin Yang, Hang Zhou, Fang Fu, Ken Cheng, You Wang, Chunling Ma, Ru Li, Xiangyi Jing, Jin Han, and et al. 2022. "Prenatal Diagnosis of Talipes Equinovarus by Ultrasound and Chromosomal Microarray Analysis: A Chinese Single-Center Retrospective Study" Genes 13, no. 9: 1573. https://doi.org/10.3390/genes13091573
APA StyleHuang, R., Yang, X., Zhou, H., Fu, F., Cheng, K., Wang, Y., Ma, C., Li, R., Jing, X., Han, J., Zhen, L., Pan, M., Li, D., & Liao, C. (2022). Prenatal Diagnosis of Talipes Equinovarus by Ultrasound and Chromosomal Microarray Analysis: A Chinese Single-Center Retrospective Study. Genes, 13(9), 1573. https://doi.org/10.3390/genes13091573





