Plant Development in the Garden Pea as Revealed by Mutations in the Crd/PsYUC1 Gene
Abstract
:1. Introduction
2. Materials and Methods
2.1. Plant Lines
2.2. Growth Conditions
2.3. Synthesis of oxIAA-Asp
2.4. Extraction and LC-MS Analysis of IAA, IAA-Asp, and oxIAA-Asp
3. Results
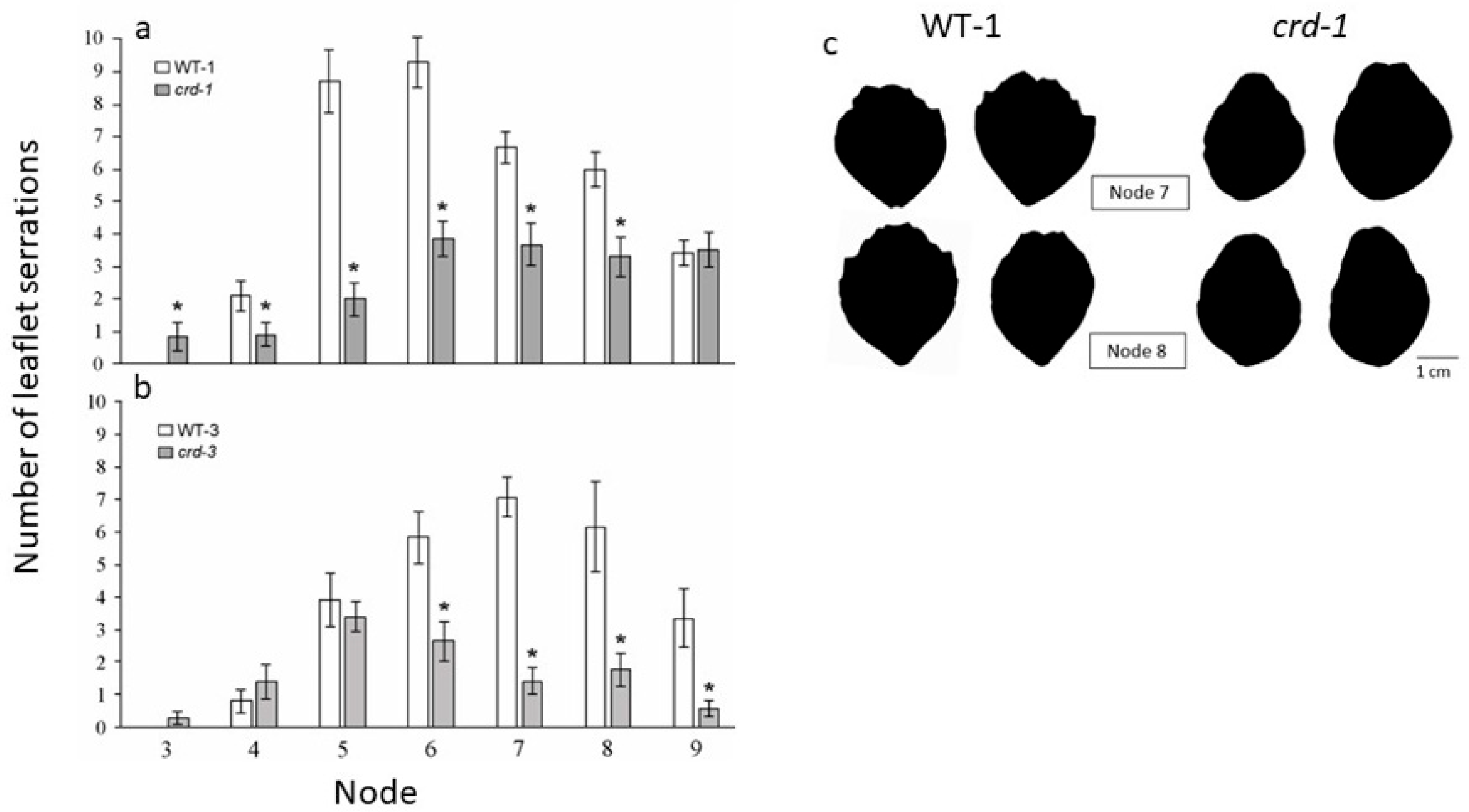
3.1. Effects of crd Mutations on Leaf Development
3.1.1. Leaf Organ Initiation Is Reduced in crd Mutants
3.1.2. Leaf Organ Identity Is Altered in crd Mutants
3.1.3. Additional Effects of the crd Mutations on Leaf Development
3.2. crd Mutants Exhibit Floral Abnormalities
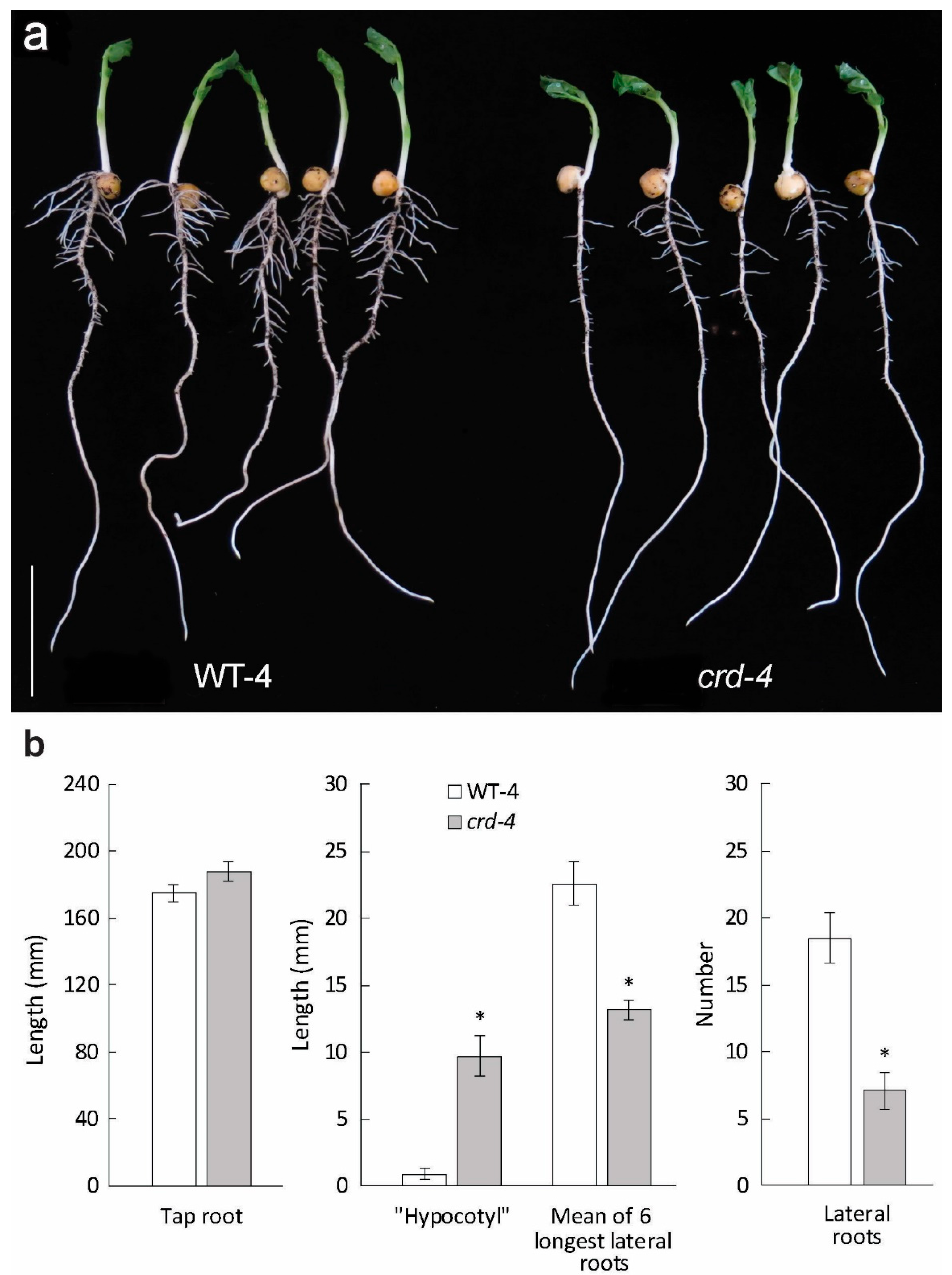
3.3. A Further crd Mutation, crd-4, Affects Plant Development Similarly to crd-1 and crd-3
3.4. Effects of crd Mutations on Root Development
3.5. Pisum YUC Genes
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Zhao, B.; He, L.; Jiang, C.; Liu, Y.; He, H.; Bai, Q.; Zhou, S.; Zheng, X.; Wen, J.; Mysore, K.S. Lateral Leaflet Suppression 1 (LLS1), encoding the MtYUCCA1 protein, regulates lateral leaflet development in Medicago truncatula. New Phytol. 2020, 227, 613–628. [Google Scholar] [CrossRef]
- Cheng, Y.; Dai, X.; Zhao, Y. Auxin biosynthesis by the YUCCA flavin monooxygenases controls the formation of floral organs and vascular tissues in Arabidopsis. Genes Dev. 2006, 20, 1790–1799. [Google Scholar] [CrossRef]
- Cheng, Y.; Dai, X.; Zhao, Y. Auxin synthesized by the YUCCA flavin monooxygenases is essential for embryogenesis and leaf formation in Arabidopsis. Plant Cell 2007, 19, 2430–2439. [Google Scholar] [CrossRef] [PubMed]
- Tobeña-Santamaria, R.; Bliek, M.; Ljung, K.; Sandberg, G.; Mol, J.N.; Souer, E.; Koes, R. FLOOZY of petunia is a flavin mono-oxygenase-like protein required for the specification of leaf and flower architecture. Genes Dev. 2002, 16, 753–763. [Google Scholar] [CrossRef] [PubMed]
- Lu, L.; Pi, M.; Liu, Z.; Kang, C. Auxin biosynthesis gene FveYUC4 is critical for leaf and flower morphogenesis in woodland strawberry. Plant J. 2023, 115, 1428–1442. [Google Scholar] [CrossRef] [PubMed]
- Phillips, K.A.; Skirpan, A.L.; Liu, X.; Christensen, A.; Slewinski, T.L.; Hudson, C.; Barazesh, S.; Cohen, J.D.; Malcomber, S.; McSteen, P. vanishing tassel2 encodes a grass-specific tryptophan aminotransferase required for vegetative and reproductive development in maize. Plant Cell 2011, 23, 550–566. [Google Scholar] [CrossRef] [PubMed]
- Woo, Y.-M.; Park, H.-J.; Su’udi, M.; Yang, J.-I.; Park, J.-J.; Back, K.; Park, Y.-M.; An, G. Constitutively wilted 1, a member of the rice YUCCA gene family, is required for maintaining water homeostasis and an appropriate root to shoot ratio. Plant Mol. Biol. 2007, 65, 125–136. [Google Scholar] [CrossRef] [PubMed]
- Yoshikawa, T.; Ito, M.; Sumikura, T.; Nakayama, A.; Nishimura, T.; Kitano, H.; Yamaguchi, I.; Koshiba, T.; Hibara, K.I.; Nagato, Y. The rice FISH BONE gene encodes a tryptophan aminotransferase, which affects pleiotropic auxin-related processes. Plant J. 2014, 78, 927–936. [Google Scholar] [CrossRef] [PubMed]
- Pacheco-Villalobos, D.; Sankar, M.; Ljung, K.; Hardtke, C.S. Disturbed local auxin homeostasis enhances cellular anisotropy and reveals alternative wiring of auxin-ethylene crosstalk in Brachypodium distachyon seminal roots. PLoS Genet. 2013, 9, e1003564. [Google Scholar] [CrossRef]
- Mashiguchi, K.; Tanaka, K.; Sakai, T.; Sugawara, S.; Kawaide, H.; Natsume, M.; Hanada, A.; Yaeno, T.; Shirasu, K.; Yao, H. The main auxin biosynthesis pathway in Arabidopsis. Proc. Natl. Acad. Sci. USA 2011, 108, 18512–18517. [Google Scholar] [CrossRef]
- Berbel, A.; Ferrándiz, C.; Hecht, V.; Dalmais, M.; Lund, O.S.; Sussmilch, F.C.; Taylor, S.A.; Bendahmane, A.; Ellis, T.N.; Beltrán, J.P. VEGETATIVE1 is essential for development of the compound inflorescence in pea. Nat. Commun. 2012, 3, 797. [Google Scholar] [CrossRef]
- Wang, Z.; Luo, Y.H.; Li, X.; Wang, L.P.; Xu, S.L.; Yang, J.; Weng, L.; Sato, S.S.; Tabata, S.; Ambrose, M.; et al. Genetic control of floral zygomorphy in pea (Pisum sativum L.). Proc. Natl. Acad Sci. USA 2008, 105, 10414–10419. [Google Scholar] [CrossRef] [PubMed]
- Coen, E.S.; Meyerowitz, E.M. The war of the whorls: Genetic interactions controlling flower development. Nature 1991, 353, 31. [Google Scholar] [CrossRef] [PubMed]
- Irish, V. The ABC model of floral development. Curr. Biol. 2017, 27, R887–R890. [Google Scholar] [CrossRef] [PubMed]
- Tucker, S.C. Overlapping organ initiation and common primordia in flowers of Pisum sativum (Leguminosae: Papilionoideae). Am. J. Bot. 1989, 76, 714–729. [Google Scholar] [CrossRef]
- Ferrandiz, C.; Navarro, C.; Gomez, M.; Canas, L.; Beltran, J. Flower development in Pisum sativum: From the war of the whorls to the battle of the common primordia. Dev. Genet. 1999, 25, 280–290. [Google Scholar] [CrossRef]
- Zhao, Y. Essential roles of local auxin biosynthesis in plant development and in adaptation to environmental changes. Annu. Rev. Plant Biol. 2018, 69, 417–435. [Google Scholar] [CrossRef] [PubMed]
- McAdam, E.L.; Meitzel, T.; Quittenden, L.J.; Davidson, S.E.; Dalmais, M.; Bendahmane, A.I.; Thompson, R.; Smith, J.J.; Nichols, D.S.; Urquhart, S.; et al. Evidence that auxin is required for normal seed size and starch synthesis in pea. New Phytol. 2017, 216, 193–204. [Google Scholar] [CrossRef]
- McAdam, S.A.M.; Eleouet, M.P.; Best, M.; Brodribb, T.J.; Carins Murphy, M.; Cook, S.D.; Dalmais, M.; Dimitriou, T.; Gélinas-Marion, A.; Gill, W.M.; et al. Linking auxin with photosynthetic rate via leaf venation. Plant Physiol. 2017, 175, 351–360. [Google Scholar] [CrossRef]
- Alves-Carvalho, S.; Aubert, G.; Carrère, S.; Cruaud, C.; Brochot, A.-L.; Jacquin, F.; Klein, A.; Martin, C.; Boucherot, K.; Kreplak, J.; et al. Full-length de novo assembly of RNA-seq data in pea (Pisum sativum L.) provides a gene expression atlas and gives insights into root nodulation in this species. Plant J. 2015, 84, 1–19. [Google Scholar] [CrossRef]
- Tivendale, N.D.; Davies, N.W.; Molesworth, P.P.; Davidson, S.E.; Smith, J.A.; Lowe, E.K.; Reid, J.B.; Ross, J.J. Reassessing the role of N-hydroxytryptamine in auxin biosynthesis. Plant Physiol. 2010, 154, 1957–1965. [Google Scholar] [CrossRef] [PubMed]
- Swiecicki, W. A new gene CRISPOID (crd) on chromosome 1. Pisum Newsl. 1989, 21, 73–74. [Google Scholar]
- Domoney, C.; Knox, M.; Moreau, C.; Ambrose, M.; Palmer, S.; Smith, P.; Christodoulou, V.; Isaac, P.G.; Hegarty, M.; Blackmore, T. Exploiting a fast neutron mutant genetic resource in Pisum sativum (pea) for functional genomics. Funct. Plant Biol. 2013, 40, 1261–1270. [Google Scholar] [CrossRef] [PubMed]
- Kai, K.; Horita, J.; Wakasa, K.; Miyagawa, H. Three oxidative metabolites of indole-3-acetic acid from Arabidopsis thaliana. Phytochemistry 2007, 68, 1651–1663. [Google Scholar] [CrossRef] [PubMed]
- Novák, O.; Hényková, E.; Sairanen, I.; Kowalczyk, M.; Pospíšil, T.; Ljung, K. Tissue-specific profiling of the Arabidopsis thaliana auxin metabolome. Plant J. 2012, 72, 523–536. [Google Scholar] [CrossRef]
- Kreplak, J.; Madoui, M.-A.; Cápal, P.; Novák, P.; Labadie, K.; Aubert, G.; Bayer, P.E.; Gali, K.K.; Syme, R.A.; Main, D. A reference genome for pea provides insight into legume genome evolution. Nat. Genet. 2019, 51, 1411–1422. [Google Scholar] [CrossRef] [PubMed]
- Lester, D.R.; Ross, J.J.; Davies, P.J.; Reid, J.B. Mendel’s stem length gene (Le) encodes a gibberellin 3 β-hydroxylase. Plant Cell 1997, 9, 1435–1443. [Google Scholar] [CrossRef] [PubMed]
- Hladik, P.; Petrik, I.; Zukauskaite, A.; Novak, O.; Pencik, A. Metabolic profiles of 2-oxindole-3-acetyl-amino acid conjugates differ in various plant species. Front. Plant Sci. 2023, 14, 1217421. [Google Scholar] [CrossRef]
- Zhang, J.; Lin, J.E.; Harris, C.; Pereira, F.C.M.; Wu, F.; Blakeslee, J.J.; Peer, W.A. DAO1 catalyzes temporal and tissue-specific oxidative inactivation of auxin in Arabidopsis thaliana. Proc. Natl. Acad. Sci. USA 2016, 113, 11010–11015. [Google Scholar] [CrossRef]
- Kumar, S.; Stecher, G.; Li, M.; Knyaz, C.; Tamura, K. MEGA X: Molecular evolutionary genetics analysis across computing platforms. Mol. Biol. Evol. 2018, 35, 1547–1549. [Google Scholar] [CrossRef]
- Davis, K.; Gkotsi, D.S.; Smith, D.R.; Goss, R.J.; Caputi, L.; O’Connor, S.E. Nicotiana benthamiana as a transient expression host to produce auxin analogs. Front. Plant Sci. 2020, 11, 581675. [Google Scholar] [CrossRef] [PubMed]
- Kaur, H.; Ozga, J.A.; Reinecke, D.M. Balancing of hormonal biosynthesis and catabolism pathways, a strategy to ameliorate the negative effects of heat stress on reproductive growth. Plant Cell Environ. 2021, 44, 1486–1503. [Google Scholar] [CrossRef] [PubMed]
- Chabikwa, T.G.; Brewer, P.B.; Beveridge, C.A. Initial bud outgrowth occurs independent of auxin flow from out of buds. Plant Physiol. 2019, 179, 55–65. [Google Scholar] [CrossRef] [PubMed]
- DeMason, D.A.; Chawla, R. Roles for auxin during morphogenesis of the compound leaves of pea (Pisum sativum). Planta 2004, 218, 435–448. [Google Scholar] [CrossRef] [PubMed]
- Givnish, T.J.; Kriebel, R. Causes of ecological gradients in leaf margin entirety: Evaluating the roles of biomechanics, hydraulics, vein geometry, and bud packing. Am. J. Bot. 2017, 104, 354–366. [Google Scholar] [CrossRef] [PubMed]
- Mentink, R.A.; Tsiantis, M. From limbs to leaves: Common themes in evolutionary diversification of organ form. Front. Genet. 2015, 6, 284. [Google Scholar] [CrossRef] [PubMed]
- Guerra, S.; Peressotti, A.; Peressotti, F.; Bulgheroni, M.; Baccinelli, W.; D’Amico, E.; Gómez, A.; Massaccesi, S.; Ceccarini, F.; Castiello, U. Flexible control of movement in plants. Sci. Rep. 2019, 9, 16570. [Google Scholar] [CrossRef]
- Gerrath, J.M.; Côté, R.; Farquhar, M. Pea (Pisum sativum L.) tendril-surface changes are correlated with changes in functional development. Int. J. Plant Sci. 1999, 160, 261–274. [Google Scholar] [CrossRef]
- Jaffe, M.; Galston, A. Physiological studies on pea tendrils. I. Growth and coiling following mechanical stimulation. Plant Physiol. 1966, 41, 1014–1025. [Google Scholar] [CrossRef]
- Engelberth, J. Mechanosensing and signal transduction in tendrils. Adv. Space Res. 2003, 32, 1611–1619. [Google Scholar] [CrossRef]
- Jaffe, M. The role of auxin in the early events of the contact coiling of tendrils. Plant Sci. Lett. 1975, 5, 217–225. [Google Scholar] [CrossRef]
- He, L.; Lei, Y.W.; Li, X.; Peng, Q.C.; Liu, W.; Jiao, K.Y.; Su, S.H.; Hu, Z.B.; Shen, Z.G.; Luo, D. SYMMETRIC PETALS 1 encodes an ALOG domain protein that controls floral organ internal asymmetry in pea (Pisum sativum L.). Int. J. Mol. Sci. 2020, 21, 4060. [Google Scholar] [CrossRef] [PubMed]
- Jia, Z.; Giehl, R.F.; von Wirén, N. Local auxin biosynthesis acts downstream of brassinosteroids to trigger root foraging for nitrogen. Nat. Commun. 2021, 12, 5437. [Google Scholar] [CrossRef] [PubMed]
- Choudry, S.G.; Husbands, A.Y. Pick a side: Integrating gene expression and mechanical forces to polarize aerial organs. Curr. Opin. Plant Biol. 2023, 2023, 102460. [Google Scholar] [CrossRef] [PubMed]
- DeMason, D.A.; Polowick, P.L. Patterns of DR5:: GUS expression in organs of pea (Pisum sativum). Int. J. Plant Sci. 2009, 170, 1–11. [Google Scholar] [CrossRef]
- Brumos, J.; Robles, L.M.; Yun, J.; Vu, T.C.; Jackson, S.; Alonso, J.M.; Stepanova, A.N. Local auxin biosynthesis is a key regulator of plant development. Dev. Cell 2018, 47, 306–318.e5. [Google Scholar] [CrossRef]
- Band, L.R.; Wells, D.M.; Fozard, J.A.; Ghetiu, T.; French, A.P.; Pound, M.P.; Wilson, M.H.; Yu, L.; Li, W.; Hijazi, H.I. Systems analysis of auxin transport in the Arabidopsis root apex. Plant Cell 2014, 26, 862–875. [Google Scholar] [CrossRef]
- Grieneisen, V.A.; Xu, J.; Marée, A.F.; Hogeweg, P.; Scheres, B. Auxin transport is sufficient to generate a maximum and gradient guiding root growth. Nature 2007, 449, 1008–1013. [Google Scholar] [CrossRef]
- Adamowski, M.; Friml, J. PIN-dependent auxin transport: Action, regulation, and evolution. Plant Cell 2015, 27, 20–32. [Google Scholar] [CrossRef]
- Won, C.; Shen, X.; Mashiguchi, K.; Zheng, Z.; Dai, X.; Cheng, Y.; Kasahara, H.; Kamiya, Y.; Chory, J.; Zhao, Y. Conversion of tryptophan to indole-3-acetic acid by TRYPTOPHAN AMINOTRANSFERASES OF ARABIDOPSIS and YUCCAs in Arabidopsis. Proc. Natl. Acad. Sci. USA 2011, 108, 18518–18523. [Google Scholar] [CrossRef]
- Gélinas-Marion, A.; Nichols, D.S.; Ross, J.J. Conversion of unstable compounds can contribute to the auxin pool during sample preparation. Plant Physiol. 2020, 183, 1432–1434. [Google Scholar] [CrossRef]
- Hayashi, K.-i.; Arai, K.; Aoi, Y.; Tanaka, Y.; Hira, H.; Guo, R.; Hu, Y.; Ge, C.; Zhao, Y.; Kasahara, H. The main oxidative inactivation pathway of the plant hormone auxin. Nat. Commun. 2021, 12, 6752. [Google Scholar] [CrossRef] [PubMed]
- Ross, J.J.; Gélinas-Marion, A. Two pathways become one. Nat. Plants 2021, 7, 1546–1547. [Google Scholar] [CrossRef] [PubMed]
- Serra-Picó, M.; Hecht, V.; Weller, J.; Benlloch, R.; Madueño, F. Identification and characterization of putative targets of VEGETATIVE1/FULc, a key regulator of development of the compound inflorescence in pea and related legumes. Front. Plant Sci. 2022, 13, 765095. [Google Scholar] [CrossRef] [PubMed]



| Genotype | Number of Leaflets Per Plant | Number of Tendrils Per Plant | Number of Protuberances and Hair-like Organs Per Plant | Total Number of Leaflets, Tendrils, Protuberances, and Hair-like Organs Per Plant |
|---|---|---|---|---|
| WT-3 | 28.4 ± 0.5 | 35.5 ± 0.8 | 0 | 63.9 ± 1.2 |
| crd-3 | 19.3 ± 0.7 | 21.0 ± 1.0 | 5.0 ± 0.8 | 45.2 ± 1.2 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Gélinas-Marion, A.; Eléouët, M.P.; Cook, S.D.; Vander Schoor, J.K.; Abel, S.A.G.; Nichols, D.S.; Smith, J.A.; Hofer, J.M.I.; Ross, J.J. Plant Development in the Garden Pea as Revealed by Mutations in the Crd/PsYUC1 Gene. Genes 2023, 14, 2115. https://doi.org/10.3390/genes14122115
Gélinas-Marion A, Eléouët MP, Cook SD, Vander Schoor JK, Abel SAG, Nichols DS, Smith JA, Hofer JMI, Ross JJ. Plant Development in the Garden Pea as Revealed by Mutations in the Crd/PsYUC1 Gene. Genes. 2023; 14(12):2115. https://doi.org/10.3390/genes14122115
Chicago/Turabian StyleGélinas-Marion, Ariane, Morgane P. Eléouët, Sam D. Cook, Jacqueline K. Vander Schoor, Steven A. G. Abel, David S. Nichols, Jason A. Smith, Julie M. I. Hofer, and John J. Ross. 2023. "Plant Development in the Garden Pea as Revealed by Mutations in the Crd/PsYUC1 Gene" Genes 14, no. 12: 2115. https://doi.org/10.3390/genes14122115
APA StyleGélinas-Marion, A., Eléouët, M. P., Cook, S. D., Vander Schoor, J. K., Abel, S. A. G., Nichols, D. S., Smith, J. A., Hofer, J. M. I., & Ross, J. J. (2023). Plant Development in the Garden Pea as Revealed by Mutations in the Crd/PsYUC1 Gene. Genes, 14(12), 2115. https://doi.org/10.3390/genes14122115








